Chinese Journal of Tissue Engineering Research ›› 2015, Vol. 19 ›› Issue (11): 1774-1781.doi: 10.3969/j.issn.2095-4344.2015.11.025
Previous Articles Next Articles
Neuroprotective effect of Chinese herbal monomers and extracts via activation of Nrf2 signal pathway
Zhang Li-na1, Wang Yuan-yuan2, Huang Ying-juan2, Jin Ming-hua2
- 1 Department of Traditional Chinese Medicine, Sixth Affiliated Hospital of Sun Yat-sen University, Guangzhou 510655, Guangdong Province, China; 2 Department of Traditional Chinese Medicine, First Affiliated Hospital of Sun Yat-sen University, Guangzhou 510080, Guangdong Province, China
-
Revised:2015-01-24Online:2015-03-12Published:2015-03-12 -
Contact:Jin Ming-hua, M.D., Associate chief physician, Department of Traditional Chinese Medicine, First Affiliated Hospital of Sun Yat-sen University, Guangzhou 510080, Guangdong Province, China -
About author:Zhang Li-na, Master, Physician, Department of Traditional Chinese Medicine, Sixth Affiliated Hospital of Sun Yat-sen University, Guangzhou 510655, Guangdong Province, China
CLC Number:
Cite this article
Zhang Li-na, Wang Yuan-yuan, Huang Ying-juan, Jin Ming-hua. Neuroprotective effect of Chinese herbal monomers and extracts via activation of Nrf2 signal pathway[J]. Chinese Journal of Tissue Engineering Research, 2015, 19(11): 1774-1781.
share this article
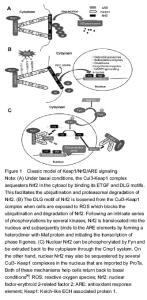
The general information of the included articles There are 14 articles included because of background and mechanism[1-14]; 3 articles of alkaloids[15-17]; 5 articles of organic acids[18-22]; 2 articles of phenols[23-24]; 1 articles of saponins and terpenes[25]; 12 articles of flavonoids[26-37]; 7 articles of alkaloids[38-44]; 2 articles of other single and composite components[45-46]. The evaluation methods involved in the included studies Methods in the included studies include immunohistochemistry, PCR, western blot and wlectron microscope, etc. Neuroprotection of alkaloids as monomers Oxymatrine Oxymatrine is the major quinolizidine alkaloid extracted from the root of sophora flavescens ait, and has been proved to be protective function after brain ischemia in recent studies. The results indicated that the ischemic infarct and edema were significantly reduced in rats that received oxymatrine, with a corresponding improvement in neurological function after cerebral ischemia- reperfusion. In immunohistochemistry and western blot analyses, Nrf2 and HO-1 were up-regulated in the ischemic cortex, beginning at 6 hours, peaking at 48 hours and declining at 72 hours after cerebral ischemia-reperfusion. Intraperitoneal injection of oxymatrine inhibited the production of lipid peroxidation and increased the activities of Nrf2 and HO-1 in rat’s brain after cerebral ischemia-reperfusion. Taken together, these results suggest that oxymatrine administered systemically protects the brain against focal ischemia-reperfusion damage at the early stage of stroke, and activating Nrf2/HO-1 pathway may contribute to the neuroprotective action of oxymatrine in the rat focal brain ischemia-reperfusion model. Thus, treatment of stroke with oxymatrine may prevent severe consequences after brain attack[15]. Sulforaphane Sulforaphane is a multifunctional enzyme inducer and induce the body to produce a II type detoxification enzyme of glutathione transferase and quinone reductase against free radicals injuries. It commonly exists in cruciferous vegetables such as carrots, cabbage, broccoli and other vegetables and in one of Chinese herbs of radish seed. We have tested the hypothesis that sulforaphane, a naturally occurring isothiocyanate that is also a known activator of the ARE/Nrf2 antioxidant pathway, can protect immature neurons from oxidative stress-induced death. The hypothesis was tested with primary mouse hippocampal neurons exposed to either oxygen and glucose deprivation or hemin. Treatment of immature neurons with sulforaphane immediately after the oxygen and glucose deprivation during reoxygenation was effective in protecting immature neurons from delayed cell death. Exposure of immature hippocampal neurons to hemin induced significant cell death, and both pre- and co-treatment with sulforaphane were remarkably effective in