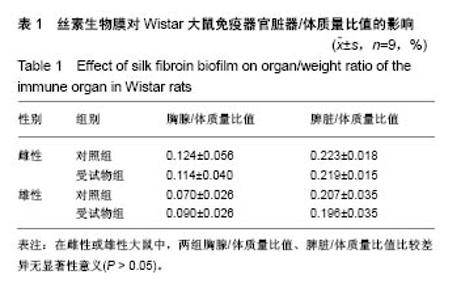
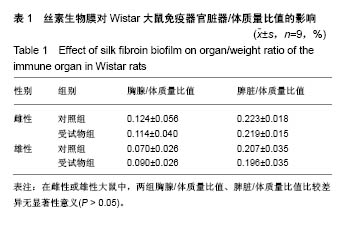
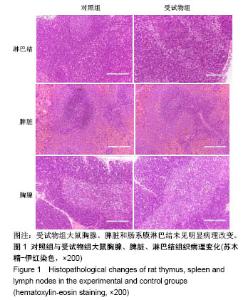
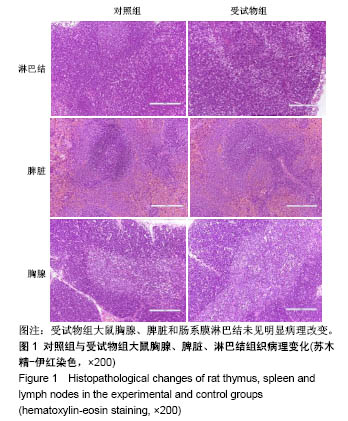
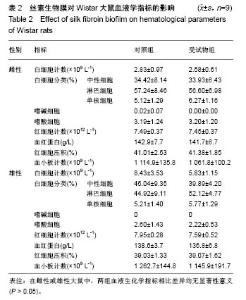
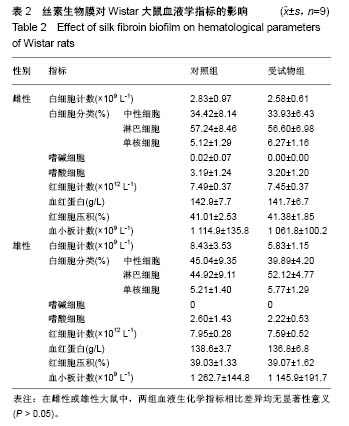
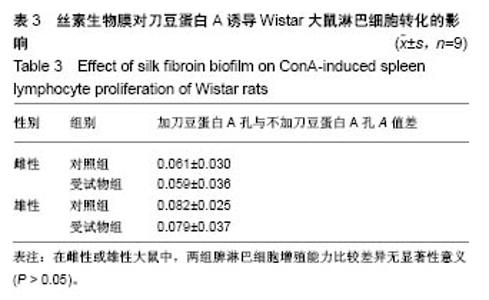
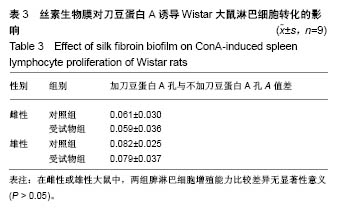
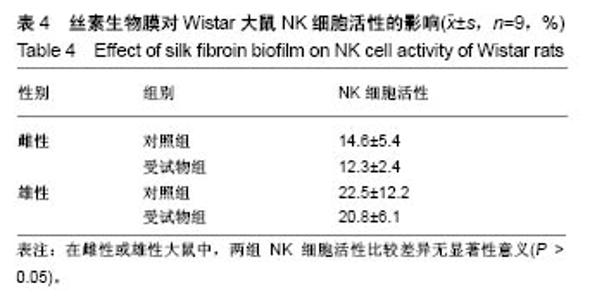
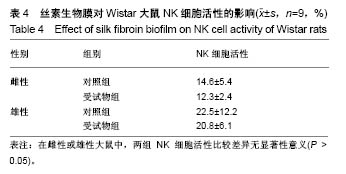
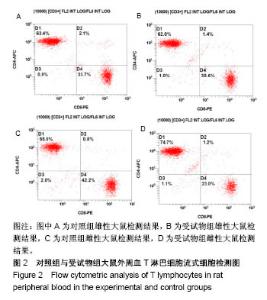
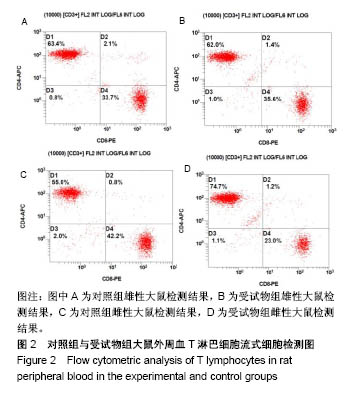
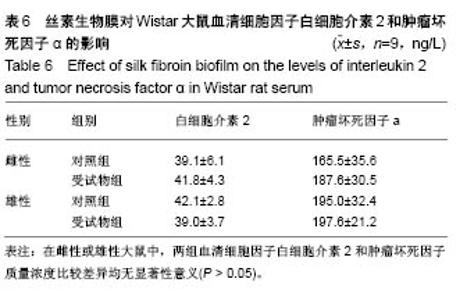
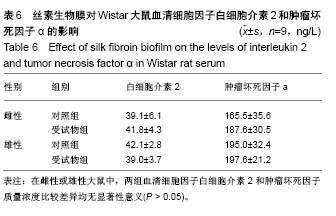
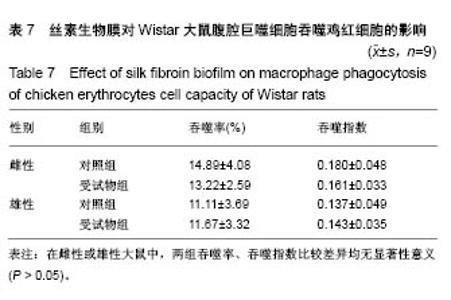
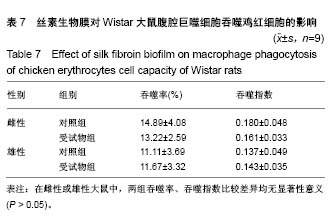
| [1] Li MZ,Tao W,Lu SZ,et al.Compliant film of regenerated Antheraea pernyi silk fibroin by chemical crosslinking. Int J Biol Macromol. 2003;32(3-5): 159-163. [2] Cha BG,Kwak HW,Park AR,et al. Structural characteristics and biological performance of silk fibroin nanofiber containing microalgae Spirulina extract.Biopolymers. 2014;101(4):307-318.[3] Luangbudnark W,Viyoch J,Laupattarakasem W,et al. Properties and biocompatibility of chitosan and silk fibroin blend films for application in skin tissue engineering. ScientificWorldJournal. 2012;2012:697201. [4] 王宏昕,李敏.丝素蛋白作为组织工程生物材料的研究进展[J].中国修复重建外科杂志,2008,22(2):192-195.[5] Wang J,Wei Y,Yi H,et al. Cytocompatibility of a silk fibroin tubular scaffold.Mater Sci Eng C Mater Biol Appl. 2014;34:429-436.[6] Calamak S,Erdo?du C,Ozalp M,et al. Silk fibroin based antibacterial bionanotextiles as wound dressing materials. Mater Sci Eng C Mater Biol Appl.2014;43:11-20.[7] 陆艳,赵霞,邵正中,等.多孔丝素材料组织相容性的初步研究[J].中国组织工程研究和临床康复,2011,15(34):6364-6367.[8] ISO/TS l0993-20:2006,Biological evaluation of medical Devices-Part 20:Principles and methods for immunotoxicology testing of medical device.