Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (29): 4736-4741.doi: 10.3969/j.issn.2095-4344.2014.29.025
Previous Articles Next Articles
Erosion prevention and remineralization of human teeth
Wu Rui, Shi Mao-yu, Zheng Jing
- Tribology Research Institute, Key Laboratory of Advanced Technologies of Materials, Ministry of Education, Southwest Jiaotong University, Chengdu 610031, Sichuan Province, China
-
Revised:2014-04-22Online:2014-07-09Published:2014-07-09 -
About author:Wu Rui, Master, Tribology Research Institute, Key Laboratory of Advanced Technologies of Materials, Ministry of Education, Southwest Jiaotong University, Chengdu 610031, Sichuan Province, China -
Supported by:the National Natural Science Foundation of China, No. 51222511, 51305366; Excellent Youth Scientific Fund of Sichuan Province, No. 2012JQ0016
CLC Number:
Cite this article
Wu Rui, Shi Mao-yu, Zheng Jing. Erosion prevention and remineralization of human teeth[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(29): 4736-4741.
share this article
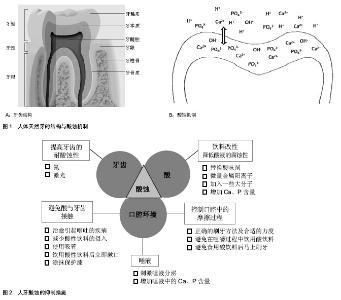
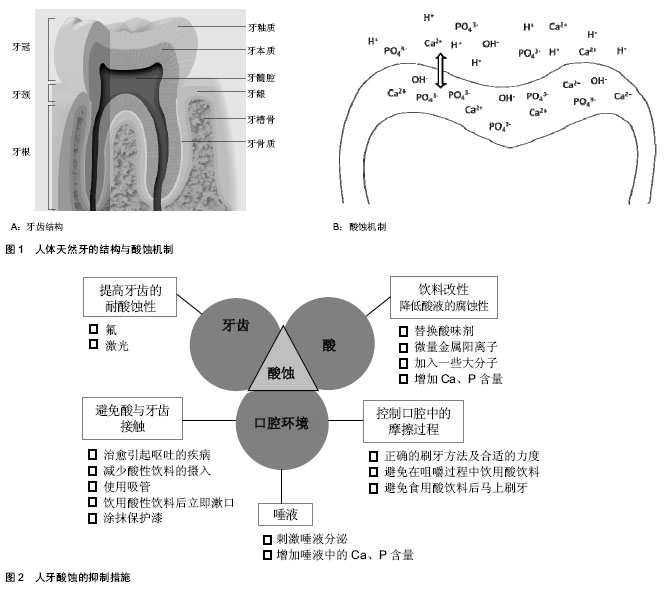
2.1 人牙酸蚀的机制与诱因 2.1.1 酸蚀产生的机制 人体天然牙由牙釉质、牙本质、牙骨质3种硬组织和一种软组织—牙髓构成[10](图1A)。通常,外层的牙釉质暴露在口腔环境下,充当咬合面,在咀嚼过程中直接与食物和对颌牙接触,承受咀嚼磨损和酸性介质的腐蚀。牙釉质是人体内最硬的组织,无机物含量高达92%-96%(质量分数),有机物和水的质量分数分别占1.0%-2.0%和3.0%-4.0% [11]。这些无机物以磷酸钙盐为主,它们以大量紧密排列的六边形羟基磷灰石[Ca10(PO4)6(OH)2]晶体的形式存在,并以此构成釉柱。釉柱是牙釉质的基本结构单元,呈细长的柱状结构,从牙本质釉质界发出,与牙本质表面垂直,呈放射状排列,一直延伸到牙齿表面,贯穿釉质全层。釉柱与釉柱之间是富含有机蛋白的釉间质,其所含的无机相成分与釉柱所含成分相同,只是含量和晶体排列方式不同。 含量高且排列精密的羟基磷灰石赋予牙釉质优异的机械性能与摩擦学性能,为咀嚼功能的完成和长期服役提供了保证。但是,高含量的羟基磷灰石也使得牙齿对酸十分敏感,容易发生酸蚀。牙齿酸蚀症是指在无细菌参与的情况下,由于接触牙面的酸或其螯合物的化学侵蚀作用而引起的一种病理的、慢性的牙体硬组织表面层丧失[12](图1B)。 当酸性物质进入口腔,牙釉质表面游离的PO43-、OH-、CO32-与H+结合,浓度下降;或者某些对钙离子有极强螯合能力的酸(比如柠檬酸)进入口腔,使Ca2+浓度迅速降 低[13],这些都会导致磷酸钙在牙釉质周围液体中的饱和程度下降。在不饱和状态下,牙釉质中的磷酸钙发生溶解,Ca2+﹑PO43-从牙釉质表面脱出,牙釉质发生脱矿。与脱矿相对应的是再矿化,即在脱矿初期,从牙齿表面溶解脱出的无机物重新回到牙齿表面的现象。这是因为牙齿无机相在酸性介质中的溶解行为具有动态可逆性[14-15]。一般情况下,牙釉质的脱矿与再矿化过程保持着动态平衡,牙齿不会出现明显的损伤,而当这一平衡向脱矿方向移动,牙齿将出现实质性的损伤,即酸蚀,临床上通常称之为牙齿酸蚀症。 2.1.2 酸性介质的来源 引起牙齿酸蚀的酸性物质可以分为内源性酸和外源性酸(表1)。从酸的来源来看,需要长期服用酸性药物的患者、胃食管返流疾病的患者、电池厂或磷酸厂的工作人员等都是发生牙齿酸蚀的高危人群。随着人们生活质量的提高和饮食模式的改变,酸性软饮料的摄入量与日俱增,由此导致的牙齿酸蚀问题也越来越严重。通常,饮料为了获得清爽的口感,包括碳酸饮料、果汁和运动饮料,几乎全部为酸性,pH值介于1-6之间[2],且绝大多数低于牙釉质脱矿的临界pH值(pH=5.5)[16],具有极强的酸蚀潜力[17-19]。 2.1.3 酸蚀的危害 酸蚀不仅导致牙齿硬组织的直接丧失,而且会显著降低牙齿表面的机械性能,使其更容易发生磨损[20]。研究显示,经酸软化后的人牙表面比紧实的牙齿表面更易发生三体磨粒磨损和两体接触磨损[21],因此,酸性饮食嗜好者的牙齿磨损率是正常人的10倍。如果对牙釉质的酸蚀损伤不加以控制,随着损伤程度加重,往往会诱发牙本质或牙髓受损,从而导致牙齿过敏,甚至牙髓暴露。严重的酸蚀症还可能造成牙齿折断、牙齿早失及咬合关系紊乱等,对患者的咀嚼功能、面形和心理等方面都会产生不良影响[22]。另外,酸蚀脱矿的牙面,由于釉质结构遭到破坏,表面粗糙,更容易罹患龋病[23]。 