blocking cytotoxicity. RT-PCR analysis showed that several Nrf2-dependent cytoprotective genes, including NAD(P)H quinone oxidoreductase 1, HO-1, and glutamate-cysteine ligase modifier subunit, which are involved in glutathione biosynthesis, were up-regulated following sulforaphane treatment both in control neurons and following exposure to oxygen and glucose deprivation and hemin. These results indicate that sulforaphane activates the ARE/Nrf2 pathway of antioxidant defense and protects immature neurons from death caused by stress paradigms relevant to those associated with ischemic and traumatic injury to the immature brain[16]. Berberine Berberine is one of the major alkaloids and has been reported to have a variety of pharmacologic effects, including inhibition of cell cycle progression. Here, we investigated the mechanisms of berberine protection of neuronal cells from cell death induced by the Parkinson’s disease-related neurotoxin 6-hydroxydopamine (6-OHDA). Pretreatment of SH-SY5Y cells with berberine significantly reduced 6-OHDA-induced generation of ROS, caspase-3 activation, and subsequent cell death. Berberine also upregulated HO-1 expression, which conferred protection against 6-OHDA-induced dopaminergic neuron injury and besides, effect of berberine on HO-1 was reversed by siRNA-Nrf2. Furthermore, berberine induced PI3K/Akt and p38 activation, which is involved in the induction of Nrf2 expression and neuroprotection. These results suggest that berberine may be useful as a therapeutic agent for the treatment of dopaminergic neuronal diseases[17]. Neuroprotection of organic acids as monomers Ferulic acid Angelica, prepared from the rhizome of the plant Angelica sinesis, is a Chinese herb traditionally used in the treatment of women’s menopausal symptoms and is one of the herbs in Shiquan Dabu Tang. Angelica has been shown to hold anti-inflammatory properties and antioxidant activities, especially when used concurrently with other herbs[18-19]. It has also been found that Angelica may exert protection against neuronal oxidative stress on rat cerebral ischemia/reperfusion models[20]. Dietz and colleagues have reported that the major lipophilic constituent of Angelica, Z-lingustilide, reduces oxidative stress through up-regulation of antioxidant enzymes such as quinone oxidoreductase 1, a Nrf2 pathway gene[21]. The study showed that both angelica extract and Z-lingustilide induced a dose-dependent increase of quinone oxidoreductase 1 in an ARE luciferase reporter assay. Further matrix assisted laser desorption/ionization time-of-flight mass spectrometry and liquid chromatography-tandem mass spectrometry analysis of the incubation mixture reveals that Z-lingustilide is able to alkylate cysteine residues in the KEAP1 protein which allows Nrf2 to bind to the ARE and activate transcription of Nrf2-ARE genes[21]. In addition, this Chinese herb can inhibit cyclooxygenase-2 expression at mRNA and protein levels[19]. Ursolic acid We demonstrated the hypothesis that ursolic acid, a natural pentacyclic triterpenoid acid, isolated from edible plants in the Oleaceae family, a well-know antioxidative and anti-inflammatory reagent, protects the brain against ischemic injury by activating the Nrf2 pathway. Nrf2-/- and wild-type mice were induced into focal cerebral ischemia by transient middle cerebral artery occlusion, and received ursolic acid treatment immediately after middle cerebral artery occlusion. The behavioral dysfunction, infarct size, and the expression of Nrf2, HO-1 and inflammatory factors (Toll-like receptor 4 and nuclear factor-κB) in ischemic brain were measured at 24 hours after stroke. Ursolic acid treatment significantly improved neurological deficit and reduced infarct size in wild-type mice after middle cerebral artery occlusion. Administration of ursolic acid also decreased the product of lipid peroxidation, promoted the actibation of Nrf2 pathway and decreased the expression of Toll-like receptor 4 and nuclear factor-κB after stroke in wild-type mice. However, Nrf2-/- mice demonstrated more severe neurologic deficits, infarct size and inflammatory damage after middle cerebral artery occlusion, and did not benefit from the protective effect of ursolic acid. The