Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (11): 2060-2066.doi: 10.3969/j.issn.2095-4344.2013.11.024
Previous Articles Next Articles
Osteocytes and bone remodeling
Wang Si-sheng, Gui Bin-jie, Zhou Jian
- The First Affiliated Hospital of Anhui Medical University, Hefei 230022, Anhui Province, China
-
Received:2012-09-16Revised:2012-11-12Online:2013-03-12Published:2013-03-12 -
Contact:Gui Bin-jie, Doctor, Associate chief physician, the First Affiliated Hospital of Anhui Medical University, Hefei 230022, Anhui Province, China guibinjie@163.com -
About author:Wang Si-sheng★, Studying for master’s degree, Physician, the First Affiliated Hospital of Anhui Medical University, Hefei 230022, Anhui Province, China wssd1988@yahoo.com.cn
CLC Number:
Cite this article
Wang Si-sheng, Gui Bin-jie, Zhou Jian. Osteocytes and bone remodeling[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(11): 2060-2066.
share this article
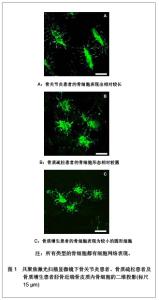
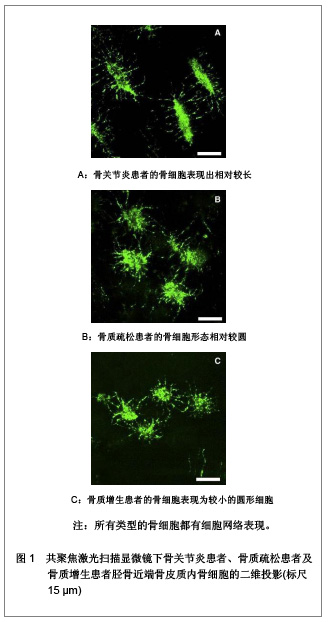
结果 2.1 应力感受细胞 骨细胞:占骨组织中细胞总数90%的骨细胞是由成骨细胞分化而来的。当成骨细胞包埋于它们自己合成的骨基质内,便分化成了拥有大量突触结构的骨细胞[3]。而每立方毫米骨组织内大约有10 000个骨细胞,每个骨细胞平均有50个细胞突触[4]。如此大量的骨细胞均匀地分布于矿化骨基质中,通过包膜和突触相互连接并与骨表面的细胞连接形成了三维网络状的细胞结构[5]。这个庞大的细胞结构位于骨基质中的骨陷窝-微管系统(lacuna- canalicular system)内,并通过骨陷窝-微管系统获得营养并排泄废物[6]。 一般来说,真核细胞对应力的刺激都有反应,这也意味着骨骼内的所有细胞都应当包含在力的感应细胞内。然而应力是作用于整个骨组织上的,利用计算机仿真模型比较骨细胞模型和骨表面细胞模型,发现骨表面的应力信息可能不足以充分的调节骨的适应性功能[7]。因此,相比于分布在骨表面的成骨细胞和破骨细胞,均匀分布在矿化骨基质中并形成细胞网络的骨细胞也许是更加理想的力学感应器。大量的研究表明,在体外培养试验中骨细胞感应到流体剪切力,并产生与体内骨受力相关的应答,例如产生一氧化氮[8]。通过三维荧光形态计量法研究鼠的颅骨与长骨间骨细胞网络结构发现,鼠胫骨的骨细胞突触的数量以及每一个突触分叉的数量显著高于顶骨。且胫骨骨细胞突触的长度约是顶骨的2倍,但由于突触直径较细,胫骨骨细胞的表面积和体积却小顶骨骨细胞。鼠不同部位间骨细胞形态以及细胞网络上的不同,一定程度上反应了骨细胞对于生物力学的敏感性以及适应性[9]。而实验也发现将动物体内的骨细胞去除将会造成骨丢失。同时,失去骨细胞的骨组织对力学刺激也丧失了反应。这些的实验显示针对正常的力刺激要维护骨量必须要骨细胞的参与[10]。因此,可以认为骨细胞是骨组织中最主要的应力感受器。 2.2 骨细胞如何感受应力 应力的感受是骨组织重建的第1步。因此,关于骨细胞是如何感受应力的问题,一直是该领域研究的重点。位于矿化骨基质内的骨细胞四周被坚硬的骨质包裹着。这些骨质阻止应力直接传导至骨细胞。然而,骨组织是一种包含液体、固体基质和细胞的多孔隙组织。在孔隙中运动的液体作为一种偶联性的中间介质,能够将外界动态环境中内生和外生的生物力学、生物化学和电化学信号传递给骨基质内的细胞[11]。理论模型和实验表明,由胞外压力和应变力变化产生的陷窝小管系统内的组织间液流量变化将作用于骨组织和骨细胞上,致使不同部位骨组织由于所受应力不同,骨细胞的三维形态及结构亦不相同,见图1[12]。施加至骨骼上的机械力诱导组织间液沿着陷窝-微管系统流动,产生细胞水平的剪切力并引起骨细胞质膜的变形[13]。理论模型研究发现,在生理负荷下作用于骨细胞质膜上的剪切力有0.8- 3 Pa[14]。而有研究表明,这种强度的机械信号足以直接被细胞膜上的纤毛或鞭毛直接感应到[15-16]。但是,纤毛或鞭毛是如何应答并传导力学信号的还不清楚。另一种观点认为,骨细胞感受流体剪切力主要是沿着树突进行的。在利用微探针和微粒对孤立的骨细胞实施局部变形,并分析变形中骨细胞胞内钙动态的研究发现,细胞突触的敏感性比胞体要高。这表明骨细胞的突触可能作为应力造成细胞变形的直接感受器[17]。"