2.2 牙齿酸蚀的抑制措施 随着牙齿酸蚀问题日趋严重,越来越多的学者开始关注牙齿酸蚀的防护与抑制措施。目前,牙齿酸蚀的相关研究主要分为3类:①临床观察与统计[24-25],这方面的研究主要由临床牙科医生完成,调查不同人群牙齿酸蚀损伤等级与流行性,揭示牙齿酸蚀与内源酸或外源酸的相关性,并给予公众建议,以维护口腔健康。②酸性介质的酸蚀潜力[21,26-27],这些研究多采用体外模拟试验,考察牙齿在不同酸性介质中的腐蚀损伤行为,评价酸性介质对牙齿的腐蚀能力,研究酸的种类、酸度、接触时间等因素对牙齿酸蚀损伤程度的影响。③酸蚀的抑 制[28-30],这类研究是牙齿酸蚀研究的热点,在牙齿酸蚀相关的研究中所占的比重最大。目前,研究者已经总结和发展了很多抑制酸蚀的措施,而且新的方法也不断出现。这些抑制措施主要围绕牙齿、酸和口腔环境3个因素展开(图2)。 2.2.1 调控口腔环境 避免或减少牙齿与酸性物质的直接接触:治愈能够引起呕吐的疾病,避免胃酸进入口腔,减少内源性酸对牙齿的酸蚀。佩戴面罩或者使用中和药剂避免环境中的酸性介质进入口腔。遵循更健康的生活习惯。如:减少酸性饮食的摄入频率和次数;不要将酸性饮料长时间含在口中;使用吸管能够缩短酸性饮料与牙齿的接触时间;饮用酸性饮料之后用清水或苏打水漱口,加速清除口腔中残留的酸性物质,使口腔pH值回归中性;如果可能,避免嚼服和含服酸性药物,采取吞咽服用。 此外,在牙齿表面涂抹保护漆,形成保护膜,也能有效避免酸与牙齿表面的直接接触。这种方法的局限性在于,操作需要由牙科医生完成,过程复杂,一般只针对酸蚀易感的牙齿,而且保护层容易脱落,不能保证长期效果。 唾液:刺激唾液分泌,保证唾液在口腔中的流量,能有效抑制酸蚀。唾液在抑制牙齿酸蚀方面扮演着重要的角色:唾液可作为缓冲溶液,保持口腔的中性环境,降低牙齿酸蚀的可能性;唾液的吞咽能加速清除口腔中残留的酸性物质,缩短酸液在口腔中的停留时间[13],减小牙齿酸蚀损伤;唾液中含有钙、磷等离子,能够为酸蚀牙釉质的再矿化过程提供矿物质,在一定程度上修复牙釉质的酸蚀损伤。刺激唾液分泌的具休方法,如咀嚼口香糖、奶酪等。对于某些特殊情况,如服用利尿药物导致的口腔干燥,刺激唾液分泌对酸蚀的抑制十分有效。 增加唾液中的钙、磷含量,也可有效修复牙齿的酸蚀损伤。对于酸蚀后的牙齿,增加唾液中的钙、磷含量,可以促进牙齿的再矿化和重建,修复牙齿的酸蚀损伤。研究表明酪蛋白磷酸肽对牙齿酸蚀损伤有明显的修复能力[31]。同样,NovaMin(诺华敏)即磷硅酸钙钠,是一种在生物活性玻璃基础上开发的口腔保健材料,也可在牙体表面释放钙和磷,促进牙体组织矿化和重建[23]。 口腔中的摩擦过程:避免酸蚀牙齿表面发生摩擦。酸蚀导致牙齿外表层硬组织部分脱矿,机械性能显著下降,称之为软化层。通常情况下,软化层仍然可以保留牙釉质晶相结构的基本框架,在唾液中可以通过再矿化机制来修复酸蚀损伤[32]。另一方面,在酸蚀的后续过程中,软化层作为钙、磷离子脱出的空间障碍,可以抑制次表层牙釉质的脱矿,从而减缓牙齿酸蚀的进一步发生[33]。因此,软化层的存在对牙釉质酸蚀损伤的抑制和修复是有利的。不过,由于软化层的机械性能和耐磨性明显逊于正常牙釉质,即使在正常的牙齿与牙齿、牙齿与口腔软组织、牙齿与牙刷的接触过程中,也很容易被摩擦去除。因此,应在咀嚼过程中应该尽量避免饮用酸性饮料,并避免在酸性饮食后马上刷牙。此外,应对酸蚀冲击时,牙齿表面的获得性唾液吸附膜及牙菌斑也对牙齿有一定的保护作用[12],因此,日常应采用正确的刷牙方法及合适的力度刷牙,尽量选用刷毛柔软的牙刷和对牙齿磨损较小的牙膏,以减少对牙齿表面唾液吸附膜和软化层的破坏。 2.2.2 降低酸的腐蚀能力 这方面的研究主要集中在酸性饮料配方的改性设计上,具体方法包括:①寻找腐蚀性较低的酸味剂替代腐蚀性较高的酸味剂,降低饮料的酸蚀能力。②饮料中加入微量金属阳离子,如Fe2+,可以有效降低酸性饮料的腐蚀性,抑制牙齿酸蚀的发生[34]。③当一些大分子被加入到酸性饮料中,如木糖醇[35],可以渗透到脱矿表面内部,阻断离子扩散通路,减少钙、磷离子的脱出,抑制酸蚀。④增加酸性饮料中的钙、磷含量,能够降低牙釉质在酸性介质中的脱矿程度,显著降低饮料的腐蚀能力[36-39]。 2.2.3 提高牙齿抵抗酸蚀的能力 氟在口腔健康中有着举足轻重的地位,在临床防龋方面有悠久的历史,即使在龋病预防方法多元化的今天,氟化物在龋病预防中仍然发挥着重要作用。鉴于氟在防龋方面取得的显著效果,近来一些学者对氟抑制牙齿酸蚀的能力进行了研究。研究结果显示,当以保护漆[40-41]、牙膏[42-43]、漱口水[44]、凝胶的形式使用氟时[45-46],牙齿的酸蚀都能得到抑制。氟的酸蚀抑制作用机制主要表现在两个方面,一方面氟离子(F-)与羟基磷灰石晶体中的OH-发生交换,或者与口腔中的Ca2+和PO43-结合形成氟磷灰石,氟磷灰石晶体在酸液中溶解的临界pH值显著低于羟基磷灰石晶体,能提高牙齿的抗酸能力;另一方面,F-与游离Ca2+能结合生成氟化钙沉淀(CaF2),阻止钙离子的流失,为牙齿提供一个钙离子的储库,当酸攻击釉质时,釉质表面沉积的氟化钙颗粒能够中和酸,缓冲口腔pH,成为酸攻击时的暂时屏障[47]。研究发现,激光处理也能减少牙齿的脱矿[48-50],其龋齿防治能力已经被证实[51-52],是一种切实可行的龋齿防治手段[53]。相比而言,激光处理用于酸蚀抑制的研究还比较少。Liu等[54]报道了Er:YAG激光在以相同能量处理牙齿时抑制了19.2-32.78%的酸蚀损伤。Esteves-Oliveira等[55]在体外试验中使用CO2激光处理牙齿,抑制了牙齿硬度因酸蚀导致的下降,效果高达97%,并在后续的试验中证明激光处理能显著增加酸蚀后软化表面的耐磨性。激光能够抑制酸蚀的原因在于,热效应使牙齿的温度急剧上升,最高温度超过1 600 ℃[56]。在这样的高温下,牙釉质内的羟基磷灰石晶体熔化,随后凝结成更加致密的结构,降低了牙齿的渗透性,从而抑制牙齿的酸蚀损伤[57]。"
