Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (10): 1597-1603.doi: 10.3969/j.issn.2095-4344.2247
Previous Articles Next Articles
Preparation method of acellular matrix and its application in tissue engineering of parotid gland
Zhang Lin1, Wang Shuai2, Zhang Nini3, Huang Guilin3
- 1Hospital of Stomatology, Zunyi Medical University, Zunyi 563000, Guizhou Province, China; 2Department of Scientific Research, 3Department of Oral and Maxillofacial surgery, Hospital of Stomatology, Zunyi Medical University, Zunyi 563000, Guizhou Province, China
-
Received:2019-08-29Revised:2019-08-31Accepted:2019-10-21Online:2020-04-08Published:2020-02-18 -
Contact:Huang Guilin, MD, Professor, Department of Oral and Maxillofacial surgery, Hospital of Stomatology, Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
About author:Zhang Lin, Master candidate, Hospital of Stomatology, Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 81760201 and 81860198
CLC Number:
Cite this article
Zhang Lin, Wang Shuai, Zhang Nini, Huang Guilin. Preparation method of acellular matrix and its application in tissue engineering of parotid gland[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(10): 1597-1603.
share this article

2.1 脱细胞基质的制备方法 所有脱细胞方法均会破坏细胞外基质结构和组成,脱细胞的目的在于尽可能保留细胞外基质三维超微结构和成分,同时能彻底清除细胞及细胞残余。最佳脱细胞方法因组织与器官具体因素而异,如细胞和基质密度、组织厚度及形状等几何因素。完全清除所有细胞残留是不可能的,脱细胞过程中会不可避免的引起基质三维结构破坏及不良的炎症反应[2]。近些年来已研究出多种脱细胞方法,以最大限度保留细胞外基质的完整性。通常脱细胞方法可分为3种类型,见表1,包括化学方法(如酸和碱、低渗和高渗溶液、十二烷基硫酸钠和TritonX-100)、物理方法(如冻融、高静水压力学加压、超临界二氧化碳及电穿孔)、生物(酶)方法(如胰蛋白酶),上述脱细胞方法可单独或联合使用,以便提高脱细胞效率。 "

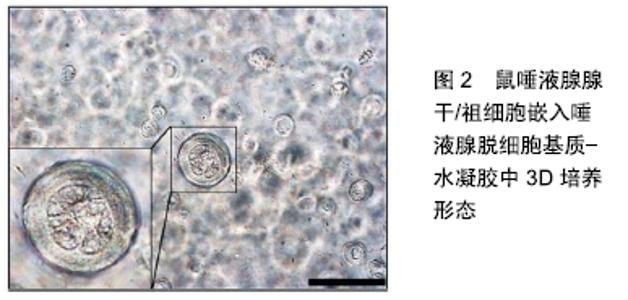
2.1.1 化学脱细胞方法 化学脱细胞方法中有几种常用洗涤剂,包括表面活性剂、酸、碱、高渗和低渗溶液及螯合剂;表面活性剂根据其电荷又可分为离子型、非离子型或两性离子型,这些洗涤剂均可溶解细胞膜并降解DNA。 表面活性剂:①离子型表面活性剂:含有带负电荷的亲水基团,可以溶解细胞质及核膜,使基质蛋白变性,破坏基质的天然结构[3]。广泛应用的离子型表面活性剂SDS能够有效去除致密组织中细胞核及细胞质蛋白,但基质超微结构、糖胺聚糖、生长因子及胶原蛋白也会相应损伤[4-7]。例如经SDS处理后的心脏瓣膜基质中胶原变得更加致密[7],同样,人及猪肺组织经脱细胞后纤维结构也将相应增加[8],糖胺聚糖和血管内皮生长因子等成分却相应减少,从而影响细胞功能及支架稳定性[9-10]。糖胺聚糖的减少可能是由于这些分子在细胞膜中的位置,使其在脱细胞过程中被破坏[11]。GILPIN等[12]用SDS灌注人及猪肺组织以制备肺脱细胞基质,结果基质保留肺血管结构及肺泡原有形态,该方法对肺脱细胞基质接种干细胞的营养及功能恢复至关重要。但SDS具有细胞毒性,因此必须彻底清洗组织以确保接种细胞的活性[13-14]。虽然经大多数表面活性剂处理过的组织通常需用PBS等溶液清洗,但SDS由于其离子性能较难去除。另一种常用离子型洗涤剂SDC已被用于脱细胞肺(如人、猪、鼠)和猪心脏瓣膜[15]。已经证明与非离子表面活性剂TritonX-100相比,SDC用于心脏瓣膜脱细胞的效果可能欠佳,因其核物质不能完全去 除[16],其他研究表明SDC是一种完全有效的肺组织脱细胞剂,其核物质去除效果与SDS、TritonX-100和CHAPS不相上下[12]。②非离子型表面活性剂:含有不带电荷的亲水性基团,破坏脂质与脂质蛋白之间相互作用,保留蛋白质之间的相互作用[17]。非离子型表面活性剂Triton X-100常用于去除组织中残留的SDS。与离子型表面活性剂相比,Triton X-100脱细胞后组织结构完整性的破坏相对较小,但会降低基质中糖胺聚糖含量[18]。Triton X-100 结合氢氧化铵可完全去除组织中所有DNA成分并保留更多的Ⅰ型胶原蛋白和超微结构,力学性能同样得以保留[19]。ROTH等[20]通过冻融循环和非离子型表面活性剂Triton X-100联合制备肌腱脱细胞基质。TAVASSOLI等[21]采用物理和化学相结合方法制备肌腱脱细胞基质,结果表明5个周期的液氮速冻与4 h的2.5% SDS处理相结合获得了最佳的脱细胞效果。③两性离子型表面活性剂:兼具离子型及非离子型表面活性剂特征,如HAPS、SB-10和SB-16。组织学分析证实CHAPS是薄组织较为理想的洗涤剂,如周围神经[22-23]、血管或肺[24-25],且不损害基质中的胶原蛋白和弹性蛋白,但会降低支架的机械性能,这与Triton X-100处理组织引起的力学性能下降相当[26];用于较厚组织可能无效,即使与SDS联合或单独使用[27],但与SDS等其他洗涤剂相比,其去除残余DNA方面往往表现出更高的效率。