results indicated that ursolic acid protected the brain against ischemic injury in mice by anti-oxidative and anti-inflammatory effects after middle cerebral artery occlusion. Activation of the Nrf2 pathway contributes to the neuroprotective effects induced by ursolic acid in cerebral ischemia[22]. Neuroprotection of phenolic substances as monomers Curcumin Oxidative and cytotoxic damage plays an important role in cerebral ischemic pathogenesis and may represent a target for treatment. Curcumin is proved to elicit a vanity of biological effects through its antioxidant and anti-inflammatory properties. Curcumin protect the brain from damage caused by middle cerebral artery occlusion, and this effect may be through upregulation of the transcription factor Nrf2 expression. Nrf2 may be one of the strategic targets for cerebral ischemic therapies[23]. Epicatechin Catechin is also called tea tannin. The polyphenols content of catechin in tea is 75% to 80%. In one study, wild-type mice pretreated orally with 5, 15, or 30 mg/kg epicatechin before middle cerebral artery occlusion had significantly smaller brain infarcts and decreased neurologic deficit scores than did the vehicle-treated group. Mice that were posttreated with 30 mg/kg of epicatechin at 3.5 hours after middle cerebral artery occlusion also had significantly smaller brain infarcts and decreased neurologic deficit scores. Similarly, wild-type mice pretreated with 30 mg/kg of epicatechin and subjected to N-methyl-D-aspartate-induced excitotoxicity had significantly smaller lesion volumes. Cell viability assays with neuronal cultures further confirmed that epicatechin could protect neurons against oxidative insults. Interestingly, the epicatechin-associated neuroprotection was mostly abolished in mice lacking the HO-1 or Nrf2, and in neurons derived from these knockout mice. These results suggest that epicatechin exerts part of its beneficial effect through activation of Nrf2 and an increase in the neuroprotective HO-1[24]. Neuroprotection of terpenoids and saponins as monomers Ginsenoside Intestinal ischemia reperfusion is a serious clinical condition associated with simultaneous multiple organ dysfunction. In a study aiming to investigate the effects of ginsenoside Rb1 on intestinal ischemia reperfusion induced renal injury in mice, an intestinal ischemia reperfusion mouse model was established by superior mesenteric artery occlusion for 45 minutes, followed by reperfusion for 2 hours. Intestinal ischemia reperfusion induced renal injury characterized by increase of blood urea nitrogen, creatinine and neutrophil gelatinase associated lipocalin in serum, malonaldehyde levels and decrease of superoxide dismutase levels in the renal tissues. Ginsenoside Rb1 (30, 60 mg/kg) given intraperitoneally before reperfusion attenuated renal injury, which was associated with decrease of blood urea nitrogen, creatinine and neutrophil gelatinase associated lipocalin in serum, malonaldehyde levels and increase of superoxide dismutase levels in the renal tissues. Furthermore, the immunohistochemistry and western blot data showed that ginsenoside Rb1 dramatically reversed intestinal ischemia reperfusion induced renal injury, associated with upregulated Nrf2 and HO-1 in renal tissues. These data suggests that ginsenoside Rb1 attenuates acute renal injury induced by intestinal ischemia reperfusion by activating the Nrf2/ARE pathway[25]. Neuroprotection of flavonoids as monomers Quercetin Some research showed rapid quercetin internalization into neurons, reaching the nucleus after its addition to the culture. Quercetin pretreatment increased total glutathione levels, but did not increase Trx2. Interestingly it caused Nrf2 nuclear translocation and significantly increased GCLC gene expression. At the moment of hydrogen peroxide addition, intracellular quercetin or related metabolites were undetectable in the cultures although quercetin pretreatment prevented neuronal death from the oxidant exposure. The findings suggest alternative mechanisms of quercetin neuroprotection beyond its long-established ROS scavenging properties, involving Nrf2-dependent modulation of the glutathione redox system[26]. EGb 761 Ginkgo biloba/EGb 761® (EGb 761) is