| [1] Ren YF, Zhao Q, Malmstrom H, et al. Assessing fluoride treatment and resistance of dental enamel to soft drink erosion in vitro: Applications of focus variation 3D scanning microscopy and stylus profilometry.J Dent.2009;37(3):167-76. [2] Ehlen LA, Marshall TA, Qian F, et al.Warren. Acidic beverages increase the risk of in vitro tooth erosion.Nutr Res. 2008;28(5): 299-303. [3] Ren YF.Dental Erosion: Etiology, Diagnosis and Prevention. The Academy of Dental Therapeutics and Stomatology.2011. [4] Ehlen LA, Marshall TA, Qian F, et al. Acidic beverages increase the risk of in vitro tooth erosion. Nutr Res. 2008; 28: 299-303. [5] Zhou ZR, Zheng J. Tribology of dental materials: a review.Journal of Physics D: Applied Physics. 2008;41(11): 113001. [6] Zheng J, Xiao F, Qian LM, et al. Erosion behavior of human tooth enamel in citric acid solution.Tribology International. 2009; 42(11-12):1588-1564. [7] Van Eygen I, Vannet BV, Wehrbein H.Influence of a soft drink with low pH on enamel surfaces: an in vitro study.Am J Orthod Dentofacial Orthop.2005;128(3):372-377. [8] Devlin H, Bassiouny MA, Boston D.Hardness of enamel exposed to Coca-Cola and artificial saliva.J Oral Rehabil. 2006; 33(1):26-30. [9] Hannig C, Hamkens A, Becker K, et al. Erosive effects of different acids on bovine enamel: release of calcium and phosphate in vitro.Arch Oral Biol.2005;50(6):541-552. [10] 于世凤.口腔组织病理学[M].北京:人民卫生出版社,2000. [11] Gwinnett AJ. Structure and composition of enamel. Oper Dent.1992;17:10-17. [12] 高晋华,杜民权,台保军.牙齿酸蚀症[J].国外医学:口腔医学分册, 2005;32(5):388-390. [13] Bashir E, Ekberg O, Lagerlöf F. Salivary clearance of citric acid after an oral rinse.J Dent.1995;23(4):209-212. [14] Holbrook WP, Arnadóttir IB, Kay EJ.Prevention. Part 3: prevention of tooth wear.Br Dent J.2003;195(2):75-81. [15] Tahmassebi J, Duggal M, Malik-Kotru G,et al.Soft drinks and dental health: a review of the current literature.J Dent. 2006; 34(1):2-11. [16] Hicks J, Garcia-Godoy F, Flaitz C. Biological factors in dental caries enamel structure and the caries process in the dynamic process of demineralization and remineralization (part 2).J Clin Pediatr Dent.2004;28(2):119-124. [17] Devlin H, Bassiouny M, Boston D. Hardness of enamel exposed to Coca‐Cola® and artificial saliva. J Oral Rehabil. 2006;33(1):26-30. [18] Kitchens M, Owens BM. Effect of carbonated beverages, coffee, sports and high energy drinks, and bottled water on the in vitro erosion characteristics of dental enamel.J Clin Pediatr Dent.2007;31(3):153-159. [19] Moazzez R, Smith B, Bartlett D. Oral pH and drinking habit during ingestion of a carbonated drink in a group of adolescents with dental erosion.J Dent.2000;28(6):395-397. [20] 黄欢.