刘芬等[28]采用4种不同方法制备肺脱细胞基质,最后对脱细胞肺基质分别进行大体形态观察、苏木精-伊红染色、弹性纤维、Masson染色、Ⅰ型胶原及Ⅲ型胶原免疫组织化学染色,结果显示经CHAPS处理的肺组织大体形态最为透明,染色结果显示肺基质构成的肺泡、细支气管、胶原纤维保留较完整。 酸和碱:酸和碱使组织与器官中的蛋白质变性、细胞成分溶解,从而破坏细胞。如脱细胞过程中常使用的酸性洗涤剂过氧乙酸处理小肠黏膜下层[29]、膀胱等较薄组织时[30],虽然脱细胞后组织仍具有生物相容性,但细胞并未完全去除[29],对基质中糖胺聚糖也有一定影响,特别是胶原蛋白和生长因子[31]。此外,组织的机械性能随屈服应力和弹性模量的增加而改变,特别是纵向而非圆周方向,由于组织机械性能改变,故其不适合具有膨胀性和顺应性的组织。此外,过氧乙酸也具有杀菌作用[31]。碱性洗涤剂通常对组织具有刺激性,因此通常用于脱细胞前从皮肤样本中去除毛发等物质,对基质成分同样产生破坏作用,如破坏胶原纤维后导致力学性能下降[9]。 高渗和低渗溶液:也是一个从组织中提取细胞的化学方法,通过渗透裂解作用去除细胞的同时对细胞外基质结构影响较小[32-35]。MóZES等[36]研究使用低渗及高渗溶液对大鼠鼠肾髓质转化生长因子β和早期生长反应基因1表达的影响,结果显示持续使用高渗溶液能上调大鼠肾脏髓质中的转化生长因子β和早期生长反应基因1表达,并且能促进细胞外基质成分的持续表达。VAFAEE等[37]采用低渗缓冲液、1 g/L十二烷基硫酸钠和核酸酶消化法制备人主动脉和肺动脉脱细胞基质,采用组织学、免疫组化、定量测定总脱氧核糖核酸、胶原蛋白和糖胺聚糖含量、体外细胞毒性、单轴拉伸和小鼠皮下移植等方法将脱细胞组织与冷冻保存的瓣膜组织进行对比,结果所有区域(动脉壁、肌肉、小叶)97%的脱氧核糖核酸被清除,保留了Ⅳ型胶原,纤维连接蛋白、层粘连蛋白和硫酸软骨素染色见部分缺失;脱细胞肺动脉瓣组织未见主要组织相容性复合体Ⅰ级染色,仅在脱细胞主动脉瓣组织孤立区有染色残留,脱细胞后胶原含量没有降低,而糖胺聚糖含量降低,脱细胞组织在体外无细胞毒性,小鼠皮下植入模型检测具有生物相容性。 螯合剂:另一种常用于脱细胞处理的化学试剂(例如乙二胺四乙酸EDTA)。螯合剂与中心金属离子形成一个或两个配位键,杂环包括一个中心金属原子作为环的一个组成部分。在微生物方面化学螯合剂用于从溶液中去除离子,同样用于金属和化学(化疗)中毒的治疗。EDTA的作用原理是将多价离子从溶液中分离出来,形成实质上的永久键,从而抑制金属蛋白酶的活性[38]。已有研究表明,乳腺上皮细胞在致密的Ⅰ型胶原凝胶内侵袭/分支需要金属蛋白酶的表达和活性[39],以这种方式,螯合剂通过细胞与细胞外基质黏附位点上结合金属离子将细胞与细胞外基质分离。 2.1.2 生物(酶)脱细胞方法 生物(酶)选择性的裂解精氨酸或赖氨酸羧基侧细胞黏附蛋白,导致细胞从邻近的基质中分离和裂解。通常使用的酶包括胰蛋白酶、胶原酶、核酸酶、嗜热菌蛋白酶和分散酶[40-41]。酶使用时间过长会破坏胶原蛋白、弹性蛋白、糖胺聚糖等天然基质成分[42]。组织经酶脱细胞后必须彻底冲洗,以去除或灭活任何酶残留成分并清除细胞碎片[43]。因此建议短期使用,以防对基质成分造成损害。胰蛋白酶不会对生物工程材料产生细胞毒性作用,而这对体外培养细胞方面而言至关重要,制备无细胞毒性的组织与器官是生物工程领域的一个重要方面[44-46]。 胰蛋白酶是最常使用的酶,采用胰蛋白酶处理猪肺动脉瓣膜24 h后,组织中的细胞完整去除[47]。较短的处理时间不能完整去除细胞,然而较长的处理时间会导致糖胺聚糖、胶原蛋白和弹性蛋白含量下降,进而导致基质机械强度下降。一项关于猪心脏瓣膜脱细胞的研究中,胰蛋白酶结合乙二胺四乙酸再用Triton X-100、脱氧核糖核酸酶和核糖核酸酶处理[48],基质中无细胞核残留且弹性纤维得以完整保留。RAHMAN等[49]用几种不同脱细胞试剂制备人脱细胞弹性软骨,结果显示用胰蛋白酶处理的软骨在脱细胞14 d内产生具有最佳结构及生物力学性能的细胞外基质支架。 虽然化学和生物(酶)脱细胞方法应用比较广泛,但关于化学物质毒性和基质蛋白破坏的担忧同样存在,因此物理或机械方法得到了发展。 2.1.3 物理脱细胞方法 物理方法通过分解细胞和细胞基质黏附蛋白的共同作用去除细胞。常用的物理方法包括冻融、高静水压力学加压、超临界二氧化碳及电穿孔。 冻融:冻融过程通常涉及约-80 ℃的冷冻温度及约37 ℃的生物温度,两种温度之间交替进行。具体方法可以通过增加温差或改变冻融循环次数,重复的冻融循环可用于整个脱细胞过程,并不会增加基质蛋白的丧失[40]。研究表明冻融循环能最大程度降低化学溶剂使用的数量[50],对组织超微结构同样产生轻微破坏,在保证细胞裂解的情况下应尽量减少对组织结构的不良影响,建议使用5%海藻糖等冷冻保护剂[51]。冻融组织或器官内细胞形成冰晶,从而破坏细胞膜,然而这种方法还没报道可以完全去除核物质[52]。两项特殊的冻融研究涉及成纤维细胞进行3次和犬腰椎节段进行一次冻融循环[53-54],结果基质中胶原蛋白、糖胺聚糖及机械强度与原组织相似[54-55]。然而,成纤维细胞脱细胞后仍残留88%DNA[55]。因此,尽管冻融过程有利于生物化学成分和力学性能的保留,但由于遗传物质去除不充分可能导致潜在的免疫排斥反应。 高静水力学加压:高静水力学加压施加大于600 MPa的压力来破坏细胞膜。一项研究在10 ℃或30 ℃温度下用980 MPa压力行猪角膜脱细胞10 min,对猪血管也进行了同样实验,结果显示组织中细胞均破坏,却残留了细胞成分。温度的差异能改变基质蛋白及结构,猪角膜经10 ℃脱细胞后基质胶原蛋白和糖胺聚糖含量比30 ℃时更 高[56],然而基质结冰导致组织结构破坏。此外,高压本身将导致蛋白变形,如脱细胞血管中观察到胶原蛋白和弹性纤维变形,纤维极限拉伸强度比原组织降低约50%[57]。 超临界二氧化碳:具有低黏度和高运输性能,当以类似于临界点受控速度通过组织时,细胞从组织中去除,同时组织的机械性能破坏最小。超临界二氧化碳的临界温度为31.1 ℃,临界压力为7.40 MPa,这在生物学是许可的。因此,超临界二氧化碳以15 MPa和37 ℃行主动脉脱细胞实验时,组织中胶原蛋白、弹性蛋白及纤维机械强度在脱细胞后不变,证明超临界二氧化碳可以使组织中纤维具有不变形能力[58]。此外,由于二氧化碳具有扩散性,溶剂可以快速释放且在组织内无残留,从而不需涉及表面活性剂等溶液的大量洗涤过程。因为二氧化碳是非极性的,所以加入乙醇作为夹带剂以去除极性磷脂细胞膜,脱细胞后组织中细胞核膜均未残留,说明添加是有效的。缺点是超临界二氧化碳对高分子和极性物质的溶解力较差,虽通过加入夹带剂来提高溶解能力,但又会引进新的杂质[59]。 电穿孔:也称非热不可逆电穿孔,通常使用微秒电脉冲来破坏跨膜电势,诱导质膜上微孔的形成,并最终通过改变其稳态电平衡导致细胞死亡。该方法在很大程度上保留了组织与器官的三维结构。