Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (26): 4255-4259.doi: 10.3969/j.issn.2095-4344.1369
Previous Articles Next Articles
Research status and prospects of tissue engineering technology for repairing intervertebral disc annulus fibrosus
-
Received:2019-03-25Online:2019-09-18Published:2021-04-29 -
Contact:Ye Xiaojian, MD, Professor, Chief physician, Center for Minimally Invasive Spine Surgery, Changzheng Hospital, Second Military Medical University, Shanghai 200003, China -
About author:Wang Shuang, Doctoral candidate, Attending physician, Center for Minimally Invasive Spine Surgery, Changzheng Hospital, Second Military Medical University, Shanghai 200003, China -
Supported by:the National Natural Science Foundation of China, No. 81772445 (to YXJ)
CLC Number:
Cite this article
Wang Shuang, Yu Lei, He Yunfei, Ma Jun, Wen Jiankun, Ye Xiaojian. Research status and prospects of tissue engineering technology for repairing intervertebral disc annulus fibrosus[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(26): 4255-4259.
share this article
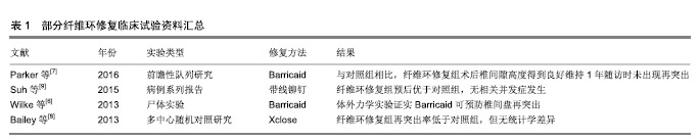
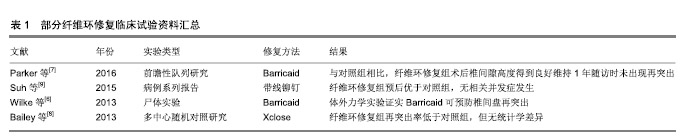
2.1 纤维环缝合、封堵技术 以往,有研究者尝试通过物理缝合、封堵等方式修复纤维环,BarricaidTM(美国Intrinsic Therapeutics Inc.)和Xclose Tissue Repair SystemTM(美国Anulex Technologies)是研究较多的纤维环缝合和修复产品[6],已部分应用于临床。有研究表明,应用此类修复方法后,患者症状改善程度、并发症风险等方面与单纯行髓核摘除而未修复纤维环的对照组接近,但术后短期随访证实纤维环修复组再手术风险明显降低[7-8]。此类直接缝合技术的中远期效果目前报道有限。在一项临床研究中,拟行纤维环修复的478例患者中,有多达137例(28.7%)无法完成修复而退出实验,失败原因包括纤维环组织条件差、突出范围过大、星芒状撕裂、解剖因素导致操作困难等,提示这一方法在操作技术方面仍需改进[9]。近年来,有研究者尝试以带线铆钉直接缝合纤维环破口,经3年随访未发现复发和再突出,但仍强调对适应证的严格选择是确保该技术修复效果的前提[9],见表1。 凝胶等修复材料可用于封堵纤维环缺损,但其力学性能有限,单独应用时难以取得理想的效果,因此目前多将其与其他材料复合使用以提高力学性能,如核黄素交联后的凝胶修复损伤纤维环的效果优于非交联凝胶[10]。也有研究者尝试在凝胶与其他材料构成复合材料的基础上向材料内载入纤维环细胞,形成交联强化凝胶+细胞复合体,且其植入尾椎间盘退变大鼠模型中取得了理想的修复效果[11],此类方法已接近组织工程技术流程,虽目前仍处于动物实验阶段,但已逐渐接近临床。 尽管获得了以上成果,直接修复技术的主要作用仍是直接修复纤维环,以恢复纤维环的完整性。但是直接修复仅可恢复和维持椎间盘的微环境,不能起到促进椎间盘再生的目的,且受术野和技术所限,对浅层纤维环缝合效果好,深层纤维环的修复作用有限[12]。 缝合和封堵技术的发展可为其他修复材料或支架提供恰当的固定方式,确保其稳妥地固定于缺损部位而发挥作用。理想的缝合或修复方式应满足以下条件:操作简单,便于在狭小的术野中展开;所使用材料的生物相容性好、炎症反应小,尽可能不与周围组织发生炎症反应;尽可能避免使用金属材料以免影响术后MRI等检查。 2.2 组织工程修复纤维环技术 近年来,组织工程技术为这一问题的解决提供了新的思路,其基本思路是将种子细胞与纤维环修复支架共培养后植入纤维环破口,借助植入细胞的再生长使纤维环破口得到生物学修复[13]。针对纤维环修复的组织工程技术近年的发展主要集中于支架的制备、种子细胞的获取、微环境的优化等方面。 2.2.1 纤维环修复支架的构建 组织工程技术首先需根据纤维环的特殊结构构建仿生化支架。纤维环的主要成分为富含胶原纤维的纤维软骨,位于椎间盘外周,环绕髓核,主要发挥限制髓核、连接相邻椎体的作用,并维持椎间盘运动单元的功能[14],其完整性对维持椎间盘内压、保护髓核组织具有重要作用。在物理结构上,纤维环在纵切面上纤维在椎体间斜行,相邻板层间纤维排列方向相反并形成交叉;横切面上各层纤维排列成同心环,层间还有许多胶原纤维交联[15],具有各向异性、非线性、黏弹性等特点[16]。为模拟纤维环胶原纤维定向排列的结构特点,现多采用静电纺丝技术进行支架构建[5,17],可用于构建支架的材料既包括蚕丝、海藻酸/壳聚糖等天然材料[18-20],也包括聚丙烯甲酯、聚已内酯等合成材料[21-24]。制备支架时需兼顾支架孔隙率和生物力学强度间的平衡[25-26],以达到既利于细胞长入又确保支架可承担相应力学负荷的目的。