人体天然牙在柠檬酸介质中腐蚀磨损特性研究[D].成都:西南交通大学, 2010. [21] Hannig C, Hamkens A, Becker K,et al. Erosive effects of different acids on bovine enamel: release of calcium and phosphate in vitro.Arch Oral Biol.2005;50(6):541-552. [22] 李文卿,赵玮.控制牙齿酸蚀症的研究进展[J].国际口腔医学杂志, 2008,35(4):3. [23] 蔡晨星,朱玲,吴婴南.奥威尔牙齿脱敏剂对抗可乐酸蚀作用研究[J]. 实用口腔医学杂志, 2011,27(4):518-521. [24] Tahmassebi JF, Duggal MS, Malik-Kotru G, et al.Soft drinks and dental health: a review of the current literature.J Dent. 2006;34(1):2-11. [25] Milosevic A.Dental Erosion in a Series of Referred Patients was Statistically Associated With Gastric Reflux, Acidic Drink Intake of more than 0.5 L per day, and Low Salivary Buffering Capacity.J Evid Based Dent Pract.2010;10(3):176-178. [26] Beyer M, Reichert J, Bossert J,et al.Acids with an equivalent taste lead to diferent erosion of human dental enamel original. Dent Mater.2011;27(10):1017-1023. [27] Ablal MA, Kaur JS, Cooper L, et al.The erosive potential of some alcopops using bovine enamel: An in vitro study. J Dent.2009;37(11):835-839. [28] Hooper SM, Newcombe RG, Faller R,et al.The protective effects of toothpaste against erosion by orange juice: Studies in situ and in vitro.J Dent.2007;35(6):476-481. [29] Tschoppe P, Zandim DL, Sampaio JE,et al.Saliva substitute in combination with high-concentrated fluoride toothpaste: Effects on demineralised dentin in vitro.J Dent. 2010;38(3): 207-213. [30] Wang X, Megert B, Hellwig E, et al. Preventing erosion with novel agents.J Dent.2011;39(2):163-170. [31] Reynolds EC. Remineralization of enamel subsurface lesions by casein phosphopeptide-stabilized calcium phosphate solutions.J Dent Res.1997;76(9):1587-1595. [32] Collys K, Cleymaet R, Coomans D, et al.Rehardening of surface softened and surface etched enamel in vitro and by intraoral exposure. Caries Res. 2009;27:15-20. [33] 肖枫.人牙釉质的酸蚀特性研究[D].成都:西南交通大学, 2008. [34] Kato MT, Sales-Peres SH, Buzalaf MA.Effect of iron on acid demineralisation of bovine enamel blocks by a soft drink.Arch Oral Biol.2007;52(11):1109-1111. [35] Amaechi B, Higham S, Edgar W. The influence of xylitol and fluoride on dental erosion in vitro.Arch Oral Biol. 1998;43(2): 157-161. [36] Grenby TH. Lessening dental erosive potential by product modification.Eur J Oral Sci.1996;104(2 ( Pt 2)):221-228. [37] Larsen MJ.Degrees of saturation with respect to apatites in fruit juices and acidic