GOLBERG等[60]使用电穿孔法对正常肝脏行消融处理,结果显示细胞外基质及肝脏结构得以保留,并且祖细胞通过YAP和notch途径分化及肝细胞扩张使消融后的肝脏几乎完全再生,在消融区形成肝细胞样细胞,术后6个月未见再生区明显瘢痕或肿瘤形成。与此相似的研究使用电穿孔法制备大鼠皮肤细胞外基质,消融6个月后,整个消融区域的表皮、皮脂腺、肉圆肌、毛囊、微血管和竖毛肌全部重新形成[61]。 由于上述每种方法都有其优缺点,为了探索更有效的脱细胞过程及制备更理想的脱细胞基质,根据组织与器官的厚度、组成和性质,采用物理、化学、生物(酶)及上述几种方法联合使用去除组织与器官中细胞物质(如DNA、线粒体、膜脂和胞质蛋白)的同时保留其结构和功能,为了评估脱细胞基质效果,可从细胞、遗传物质、蛋白及机械性能4个方面进行评估,评估标准:①每毫克干质量脱细胞基质少于50 ng双链DNA;②DNA片段长度小于200个碱基对;③组织学染色观察:组织中缺乏可见的核物质,如苏木精和伊红染色或4,6-二氨基-2-苯基吲哚染[62]。此外还应评估基质中的剩余蛋白含量,重点分析胶原蛋白、纤连蛋白及层粘连蛋白等结构蛋白,同时包括黏多糖及生长因子[63]。 为了防止化学和生物(酶)试剂残留引起的毒副作用,脱细胞过程应包括足够的消毒步骤,通常使用氧化剂、环氧乙烷、伽玛射线或电子束照射消毒或灭菌[64]。无论采用何种方法,最终得到的基质都应无毒且具有较好的生物相容性,并保留组织的超微结构和生物力学性能[65]。 脱细胞基质从宏观和微观保留了基质超微结构,由于基质具有广阔的应用前景,因而成为最具潜能的生物支架材料,并为组织与器官的再生提供了新的思路。 2.2 脱细胞基质制备方法在涎腺组织工程中的应用 涎腺又称唾液腺,是一种复杂的分泌组织,产生唾液并维持口腔内环境稳定。放疗等原因引起的涎腺萎缩可使唾液分泌减少,进而导致龋齿、真菌感染、味觉改变、口臭、吞咽和咀嚼困难,这些症状严重影响患者的生活质量[66]。随着放疗技术的成熟,涎腺已不再受大剂量放疗射线的影响,但仍有大量患者遭受严重的涎腺损伤。因此,涎腺的修复与再生是近十年来备受关注的研究课题。为了修复涎腺功能,迄今为止已有几种治疗方案:移植自体涎腺来源的上皮干细胞/祖细胞;利用非上皮细胞和/或它们的生物活性溶解物;组织工程化3D生物材料复合涎腺细胞和/或生物因子来模拟体内涎腺[67]。 LILLIU等[68]将颌下腺脱细胞基质与天然颌下腺组织进行对比实验,用物理及生物(酶)等方法将人颌下腺组织(均无放化疗史)制备成脱细胞颌下腺组织,通过高分辨电子显微镜观察其三维结构,用苦味酸天狼星红和免疫金染色法观察细胞外基质蛋白成分及分布,将人上皮细胞及成纤维细胞接种于两种组织,在短期实验中评估细胞的黏附及增殖情况。结果显示脱细胞颌下腺组织组织纤维排列与天然颌下腺组织相当,染色结果表明Ⅰ、Ⅲ、Ⅳ型胶原存在,接种细胞黏附及增殖(14%-55%),培养4-8 d后细胞存活(86%-99%)。 戴太强等[69]利用化学及物理等不同组合方法制备颌下腺脱细胞基质,并将颌下腺细胞、脂肪干细胞及两者共培养诱导后的脂肪干细胞灌注于支架行再细胞化,结果表明:制备的颌下腺脱细胞支架在有效去除细胞成分的同时较完整的保留了其原有三维结构及细胞外基质成分;体外构建再细胞化可观察到淀粉酶阳性表达,同时形成腺泡样结构,但随培养时间增加腺泡结构逐渐减少。此方法制备的大鼠颌下腺脱细胞支架为构建组织工程涎腺提供了一定的可行性,为涎腺再生提供了技术方法和理论支持。SHIN等[70]应用化学及物理方法制备了唾液腺脱细胞基质支架材料并与水凝胶进行交联,随后将鼠唾液腺腺干/祖细胞嵌入其中进行三维培养,见图2,结果显示细胞在支架中表达唾液腺功能性标志物,蛋白水平α-淀粉酶酶活性增加,但唾液腺干/祖细胞标志物水平降低,包括c-Kit、c-Met和CD44。结果表明,唾液腺脱细胞基质-水凝胶可以为干/祖细胞存活提供合适的微环境,并为唾液腺干/祖细胞分化提供来源。 "

BURGHARTZ等[71]用生物(酶)和化学联合方法制备猪肠道脱细胞基质,将人唾液腺上皮细胞及微血管内皮细胞(在猪肠道脱细胞基质中进行培养,其细胞生长良好并在体外成功形能性人工唾液腺组织,该三维组织可用于肿瘤放疗术后口干症患者的进一步治疗。 作者所在课题组采用冻融、化学、机械等联合方法制备肝脏脱细胞基质并用肝脏基质与肝素和透明质酸水凝胶进行交联(肝素-透明质酸-肝细胞外基质水凝胶),后将肝素-透明质酸-肝细胞外基质水凝胶与大鼠颌下腺细胞进行细胞相容性检测[72]。结果显示,肝素-透明质酸-肝细胞外基质水凝胶具有良好的细胞相容性,而该实验不足之处未涉及涎腺肝细胞外基质与颌下腺的细胞相容性研究。综上所述,涎腺脱细胞基质可能为涎腺前体细胞提供了构建涎腺类器官必要的生物活性因子/蛋白和三维生长的微环境,促进形成具有生理结构和分泌功能的涎腺类器官,为涎腺类器官治疗临床上各种原因引起的涎腺功能降低或丧失提供一定的理论基础和实验依据。然而,由于涎腺结构复杂和组织工程化在临床应用中的挑战,组织工程化涎腺应用于患者的临床移植进展有限,该领域的体内研究仅限于动物[73-74]。 "

| [1] RANA D, ZREIQAT H, BENKIRANE-JESSEL N, et al. Development of decellularized scaffolds for stem cell-driven tissue engineering.J Tissue Eng Regen Med.2017;11:942-965. [2] KEANE TJ, SWINEHART IT, BADYLAK SF. Methods of tissue decellularization used for preparation of biologic scaffolds and in vivo relevance.Methods.2015;84:25-34. [3] KARNIK AV, MALVIYA NJ, KULKARNI AM, et al.Synthesis and Antibacterial Studies of 6-Aryl-2,2a-dihydro-naphtho[2′,1′-5,6]pyrano[4,3-e][1,2,4]triazolo [3,4-b][1,3,4]thiadiazine and Its Derivatives.Cheminform.2010;37(30):489-493. [4] BROWN BN. Macrophage phenotype as a predictor of constructive remodeling following the implantation of biologically derived surgical mesh materials.Acta Biomaterialia.2012; 8(3): 978-987. [5] FAULK DM, WILDEMANN JD, BADYLAK SF. Decellularization and cell seeding of whole liver biologic scaffolds composed of extracellular matrix.J Clin Exp Hepatol.2015;5:69-80. [6] HUANG Y, LU MQ, LI H, et al.Occurrence of cGMP/nitric oxide-sensitive store-operated calcium entry in fibroblasts and its effect on matrix metalloproteinase secretion.World J Gastroenterol. 