Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (25): 4081-4087.doi: 10.3969/j.issn.2095-4344.2084
Previous Articles Next Articles
Research progress in mesenchymal stem cells for treating spinal cord injury
Shi Xu, Liu Jingsong, Wan Ran, Wang Yansong
Fifth Department of Orthopedics, the First Affiliated Hospital of Harbin Medical University, Harbin 150000, Heilongjiang Province, China
-
Received:2019-10-28Revised:2019-10-31Accepted:2019-11-29Online:2020-09-08Published:2020-08-26 -
Contact:Wang Yansong, MD, Chief physician, Fifth Department of Orthopedics, the First Affiliated Hospital of Harbin Medical University, Harbin 150000, Heilongjiang Province, China -
About author:Shi Xu, Master candidate, Fifth Department of Orthopedics, the First Affiliated Hospital of Harbin Medical University, Harbin 150000, Heilongjiang Province, China -
Supported by:the National Natural Science Foundation of China, No. 81871781
CLC Number:
Cite this article
Shi Xu, Liu Jingsong, Wan Ran, Wang Yansong.
Research progress in mesenchymal stem cells for treating spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(25): 4081-4087.
share this article
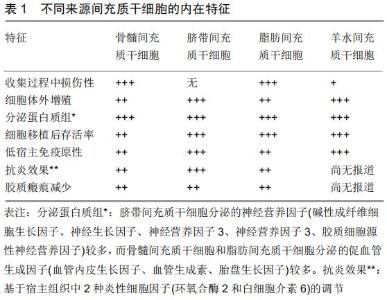
2 结果 Results 2.1 脊髓损伤过程 2.1.1 脊髓损伤急性期 脊髓损伤急性期的情况取决 于创伤的机制,创伤可能由挫伤、撕裂、拉伸、压迫或直接的大规模破坏引起,与创伤相关的事件构成主要损伤,并伴有神经通路的中断。在急性期(2 h内),病变部位的神经元和胶质细胞坏死或凋亡。因此,脊髓修复首先应以恢复椎管内回路为目标,然后再获得下行通路的再生,以恢复对椎管内回路的自主控制。 众所周知,脊髓损伤病理生理过程中最关键的阶段是继发性损伤,即损伤后产生的异常分子信号、炎症、血管改变和继发性细胞功能障碍等不受控制的、破坏性的级联反应[5]。 2.1.2 脊髓损伤后的继发损伤 脊髓继发性损伤的程度或大或小取决于原发损伤的严重程度。由于损伤部位血管痉挛、局灶性微出血或血栓形成,可观察到损伤部位血流减少,从而导致血脊髓屏障的整体功能丧失[5-6]。级联事件也影响细胞膜及离子泵/转运蛋白周围的电解稳态。钾离子(K+)增加细胞外浓度,钠离子(Na+)和钙离子(Ca2+)增加胞内浓度[7-8],这导致了神经传递的阻塞。酸中毒和高渗透压更促进Na+、水内流,导致细胞毒性水肿[9-10]。脊髓损伤后血脊髓屏障被破坏,局部水肿、细胞坏死,发生脱髓鞘,产生大量鞘磷脂,激活损伤部位残存的胶质细胞、血管内皮细胞,这些细胞产生并释放大量的炎症趋化因子及炎性细胞因子,如白细胞介素1β、白细胞介素1α、肿瘤坏死因子α和白细胞介素6等,趋化炎症细胞进入损伤部位,进而导致神经的退化变 性[11-12]。 2.1.3 脊髓损伤慢性期和神经变性 慢性期的特征是胶质增生和细胞外基质沉积后瘢痕形成。原发性损伤中少突胶质细胞的死亡似乎是脊髓损伤的一个关键点,髓鞘碎片中含有抑制分子,阻止轴突的生长,如Nogo-A蛋白或髓鞘相关糖蛋白[13],在病理生理过程中表现出不同的功能模式;虽然它们中的大多数具有抑制作用,如限制轴突再生,但也限制了损伤部位瘢痕的形成,阻止组织损伤的进一步扩大[14-15]。 