drinks.Scand J Dent Res. 1975;83(1): 13-17. [38] Magalhães A, Moraes S, Rios D, et al.Effect of ion supplementation of a commercial soft drink on tooth enamel erosion.Food Addit Contam Part A Chem Anal Control Expo Risk Assess.2009;26(2):152-156. [39] Ramalingam L, Messer L, Reynolds E.Adding casein phosphopeptide-amorphous calcium phosphate to sports drinks to eliminate in vitro erosion.Pediatr Dent. 2005;27(1): 61-67. [40] Murakami C, Bönecker M, Corrêa MSNP,et al. Effect of fluoride varnish and gel on dental erosion in primary and permanent teeth.Arch Oral Biol.2009;54(11):997-1001. [41] Vieira A, Ruben JL, Huysmans MC.Effect of titanium tetrafluoride, amine fluoride and fluoride varnish on enamel erosion in vitro.Caries Res.2005;39(5):371-379. [42] Barlow A, Sufi F, Mason S. Evaluation of different fluoridated dentifrice formulations using an in situ erosion remineralization model.J Clin Dent.2009;20(6):192-128. [43] Zero D, Hara A, Kelly S, et al. Evaluation of a desensitizing test dentifrice using an in situ erosion remineralization model. J Clin Dent.2006;17(4):112-116. [44] Schlueter N, Klimek J, Ganss C.In vitro efficacy of experimental tin-and fluoride-containing mouth rinses as anti-erosive agents in enamel.J Dent.2009;37(12):944-948. [45] Jones L, Lekkas D, Hunt D, et al.Studies on dental erosion: An in vivo‐in vitro model of endogenous dental erosion‐its application to testing protection b flouride gel application. Aust Dent J.2002;47(4):304-308. [46] Lagerweij M, Buchalla W, Kohnke S, et al. Prevention of erosion and abrasion by a high fluoride concentration gel applied at high frequencies. Caries Res. 2006;40:148-53. [47] Petzold M. The influence of different fluoride compounds and treatment conditions on dental enamel: a descriptive in vitro study of the CaF2 precipitation and microstructure. Caries Res. 2001;35:45-51. [48] Esteves-Oliveira M, Zezell D, Meister J, et al. CO2 laser (10.6 μm) parameters for caries prevention in dental enamel.Caries Res.2009;43(4):261-268. [49] Rodrigues L, Dos Santos MN, Featherstone J.In situ mineral loss inhibition by CO2 laser and fluoride.J Dent Res. 2006; 85(7):617-621. [50] Steiner-Oliveira C, Nobre-dos-Santos M, Zero DT, et al. Effect of a pulsed CO2 laser and fluoride on the prevention of enamel and dentine erosion.Arch Oral Biol. 2010;55(2): 127-133. [51] Antunes A, Salvador V, Scapin M, et al.Nanosecond Nd: YAG laser on dental enamel: compositional analysis by X-ray fluorescence.Laser Physics Letters.2005;2:318-323. [52] L. E. H. de