a popular and standardized natural extract used worldwide for the treatment of many ailments. Although EGb 761 is purported to have a plethora of benefits, scholars are interested to study the neuroprotective properties of EGb 761 and its components and determine whether Nrf2/HO-1 induction of the collapsin response mediator protein 2 pathway contributes to neuroprotection. Mice were pretreated with EGb 761 or one of its constituents (bilobalide, ginkgolide A, ginkgolide B, and terpene free material) for 7 days and then subjected to transient middle cerebral artery occlusion and 48 hours of reperfusion. All components except terpene free material significantly reduced infarct volumes and neurologic deficits. The antioxidant and neuritogenic properties of EGb 761 in primary neurons were examined. Compared with vehicle-treated cells, pretreatment with EGb 761 significantly enhanced the survival of neurons exposed to tertiary butylhydroperoxide, hydrogen peroxide, and N-methyl-D-aspartate. Bilobalide and ginkgolide A also protected cells against N-methyl-D- aspartate-induced excitotoxicity. Immunofluorescence and Western blot analysis showed that EGb 761 pretreatment significantly increased the protein expression levels of Nrf2, HO-1, GAPDH, β-actin, collapsin response mediator protein 2, and histone H3 during tertiary butylhydroperoxide- induced oxidative stress. These findings suggest that EGb 761 not only has antioxidant activity but also possesses neuritogenic potential. Demonstrating such effects for possible drug discovery may prove beneficial in stroke and ischemic brain injury[27]. Tanshinone The root of the plant Salvia miltiorrhiza, known as Danshen or red sage root, is a widely used in TCM clinic for treatment of cardiovascular disorders by improving blood circulation[28]. Its major chemical constituents have been found to be lipophilic tanshinones, mainly tanshinone IIA and the hydrophilic compounds tanshinol (also known as salvianolic acid A) and salvianolic acid B[29]. Recent studies have also shown that Danshen and its constituents possess anti-inflammatory effects through the inhibition of inducible nitric oxide synthase expression and cytokine secretion[30-31]. Some studies have shown that tanshinone IIA induces apoptosis and inhibits growth in leukemia THP-1 and human colon adenocarcinoma cells as well[32]. Tanshinone IIA also displays antioxidant protection against ROS-nduced oxidative stress through stress-activated kinases JNKs and p38 MAPK and by an increase in scavenging of oxygen free radicals[33-34]. The Nrf2 pathway has been known to interact with JNKs and p38 MAPK kinases which indirectly regulate the Nrf2 pathway through phosphorylation of Nrf2[33-34]. Zhang and colleagues showed that Nrf2 is involved in the effects of tanshinone IIA by reversing tumor necrosis factor-induced down-regulation of glutathione, NADPH, and glucose 6-phosphate dehydrogenase[35]. Small interfering RNA silencing of Nrf2 abolishs tanshinone IIA-induced up-regulation of glutathione and glucose 6-phosphate dehydrogenase[35]. It has also been found that the anti-inflammatory effects of tanshinone IIA directly result from the up-regulation of HO-1 through PI3-K/Akt and ERK pathways induced higher levels of Nrf2[36]. Tanshinone IIA activates the Nrf2 pathway through disruption of the Nrf2-KEAP1 complex and through kinase signaling pathways such as JNKs, p38 MAPK, PI3-K/Akt, and ERK to assist in the release of Nrf2. Based on the effectiveness on Nrf2, Danshen is also a promising medicine for the treatment of neuroprotection after stroke. Naringin In order to investigate neuroprotective role of naringin and delineate the mechanismof action on 3-nitropropionic acid-induced neurodegeneration, the rats were injected with 3- nitropropionic acid (10 mg/kg body weight/day, i.p.) for 2 weeks to develop neurodegeneration, while naringin (80 mg/kg body weight/day, orally) was administered throughout the experimental period, 1 hour prior to 3-nitropropionic acid exposure. Thereafter rats were euthanized for biochemical, histological, and molecular studies. Treatment with naringin ameliorated the reduced glutathione/oxidized glutathione ratio with concomitant decrease in the levels of hydroxyl