在这种情况下,清除细胞碎片和改变细胞微环境是神经再生的关键;巨噬细胞具有不同的表型(M1和M2),具有支持神经保护或促进炎症的作用,因此调节巨噬细胞也是决定脊髓损伤恢复程度的关键因素。 脊髓损伤后脊髓内神经元的再生是很难的[16],因此识别控制神经再生的细胞机制是促进损伤后脊髓修复的基础。调节神经元内信号通路和细胞微环境对促进轴突再生至关重要,从而促进髓内通路的再生[17]。脊髓损伤后神经胶质瘢痕的形成和神经网络的调节以及脊髓损伤后神经炎症的控制是脊髓修复的必要条件[18]。轴突的再生、突触的可塑性和重塑,部分是细胞自主的行为,也可能是受损脊髓不同区域的细胞和分子的调控[19]。 尽管对神经系统损伤的发病机制有了进一步的了解,但目前对脊髓损伤的治疗还不能令人十分满意。 2.2 干细胞治疗脊髓损伤及间充质干细胞的特点 干细胞疗法是一种相对较新的治疗方案,在很多医学领域如治疗心肌梗死、骨关节炎、移植物抗宿主病、糖尿病、系统性红斑狼疮、肌萎缩性脊髓侧索硬化症、多发性硬化症、脊髓损伤等都有报道并取得较为满意的结果[20]。 干细胞能够分化成不同的表型,从而决定其在不同疾病中的作用。干细胞在脊髓损伤中的应用旨在通过引导轴突生长,维持轴突髓鞘的再生,促进神经元再生,神经元的营养供应以及促进脊髓自身内源性干细胞的增殖来治疗脊髓损伤。 与其他神经退行性疾病一样,对于干细胞移植治疗脊髓损伤的机制还远未阐明,但干细胞功能似乎主要与其旁分泌作用和营养支持有关[6]。 间充质干细胞因其易于分离和保存的特点而受到关注,其没有引起伦理争议[21],也没有引起发展为肿瘤的风险[22]。而胚胎干细胞存在许多伦理争议限制了其广泛应用[23]。此外,即使在-80 ℃的低温保存条件下,间充质干细胞仍能保持其再生潜能。间充质干细胞增殖速度极快,具有较高的多向分化率。 间充质干细胞表现出归巢性,能够向病灶部位迁 移[24]。这种细胞行为是由多种炎症或趋化因子介导的,例如在损伤部位释放的血管内皮生长因子和肝细胞生长因子实际上可以吸引间充质干细胞的聚集[25-26]。此外,基质细胞衍生因子1α/趋化因子受体4(SDF-1α/ CXCR4)轴在这些机制中起着重要的作用[27],可影响间充质干细胞的归巢能力[28]。当间充质干细胞暴露于条件培养基时,可通过激活Akt和Erk的胞内信号通路——PI3K/Akt通路和Erk1/2通路,增强Akt和Erk的磷酸化水平,从而增强间充质干细胞的迁移[27]。 虽然证明间充质干细胞具有归巢能力,但确切的机制在很大程度上仍是未知的。经静脉植入的间充质干细胞能够有针对性地向脊髓损伤中心迁移[29-30],并表现出良好的存活率[31]。 2.3 间充质干细胞 间充质干细胞与胚胎干细胞、诱导多能干细胞等干细胞相比,涉及较少的伦理问题和安全问题,因此在临床上得到广泛应用[20]。人脐带间充质干细胞来源于脐带的华通胶,是一种来源丰富的原始细胞。在适当的条件下,可以通过诱导分化成为神经干细胞,治疗神经系统疾病具有良好的临床应用前景。不同来源间充质干细胞的内在特征,见表1。脐带间充质干细胞相比于骨髓间充质干细胞、成体器官干细胞、胎儿间充质干细胞、胚胎干细胞、多能诱导干细胞等,其侵袭性分离、伦理约束的弊端、免疫原性、诱导肿瘤发生的程度明显降低[32]。骨髓间充质干细胞和人脐带间充质干细胞移植都可减轻脊髓损伤后神经性疼痛症状并可促进脊髓损伤后的运动功能恢复,人脐带间充质干细胞的存活率和电生理表现明显优于骨髓间充质干细胞[33]。在脊髓损伤大鼠或小鼠动物模型中,脐带间充质干细胞显示出良好的神经营养、抗凋亡和抗炎作用[34]。 虽然不同来源的间充质干细胞存在差异,但都表现出显著的自分泌和旁分泌活性,包括免疫调节和营养因子以及抗炎细胞因子,移植的细胞可以对宿主环境产生积极的影响[35-36]。此外,间充质干细胞分泌体还可对宿主微环境产生免疫调节、抗炎、神经营养/神经保护和血管生成等作用。间充质干细胞的免疫调节作用主要是通过组织相容性复合体Ⅰ在间充质干细胞表面的表达来实现的,这样可以防止T细胞识别和诱导宿主免疫应 答[37]。此外,间充质干细胞还能抑制T细胞的增殖、活化和分化[38]。间充质干细胞具有抗炎作用,可分泌多种可溶性分子,抗炎细胞因子包括肿瘤坏死因子β1、白细胞介素13、白细胞介素18结合蛋白、白细胞介素10等;间充质干细胞还可以调节细胞因子的产生和释放,例如抑制促炎细胞因子肿瘤坏死因子α的释放或增加抗炎因子白细胞介素10的释放[39]。 2.4 间充质干细胞移植治疗脊髓损伤后的分化方向 脊髓损伤部位功能细胞的缺失是导致病症的根本原因,若能够弥补细胞的缺失,就可以重塑脊髓功能的完整,这就需要建立可靠的培养条件来促进干细胞向特定的神经元和胶质亚型的有效分化。 