Andrade, R. F. Z. Lizarelli, J. E. P. Pelino, et al. Enamel caries resistance accidentally irradiated by the Nd: YAG laser.Laser Physics Letters.2007;4:457-463. [53] Tsai CL, Lin YT, Huang ST, et al. In vitro Acid Resistance of CO2and Nd-YAG Laser-Treated Human Tooth Enamel.Caries Res.2002 ;36(6):423-429. [54] Liu JF, Liu Y, Stephen HC.Optimal Er: YAG laser energy for preventing enamel demineralization. J Dent. 2006;34(1):62- 66. [55] Esteves-Oliveira M, Pasaporti C, Heussen N,et al. Rehardening of acid-softened enamel and prevention of enamel softening through CO2 laser irradiation.J Dent.2011; 39(6):414-421. [56] Hirota F, Furumoto K. Temperature rise caused by laser (CO2, Nd: YAG, Er: YAG) irradiation of teeth. International Congress Series.2003;1248:301-304. [57] 刘莉,黄楚云,李正佳. Nd:YAG激光提高牙齿抗酸能力实验与机制研究[J].激光杂志,2005,26(1):83-84. [58] Attin T, Knöfel S, Buchalla W, et al.In situ evaluation of different remineralization periods to decrease brushing abrasion of demineralized enamel. Caries Res. 2001;35(3): 216-222. |
| [1] | Chen Ziyang, Pu Rui, Deng Shuang, Yuan Lingyan. Regulatory effect of exosomes on exercise-mediated insulin resistance diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 4089-4094. |
| [2] | Chen Yang, Huang Denggao, Gao Yuanhui, Wang Shunlan, Cao Hui, Zheng Linlin, He Haowei, Luo Siqin, Xiao Jingchuan, Zhang Yingai, Zhang Shufang. Low-intensity pulsed ultrasound promotes the proliferation and adhesion of human adipose-derived mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3949-3955. |
| [3] | Yang Junhui, Luo Jinli, Yuan Xiaoping. Effects of human growth hormone on proliferation and osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3956-3961. |
| [4] | Sun Jianwei, Yang Xinming, Zhang Ying. Effect of montelukast combined with bone marrow mesenchymal stem cell transplantation on spinal cord injury in rat models [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3962-3969. |
| [5] | Gao Shan, Huang Dongjing, Hong Haiman, Jia Jingqiao, Meng Fei. Comparison on the curative effect of human placenta-derived mesenchymal stem cells and induced islet-like cells in gestational diabetes mellitus rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3981-3987. |
| [6] | Hao Xiaona, Zhang Yingjie, Li Yuyun, Xu Tao. Bone marrow mesenchymal stem cells overexpressing prolyl oligopeptidase on the repair of liver fibrosis in rat models [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3988-3993. |