2006;12:5483-5489. [7] ZHOU J, FRITZE O, SCHLEICHER M, et al.Impact of heart valve decellularization on 3-D ultrastructure, immunogenicity and thrombogenicity.Biomaterials.2010;31(9):2549-2554. [8] O'NeILL JD, ANFANG R, ANANDAPPA A, et al.Decellularization of human and porcine lung tissues for pulmonary tissue engineering.Ann Thorac Surg.2013;96:1046-1055; discussion 1055-1056. [9] GILBERT TW, SELARO TL, BADLYAK SF. Decellularization of tissues and organs. Biomaterials.2006;19(27):3675-3683. [10] COURTMAN DW, PEREIRA CA, KASHEF V, et al.Development of a pericardial acellular matrix biomaterial: biochemical and mechanical effects of cell extraction.J Biomed Mater Res. 1994; 28(6):655-666. [11] UYGUN BE, SOTO-GUTIERREZ A, YAGI H, et al.Organ reengineering through development of a transplantable recellularized liver graft using decellularized liver matrix.Nat Med. 2010;16: 814-820. [12] GILPIN SE, GUYETTE JP, GONZALEZ G, et al.Perfusion decellularization of human and porcine lungs: bringing the matrix to clinical scale.J Heart Lung Transplant.2014;33(3):298-308. [13] ORLANDO G, BAPTISTA P, BIRCHALL M, et al.Regenerative medicine as applied to solid organ transplantation: current status and future challenges.Transpl Int.2011;24:223-232. [14] SYED O, WALTERS NJ, DAY R, et al.Evaluation of decellularization protocols for production of tubular small intestine submucosa scaffolds for use in oesophageal tissue engineering. Acta Biomaterialia.2014;10(12):5043-5054. [15] ZVAROVA B, UHL FE, URIARTE JJ, et al.Residual detergent detection method for non-destructive cytocompatibility evaluation of decellularized whole lung scaffolds.Tissue Eng Part C Methods. 2016;22:418-428. [16] ROOSENS A, SOMERS P, DE SOMER F, et al.Impact of detergent-based decellularization methods on porcine tissues for heart valve engineering.Ann Biomed Eng.2016;44:2827-2839. [17] MEDBERRY CJ, CRAPO PM, SIU BF, et al.Hydrogels derived from central nervous system extracellular matrix.Biomaterials. 2013;34(4):1033-1040. [18] GRAUSS RW, HAZEKAMP MG, OPPENHUIZEN F, et al. Histological evaluation of decellularised porcine aortic valves: matrix changes due to different decellularisation methods.Eur J Cardiothorac Surg.2005;27(4):566-571. [19] MENDOZA-NOVELO B, AVILA EE, CAUICH-RODRÍGUEZ JV, et al. Decellularization of pericardial tissue and its impact on tensile viscoelasticity and glycosaminoglycan content.Acta Biomaterialia. 