间充质干细胞具有良好的可塑性,且在不同条件下均可经诱导后向神经细胞分化,其向非中胚层细胞(如神经元)分化的能力已被广泛报道[40]。脐带间充质干细胞、骨髓间充质干细胞及脂肪间充质干细胞都具有相似的神经元分化潜能[41]。间充质干细胞在神经分化培养基培养3周后,细胞开始成簇,表现出神经元样突起,并检测细胞微管相关蛋白2(成熟神经元特异性标记物)表达阳性[41]。BORKOWSKA等[42]研究认为间充质干细胞能向神经元样细胞分化,且在不同药物的干预下还可提高间充质干细胞向神经元样细胞的分化程度。间充质干细胞同样具有分化为少突胶质样谱系的潜力[43],干细胞分化后能够阳性表达成熟少突胶质细胞标志物髓鞘碱性蛋白,但与真正的少突胶质细胞相比在形态学和功能上似乎还不够成熟。间充质干细胞虽然已经被证明能够分化成许多细胞谱系,但这些分化的细胞还不够成熟,不能像终末分化细胞那样发挥作用[41]。目前间充质干细胞分化的机制也仍不清楚[44]。 2.5 间充质干细胞的细胞外基质对脊髓损伤的作用 在中枢神经系统中,细胞外基质形成了一个复杂的三维网络,锚定和支持神经元细胞和非神经元细胞,这些细胞也分泌各种细胞外基质成分,它们的命运和功能在脊髓损伤中起着至关重要的作用。细胞外基质是组织中细胞生态位的重要组成部分,为启动和维持细胞功能提供重要的生化和物理信号。间充质干细胞能够分泌层粘连蛋白、纤连蛋白、硫酸软骨素蛋白多糖2、Ⅰ型胶原蛋白、Ⅲ型胶原蛋白、Ⅴ型胶原蛋白以及各种类型的蛋白多糖等,以维持干细胞的干性[45]。 近年来,细胞来源的细胞外基质已成为体外细胞培养的底物,以更好地还原体外的干细胞微环境[46]。成体神经干细胞的迁移和分化受到细胞外基质蛋白在干细胞生态位空间分布的影响[47]。细胞外基质已被证明可以调节细胞黏附、增殖、迁移、分化和相互作用,细胞外基质也提供了一种结构支持[48]。GU等[49]研究证明间充质干细胞来源的细胞外基质对轴突再生和功能恢复具有良好的作用,细胞外基质支架是有利于建立神经再生所需要的内部微环境,有利于促进细胞黏附、增殖和迁移。与其他细胞相比,间充质干细胞源性的细胞外基质能显著促进细胞增殖,促进间充质干细胞的迁移和向神经干细胞分化,具有重建神经和其他软组织的能力[50]。纤维粘连蛋白是中枢神经系统中一种重要的糖蛋白,参与细胞迁移。纤维粘连蛋白具有细胞黏附性、隔离营养物质和生长因子的能力,在组织修复中具有重要作 用[51-52]。层粘连蛋白以片状聚合物的形式存在,参与细胞在发育过程中的生长和迁移,在中枢及外周神经系统中层粘连蛋白都维持在一个较高的水平[53]。当层粘连蛋白被引入受损脊髓时,可促进轴突生长,同时伴有功能恢复[54]。共培养对胶质标志物表达的影响更为显著,表达的增强和成熟可以归因于间充质干细胞的细胞外基质层粘连蛋白[55]。 脊髓损伤后,在干细胞分泌的细胞外基质作用下,形成胶原纤维和连接神经间隙的基板管,随后新生的轴突沿着新的细胞外基质支架通过神经间隙生长[48]。 2.6 间充质干细胞旁分泌作用对脊髓损伤的作用 旁分泌作用可能是间充质干细胞在周围神经修复中的作用机制之一[56]。移植干细胞治疗脊髓损伤,虽然干细胞可以向神经细胞分化,但其不是通过细胞分化直接替代失去的细胞,而是通过神经保护手段,提供神经营养和免疫调节等因素来促进轴突生长,调节微环境,并减少神经炎症[57]。 间充质干细胞可能是通过其旁分泌的众多因子来调节受损组织的修复过程[58]。神经营养因子是神经干细胞能够增殖传代的必要条件,这些因子支持中枢神经系统受损后神经元的存活。神经元丢失的增加是由于神经干细胞后代失去了神经营养支持[59]。为了发挥神经保护作用,间充质干细胞分泌多种神经营养因子,如脑源性生长因子、胶质源性生长因子、神经生长因子、碱性成纤维细胞生长因子等。通过这些因素,间充质干细胞一方面可以防止神经变性和凋亡,另一方面可以支持神经发生、轴突生长、髓鞘再生和细胞代谢[60-61]。 移植细胞经基因修饰生长因子或神经营养因子来调节微环境和/或提供营养支持已被证明可改善轴突生长[62]。研究表明间充质干细胞所分泌的因子中含有高水平的脑源性生长因子、胶质源性生长因子、碱性成纤维细胞生长因子等,而且多因子共同作用下能够更大程度促进神经干细胞的增殖分化能力[56]。CHEN等[63]研究发现神经生长因子/脑源性生长因子/碱性成纤维细胞生长因子联合应用能显著提高神经干细胞增殖分化能力[64]。RAO等[65]在猴子脊髓半切实验中证明神经营养因子3够在运动和感觉功能恢复的同时实现轴突再生。碱性成纤维细胞生长因子在脊髓损伤恢复中通过激活PI3K/Akt/mTOR信号通路抑制过度自噬和增强泛素化蛋白清除来促进脊髓损伤神经元的恢复和存活[66]。胶质源性生长因子是运动神经元存活、连接和轴突生长最重要的营养因子之一。