radical, hydroperoxide and nitrite in 3-nitropropionic acid-induced rats. Nissl staining and transmission electron microscopic studies showed that naringin modulated 3-nitropropionic acid- induced histological changes. Naringin induces NAD(P)H: quinone oxidoreductase-1, HO-1, glutathione S-transferase P1 and gamma-glutamylcysteine ligase mRNA expressions through the activation of Nrf2 and decreases the expressions of pro-inflammatory mediators, such as tumor necrosis factor-alpha, cyclooxygenase-2 and inducible nitric oxide synthase. These results indicate that naringin may be beneficial in mitigating 3-nitropropionic acid-induced neurodegeneration through the enhancement of phase II and antioxidant gene expressions via Nrf2 activation, thereby modulating the oxidative stress and inflammatory responses[37]. Neuroprotection of alkaloids of essential oils as monomers Borneol Borneol is proved to protect human neuroblastoma cells (SH-SY5Y) against Aβ-induced toxicity, exert an antioxidative effect and suppress apoptosis, which is hopeful to be a candidate compound for developing therapeutic drug for the prevention and treatment of Alzheimer’s disease and other Aβ-related neurodegenerative diseases[38]. Notopterygium forbesii Boiss extract Qianghuo is one of Chinese herbs and prepared from the root and rhizome of the plant Notopterygium forbesii Boiss, which belongs to the umbelliferae family. Qianghuo has been used to treat the common cold, headache, rheumatism. Its extract contained sweet bean pure compounds and may possibly possess anti-inflammatory, diaphoretic, and analgesic properties[39-41]. Nine chemical constituents of Notopterygium forbesii Boiss have been isolated and identified[42]. Tang and co-workers found that the mechanism for Notopterygium forbesii Boiss underlying cancer preventive effects is correlated to the up-regulation of HO-1 and Notopterygium forbesii Boiss also has effects on oxidative stress[43-44]. It is found that Notopterygium forbesii Boiss induces oxidative stress and a rise in HO-1 proteins in human fetal hepatocytes through the activation of the Nrf2 pathway by the p38 MAPK pathway and reactive species. The active constituents are identified to be phenethyl ferulate, bergaptol, and isoimperatorin which contribute to the increased HO-1 levels. Extracts of Notopterygium forbesii Boiss also induce a six-fold increase in quinone oxidoreductase 1 levels. Notopterygium forbesii Boiss and its constituents also attenuate lipopolysaccharide-induced pro-inflammatory responses and inducible nitric oxide synthase and cyclooxygenase-2 overexpression[44]. Neuroprotection of other monomers and compound preparations Lipoic acid R-a-lipoic acid has a dramatic neuroprotective effect against oxidative stress-induced death of the retinal neuronal RGC-5 cell line. R-a-lipoic acid induces the expression of HO-1 by promoting the translocation of Nrf2 to the nucleus, and the mechanism underlying HO-1 induction by R-a-lipoic acid is examined by focusing on downstream signaling pathways. R-a-lipoic acid activates Akt, and HO-1 induction by R-a-lipoic acid (involving Nrf2 translocation to the nucleus) suppressed by PI3-K inhibitors. In addition, R-a-lipoic acid produces ROS, including hydrogen peroxide. Pretreatment with a ROS scavenger or a NADPH oxidase inhibitor can suppress R-a-lipoic acid-induced Nrf2 translocation to the nucleus and HO-1 induction. These results suggest that ROS production triggered by R-a-lipoic acid may modify keap1, which in turn induces HO-1 expression through the PI3K signaling pathway. Furthermore, R-a-lipoic acid significantly attenuates cell death and accumulation of 4-hydroxy-2-nonenal in the ertina induced by optic nerve injury in vivo through an HO-1 activity-dependent mechanism. These data demonstrate for the first time that R-a-lipoic acid exerts a neuroprotective effect against oxidative stress in retinal neurons in vitro and in vivo by inducing HO-1 through keap1/Nrf2 signaling[45]. Shiquan Dabu Tang Shiquan Dabu Tang is a TCM formula consisting of 10 ingredients, originally formulated in the China Song Dynasty in AD 1200. It has been used to help treat anemia, anorexia, exhaustion, fatigue, and general weakness[46]."