研究表明,将不同性质的材料复合在一起构建复合材料支架的方法能发挥不同材料的优势[14,27-28]。 近年来,有研究证实纳米纤维支架的拓扑结构能诱导粘附于其表面的干细胞发生形态改变,并进而调控其定向分化[29]。虽然纳米纤维调节细胞形态的现象已经被广泛认可,但细胞发生这些形态变化的分子机制还有待研究。 2.2.2 种子细胞的获得 究竟何种细胞是纤维环修复的最理想的种子目前尚无定论,有研究表明,即使是退变间盘的纤维环细胞,也保留有一定的增殖、迁移和形成细胞外基质的潜力[30],这些潜力能够被不同因素激发或上调。因此,自体纤维环细胞虽有可能成为合适的修复纤维环的细胞来源[31],但因其数量过少,仅占组织容量的1%,在实验条件下无法获取足够数量,严重限制了其应用前景。 干细胞可分化为纤维环细胞和髓核细胞,且来源丰富、获取和培养条件成熟,易应用于体外实验,作为修复纤维环的种子细胞具有较大优势,但如何使其定向分化仍是急需解决的问题。有研究者通过将纤维环细胞或髓核细胞与干细胞以一定比例共同培养并给予动态加压条件来促进其定向分化[32],获得了较好的效果。也有研究者尝试应用非接触共培养体系,在纤维环细胞的液体环境中培养骨髓干细胞,发现其可分化为纤维环样细胞[33],为这一问题的研究提供了新的思路。另外,动物实验表明同种异体干细胞对于纤维环和髓核的修复均有积极作用[34],但这一方法目前尚处实验阶段,且其应用必然面临一系列技术和伦理问题。 基于细胞外基质组成和细胞表型的不同,可将纤维环细胞大致分为外层和内层两部分,外层纤维环富含胶原纤维(主要为Ⅰ型胶原),细胞沿着胶原纤维取向分布,拉伸呈梭形,细胞形态功能和成纤维细胞类似,主要分泌Ⅰ型胶原;内层纤维环与髓核交界,细胞形态和功能逐渐与髓核细胞相似,接近软骨细胞,主要分泌Ⅱ型胶原和蛋白多糖。从外层到内层,纤维环细胞数量不断减少[35-36],且Ⅰ型胶原含量不断降低,而Ⅱ型胶原和蛋白多糖的含量逐渐升高。内外层纤维环基质成分和表型的差异决定了两者在生物力学性质上的不同:外层纤维环主要发挥抗张力和剪切力的作用,而内层纤维环主要承担抗压力的作用。纤维环细胞是高度分化细胞,临床取材困难,目前多采用骨髓间充质干细胞作为种子细胞[37],利用其可多向分化的特点通过诱导使其向所需方向分化。 2.2.3 干细胞差异分化的调控 干细胞的分化主要包括形态改变、骨架重组、基因表达和细胞功能的改变,使细胞感应微环境并将信号传递到细胞核、激发一系列细胞响应是研究纤维环再生材料的关键。 如前所述,组织工程支架表面的拓扑结构与干细胞定向分化有密切关系,这一过程可能主要是通过非直接力传导机制完成[38-39]。黏着斑激酶可作为整合素信号转导通路的关键黏附分子,在这一过程中发挥了重要作用[13]。以往研究表明,黏着斑激酶可与多种下游分子结合,并使其激活,如丝裂原活化蛋白激酶[40],丝原活化蛋白激酶P38[41-43]、细胞外信号调节蛋白激酶1/2、C6N末端激酶等[44-45]。由此可推断,支架的局部形貌可调控干细胞黏着斑激酶的表达,进而激活或者抑制其下游丝裂原活化蛋白激酶、细胞外信号调节蛋白激酶等信号通路,从而影响干细胞分化。尽管已经发现以上研究结果,支架表面拓扑结构调节干细胞分化的具体机制仍有待研究,各种信号通路及非编码RNA在其中发挥的作用也有待明确。 2.2.4 细胞因子的参与和调控 与维持细胞表型和诱导细胞分化相关的因子包括转化生长因子、生长分化因子等,其作用包括诱导细胞向椎间盘细胞分化[46]、同时降低间盘退变带来的损害等,因此被认为有助于椎间盘修复或再生[47-49]。另外,也有研究发现,转化生长因子β1和生长分化因子5可以协同促进干细胞向髓核细胞分化,血清、转化生长因子β3均有助于纤维环细胞生长[50]。早期的有研究发现,动态加载的压力有助于调节细胞行为[51]。通过引入趋化因子吸引周围纤维环细胞的思路目前尚处于研究阶段,即使是某些已被证实具有趋化作用的细胞因子,与凝胶等材料结合后也并未表现出对纤维环细胞再生的支持作用[52],要想借助趋化作用,仍需继续寻找趋化作用更强的细胞因子。 "
| [1]Carragee EJ, Spinnickie AO, Alamin TF, et al. A prospective controlled study of limited versus subtotal posterior discectomy: short-term outcomes in patients with herniated lumbar intervertebral discs and large posterior anular defect. Spine (Phila Pa 1976). 2006;31(6): 653-657.[2]McGirt MJ, Eustacchio S, Varga P, et al. A prospective cohort study of close interval computed tomography and magnetic resonance imaging after primary lumbar discectomy: factors associated with recurrent disc herniation and disc height loss. Spine (Phila Pa 1976). 