胶质源性生长因子的作用已被证明可以减少病变的大小,改善脊髓损伤所带来的疼痛,再生中枢神经系统和周围神经系统的轴突[67]。KITAMURA等[68]研究证明通过注射肝细胞生长因子表达的病毒载体将外源性肝细胞生长因子引入脊髓,可显著增加脊髓损伤大鼠神经元和少突胶质细胞的存活、血管生成和轴突再生,减少损伤面积,促进脊髓损伤后大鼠的功能恢复,在脊髓损伤急性期外源性给予肝细胞生长因子可以减少星形细胞的激活,减少胶质瘢痕的形成并发挥抗炎作用,减少白细胞的浸润。神经营养因子3则可以促进皮质脊髓轴突的生长,改善慢性瘢痕切除后的功能缺 损[69]。虽然很多营养因子对脊髓损伤的治疗都有一定程度的作用,但与其他神经营养因子联合使用时能够更大程度的发挥作用[67]。 间充质干细胞移植促进受损神经功能修复的机制与通过促进损伤部位血管内皮生长因子的表达和血管新生有关[70]。ZHAO等[71]通过静脉注射间充质干细胞治疗缺血性脑卒中大鼠模型,研究发现间充质干细胞分泌神经营养生长因子和促血管生成的细胞因子,促进血管生成。 很多研究表明外泌体在细胞间通讯中起着重要的作用[72]。间充质干细胞分泌外泌体也是旁分泌作用的一种,外泌体参与到机体的免疫应答、抗原提呈、细胞迁移、细胞分化及肿瘤侵袭等各方面。间充质干细胞来源外泌体具有与间充质干细胞相似的功能,且免疫原性低,未发生肿瘤化[73]。外泌体通过生物分子传递改变受体细胞的生化特性,并在细胞通讯中发挥作用。间充质干细胞衍生的外泌体可以通过减轻损伤区域的炎症来促进脊髓损伤的愈合[74]。XIN等[75]通过外泌体将miR-133b转移到神经细胞,从而调控神经元突起的生长。在缺氧条件下,外泌体可以通过PTEN/AKT通路保护脊髓微血管内皮细胞的屏障[76]。 2.7 间充质干细胞的局限性和未来的发展方向 间充质干细胞或其他干细胞在动物模型中显示出了良好的结 果[77],但还有许多问题需要进一步研究。动物模型研究通常应用标准化方法制备,其损伤程度、治疗方案和移植的具体时间都是相对固定的,而人类脊髓损伤患者的各种复杂多变情况往往是不可模仿的,目前还有一定差距。啮齿动物和人类在大脑解剖和行为学特征上有一定区别,因此在体外实验或动物体内实验有进展的前提下,进行人类临床试验并最终应用临床发挥疗效应该是干细胞研究的最终目标。 DAI等[78]在一项随机研究中对脊髓损伤患者进行了间充质干细胞移植治疗。采用AIS分级、ASIA评分、残余尿量、神经生理检查进行神经功能评估,随访6个月时其治疗效果有限。EL-KEHIR等[79]试验也无更多令人满意的结果。GEFFNER等[80]对8例脊髓损伤患者(4例急性,4例慢性)进行干细胞移植有一定的疗效。 免疫化学、分子标志物和细胞形态表明,间充质干细胞一旦移植后出现神经元样特征,也很难将其视为神经元样细胞[6,81]。事实上,神经元抗原的表达可能仅仅是由于间充质干细胞不成熟的特性[82]。当通过化学手段(如二甲基亚砜)进行分化时,间充质干细胞表现出明显的形态变化,最终归结为细胞收缩和细胞骨架的变 化[83],并不能像真正的神经细胞一样发挥应有的作用。为了将间充质干细胞转化为神经元,更复杂的实验方案不断被开发出来[84],就目前结论而言,间充质干细胞的疗效似乎仍与其旁分泌活性相关,而与细胞替代机制关系甚微[85]。 事实上,仍有许多问题需要进一步摸索和证实,如关于干细胞的制备、类型和移植数量的最佳治疗方案;移植时机及给药途径;旁分泌作用及其对功能恢复的影响;生物材料和支架的重要性;微环境的重要性;可塑性和重建神经细胞连接的能力等。 "
|
[1] KALRA H, SIMPSON RJ, JI H, et al. Vesiclepedia: a compendium for extracellular vesicles with continuous community annotation. PLoS Biol. 2012;10(12):e1001450.
[2] YILMAZ T, KAPTANOĞLU E. Current and future medical therapeutic strategies for the functional repair of spinal cord injury. World J Orthop. 2015;6(1):42-55.
[3] WILSON JR, TETREAULT LA, KWON BK, et al. Timing of Decompression in Patients With Acute Spinal Cord Injury: A Systematic Review. Global Spine J. 2017;7(3 Suppl): 95S-115S.