2011;7(3):1241-1248. [20] ROTH SP, ERBE I, BURK J. Decellularization of Large Tendon Specimens: Combination of Manually Performed Freeze-Thaw Cycles and Detergent Treatment.Methods Mol Biol. 2018;1577: 227-237. [21] TAVASSOLI A, MATIN M M, NIAKI MA, et al.Mesenchymal stem cells can survive on the extracellular matrix-derived decellularized bovine articular cartilage scaffold.Iran J Basic Med Sci. 2015;18: 1221-1227. [22] HUDSON TW, ZAWKO S, DEISTER C, et al.Optimized acellular nerve graft is immunologically tolerated and supports regeneration. Tissue Eng.2004;10:1641-1651. [23] HUDSON TW, LIU SY, SCHMIDT CE.Engineering an improved acellular nerve graft via optimized chemical processing.Tissue Eng Part A.2004;10(10):1346-1358. [24] DAHL SLM, KOH J, PRABHAKAR V, et al.Decellularized Native and Engineered Arterial Scaffolds for Transplantation.Cell Transplant. 2003;12(6):659-666. [25] WALLIS JM, BORG ZD, DALY AB, et al.Comparative assessment of detergent-based protocols for mouse lung de-cellularization and re-cellularization.Tissue Eng Part C Methods.2012;18:420-432. [26] HREBIKOVA H, DIAZ D, MOKRY J.Chemical decellularization: a promising approach for preparation of extracellular matrix.Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub.2015;159:12-17. [27] PETERSEN TH, CALLE EA, ZHAO L, et al.Tissue-Engineered Lungs for in Vivo Implantation. Science. 2010;329(5991):538-541. [28] 刘芬,董明清,邢书娟,等.4种肺脏全器官脱细胞支架制备方法的比较[J].生物医学工程与临床,2019,23(2):121-128. [29] SYED O, WALTERS NJ, DAY R, et al.Evaluation of decellularization protocols for production of tubular small intestine submucosa scaffolds for use in oesophageal tissue engineering. Acta Biomaterialia.2014;10(12):5043-5054. [30] GILBERT TW, WOGNUM S, JOYCE EM, et al.Collagen fiber allgnment and biaxial mechanical behavior or porcine urinary bladder derived extracell ular matrix.Biomaterials.2008;29(36):4775-4782. [31] GILBERT TW, FREUND JM, BADYLAK SF.Quantification of DNA in biologic scaffold materials.J Surg Res.2009;152:135-139. [32] XU CC,CHAN RW,TIRUNAGARI N.A biodegradable, acellular xenogeneic scaffold for regeneration of the vocal fold lamina propria.Tissue Eng.2007;13:551-566. [33] ISHIDA Y, SAKAKIBARA S, TERASHI H, et al.Development of a novel method for decellularizing a nerve graft using a hypertonic sodium chloridesolution.Int J Artif Organs.2014;37:854-860. [34] GENOVESE L, ZAWADA L, TOSONI A, et al.Cellular localization, invasion, and turnover are differently influenced by healthy and tumorderived extracellular matrix.Tissue Eng Part A. 2014;20: 2005-2018. [35] REMLINGER NT, WEARDEN PD, GILBERT TW. Procedure for decellularization of porcine heart by retrograde coronary perfusion. J Vis Exp.2012;70:e50059. [36] MÓZES MM, SZOLECZKY P, ROSIVALL L, et al.Sustained hyperosmolarity increses TGF-ß1 and Egr-1 expression in the rat renal medulla.BMC Nephrol.2017;18(1):209. [37] VAFAEE T, THOMAS D, DESAI A, et al. Decellularization of human donor aortic and pulmonary valved conduits using low concentration sodium dodecyl