[9] ICH:S8(Step 4)Immunoxicity Studies For Human Pharmaceutical:2005: 1-11.[10] 雷静,李奕恒,刘旭昭,等.动物源I型胶原蛋白可引起BALBlc小鼠细胞免疫反应和组织免疫毒性[J].中国组织工程研究, 2015,19(34):5506-5512.[11] 梅昕,马凤森,喻炎,等.高分子可降解生物材料的降解研究进展[J].材料导报, 2016,30(S1):298-303.[12] 李君涛,陈周煜.可降解生物医用材料研究现状与展望[J].新材料产业, 2016, 18(1):32-35.[13] 李云飞.医疗器械化学性能检测的重要性分析[J].化工设计通讯, 2017, 43(12):169-171.[14] 陈亮,奚廷斐,王春仁.组织工程医疗产品免疫学评价研究[J].组织工程与重建外科杂志,2009,5(1):56-58.[15] 邹文,袁暾,蔡永福,等.组织诱导性材料生物安全性评价研究策略与实践[J].中国组织工程研究,2018, 22(10):1534-1539.[16] 戴菁,孙旖,高婷婷,等.免疫毒性生物标志研究进展[J].毒理学杂志, 2012, 25(6):463-465.[17] Pulendran B,Artis D.New paradigms in type 2 immunity. Science. 2012; 337(6093):431-435.[18] Palm NW,Rosenstein RK,Medzhitov RAllergic host defences. Nature. 2012;484(7395):465-472.[19] Holsapple MP,Burns-Naas LA,Hastings KL,et al.Aproposed testing framework for developmental immunotoxicology(DIT). Toxicol Sci. 2005; 83(1):18-24.[20] 陈成章.免疫毒理学[M].郑州:郑州大学出版社,2008:171-174.[21] 杨晓芳,奚廷斐.医疗器械的免疫毒性评价[J].生物医学工程学杂志, 2007, 24(5):1191-1195.[22] 王延琳,马玉媛,赵雄,等.植人性医疗器械的临床前免疫毒性评价[J].医疗卫生装备,2017,38(8):117-126.[23] Germolec DR.Sensitivity and predicitivity in immunotoxicity testing: immune endpoints and disease resistance.Toxicol Lett. 2004;149(1-3): 109-114.[24] Luebke R,House R,Kimber I.Emanuela corsini animal and in vitro models of immunotoxicity.New York:CRC Press, 2007:66-68.[25] 潘东升,范玉明,李波.新药免疫毒性评价研究进展[J].中国新药杂志, 2018, 27(13):1491-1495.[26] 薛旸,丁婷婷,孙皎.生物材料免疫抑制效应评价的方法初探[J].中国医疗器械杂志,2009,33(1):20-22.[27] Hilal AA,Gaylor JDS.Bioartificial liver:review of science requirements and technology. World Review ofScience, Technology, and Sustainable Development.2006;3(1):80-83.[28] 谢从平.活化的非特异性细胞毒系统[J].国外医学(免疫学分册), 1987,14(4): 176. [29] Singh J,Suruchi A.Anti TNF-a strategy:present status of this therapeutic paradigm.Indian J Pharmacol.2004;36(1):10-14.[30] 吕志敢,郭政.肿瘤坏死因子的研究进展[J].山西医科大学学报, 2006,37(3): 311-314. |