[4] COFANO F, BOIDO M, MONTICELLI M, et al. Mesenchymal Stem Cells for Spinal Cord Injury: Current Options, Limitations, and Future of Cell Therapy. Int J Mol Sci. 2019;20(11):E2698.
[5] SILVA NA, SOUSA N, REIS RL, et al. From basics to clinical: a comprehensive review on spinal cord injury. Prog Neurobiol. 2014;114: 25-57.
[6] VISMARA I, PAPA S, ROSSI F, et al. Current Options for Cell Therapy in Spinal Cord Injury. Trends Mol Med. 2017;23(9):831-849.
[7] FEHLINGS MG, NAKASHIMA H, NAGOSHI N, et al. Rationale, design and critical end points for the Riluzole in Acute Spinal Cord Injury Study (RISCIS): a randomized, double-blinded, placebo-controlled parallel multi-center trial. Spinal Cord. 2016;54(1):8-15.
[8] GAZDIC M, VOLAREVIC V, HARRELL CR, et al. Stem Cells Therapy for Spinal Cord Injury. Int J Mol Sci. 2018;19(4): E1039.
[9] GARCIA E, AGUILAR-CEVALLOS J, SILVA-GARCIA R, et al. Cytokine and Growth Factor Activation In Vivo and In Vitro after Spinal Cord Injury. Mediators Inflamm. 2016;2016:9476020.
[10] HAYTA E, ELDEN H. Acute spinal cord injury: A review of pathophysiology and potential of non-steroidal anti-inflammatory drugs for pharmacological intervention. J Chem Neuroanat.2018; 87:25-31.
[11] PAPA S, CARON I, ERBA E, et al. Early modulation of pro-inflammatory microglia by minocycline loaded nanoparticles confers long lasting protection after spinal cord injury. Biomaterials. 2016;75:13-24.
[12] PAPA S, CARON I, ROSSI F, et al. Modulators of microglia: a patent review. Expert Opin Ther Pat. 2016;26(4):427-437.
[13] SCHWAB ME, STRITTMATTER SM. Nogo limits neural plasticity and recovery from injury. Curr Opin Neurobiol. 2014;27:53-60.
[14] BELLER JA, SNOW DM. Proteoglycans: road signs for neurite outgrowth. Neural Regen Res. 2014;9(4):343-355.
[15] YUAN YM, HE C. The glial scar in spinal cord injury and repair. Neurosci Bull. 2013;29(4):421-435.
[16] SABIN KZ, JIANG P, GEARHART MD, et al. AP-1cFos/JunB/miR-200a regulate the pro-regenerative glial cell response during axolotl spinal cord regeneration. Commun Biol. 2019;2:91.
[17] VAN NIEKERK EA, TUSZYNSKI MH, LU P, et al. Molecular and Cellular Mechanisms of Axonal Regeneration After Spinal Cord Injury. Mol Cell Proteomics. 2016;15(2):394-408.
[18] TRAN AP, WARREN PM, SILVER J. The Biology of Regeneration Failure and Success After Spinal Cord Injury. Physiol Rev. 2018;98(2): 881-917.
[19] O'SHEA TM, BURDA JE, SOFRONIEW MV. Cell biology of spinal cord injury and repair. J Clin Invest. 2017;127(9):3259-3270.
[20] LIAU LL, RUSZYMAH BHI, NG MH, et al. Characteristics and clinical applications of Wharton's jelly-derived mesenchymal stromal cells. Curr Res Transl Med. 2019 Sep 19. doi: 10.1016/j.retram.2019.09.001. [Epub ahead of print]
[21] DASARI VR, VEERAVALLI KK, DINH DH. Mesenchymal stem cells in the treatment of spinal cord injuries: A review. World J Stem Cells. 2014;6(2):120-133.
[22] LU LL, LIU YJ, YANG SG, et al. Isolation and characterization of human umbilical cord mesenchymal stem cells with hematopoiesis- supportive function and other potentials. Haematologica. 2006;91(8): 1017-1026.
[23] LO B, PARHAM L. Ethical issues in stem cell research. Endocr Rev. 2009;30(3):204-213.
[24] FILIPPI M, BOIDO M, PASQUINO C, et al. Successful in vivo MRI tracking of MSCs labeled with Gadoteridol in a Spinal Cord Injury experimental model. Exp Neurol. 2016;282:66-77.
[25] ZACHAR L, BAČENKOVÁ D, ROSOCHA J. Activation, homing, and role of the mesenchymal stem cells in the inflammatory environment. J Inflamm Res. 2016;9:231-240.
[26] QU J, ZHANG H. Roles of Mesenchymal Stem Cells in Spinal Cord Injury. Stem Cells Int. 2017;2017:5251313.
[27] PELAGALLI A, NARDELLI A, LUCARELLI E, et al. Autocrine signals increase ovine mesenchymal stem cells migration through Aquaporin-1 and CXCR4 overexpression. J Cell Physiol. 2018;233(8): 6241-6249.