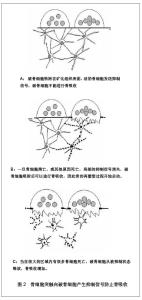
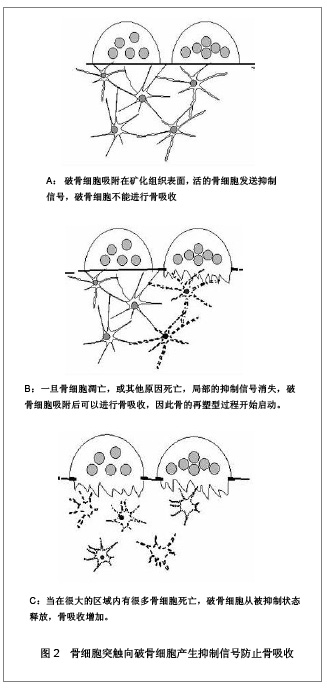
另一方面,骨细胞的分离和体外培养技术使得这一领域的研究变得更加深入。在体外培养中,骨细胞对机械刺激表现出较高的反应性。但在不同应力方式刺激下所表现出的反应并不相同。在液体脉冲处理后,一氧化氮和前列腺素E2值增加了2-9倍。循环拉伸刺激产生了2倍的一氧化氮,但不影响前列腺素E2表达。此外,基板的拉伸使骨基质Ⅰ型胶原蛋白升高的2倍,而流体剪切力造成了Ⅰ型蛋白胶原减少50%。这表明不同的应力刺激方式可能启动了不同的信号转导途径[18]。而目前,对于骨细胞将力学刺激转化为生物信号的途径仍不清楚。在今后的研究中,对骨细胞的各细胞成分,如细胞膜、细胞骨架和胞浆等,在应力的感受和传到中作用需要更进一步的探讨。 2.3 骨细胞感受应力后的反应 骨细胞能够在感受机械应力刺激后分泌并调整了一些重要的分子的表达。这些分子在骨的重建过程中有着重要的作用,影响着骨组织的代谢并调节各种骨重建相关细胞的活性。体外研究实验为此提供了大量的证据。在骨细胞的体外培养中,钙离子的涌入是感受机械应力早期重要的应答。钙离子可能直接通过细胞膜上的机械敏感性通道开放而涌入。同样也可能通过间接释放ATP和NAD+的半通道的开放而进入胞内,并导致胞内钙水平升高以增加钙通道的开放[19]。胞内钙浓度的升高是激活钙调蛋白所必须的,比如一氧化碳合酶。而在体外试验中也证明,流体剪切力可以刺激骨细胞产生高水平的钙离子和前列腺素E2[20-21]。此外,骨细胞体外培养中,应力刺激诱导产生环氧合酶的一种异构体环氧合酶2。在15 min后的脉冲流体刺激后骨细胞内的环氧合酶2的mRNA表达增加3倍。而停止刺激后1 h内环氧合酶2仍持续表达[22]。而诱导生成环氧合酶2是体内机械应力下诱导骨形成的关键步骤之一[23]。近期的研究表明,另一种分子家族被确定为骨机械应力适应性的调节者。这种分子就是Wnt蛋白家族。Wnt蛋白是一种与骨适应性应答相关的分泌糖蛋白。在体外培养试验中,骨细胞能够通过调节Wnt信号分子的mRNA来对机械应力产生应答。重要的是流体剪切力同样上调已知的Wnt靶基因的表达,例如Cx43, c-jun和CD44。这表明机械应力激活了经典的Wnt途径[24]。有实验证明骨细胞受应力刺激后可能激活Wnt/Lrp通路作为硬化蛋白的负调节因子,而硬化蛋白本身是骨形成的负调节因子[25]。 而研究也发现,在应力改变的情况下骨细胞中一些基因和蛋白质的表达发生了改变。骨细胞能够通过在早期产生c-fos mRNA对应力刺激产生应答[26]。而在失重情况下则会引起骨细胞中骨桥蛋白的表达[27]。在牙运动模型中,主要由分化后期的成骨细胞和骨细胞分泌的细胞外基质蛋白——牙本质基质蛋白1在应答机械应力几小时内被激活[28]。骨细胞分泌的牙本质基质蛋白1与新骨形成、动态骨改建及机械应力传导都密切相关。 有实验表明,牙本质基质蛋白1的基因失活突变与常染色体隐性遗传的低磷性佝偻病的发生密切相关。小鼠的佝偻病模型中,在牙本质基质蛋白1失活的小鼠体内成纤维细胞生长因子23大量表达于骨细胞。这表明牙本质基质蛋白1通过抑制骨细胞的成纤维细胞生长因子23表达而参与全身钙磷的调节。此外,牙本质基质1蛋白启动子经常被用来作为一个骨细胞特异性启动子。然而,并不是所有骨细胞都分泌牙本质基质蛋白1。牙本质基质蛋白1的表达多是在骨皮质浅层的成骨样及骨样的年轻骨细胞中,而在分布于骨质深层的成熟骨细胞内表达却明显减少。这表明,骨细胞分泌的DMP-1蛋白与新骨形成、动态骨改建及机械应力传导都密切相关[29]。 2.4 骨细胞于重建中的作用 在关于骨细胞早期的研究中,骨细胞被认为通过细胞突触向破骨细胞产生抑制信号防止骨吸收[30],见图2。 例如,通过一氧化氮途径抑制破骨细胞活性和防止骨吸收[31]。而这一概念进一步发展成为骨细胞下调骨重建的假说。该假说认为破骨细胞接受到的抑制信号的强度与接触骨表面细胞的骨细胞数目成正比,与骨细胞至骨表面的距离成反比[32]。局部高频率的骨重建与活性骨细胞数量相对较低表现出相关性。这表明骨细胞密度的降低减少了对于重建的抑制[33]。而关于微裂纹的研究可以更进一步的解释该假说。骨在不断应力适应的过程中会发生损伤,在骨基质上产生细小的微裂纹。而微裂纹所造成的骨细胞陷窝-微管网络的断裂在骨重建中可能有着重要的作用。据观察发现微裂缝旁周围1.0-2.0 mm内骨细胞的促凋亡因子表达升高[34],这表明骨细胞凋亡仅限于微裂纹附近。骨细胞凋亡的信息通过微管网络传导至骨组织表面及骨祖细胞。该信息的本质并不清楚,可能由液体流动、生物信号和电刺激构成。近期有研究发现,肿瘤坏死因子α诱导凋亡的骨细胞的条件培养基中会促进破骨细胞前体黏附到内皮细胞。而阻断骨细胞凋亡则破骨细胞前的体黏附也相应减少。该研究发现,骨细胞凋亡的条件培养基比骨细胞未凋亡的培养基中包含更多的白细胞介素6和可溶性白细胞介素6受体(sIL-6R)。而骨细胞凋亡条件培养基中血管内皮细胞表面表达细胞间黏附分子1增加,而阻断细胞间黏附分子1则会消除凋亡介导的破骨细胞前体黏附。该研究表明骨细胞凋亡其介导的细胞间黏附分子1表达可调节破骨细胞前体黏附,而这可能是通过白细胞介素6分泌增加实现的[35]。"