2009;34(19): 2044-2051. [3]Bron JL, Helder MN, Meisel HJ, et al. Repair, regenerative and supportive therapies of the annulus fibrosus: achievements and challenges. Eur Spine J. 2009;18(3):301-313. [4]Melrose J, Smith SM, Little CB, et al. Recent advances in annular pathobiology provide insights into rim-lesion mediated intervertebral disc degeneration and potential new approaches to annular repair strategies. Eur Spine J. 2008;17(9):1131-1148. [5]Bron JL, Helder MN, Meisel HJ, et al. Repair, regenerative and supportive therapies of the annulus fibrosus: achievements and challenges. Eur Spine J. 2009;18(3):301-313. [6]Wilke HJ, Ressel L, Heuer F, et al. Can prevention of a reherniation be investigated? Establishment of a herniation model and experiments with an anular closure device. Spine (Phila Pa 1976). 2013;38(10): E587-593.[7]Parker SL, Grahovac G, Vukas D, et al. Effect of an Annular Closure Device (Barricaid) on Same-Level Recurrent Disk Herniation and Disk Height Loss After Primary Lumbar Discectomy: Two-year Results of a Multicenter Prospective Cohort Study. Clin Spine Surg. 2016;29(10): 454-460.[8]Bailey A, Araghi A, Blumenthal S, et al. Prospective, multicenter, randomized, controlled study of anular repair in lumbar discectomy: two-year follow-up. Spine (Phila Pa 1976). 2013;38(14):1161-1169. [9]Suh BG, Uh JH, Park SH, et al. Repair using conventional implant for ruptured annulus fibrosus after lumbar discectomy: surgical technique and case series. Asian Spine J. 2015;9(1):14-21.[10]Grunert P, Borde BH, Hudson KD, et al. Annular repair using high-density collagen gel: a rat-tail in vivo model. Spine (Phila Pa 1976). 2014;39(3):198-206. [11]Moriguchi Y, Borde B, Berlin C, et al. In vivo annular repair using high-density collagen gel seeded with annulus fibrosus cells. Acta Biomater. 2018;79:230-238. [12]Bron JL, Helder MN, Meisel HJ, et al. Repair, regenerative and supportive therapies of the annulus fibrosus: achievements and challenges. Eur Spine J. 2009;18(3):301-313.[13]Pirvu T, Blanquer SB, Benneker LM, et al. A combined biomaterial and cellular approach for annulus fibrosus rupture repair. Biomaterials. 2015;42:11-19. [14]Yao H, Justiz MA, Flagler D, et al. Effects of swelling pressure and hydraulic permeability on dynamic compressive behavior of lumbar annulus fibrosus. Ann Biomed Eng. 2002;30(10):1234-1241.[15]Schollum ML, Robertson PA, Broom ND. ISSLS prize winner: microstructure and mechanical disruption of the lumbar disc annulus: part I: a microscopic investigation of the translamellar bridging network. Spine (Phila Pa 1976). 2008;33(25):2702-2710. [16]Nerurkar NL, Elliott DM, Mauck RL. Mechanical design criteria for intervertebral disc tissue engineering. J Biomech. 2010;43(6): 1017-1030.[17]Bhardwaj N, Kundu SC. Electrospinning: a fascinating fiber fabrication technique. Biotechnol Adv. 2010;28(3):325-347. [18]Bhattacharjee M, Miot S, Gorecka A, et al. Oriented lamellar silk fibrous scaffolds to drive cartilage matrix orientation: towards annulus fibrosus tissue engineering. Acta Biomater. 