[28] MARQUEZ-CURTIS LA, GUL-ULUDAG H, XU P, et al. CXCR4 transfection of cord blood mesenchymal stromal cells with the use of cationic liposome enhances their migration toward stromal cell-derived factor-1. Cytotherapy. 2013;15(7):840-849.
[29] KIM Y, JO SH, KIM WH, et al. Antioxidant and anti-inflammatory effects of intravenously injected adipose derived mesenchymal stem cells in dogs with acute spinal cord injury. Stem Cell Res Ther. 2015;6:229.
[30] RAMALHO BDS, ALMEIDA FM, SALES CM, et al. Injection of bone marrow mesenchymal stem cells by intravenous or intraperitoneal routes is a viable alternative to spinal cord injury treatment in mice. Neural Regen Res. 2018;13(6):1046-1053.
[31] YANG C, WANG G, MA F, et al. Repeated injections of human umbilical cord blood-derived mesenchymal stem cells significantly promotes functional recovery in rabbits with spinal cord injury of two noncontinuous segments. Stem Cell Res Ther. 2018;9(1):136.
[32] WATSON N, DIVERS R, KEDAR R, et al. Discarded Wharton jelly of the human umbilical cord: a viable source for mesenchymal stromal cells. Cytotherapy. 2015;17(1):18-24.
[33] YOUSEFIFARD M, NASIRINEZHAD F, SHARDI MANAHEJI H, et al. Human bone marrow-derived and umbilical cord-derived mesenchymal stem cells for alleviating neuropathic pain in a spinal cord injury model. Stem Cell Res Ther. 2016;7:36.
[34] CARON I, ROSSI F, PAPA S, et al. A new three dimensional biomimetic hydrogel to deliver factors secreted by human mesenchymal stem cells in spinal cord injury. Biomaterials. 2016;75:135-147.
[35] SOBACCHI C, PALAGANO E, VILLA A, et al. Soluble Factors on Stage to Direct Mesenchymal Stem Cells Fate. Front Bioeng Biotechnol. 2017;5:32.
[36] BAEZ-JURADO E, HIDALGO-LANUSSA O, BARRERA-BAILÓN B, et al. Secretome of Mesenchymal Stem Cells and Its Potential Protective Effects on Brain Pathologies. Mol Neurobiol. 2019;56(10):6902-6927.
[37] RYAN JM, BARRY FP, MURPHY JM, et al. Mesenchymal stem cells avoid allogeneic rejection. J Inflamm (Lond). 2005;2:8.
[38] LIANG X, DING Y, ZHANG Y, et al. Paracrine mechanisms of mesenchymal stem cell-based therapy: current status and perspectives. Cell Transplant. 2014;23(9):1045-1059.
[39] VIZOSO FJ, EIRO N, CID S, et al. Mesenchymal Stem Cell Secretome: Toward Cell-Free Therapeutic Strategies in Regenerative Medicine. Int J Mol Sci. 2017;18(9): E1852.
[40] SALEHI H, AMIRPOUR N, NIAPOUR A, et al. An Overview of Neural Differentiation Potential of Human Adipose Derived Stem Cells. Stem Cell Rev Rep. 2016;12(1):26-41.
[41] CHEN S, ZHANG W, WANG JM, et al. Differentiation of isolated human umbilical cord mesenchymal stem cells into neural stem cells. Int J Ophthalmol. 2016;9(1):41-47.
[42] BORKOWSKA P, KOWALSKA J, FILA-DANILOW A, et al. Affect of antidepressants on the in vitro differentiation of rat bone marrow mesenchymal stem cells into neuronal cells. Eur J Pharm Sci. 2015;73:81-87.
[43] LEITE C, SILVA NT, MENDES S, et al. Differentiation of human umbilical cord matrix mesenchymal stem cells into neural-like progenitor cells and maturation into an oligodendroglial-like lineage. PLoS One. 2014;9(10):e111059.
[44] HERNÁNDEZ R, JIMÉNEZ-LUNA C, PERALES-ADÁN J, et al. Differentiation of Human Mesenchymal Stem Cells towards Neuronal Lineage: Clinical Trials in Nervous System Disorders. Biomol Ther (Seoul). 2019 Oct 25. doi: 10.4062/biomolther.2019.065. [Epub ahead of print]
[45] ZENG X, MA YH, CHEN YF, et al. Autocrine fibronectin from differentiating mesenchymal stem cells induces the neurite elongation in vitro and promotes nerve fiber regeneration in transected spinal cord injury. J Biomed Mater Res A. 2016;104(8):1902-1911.
[46] RAGELLE H, NABA A, LARSON BL, et al. Comprehensive proteomic characterization of stem cell-derived extracellular matrices. Biomaterials. 2017;128:147-159.
[47] JOO S, YEON KIM J, LEE E, et al. Effects of ECM protein micropatterns on the migration and differentiation of adult neural stem cells. Sci Rep. 2015;5:13043.
[48] XIAO B, RAO F, GUO ZY, et al. Extracellular matrix from human umbilical cord-derived mesenchymal stem cells as a scaffold for peripheral nerve regeneration. Neural Regen Res. 2016;11(7): 1172-1179.