sulfate.J Tissue Eng Regen Med. 2018;12(2):e841-e853. [38] BALLAL NV, TWEENY A, BAUMGARTNER JC, et al.Effect of maleic acid and ethylenediaminetetraacetic acid on the shear bond strength of RealSeal SE sealer to root canal dentin.Eur J Prosthodont Restor Dent.2013;21:152-156. [39] GOMES AM, BHAT R, CORREIA AL, et al.Mammary Branching Morphogenesis Requires Reciprocal Signaling by Heparanase and MMP-14.J Cell Biochem.2015;116:1668-1679. [40] CRAPO PM, GILBERT TW, BADYLAK SF. An overview of tissue and whole organ decellularization processes.Biomaterials. 2011; 32(12):3233-3243. [41] SAYK F, BOS I, SCHUBERT U, et al.Histopathologic Findings in a Novel Decellularized Pulmonary Homograft: An Autopsy Study. Ann Thorac Surg.2005;79(5):1755-1758. [42] YANG M, CHEN CZ, WANG XN, et al.Favorable effects of the detergent and enzyme extraction method for preparing decellularized bovine pericardium scaffold for tissue engineered heart valves. J Biomed Mater Res B Appl Biomater. 2009;91B(1):354-361. [43] FAULK DM, JOHNSON SA, ZHANG L, et al. Role of the extracellular matrix in whole organ engineering.J Cell Physiol. 2014;229:984-989. [44] RODRIGUEZ KJ, PIECHURA LM, PORRAS AM, et al. Manipulation of valve composition to elucidate the role of collagen in aortic valve calcification.BMC Cardiovasc Disord.2014;1:29. [45] KHAN AA, VISHWAKARMA SK, BARDIA A.Venkateshwarulu J.Repopulation of decellularized whole organ scaffold using stem cells: An emerging technology for the development of neoorgan.J Artif Organs.2014;17:291-300. [46] CARRUTHERS CA, DEARTH CL, REING JE, et al.Histologic characterization of acellular dermal matrices in a porcine model of tissue expander breast reconstruction.Tissue Eng Part A. 2015; 21:35-44. [47] OTT HC, MATTHIESEN TS, GOH SK, et al. Perfusion- decellularized matrix: Using nature's platform to engineer a bioartificial heart.Nat Med.2008;14(2):213-221. [48] LU TY, LIN B, KIM J, et al.Repopulation of decellularized mouse heart with human induced pluripotent stem cell-derived cardiovascular progenitor cells.Nat Commun.2013;4:2307. [49] RAHMAN S, GRIFFIN M, NAIK A, et al.Optimising the decellularization of human elastic cartilage with trypsin for future use in ear reconstruction.Sci Rep.2018;8:3097. [50] CEBOTARI S, TUDORACHE I, JAEKEL T, et al.Detergent decellularization of heart valves for tissue engineering: toxicological effects of residual detergents on human endothelial cells.Artif Organs.2010;34:206-210. [51] PULVER, SHEVTSOV A, LEYBOVICH B, et al.Production of organ extracellular matrix using a freeze-thaw cycle employing extracellular cryoprotectants.Cryo Letters.2014;35(4):400-406. [52] GUPTA SK, MISHRA NC.Decellularization Methods for Scaffold Fabrication.Methods Mol Biol. 2018;1577:1-10. [53] ROSS EA, WILLIAMS MJ, HAMAZAKI T, et al.Embryonic stem cells proliferate and differentiate when seeded into kidney scaffolds.J Am Soc Nephrol.2009;20:2338-2347. [54] ELDER