2012;8(9):3313-3325. [19]Park SH, Gil ES, Cho H, et al. Intervertebral disk tissue engineering using biphasic silk composite scaffolds. Tissue Eng Part A. 2012; 18(5-6):447-458. [20]Shao X, Hunter CJ. Developing an alginate/chitosan hybrid fiber scaffold for annulus fibrosus cells. J Biomed Mater Res A. 2007; 82(3):701-710.[21]Yang L, Kandel RA, Chang G, et al. Polar surface chemistry of nanofibrous polyurethane scaffold affects annulus fibrosus cell attachment and early matrix accumulation. J Biomed Mater Res A. 2009;91(4):1089-1099. [22]Yeganegi M, Kandel RA, Santerre JP. Characterization of a biodegradable electrospun polyurethane nanofiber scaffold: Mechanical properties and cytotoxicity. Acta Biomater. 2010;6(10): 3847-3855. [23]Nerurkar NL, Mauck RL, Elliott DM. ISSLS prize winner: integrating theoretical and experimental methods for functional tissue engineering of the annulus fibrosus. Spine (Phila Pa 1976). 2008;33(25): 2691-2701.[24]Nerurkar NL, Baker BM, Sen S, et al. Nanofibrous biologic laminates replicate the form and function of the annulus fibrosus. Nat Mater. 2009;8(12):986-992. [25]Baker BM, Handorf AM, Ionescu LC, et al. New directions in nanofibrous scaffolds for soft tissue engineering and regeneration. Expert Rev Med Devices. 2009;6(5):515-532. [26]Xu Y, Wu J, Wang H, et al. Fabrication of electrospun poly(L-lactide-co-ε-caprolactone)/collagen nanoyarn network as a novel, three-dimensional, macroporous, aligned scaffold for tendon tissue engineering. Tissue Eng Part C Methods. 2013;19(12):925-936.[27]Guillaume O, Naqvi SM, Lennon K, et al. Enhancing cell migration in shape-memory alginate-collagen composite scaffolds: In vitro and ex vivo assessment for intervertebral disc repair. J Biomater Appl. 2015;29(9):1230-1246. [28]Pereira DR, Silva-Correia J, Oliveira JM, et al. Nanocellulose reinforced gellan-gum hydrogels as potential biological substitutes for annulus fibrosus tissue regeneration. Nanomedicine. 2018;14(3): 897-908. [29]Li WJ, Jiang YJ, Tuan RS. Chondrocyte phenotype in engineered fibrous matrix is regulated by fiber size. Tissue Eng. 2006;12(7): 1775-1785.[30]Henriksson H, Thornemo M, Karlsson C, et al. Identification of cell proliferation zones, progenitor cells and a potential stem cell niche in the intervertebral disc region: a study in four species. Spine (Phila Pa 1976). 2009;34(21):2278-2287.[31]Hondke S, Cabraja M, Krüger JP, et al. Proliferation, Migration, and ECM Formation Potential of Human Annulus Fibrosus Cells Is Independent of Degeneration Status. Cartilage. 