[49] GU Y, LI Z, HUANG J, et al. Application of marrow mesenchymal stem cell-derived extracellular matrix in peripheral nerve tissue engineering. J Tissue Eng Regen Med. 2017;11(8):2250-2260.
[50] KOČÍ Z, VÝBORNÝ K, DUBIŠOVÁ J, et al. Extracellular Matrix Hydrogel Derived from Human Umbilical Cord as a Scaffold for Neural Tissue Repair and Its Comparison with Extracellular Matrix from Porcine Tissues. Tissue Eng Part C Methods. 2017;23(6): 333-345.
[51] CHEN ZL, STRICKLAND S. Laminin gamma1 is critical for Schwann cell differentiation, axon myelination, and regeneration in the peripheral nerve. J Cell Biol. 2003;163(4):889-899.
[52] ROGERS SL, LETOURNEAU PC, PALM SL,et al. Neurite extension by peripheral and central nervous system neurons in response to substratum-bound fibronectin and laminin. Dev Biol. 1983;98(1): 212-220.
[53] HAGGERTY AE, MARLOW MM, OUDEGA M. Extracellular matrix components as therapeutics for spinal cord injury. Neurosci Lett. 2017;652:50-55.
[54] MENEZES K, DE MENEZES JR, NASCIMENTO MA, et al. Polylaminin, a polymeric form of laminin, promotes regeneration after spinal cord injury. FASEB J. 2010;24(11):4513-4522.
[55] OPPLIGER B, JOERGER-MESSERLI MS, SIMILLION C, et al. Mesenchymal stromal cells from umbilical cord Wharton's jelly trigger oligodendroglial differentiation in neural progenitor cells through cell-to-cell contact. Cytotherapy. 2017;19(7):829-838.
[56] GUO ZY, SUN X, XU XL, et al. Human umbilical cord mesenchymal stem cells promote peripheral nerve repair via paracrine mechanisms. Neural Regen Res. 2015;10(4):651-658.
[57] SUN L, WANG F, CHEN H, et al. Co-Transplantation of Human Umbilical Cord Mesenchymal Stem Cells and Human Neural Stem Cells Improves the Outcome in Rats with Spinal Cord Injury. Cell Transplant. 2019;28(7):893-906.
[58] HUANG JH, YIN XM, XU Y, et al. Systemic Administration of Exosomes Released from Mesenchymal Stromal Cells Attenuates Apoptosis, Inflammation, and Promotes Angiogenesis after Spinal Cord Injury in Rats. J Neurotrauma. 2017;34(24):3388-3396.
[59] SABELSTRÖM H, STENUDD M, RÉU P, et al. Resident neural stem cells restrict tissue damage and neuronal loss after spinal cord injury in mice. Science. 2013;342(6158):637-640.
[60] HOFER HR, TUAN RS. Secreted trophic factors of mesenchymal stem cells support neurovascular and musculoskeletal therapies. Stem Cell Res Ther. 2016;7(1):131.
[61] TEIXEIRA FG, CARVALHO MM, SOUSA N, et al. Mesenchymal stem cells secretome: a new paradigm for central nervous system regeneration? Cell Mol Life Sci. 2013;70(20):3871-3882.
[62] SABELSTRÖM H, STENUDD M, FRISÉN J. Neural stem cells in the adult spinal cord. Exp Neurol. 2014;260:44-49.
[63] CHEN SQ, CAI Q, SHEN YY, et al. Combined use of NGF/BDNF/bFGF promotes proliferation and differentiation of neural stem cells in vitro. Int J Dev Neurosci. 2014;38:74-78.
[64] 黄斐,马广文,尹宗生,等.脑源性神经营养因子缓释胶原凝胶支架对神经干细胞生长和分化的影响[J].安徽医科大学学报, 2014,49(5):586-590.
[65] RAO JS, ZHAO C, ZHANG A, et al. NT3-chitosan enables de novo regeneration and functional recovery in monkeys after spinal cord injury. Proc Natl Acad Sci U S A. 2018;115(24):E5595-E5604.
[66] ZHANG HY, WANG ZG, WU FZ, et al. Regulation of autophagy and ubiquitinated protein accumulation by bFGF promotes functional recovery and neural protection in a rat model of spinal cord injury. Mol Neurobiol. 2013;48(3):452-464.
[67] ROSICH K, HANNA BF, IBRAHIM RK, et al. The Effects of Glial Cell Line-Derived Neurotrophic Factor after Spinal Cord Injury. J Neurotrauma. 2017;34(24):3311-3325.
[68] KITAMURA K, NAGOSHI N, TSUJI O, et al. Application of Hepatocyte Growth Factor for Acute Spinal Cord Injury: The Road from Basic Studies to Human Treatment. Int J Mol Sci. 2019;20(5): E1054.
[69] TUSZYNSKI MH, GRILL R, JONES LL, et al. NT-3 gene delivery elicits growth of chronically injured corticospinal axons and modestly improves functional deficits after chronic scar resection. Exp Neurol. 2003;181(1):47-56.