BD, KIM DH, ATHANASIOU KA. Developing an Articular Cartilage Decellularization Process Toward Facet Joint Cartilage Replacement.Neurosurgery.2010;66(4):722-727. [55] XING Q, YATES K, TAHTINEN M, et al.Decellularization of Fibroblast Cell Sheets for Natural Extracellular Matrix Scaffold Preparation.Tissue Eng Part C Methods.2015;21(1):77-87. [56] KISHIDA A, HONDA T, KIMURA T, et al.Preparation and characterization of decellularized cornea using high-hydrostatic pressurization for corneal tissue engineering.Biomaterials. 2010; 31(14):3941-3948. [57] FUNAMOTO S, NAM K, KIMURA T, et al.The use of high-hydrostatic pressure treatment to decellularize blood vessels. Biomaterials.2010;31(13):3590-3595. [58] SAWADA K, TERADA D, YAMAOKA T, et al.Cell removal with supercritical carbon dioxide for acellular artificial tissue.J Chem. Technol Biotechnol.2008;83(6):7. [59] GOH SK, BERTERA S, CANDIELLO JE, et al. Perfusion- decellularized pancreas as a natural 3D scaffold for pancreatic tissue and whole organ engineering.Biomaterials. 2013;34(28):6760-6772. [60] GOLBERG A, BRUINSMA BG, JARAMILLO M, et al.Rat liver regeneration following ablation with irreversible electroporation. Peer J.2016;4:e1571. [61] GOLBERG A, VILLIGER M, FELIX BG, et al.Skin regeneration with all accessory organs following ablation with irreversible electroporation.J Tissue Eng Regen Med.2018;12:98-113. [62] SAYK F, BOS I, SCHUBERT U, et al.Histopathologic Findings in a Novel Decellularized Pulmonary Homograft: An Autopsy Study. Ann Thorac Surg.2005;79(5):1755-1758. [63] CRAPO PM, GILBERT TW, BADYLAK SF. An overview of tissue and whole organ decellularization processes.Biomaterials. 2011; 32(12):3233-3243. [64] KIM BS, IM H, GAO G, et al.Decellularized extracellular matrix: a step towards the next generation source for bioink manufacturing. Biofabrication.2017;9(3):034104. [65] OTT HC, RAJAB TK. Chapter 13-Tissue-Derived Matrices.Situ Tissue Regeneration.2016:229-250. [66] TANASIEWICZ M, HILDEBRANDT T, OBERSZTYN I. Xerostomia of Various Etiologies: A Review of the Literature.Adv Clin Exp Med. 2016;25:199-206. [67] LOMBAERT I, MOVAHEDNIA MM, ADINE C, et al.Concise Review: Salivary Gland Regeneration: Therapeutic Approaches from Stem Cells to Tissue Organoids.Stem Cells.2017;35:97-105. [68] LILLIU M, SEO Y, ISOLA M, et al.Natural extracellular matrix scaffolds recycled from human salivary digests: a morphometric study.Oral Diseases.2016;22(4):313-323. [69] 戴太强,许方方,刘彦普,等.基于大鼠颌下腺脱细胞支架的组织工程涎腺再生初步研究[C].第十四次中国口腔颌面外科学术会议论文汇编,2018. [70] SHIN K, KOO KH, JEONG J, et al.Three-Dimensional Culture of Salivary Gland Stem Cell in Orthotropic Decellularized Extracellular Matrix Hydrogels..Tissue Eng Part A.2019;25: 1396-1403. [71] BURGHARTZ M, LENNARTZ S, SCHWEINLIN M, et al. Development of Human Salivary Gland-Like Tissue In Vitro. Tissue Eng Part A.2018;24:301-309. [72] 董娇.HP-HA-肝ECM水凝胶制备及细胞相容性研究[D].遵义:遵义医学院,2018. [73] ALMELA T, BROOK I M, MOHARAMZADEH K.The Significance of Cell-related Challenges in the Clinical Application of Tissue Engineering.J Biomed Mater Res A.2016;104:3157-3163. [74] HITOMI O, AYA O, YU U, et al. Regenerating Salivary Glands in the Microenvironment of Induced Pluripotent Stem Cells.BioMed Res Int.2015;2015:1-11. |