2018. doi: 10.1177/1947603518764265. [Epub ahead of print][32]Tsai TL, Nelson BC, Anderson PA, et al. Intervertebral disc and stem cells cocultured in biomimetic extracellular matrix stimulated by cyclic compression in perfusion bioreactor. Spine J. 2014;14(9):2127-2140. [33]孟格栋,吴一民,李树文,等.非接触共培养条件下山羊BMSCs向纤维环样细胞诱导分化的研究[J].中国矫形外科杂志, 2017,25(2):145-151.[34]Freeman BJ, Kuliwaba JS, Jones CF, et al. Allogeneic Mesenchymal Precursor Cells Promote Healing in Postero-lateral Annular Lesions and Improve Indices of Lumbar Intervertebral Disc Degeneration in an Ovine Model. Spine (Phila Pa 1976). 2016;41(17):1331-1339. [35]Li J, Liu C, Guo Q, et al. Regional variations in the cellular, biochemical, and biomechanical characteristics of rabbit annulus fibrosus. PLoS One. 2014;9(3):e91799.[36]Chou AI, Bansal A, Miller GJ, et al. The effect of serial monolayer passaging on the collagen expression profile of outer and inner anulus fibrosus cells. Spine (Phila Pa 1976). 2006;31(17):1875-1881.[37]Steck E, Bertram H, Abel R, et al. Induction of intervertebral disc-like cells from adult mesenchymal stem cells. Stem Cells. 2005;23(3): 403-411.[38]Davies JE, Ajami E, Moineddin R, et al. The roles of different scale ranges of surface implant topography on the stability of the bone/implant interface. Biomaterials. 2013;34(14):3535-3546. [39]Seo CH, Jeong H, Feng Y, et al. Micropit surfaces designed for accelerating osteogenic differentiation of murine mesenchymal stem cells via enhancing focal adhesion and actin polymerization. Biomaterials. 2014;35(7):2245-2252. [40]Zhang Y, Pizzute T, Pei M. A review of crosstalk between MAPK and Wnt signals and its impact on cartilage regeneration. Cell Tissue Res. 2014;358(3):633-649. [41]Li J, Zhao Z, Liu J, et al. MEK/ERK and p38 MAPK regulate chondrogenesis of rat bone marrow mesenchymal stem cells through delicate interaction with TGF-beta1/Smads pathway. Cell Prolif. 2010;43(4):333-343. [42]Li J, Zhao Z, Yang J, et al. p38 MAPK mediated in compressive stress-induced chondrogenesis of rat bone marrow MSCs in 3D alginate scaffolds. J Cell Physiol. 2009;221(3):609-617.[43]Stanton LA, Sabari S, Sampaio AV, et al. p38 MAP kinase signalling is required for hypertrophic chondrocyte differentiation. Biochem J. 2004;378(Pt 1):53-62.[44]Tuli R, Tuli S, Nandi S, et al. Transforming growth factor-beta-mediated chondrogenesis of human mesenchymal progenitor cells involves N-cadherin and mitogen-activated protein kinase and Wnt signaling cross-talk. J Biol Chem. 2003;278(42):41227-41236. [45]Oh CD, Chang SH, Yoon YM, et al. Opposing role of mitogen-activated protein kinase subtypes, erk-1/2 and p38, in the regulation of chondrogenesis of mesenchymes. J Biol Chem. 2000;275(8):5613-5619.