| [1] Chen G, Fang Q, Zhang J, et al. Role of the Nrf2-ARE pathway in early brain injury after experimental subarachnoid hemorrhage. J Neurosci Res. 2011;89(4):515-523. [2] Motohashi H, O'Connor T, Katsuoka F, et al. Integration and diversity of the regulatory network composed of Maf and CNC families of transcription factors. Gene. 2002;294(1-2):1-12. [3] Baird L, Dinkova-Kostova AT. The cytoprotective role of the Keap1-Nrf2 pathway. Arch Toxicol. 2011;85(4):241-272. [4] Zhang MJ, An CR, Gao YQ, et al. Emerging roles of Nrf2 and phase II antioxidant enzymes in neuroprotection. J Prog Neurobiol. 2013;(100):30-47. [5] Shih AY, Li P, Murphy TH. A small-molecule-inducible Nrf2-mediated antioxidant response provides effective prophylaxis against cerebral ischemia in vivo. J Neurosci. 2005;25 (44):10321-10335. [6] Zhang F, Wang S, Zhang M, et al. Pharmacological induction of heme oxygenase-1 by a triterpenoid protects neurons against ischemic injury. Stroke. 2012;43(5):1390-1397. [7] Zhao X, Sun G, Zhang J, et al. Transcription factor Nrf2 protects the brain from damage produced by intracerebral hemorrhage. Stroke. 2007;38:3280-3286. [8] Keep RF, Hua Y, Xi G. Intracerebral haemorrhage: mechanisms of injury and therapeutic targets. Lancet Neurol. 2012;11:720-731. [9] Chen M, Regan RF. Time course of increased heme oxygenase activity and expression after experimental intracerebral hemorrhage: correlation with oxidativeinjury. J Neurochem. 2007;(103):2015-2021. [10] Sun KH, Chang KH, Clawson S, et al. Glutathione-S- transferase P1 is a critical regulator of Cdk5 kinase activity. J Neurochem , 2011;118:902-914. [11] Wakade C, King MD, Laird MD, et al. Curcumin attenuates vascular inflammation and cerebral vasospasm after subarachnoid hemorrhage in mice. Antioxid Redox Signal. 2009;11:35-45. [12] Kuo CP, Lu CH, Wen LL, et al. Neuroprotective effect of curcumin in an experimental rat model of subarachnoid hemorrhage. Anesthesiology. 2011;115:1229-1238. [13] Zhang J, Zhu Y, Zhou D, et al. Recombinant human erythropoietin (rhEPO) alleviates early brain injury following subarachnoid hemorrhage in rats: possible involvement of Nrf2-ARE pathway. Cytokine. 2010;52:252-257. [14] Wang Z, Ma C, Meng CJ, et al. Melatonin activates the Nrf2-ARE pathway when it protects against early brain injury in a subarachnoid hemorrhage model. J Pineal Res. 2012;53(2):129-137. [15] Li M, Zhang XJ, Cui LL, et al. The Neuroprotection of oxymatrine in cerebral ischemia/reperfusion is related to nuclear factor erythroid 2-related factor 2 (nrf2)-mediated antioxidant response: role of nrf2 and hemeoxygenase-1 expression. Biol Pharm Bull. 2011;34(5):595-601. [16] Soane L, Li Dai W, Fiskum G, et al. Sulforaphane protects immature hippocampal neurons against death caused by exposure to hemin or to oxygen and glucose deprivation. J Neurosci Res. 2010;88:1355-1363. [17] Bae J, Lee D, Kim YK, et al. Berberine protects 6-hydroxydopamine -induced human dopaminergic neuronal cell death through the induction of heme oxygenase-1. Mol Cells. 2013;35(2):151-157. [18] Yang WJ, Li DP, Li JK, et al. Synergistic antioxidant activities of eight traditional Chinese herb pairs. Biol Pharm Bull. 2009;32(6):1021-1026. [19] Chu Q, Hashimoto K, Satoh K, et al. Effect of three herbal extracts on NO and PGE2 production by activated mouse macrophage-like cells. In vivo. 2009;23(4):537-544. [20] Lin Z, Zhu D, Yan Y, et al. An antioxidant phytotherapy to rescue neuronal oxidative stress. Evid Based Complement Alternat Med. 2011;2011:519-517. [21] Dietz BM, Liu D, Hagos GK, et al. Angelica sinensis and its alkylphthalides induce the detoxification enzyme NAD(P)H: quinine oxidoreductase 1 by alkylating Keap1. Chem Res Toxicol. 2008;21(10):1939-1948. [22] Li L, Zhang X, Cui L, et al. Ursolic acid promotes the neuroprotection by activating Nrf2 pathway after cerebral ischemia in mice. Brain Res. 2013;1497:32-39. [23] Yang C, Zhang X, Fan H, et al. Curcumin upregulates transcription factor Nrf2, HO-1 expression and protects rat brains against focal ischemia. Brain Res. 2009;1282:133-141. [24] Shah ZA, Li RC, Ahmad AS, et al. The flavanol (-)-epicatechin prevents stroke damage through the Nrf2/HO1 pathway. J Cereb Blood Flow Metab. 