| [1] | Min Youjiang, Yao Haihua, Sun Jie, Zhou Xuan, Yu Hang, Sun Qianpu, Hong Ensi. Effect of “three-tong acupuncture” on brain function of patients with spinal cord injury based on magnetic resonance technology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-8. |
| [2] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [3] | Jiang Hongying, Zhu Liang, Yu Xi, Huang Jing, Xiang Xiaona, Lan Zhengyan, He Hongchen. Effect of platelet-rich plasma on pressure ulcers after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1149-1153. |
| [4] | Wu Xun, Meng Juanhong, Zhang Jianyun, Wang Liang. Concentrated growth factors in the repair of a full-thickness condylar cartilage defect in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1166-1171. |
| [5] | Zeng Zhen, Hu Jingwei, Li Xuan, Tang Linmei, Huang Zhiqiang, Li Mingxing. Quantitative analysis of renal blood flow perfusion using contrast-enhanced ultrasound in rats with hemorrhagic shock during resuscitation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1201-1206. |
| [6] | Chai Le, Lü Jianlan, Hu Jintao, Hu Huahui, Xu Qingjun, Yu Jinwei, Quan Renfu. Signal pathway variation after induction of inflammatory response in rats with acute spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1218-1223. |
| [7] | Chen Jiming, Wu Xiaojing, Liu Tianfeng, Chen Haicong, Huang Chengshuo. Effects of silymarin on liver injury and bone metabolism induced by carbon tetrachloride in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1224-1228. |
| [8] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [9] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [10] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [11] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [12] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [13] | Zou Gang, Xu Zhi, Liu Ziming, Li Yuwan, Yang Jibin, Jin Ying, Zhang Jun, Ge Zhen, Liu Yi. Human acellular amniotic membrane scaffold promotes ligament differentiation of human amniotic mesenchymal stem cells modified by Scleraxis in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1037-1044. |
| [14] | Ma Binxiang, He Wanqing, Zhou Guangchao, Guan Yonglin. Triptolide improves motor dysfunction in rats following spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 701-706. |
| [15] | Deng Zhenhan, Huang Yong, Xiao Lulu, Chen Yulin, Zhu Weimin, Lu Wei, Wang Daping. Role and application of bone morphogenetic proteins in articular cartilage regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 798-806. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||