[70] ZANOTTI L, ANGIONI R, CALÌ B, et al. Mouse mesenchymal stem cells inhibit high endothelial cell activation and lymphocyte homing to lymph nodes by releasing TIMP-1. Leukemia. 2016;30(5):1143-1154.
[71] ZHAO J, CHEN N, SHEN N, et al. Transplantation of human umbilical cord blood mesenchymal stem cells to treat a rat model of traumatic brain injury. Neural Regen Res. 2012;7(10):741-748.
[72] RECORD M, SUBRA C, SILVENTE-POIROT S, et al. Exosomes as intercellular signalosomes and pharmacological effectors. Biochem Pharmacol. 2011;81(10):1171-1182.
[73] YAGHOUBI Y, MOVASSAGHPOUR A, ZAMANI M, et al. Human umbilical cord mesenchymal stem cells derived-exosomes in diseases treatment. Life Sci. 2019;233:116733.
[74] SUN G, LI G, LI D, et al. hucMSC derived exosomes promote functional recovery in spinal cord injury mice via attenuating inflammation. Mater Sci Eng C Mater Biol Appl. 2018;89:194-204.
[75] XIN H, LI Y, BULLER B, et al. Exosome-mediated transfer of miR-133b from multipotent mesenchymal stromal cells to neural cells contributes to neurite outgrowth. Stem Cells. 2012;30(7):1556-1564.
[76] YUAN X, WU Q, WANG P, et al. Exosomes Derived From Pericytes Improve Microcirculation and Protect Blood-Spinal Cord Barrier After Spinal Cord Injury in Mice. Front Neurosci. 2019;13:319.
[77] BOIDO M, GARBOSSA D, VERCELLI A. Early graft of neural precursors in spinal cord compression reduces glial cyst and improves function. J Neurosurg Spine. 2011;15(1):97-106.
[78] DAI G, LIU X, ZHANG Z, et al. Transplantation of autologous bone marrow mesenchymal stem cells in the treatment of complete and chronic cervical spinal cord injury. Brain Res. 2013;1533:73-79.
[79] EL-KHEIR WA, GABR H, AWAD MR, et al. Autologous bone marrow-derived cell therapy combined with physical therapy induces functional improvement in chronic spinal cord injury patients. Cell Transplant. 2014;23(6):729-745.
[80] GEFFNER LF, SANTACRUZ P, IZURIETA M, et al. Administration of autologous bone marrow stem cells into spinal cord injury patients via multiple routes is safe and improves their quality of life: comprehensive case studies. Cell Transplant. 2008;17(12):1277-1293.
[81] CORCIONE A, BENVENUTO F, FERRETTI E, et al. Human mesenchymal stem cells modulate B-cell functions. Blood. 2006; 107(1):367-372.
[82] DENG J, PETERSEN BE, STEINDLER DA, et al. Mesenchymal stem cells spontaneously express neural proteins in culture and are neurogenic after transplantation. Stem Cells. 2006;24(4):1054-1064.
[83] LU P, BLESCH A, TUSZYNSKI MH. Induction of bone marrow stromal cells to neurons: differentiation, transdifferentiation, or artifact? J Neurosci Res. 2004;77(2):174-191.
[84] CORTÉS-MEDINA LV, PASANTES-MORALES H, AGUILERA- CASTREJON A, et al. Neuronal Transdifferentiation Potential of Human Mesenchymal Stem Cells from Neonatal and Adult Sources by a Small Molecule Cocktail. Stem Cells Int. 2019;2019:7627148.
[85] PARK WS, AHN SY, SUNG SI, et al. Strategies to enhance paracrine potency of transplanted mesenchymal stem cells in intractable neonatal disorders. Pediatr Res. 2018;83(1-2):214-222. |
| [1] | He Jing, Ao Qiang. Research hotspots in tissue decellularization method for manufacturing extracellular matrices [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5413-5420. |
| [2] | Liao Jian, Huang Xiaolin, Zhou Qian, Huo Hua, Qi Yuhan, Wu Chao, Shi Qianhui, Yang Tongjing, Liao Yunmao, Liang Xing. Calcined bone/chitosan composite promotes osteogenic differentiation of bone marrow mesenchymal stem cells in Sprague-Dawley rats [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 4941-4947. |
| [3] | Wei Renyue, Li Xuechun, Li Yan, Yu Yang, Zhang Yu, Liu Zhonghua. A serum-free monolayer method for differentiation of porcine induced pluripotent stem cells into vascular endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 4971-4978. |
| [4] | Cheng Xue, Fang Hong, Zhang Yunke, Wu Yingen. Interventional mechanism of Feibi prescription on extracellular matrix transformation in a mouse model of pulmonary fibrosis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 5038-5043. |
| [5] | Xin Pengfei, Sun Youqiang, Li Jie, Chen Jianfa, Deng Baogui, Xiang Xiaobing . Research progress of biomaterials for repair of rotator cuff tear [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(28): 4459-4464. |
| [6] |
Ji Hangyu, Gu Jun, Xie Linghan, Wu Xiaotao.
Application of stem cells, tissue engineering scaffolds and neurotrophic factors in the treatment of spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(25): 4088-4093. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||