该信息进一步导致了重建信号的启动。值得一提的是该信息可以不通过包括骨衬细胞在内的通常的信号途径而直接刺激骨吸收。这种直接的启动可能与核因子κB配体激活受体相关,包括肿瘤坏死因子相关的细胞激活诱导活素、骨保护素配体以及破骨细胞分化因子[36]。研究发现,与对照组相比微损伤并没有显著的影响细胞死亡的数量。然而,通过ELISA实验分析发现骨细胞分泌了大量的集落刺激因子和核因子κB配体激活受体。换句话说,如果骨细胞突触受损引起了核因子κB配体激活受体的上调,那么这将产生一个新的通路去激活破骨细胞,从而将微损伤、骨质吸收和基本多细胞单位的形成联系到一起[37]。 骨细胞不但在骨重建的启动中起着重要的作用,在重建过程中也有着重要的作用。有假说认为,当一个成骨细胞在被足够多的新骨和类骨质覆盖后,就会通过它的树枝状细胞突触发信号给相临近的成骨细胞,使骨质沉积的速度减慢。成骨细胞很有可能是通过这种抑制作用掩埋了它的邻居,然后成骨细胞也变成新一层的骨细胞[38]。研究表明通过流体剪切力处理的骨细胞产生了极有效地条件介质抑制骨细胞的增殖,并刺激碱性磷酸酶活性作为对抗骨细胞和骨膜成纤维细胞产生的条件介质[39]。在对于骨质疏松及骨关节炎患者小梁骨的研究中发现骨细胞微管系统的过度矿物质沉积影响骨质的重建及骨的生物力学特性[40]。这表明,骨细胞在重建过程中可能还承担着控制骨基质填充速度的角色。"


总结与展望 在过去的研究中,骨细胞被认为是成骨细胞转化成的机械应力感受细胞及活跃的旁分泌细胞,在调节成骨细胞及破骨细胞的活性中起着重要的作用。越来越多的研究表明骨细胞可能是骨重建的调节中心及启动者。关于骨细胞的研究使其更多的被应用到了骨代谢性疾病的治疗中。例如,使用骨质疏松的绵羊模型研究发现,二膦酸盐治疗组对比控制组骨细胞凋亡率显著降低,骨骼微裂纹减小,良好的保护了骨骼减少了骨折的发生率[41]。而随着二膦酸盐类药品应用于临床,在治疗骨质疏松防止骨质流失方面已取得了显著的治疗效果。在未来的研究中,对关于骨细胞感受应力的主要机制、应力信号转化为生物信号的途径以及调节骨质形成和吸收的调节靶点的研究,将会更进一步的阐述骨重建的机制,有助于各种骨代谢疾病研究和提出骨代谢疾病新的治疗方案。而骨细胞对于成骨细胞及破骨细胞在骨重建中的影响可能会有助于更进一步的了解骨折术后骨不连接的机制,以及对骨折术后恢复提出新的指导方案。 作者贡献:第一作者调研、分析文献,并完成本综述,第一作者对本文负责,其他作者参与文献筛选和质量评估,通讯作者审校论文并提出了重要修改意见。 利益冲突:课题未涉及任何厂家及相关雇主或其他经济组织直接或间接的经济或利益的赞助。 伦理要求:无涉及伦理冲突的内容。 作者声明:文章为原创作品,数据准确,内容不涉及泄密,无一稿两投,无抄袭,无内容剽窃,无作者署名争议,无与他人课题以及专利技术的争执,内容真实,文责自负。"

| [1] Sikavitsas VI, Temenoff JS, Mikos AG. Biomaterials and bone mechanotransduction. Biomaterials 2001;22:2581-2593.[2] Buckwalter JA,Glimcher MJ,Cooper RR,et al.Bone biology .1. Structure,blood-supply, cells, matrix, and mineralization.J Bone Joint Surg Am.1995; 77A:1256-1257.[3] Verborgt O, Gibson GJ, Schaffler MB. Loss of osteocyte integrity in association with microdamage and bone remodeling after fatigue damage in vivo. Bone Miner Res. 2000;15:60-67.[4] Marotti G, Cane V, Palazzini S,et al. Structure-function relationships in the osteocyte. Ital Min Electro Metab. 1990;4:93-106.[5] Donahue HJ.Cap Junctions and biophysical regulation of bone cell differentiation. Bone.2000;26:417-422.[6] Schneider P, Meier M, Wepf R,et al.Towards quantitative 3D imaging of the osteocyte lacuno-canalicular network. Bone. 2010;47:848-858.[7] Mullender MG, Huiskes R.Osteocytes and Bone Lining Cells: Which are the Best Candidates for Mechano-Sensors in Cancellous Bone? Bone.1997;20(6):527-532.