2010;30:1951-1961. [25] Sun Q, Meng QT, Jiang Y, et al. Ginsenoside Rb1 Attenuates Intestinal Ischemia Reperfusion Induced Renal Injury by Activating Nrf2/ARE Pathway. Molecules. 2012;17:7195-7205. [26] Arredondo F, Echeverry C, Abin-Carriquiry JA, et al. After cellular internalization, quercetin causes Nrf2 nuclear translocation, increases glutathione levels, and prevents neuronal death against an oxidative insult. Free Radical Biol Med. 2010;49(5):738-747. [27] Nada SE, Shah ZA. Preconditioning with Ginkgo biloba (EGb 761®) provides neuroprotection through HO1 and CRMP2. Neurobiol Dis. 2012;46:180-189. [28] Cheng TO. Danshen: what every cardiologist should know about this Chinese herbal drug. Int J Cardiol. 2006;110(3):411-412. [29] Zhou L, Zuo Z, Chow MS. Danshen: an overview of its chemistry, pharmacology, pharmacokinetics, and clinical use. J Clin Pharmacol. 2005;45(12):1345-1359. [30] Park EJ, Zhao YZ, Kim YC, et al. Preventive effects of a purified extract isolated from Salvia miltiorrhiza enriched with tanshinone I, tanshinone IIA and cryptotanshinone on hepatocyte in jury in vitro and in vivo. Food Chem Toxicol. 2009;47(11):2742-2748. [31] Fan GW, Gao XM, Wang H, et al. The anti-inflammatory activities of Tanshinone IIA, an active component of TCM, are mediated by estrogen receptor activation and inhibition of iNOS. J Steroid Biochem Mol Biol. 2009;113(3-5):275-280. [32] Su CC, Chen GW, Kang JC, et al. Growth inhibition and apoptosis induction by tanshinone IIA in human colon adenocarcinoma cells. Planta Med. 2008;74(11):1357-1362. [33] Yang R, Liu A, Ma X, et al. Sodium tanshinone IIA sulfonate protects cardiomyocytes against oxidative stress-mediated apoptosis through inhibiting JNK activation. J Cardiovasc Pharmacol. 2008;51(4):396-401. [34] Fu J, Huang H, Liu J, et al. Tanshinone IIA protects cardiac myocytes against oxidative stress-triggered damage and apoptosis. Eur J Pharmacol 2007;568(1-3):213-221. [35] Zhang HS, Wang SQ. Nrf2 is involved in the effect of tanshinone IIA on intracellular redox status in human aortic smooth muscle cells. Biochem Pharmacol. 2007;73(9): 1358-1366. [36] Chen TH, Hsu YT, Chen CH, et al. Tanshinone IIA from Salvia miltiorrhiza induces heme oxygenase-1 expression and inhibits lipopolysaccharide-induced nitric oxide expression in RAW 264.7 cells. Mitochondrion. 2007;7(1-2):101-105. [37] Gopinath K, Sudhandiran G. Naringin modulates oxidative stress and inflammation in 3-nitropropionic acid-induced neurodegeneration through the activation of nuclear factor-erythroid 2-related factor-2 signalling pathway. Neuroscience. 2012;227:134-143. [38] Hur J, Pak SC, Koo BS, et al. Borneol alleviates oxidative stress via upregulation of Nrf2 and Bcl-2 in SH-SY5Y cells. Pharm Biol. 2013;51(1):30-35. [39] Guo LQ, Taniguchi M, Chen QY, et al. Inhibitory potential of herbal medicines on human cytochrome P450-mediated oxidation: Properties of umbelliferous or citrus crude drugs and their relative prescriptions. Jpn J Pharmacol 2001; 85(4):399-408. [40] Yang XW, Gu ZM, Wang BX, et al. Comparison of anti-lipid peroxidative effects of the underground parts of Notopterygium incisum and N. forbesii in mice. Planta Med. 1991;57(5):399-402. [41] Okuyama E, Nishimura S, Ohmori S, et al. Analgesic componentof Notopterygium incisum Ting. Chem Pharm Bull (Tokyo). 1993;41(5):926-929. [42] Wang S, Wang T. Chemical constituents of Notopterygium forbesii Boiss. Zhongguo Zhong Yao Za Zhi. 1996;21(5): 295-296. [43] Tang SY, Wang H, Zhang W, et al. Notopterygium forbesii boiss extract and its active constituents increase reactive species and heme oxygenase-1 in human fetal hepatocytes: Mechanisms of action. Chem Res Toxicol. 2008; 21(12): 2414-2423. [44] Tang SY, Cheah IK, Wang H, et al. Notopterygium forbesii Boiss extract and its active constituent phenethyl ferulate attenuate pro-inflammatory responses to lipopolysaccharide in RAW 264.7 macrophages. A “protective” role for oxidative stress? Chem Res Toxicol. 2009; 22(8):1473-1482. [45] Koriyama Y, Nakayama Y, Matsugo S, et al. Protective effect of lipoic acid against oxidative stress is mediated by Keap1/Nrf2-dependent heme oxygenase-1 induction in the RGC-5 cell line. Brain Research 2013;1499:145-157. [46] Zee-Cheng RK. Shi-quan-da-bu-tang (ten significant tonic decoction), SQT. A potent Chinese biological response modifier in cancer immunotherapy, potentiation and detoxification of anticancer drugs. Methods Find Exp Clin Pharmacol. 1992;14(9):725-773. |