| [7] | Liu Jianyou, Jia Zhongwei, Niu Jiawei, Cao Xinjie, Zhang Dong, Wei Jie. A new method for measuring the anteversion angle of the femoral neck by constructing the three-dimensional digital model of the femur [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3779-3783. |
| [8] | Meng Lingjie, Qian Hui, Sheng Xiaolei, Lu Jianfeng, Huang Jianping, Qi Liangang, Liu Zongbao. Application of three-dimensional printing technology combined with bone cement in minimally invasive treatment of the collapsed Sanders III type of calcaneal fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3784-3789. |
| [9] | Qian Xuankun, Huang Hefei, Wu Chengcong, Liu Keting, Ou Hua, Zhang Jinpeng, Ren Jing, Wan Jianshan. Computer-assisted navigation combined with minimally invasive transforaminal lumbar interbody fusion for lumbar spondylolisthesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3790-3795. |
| [10] | Hu Jing, Xiang Yang, Ye Chuan, Han Ziji. Three-dimensional printing assisted screw placement and freehand pedicle screw fixation in the treatment of thoracolumbar fractures: 1-year follow-up [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3804-3809. |
| [11] | Shu Qihang, Liao Yijia, Xue Jingbo, Yan Yiguo, Wang Cheng. Three-dimensional finite element analysis of a new three-dimensional printed porous fusion cage for cervical vertebra [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3810-3815. |
| [12] | Wang Yihan, Li Yang, Zhang Ling, Zhang Rui, Xu Ruida, Han Xiaofeng, Cheng Guangqi, Wang Weil. Application of three-dimensional visualization technology for digital orthopedics in the reduction and fixation of intertrochanteric fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3816-3820. |
| [13] | Sun Maji, Wang Qiuan, Zhang Xingchen, Guo Chong, Yuan Feng, Guo Kaijin. Development and biomechanical analysis of a new anterior cervical pedicle screw fixation system [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3821-3825. |
| [14] | Lin Wang, Wang Yingying, Guo Weizhong, Yuan Cuihua, Xu Shenggui, Zhang Shenshen, Lin Chengshou. Adopting expanded lateral approach to enhance the mechanical stability and knee function for treating posterolateral column fracture of tibial plateau [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3826-3827. |
| [15] | Zhu Yun, Chen Yu, Qiu Hao, Liu Dun, Jin Guorong, Chen Shimou, Weng Zheng. Finite element analysis for treatment of osteoporotic femoral fracture with far cortical locking screw [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3832-3837. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