[8] Tan SD, Bakker AD, Semeins CM, et al. Inhibition of osteocyte apoptosis by fluid flow is mediated by nitric oxide. Biochem Biophys Res Commun.2008;369(4)1150-1154.[9] Himeno-Ando A, Izumi Y, Yamaguchi A, et al. Structural differences in the osteocyte network between the calvaria and long bone revealed by three-dimensional fluorescence morphometry, possibly reflecting distinct mechano- adaptations and sensitivities.Biochem Biophys Res Commun. 2012;417: 765-770.[10] Tatsumi S,Ishii K,Amizuka N,et al.Targeted ablation of osteocyte induces osteoporosis whit defective machanotransduction. Cell Metab.2007;5: 464-475.[11] Baik AD, Lu XL, Qiu J,et al.Quasi-3D cytoskeletal dynamics of osteocytes under fluid flow. Biophys J.2010;99(9): 2812-2820.[12] van Hove RP, Nolte PA, Vatsa A, et al.Osteocyte morphology in human tibiae of different bone pathologies with different. Bone.2009;45:321-329.[13] Han Y, Cowin SC, Schaffler MB,et al.Mechanotransduction and strain amplification in osteocyte cellprocesses. Proc Natl Acad Sci USA.2004; 101:16689-16694[14] Weinbaum S, Cowin SC, Zeng Y.A model for the excitation of osteocytes by mechanical loading-induced bone fluid shear stresses.Biomech.1994;27:339-360.[15] Xiao Z, Zhang S, Mahlios J, et al. Cilia-like structures and polycystin-1 in osteoblasts/osteocytes and associated abnormalities in skeletogenesis and Runx2 expression.Biol Chem.2006;281:30884-30895.[16] Malone AM, Anderson CT,Tummala P,et al. Primary cilia mediate mechanosensing in bone cells by a calcium-independent mechanism. Proc Natl Acad Sci USA.2007; 104:13325–13330.[17] Aonuma Y, Adachi T, Tanaka M, et al. Site-Dependence of Mechanosensitivity in Isolated Osteocytes. 13th International Conference on Biomedical Engineering. 2008;23:2000-2004.[18] Mullender M, El Haj AJ, Yang Y, et al. Mechanotransduction of bone cells in vitro: mechanobiology of bone tissue. Medical & Biological Engineering & Computing.2004; 42:14-21.[19] Genetos DC, Kephart CJ, Zhang Y, et al. Oscillating fluid flow activation of gap junction hemichannels induces ATP release from MLO-Y4 osteocytes. Cell Physiol. 2007;212:207-214.[20] Rath AL, Bonewald LF, Ling J,et al.Correlation of cell strain in single osteocytes with intracellular calcium, but not intracellular nitric oxide, in response to fluid flow.J Biomech. 