[46]Clarke LE, McConnell JC, Sherratt MJ, et al. Growth differentiation factor 6 and transforming growth factor-beta differentially mediate mesenchymal stem cell differentiation, composition, and micromechanical properties of nucleus pulposus constructs. Arthritis Res Ther. 2014;16(2):R67. [47]Liu W, Zhang Y, Feng X, et al. Inhibition of microRNA-34a prevents IL-1β-induced extracellular matrix degradation in nucleus pulposus by increasing GDF5 expression. Exp Biol Med (Maywood). 2016; 241(17): 1924-1932. [48]Malonzo C, Chan SC, Kabiri A, et al. A papain-induced disc degeneration model for the assessment of thermo-reversible hydrogel-cells therapeutic approach. J Tissue Eng Regen Med. 2015; 9(12):E167-176.[49]Feng C, Liu H, Yang Y, et al. Growth and differentiation factor-5 contributes to the structural and functional maintenance of the intervertebral disc. Cell Physiol Biochem. 2015;35(1):1-16. [50]Hegewald AA, Zouhair S, Endres M, et al. Towards biological anulus repair: TGF-β3, FGF-2 and human serum support matrix formation by human anulus fibrosus cells. Tissue Cell. 2013;45(1):68-76. [51]Huang H, Kamm RD, Lee RT. Cell mechanics and mechanotransduction: pathways, probes, and physiology. Am J Physiol Cell Physiol. 2004;287(1):C1-11.[52]Zhou Z, Zeiter S, Schmid T, et al. Effect of the CCL5-Releasing Fibrin Gel for Intervertebral Disc Regeneration. Cartilage. 2018. doi: 10.1177/1947603518764263. [Epub ahead of print] |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Gao Yan, Zhao Licong, Zhao Hongzeng, Zhu Yuanyuan, Li Jie, Sang Deen. Alteration of low frequency fluctuation amplitude at brain-resting state in patients with chronic discogenic low back pain [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1160-1165. |
| [5] | Liu Zhichao, Zhang Fan, Sun Qi, Kang Xiaole, Yuan Qiaomei, Liu Genzhe, Chen Jiang. Morphology and activity of human nucleus pulposus cells under different hydrostatic pressures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1172-1176. |
| [6] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [7] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [8] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [9] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [10] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [11] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [12] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [13] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [14] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [15] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||