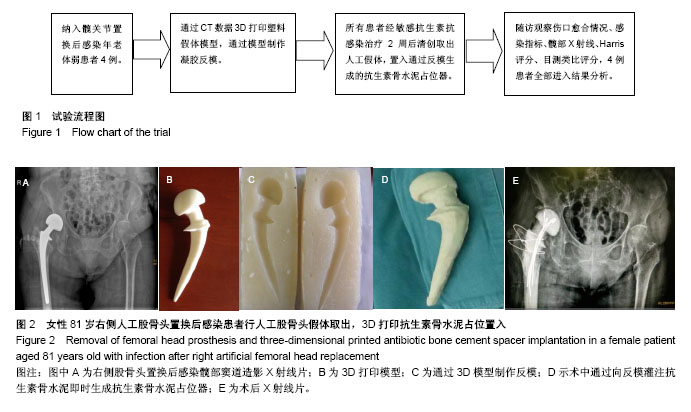
| [1] Won SH, Lee YK, Ha YC,et al.Improving pre-operative planning for complex total hip replacement with a rapid prototype model enabling surgical simulation.Bone Joint J. 2013;95-B:1458-1463.[2] Tricot M, Duy KT, Docquier PL. 3D-corrective osteotomy using surgical guides for posttraumatic distal humeral deformity. Acta Orthop Belg. 2012;78(4):538-542.[3] Corona PS, Espinal L, Rodríguez-Pardo D, et al. Antibiotic susceptibility in gram-positive chronic joint arthroplasty infections: increased aminoglycoside resistance rate in patients with prior aminoglycoside-impregnated cement spacer use. J Arthroplasty. 2014;29:1617-1621.[4] Cui Q, Mihalko WM, Shields JS, et al. Antibiotic-impregnated cement spacers for the treatment of infection associated with total hip or knee arthroplasty. J Bone Joint Surg Am. 2007; 89(4):871.[5] Mahmud T, Lyons MC, Naudie DD, et al. Assessing the gold standard: a review of 253 two-stage revisions for infected TKA. Clin Orthop Relat Res. 2012;470(10):2730–2736.[6] Iarikov D, Demian H, Rubin D, et al. Choice and doses of antibacterial agents for cement spacers in treatment of prosthetic joint infections: review of published studies. Clin Infect Dis. 2012;55(11):1474.[7] Jacobs C, Christensen CP, Berend ME. Static and mobile antibiotic-impregnated cement spacers for the management of prosthetic joint infection. J Am Acad Orthop Surg. 2009; 17(6):356.[8] 杨静,段宏,裴福兴. 髋关节置换术后慢性感染伴窦道形成的手术治疗[J]. 中华骨科杂志,2001,12(21):741-144.[9] 周勇刚,肖逸鹏,王岩,等.二期翻修在人工髋关节置换术后感染治疗中的作用[J]. 中国矫形外科杂志,2007,15(11):808-810.[10] Penner MJ, Duncan CP, MAsri BA, et al. The in vitro elution characteristics of antibiotic-loaded CMW and Palacos-R bone cements. J Arthroplasty. 1999;14:209-214.[11] 刘相成,周勇刚,王岩,等. 自制关节型抗生素骨水泥占位器在人工髋关节置换术后感染二期翻修中的应用[J].中华外科杂志, 2010,48(14):1050-1054.[12] Jiranek WA, Hanssen AD, Greenwald AS. Antibiotic-loaded bone cement for infection prophylaxis in total joint replacement. J Bone Joint Surg. 2006;88(11):2487-2500.[13] Penner MJ, Masri BA, Duncan CP. Elution characteristics of vancomycin and tobramycin combined in acrylic bone-cement. J Arthroplasty. 1996;11(8):939-944.[14] Stevens CM, Tetsworth KD, Calhoun JH, et al. An articulated antibiotic spacer used for infected total knee arthroplasty:A comparative in vitro elution study of Simplex and Palacos bone cements. J Orthop Res. 2005;23(1):27-33.[15] Masri BA, Duncan CP, Beauchamp CP. Long-term elution of antibiotics from bone-cement: An in vivo study using the prosthesis of antibiotic-loaded acrylic cement (PROSTALAC) system. J Arthroplasty. 1998;13(3):331-338.[16] Meseguer-Olmo L, Vicente-Ortega V, Alcaraz-Baños M, et al. In-vivo behavior of Si-hydroxyapatite/polycaprolactone/DMB scaffolds fabricated by 3D printing. J Biomed Mater Res A. 2013;101(7):2038-2048.[17] Cui X, Breitenkamp K, Finn MG, et al. Direct human cartilage repair using three-dimensional bioprinting technology. Tissue Eng Part A .2012;18(11):1304-1312.[18] Faulkner-Jones A, Greenhough S, King JA, et al. Development of a valve-based cell printer for the formation of human embryonic stem cell spheroid aggregates. Biofabrication. 2013;5(1):15013.[19] McGlothan KR, Gosmanova EO. A case report of acute interstitial nephritis associated with antibiotic-impregnated orthopedic bone- cement spacer. Tenn Med. 2012;105:37-40.[20] Mengnjo A, Carnero G, Govindasamy R. Acute kidney injury after total hip arthroplasty (THA) and antibiotic spacer placement. Am J Kidney Dis. 2012;59(4):B56.[21] King GS. Acute kidney injury following antibiotic spacer place-ment for two-stage arthroplasty. Int J Pharm Res Scholars (IJPRS).2014;3(2):391-395. |