2010;43(8):1560-1564.[21] Kitase Y, Barragan L, Qing H,et al. Mechanical induction of PGE2 in osteocytes blocks glucocorticoid-induced apoptosis through both the β-catenin and PKA pathways. J Bone Miner Res. 2010; 25(12):2657-2668.[22] Westbroek I, Ajubi NE, Alblas MJ, et al.Differential stimulation of prostaglandin G/H synthase-2 in osteocytes and other osteogenic cells by pulsating fluid flow. Biochem Biophys Res Commun.2000;268:414-419.[23] Forwood MR. Inducible cyclooxygenase (COX-2) mediates the induction of bone formation by mechanical loading in vivo. Bone Miner Res.1996;11:1688–1693.[24] Santos A, Bakker AD, Zandieh-Doulabi B,et al. Pulsating fluid flow modulates gene expression of proteins involved in Wnt signaling pathways in osteocytes. J Orthop Res. 2009;27(10): 1280-1287.[25] Robling AG, Niziolek PJ, Baldridge LA, et al. Mechanical stimulation of bone invivo reduces osteocyte expression of Sost/sclerostin.J Biol Chem.2008; 283:5866-5875.[26] Inaoka T, Lean JM, Bessho T, et al.Sequential analysis of gene expression after an osteogenic stimulus: c-fos expression is induced in osteocytes. Biochem. Biophys. Res Commun.1995;217:264–270.[27] Gross TS, King KA, Rabaia NA, et al. Upregulation of osteopontin by osteocytes deprived of mechanical loading or oxygen. J Bone Miner Res. 2005;20(2):250-256. [28] Toyosawa S, Oya K, Sato S,et al.Osteocyte and DMP1. Clinical Calcium. 2012;22(5):713-720.[29] Feng JQ, Ward LM, Liu S, et al.Loss of DMP1 causes rickets and osteomalacia and identifies a role for osteocytes in mineral metabolism.Nat Genet. 2006;38 (11):1310-1315.[30] Marotti G,Ferretti M,Muglia MA,et al.A quantitative evaluation of osteoblast-osteocyte relationships on growing endosteal surface of rabbit tibiae. Bone.1992;13:363-368.[31] Tan SD, de Vries TJ, Kuijpers-Jagtman AM, et al. Osteocytes subjected to fluid flow inhibit osteoclast formationand bone resorption. Bone.2007;41:745-751.[32] Martin RB. Does osteocyte formation cause the nonlinear refilling of osteons? Bone, 2000, 26:71-78[33] Metz LN, Martin RB, Turner AS. Histomorphometric analysis of the effects of osteocyte density on osteonal morphology and remodeling. Bone.2003;33:753-759.[34] Noble BS, Reeve J. Osteocyte function, osteocyte death and bone fracture resistance. Mol Cell Endocrinol.2000;159:7-13.