| [1] | Chen Ziyang, Pu Rui, Deng Shuang, Yuan Lingyan. Regulatory effect of exosomes on exercise-mediated insulin resistance diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 4089-4094. |
| [2] | Chen Yang, Huang Denggao, Gao Yuanhui, Wang Shunlan, Cao Hui, Zheng Linlin, He Haowei, Luo Siqin, Xiao Jingchuan, Zhang Yingai, Zhang Shufang. Low-intensity pulsed ultrasound promotes the proliferation and adhesion of human adipose-derived mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3949-3955. |
| [3] | Yang Junhui, Luo Jinli, Yuan Xiaoping. Effects of human growth hormone on proliferation and osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3956-3961. |
| [4] | Sun Jianwei, Yang Xinming, Zhang Ying. Effect of montelukast combined with bone marrow mesenchymal stem cell transplantation on spinal cord injury in rat models [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3962-3969. |
| [5] | Gao Shan, Huang Dongjing, Hong Haiman, Jia Jingqiao, Meng Fei. Comparison on the curative effect of human placenta-derived mesenchymal stem cells and induced islet-like cells in gestational diabetes mellitus rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3981-3987. |
| [6] | Hao Xiaona, Zhang Yingjie, Li Yuyun, Xu Tao. Bone marrow mesenchymal stem cells overexpressing prolyl oligopeptidase on the repair of liver fibrosis in rat models [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3988-3993. |
| [7] | Liu Jianyou, Jia Zhongwei, Niu Jiawei, Cao Xinjie, Zhang Dong, Wei Jie. A new method for measuring the anteversion angle of the femoral neck by constructing the three-dimensional digital model of the femur [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3779-3783. |
| [8] | Meng Lingjie, Qian Hui, Sheng Xiaolei, Lu Jianfeng, Huang Jianping, Qi Liangang, Liu Zongbao. Application of three-dimensional printing technology combined with bone cement in minimally invasive treatment of the collapsed Sanders III type of calcaneal fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3784-3789. |
| [9] | Qian Xuankun, Huang Hefei, Wu Chengcong, Liu Keting, Ou Hua, Zhang Jinpeng, Ren Jing, Wan Jianshan. Computer-assisted navigation combined with minimally invasive transforaminal lumbar interbody fusion for lumbar spondylolisthesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3790-3795. |
| [10] | Hu Jing, Xiang Yang, Ye Chuan, Han Ziji. Three-dimensional printing assisted screw placement and freehand pedicle screw fixation in the treatment of thoracolumbar fractures: 1-year follow-up [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3804-3809. |
| [11] | Shu Qihang, Liao Yijia, Xue Jingbo, Yan Yiguo, Wang Cheng. Three-dimensional finite element analysis of a new three-dimensional printed porous fusion cage for cervical vertebra [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3810-3815. |
| [12] | Wang Yihan, Li Yang, Zhang Ling, Zhang Rui, Xu Ruida, Han Xiaofeng, Cheng Guangqi, Wang Weil. Application of three-dimensional visualization technology for digital orthopedics in the reduction and fixation of intertrochanteric fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3816-3820. |
| [13] | Sun Maji, Wang Qiuan, Zhang Xingchen, Guo Chong, Yuan Feng, Guo Kaijin. Development and biomechanical analysis of a new anterior cervical pedicle screw fixation system [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3821-3825. |
| [14] | Lin Wang, Wang Yingying, Guo Weizhong, Yuan Cuihua, Xu Shenggui, Zhang Shenshen, Lin Chengshou. Adopting expanded lateral approach to enhance the mechanical stability and knee function for treating posterolateral column fracture of tibial plateau [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3826-3827. |
| [15] | Zhu Yun, Chen Yu, Qiu Hao, Liu Dun, Jin Guorong, Chen Shimou, Weng Zheng. Finite element analysis for treatment of osteoporotic femoral fracture with far cortical locking screw [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3832-3837. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||