[35] Cheung WY, Simmons CA, You L.Osteocyte apoptosis regulates osteoclast precursor adhesion via osteocytic IL-6 secretion and endothelial ICAM-1 expression. Bone.2012;50 104-110.[36] Mulcahy LE, Taylor D, Lee TC,et al.RANKL and OPG activity is regulated by injury size in networks of osteocyte-like cells. Bone.2011;48(2):182-188.[37] Kurata K, Heino HJ, Higaki H,et al. Bone marrow cell differentiation induced by mechanically damaged osteocytes in 3D gel-embedded culture. Biomed Mater Res. 2006;21: 616–625.[38] Martin R. Does osteocyte formation cause the nonlinear refilling of osteons? Bone.2000;26:71-78.[39] Vezeridis PS, Semeins CM, Chen Q, et al. Osteocytes subjected to pulsating fluid flow regulate osteoblast proliferation and differentiation. Biochem Biophys Res Commun.2005;348:1082–1088.[40] Carpentier VT, Wong J, Yeap Y, et al.Increased proportion of hypermineralized osteocyte lacunae in osteoporotic and osteoarthritic human trabecular bone: Implications for bone remodeling. Bone.2012;50:688-694.[41] Brennan O, Kennedy OD, Lee TC,et al. Effects of estrogen deficiency and bisphosphonate therapy on osteocyte viability and microdamage accumulation in an ovine model of osteoporosis. J Orthop Res. 2011;29(3): 419-424. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [3] | Li Chenjie, Lü Linwei, Song Yang, Liu Jingna, Zhang Chunqiu. Measurement and statistical analysis of trabecular morphological parameters of titanium alloy peri-prosthesis under preload [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 516-520. |
| [4] | Zhang Yu, Feng Shuo, Yang Zhi, Zhang Ye, Sun Jianning, An Lun, Chen Xiangyang. Three-dimensional gait of patients with developmental dysplasia of hip undergoing total hip arthroplasty with high hip center [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 350-355. |
| [5] | He Jie, Chang Qi. Biological reconstruction of large bone defects after resection of malignant tumor of extremities [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 420-425. |
| [6] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Comparison of the advantages and disadvantages of multiple implants in treatment of traumatic dislocation of sternoclavicular joint [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 443-448. |
| [7] | Xie Jingshu, Zhang Xianglin, Liu Jinlei, Wen Jing. Application of High Resolution reconstruction algorithm in precision CT scans of the middle and inner ears [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3614-3618. |
| [8] | Yang Caihui, Liu Qicheng, Dong Ming, Wang Lina, Zuo Meina, Lu Ying, Niu Weidong. Serine/threonine protein kinases can promote bone destruction in mouse models of chronic periapical periodontitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3654-3659. |
| [9] | Wang Shengjun, Yin Fei, Jiang Maoyu. Application of titanium ossicular prosthesis in one-stage open tympanoplasty for middle ear cholesteatoma [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3505-3509. |
| [10] | Huo Hua, Cheng Yuting, Zhou Qian, Qi Yuhan, Wu Chao, Shi Qianhui, Yang Tongjing, Liao Jian, Hong Wei. Effects of drug coating on implant surface on the osseointegration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3558-3564. |
| [11] | Chen Jie, Liao Chengcheng, Zhao Hongbo, Zhao Wei, Chen Zhiwei, Wang Yan. Application of tissue engineering urethral stent and its preparation technology in urethral reconstruction [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3591-3596. |
| [12] | Jiang Shengyuan, Li Dan, Jiang Jianhao, Shang-you Yang, Yang Shuye. Biological response of Co2+ to preosteoblasts during aseptic loosening of the prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(21): 3292-3299. |
| [13] | Zheng Weipeng, Wei Hewei, Liu Zhijun, Wan Lei, Chen Sheng, Liao Zhihao, Hu Weijian. Application of three-dimensional reconstructive CT versus X-ray in postoperative evaluation of bone tunnel and graft status after single bundle anterior cruciate ligament near-isometric reconstruction [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(18): 2881-2886. |
| [14] | Han Shichong, Li Chang, Xing Haiyang, Ge Wenlong, Wang Gang . Finite element analysis of two internal fixation methods for treating extra-articular proximal tibial fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(15): 2329-2333. |
| [15] | Wu Yukun, Han Jie, Wen Shuaibo. Mechanism of Runx2 gene in fracture healing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(14): 2274-2279. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||