Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (15): 2414-2420.doi: 10.3969/j.issn.2095-4344.0736
Previous Articles Next Articles
Management of bone defects in total knee arthroplasty
Liang Hao-dong1, Pan Jian-ke2, Xie Hui1, Huang He-tao1, Han Yan-hong1, Lin Jiong-tong1, Liu Jun2
- 1The Second School of Clinical Medicine, Guangzhou University of Chinese Medicine, Guangzhou 510405, Guangdong Province, China; 2Department of Orthopedics, Guangdong Provincial Hospital of Chinese Medicine, Guangzhou 510120, Guangdong Province, China
-
Online:2018-05-28Published:2018-05-28 -
Contact:Liu Jun, Chief physician, Professor, Doctoral supervisor, Department of Orthopedics, Guangdong Provincial Hospital of Chinese Medicine, Guangzhou 510120, Guangdong Province, China -
About author:Liang Hao-dong, Master candidate, The Second School of Clinical Medicine, Guangzhou University of Chinese Medicine, Guangzhou 510405, Guangdong Province, China -
Supported by:the National Natural Science Foundation of China, No. 81473698; the Doctoral Fund of the Ministry of Education of China, No. 20124425110004; the Standardization Project of Traditional Chinese Medicine of the State Administration of Traditional Chinese Medicine of China, No. SATCM-2015-BZ115, SATCM-2015-BZ173; the Science and Technology Planning Project of Guangdong Province, No. 2011B031700027; the Project of Guangdong Provincial Department of Finance, No. [2011]157; the Science and Technology Research Project of Guangdong Provincial Hospital of Chinese Medicine, No. YK2013B2N19, YN2015MS15.
CLC Number:
Cite this article
Liang Hao-dong, Pan Jian-ke, Xie Hui, Huang He-tao, Han Yan-hong, Lin Jiong-tong, Liu Jun. Management of bone defects in total knee arthroplasty[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(15): 2414-2420.
share this article
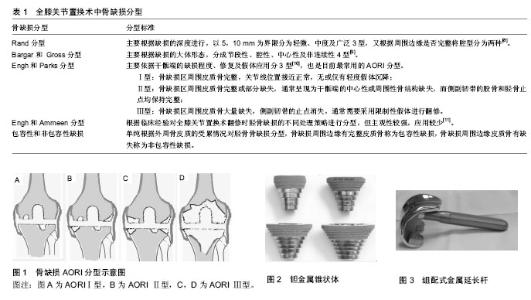
2.1 术前评估 术前评估对于全膝关节置换的成功与否起关键作用。术前评估主要症状及产生的原因、是否存在感染、手术禁忌等,了解患者的病史,进行详细的体格检查,排除其他部位病变所致膝关节疼痛以及神经肌肉疾病;完善血常规、C-反应蛋白、红细胞沉降率等辅助检查初步排查机体感染;影像学的评估主要为X射线及CT扫描,通过影像学结果了解缺损的部位、范围、程度,初步制定手术需要的工具以及手术方案。X射线评估与手术中情况的对比,对股骨及胫骨的缺损都能较准确的评估[6],但X射线检查往往容易低估骨缺损的大小,在CT上显示的骨溶解只有17%可以在平片上观察 到[7]。术中的实际情况可能更严重,因此必要时需进行CT扫描+三维重建更加准确的评估骨缺损,以更好的为骨缺损处理做准备。 2.2 骨缺损分型 目前针对不同类型骨的缺损分型方法较多,分型主要是用于全膝关节置换翻修术。因初次全膝关节置换中骨缺损的种类较少,程度亦通常较全膝关节置换翻修轻,所以初次全膝关节置换完全可以按照全膝关节置换翻修的方法进行分型,见表1。 全膝关节置换术中骨缺损分型,主要包括Rand分型、Bargar和 Gross分型、Engh 和Parks分型。Rand分型主要根据缺损的深度进行,以5,10 mm为界限分为轻微、中度及广泛3型,又根据周围边缘是否完整将腔型分为两种[8]。Bargar和Gross分型主要根据缺损的大体形态,分成节段性、腔性、中心性及非连续性4型[9]。Engh和Parks分类的主要依据是干骺端的缺损程度、修复及假体应用分3型[10],也是目前最常用的AORI (Anderson Orthopaedic Research Institute)分型(图1)。Ⅰ型:骨缺损区周围皮质骨完整,关节线位置接近正常,无或仅有轻度假体沉降;Ⅱ型:骨缺损区周围皮质骨完整或部分缺失,通常呈现为干骺端的中心性或周围性骨结构缺失,而侧副韧带的股骨和胫骨止点均保持完整;Ⅲ型:骨缺损区周围皮质骨大量缺失,侧副韧带的止点消失,通常需要采用限制性假体进行翻修。Engh和Ammeen[11]根据临床经验对全膝关节置换术翻修时胫骨缺损的不同处理策略进行分型,但主观性较强,应用较少。亦有学者单纯根据外周骨皮质的受累情况对胫骨骨缺损分为包容性和非包容性缺损。骨缺损周围边缘有完整皮质骨称为包容性缺损,骨缺损周围边缘皮质骨有缺失称为非包容性缺损。 2.3 骨缺损的处理策略 骨缺损的分型方法决定了修复的策略,目前国内外学者更倾向于根据AORI分型决定骨缺损的处理策略。AORI Ⅰ型骨缺损,可选择自体骨植骨、增加截骨量、偏移假体位置或骨水泥联合螺钉等处理;Ⅱ型骨缺损,多可以用Ⅰ型的策略,根据患者年龄、经济情况合并使用结构性植骨,使用金属假体垫块,或者金属袖套、锥形补块。对于Ⅲ型,因缺损骨质多,可考虑异体骨植骨、金属垫块,经济允许者采用结构性钽金属块修复。 增加截骨量用于缺损深度较小的包容性骨缺损,通过加深截骨的平面去除缺损,此法的缺点是降低宿主股的支持强度,需选择小号假体。假体移位的原理是避开缺损区域,把假体装在骨质情况良好的位置,但假体的位置改变将影响韧带力学性能,引起韧带激惹,且需用小号假体。目前此两种策略的相关研究较少,故本综述未进行论述。 2.3.1 初次膝关节置换 自体骨移植:自体骨移植术是对于较小的骨缺损(通常<3 mm)且骨量充足的情况下,最常用的修复方法之一,具有取材方便、成本低、无免疫排斥反应、术后易生长等优点。骨质多来源于术中的截骨,少部分为自体髂骨。Sugita等[12]对38例胫骨平台内侧骨缺损取自体骨修补骨缺损,术后随访平均65个月,移植物均与宿主骨愈合。近期亦报道了44例初次全膝关节置换,胫骨内侧平台骨缺损深度大于10 mm行自体颗粒骨移植,平均随访约6年,仅6例发现植骨与胫骨出现明显界限,但无扩大趋势[13]。马喜洪等[14]对50例采用自体骨打压移植的方法修复胫骨平台骨缺损,术后影像学检查显示胫骨假体下方骨质缺损征象消失,假体位置良好。此法缺点是植骨的骨量受到较大的限制,且术后须避免过早负重。 骨水泥+螺钉:目前单纯应用骨水泥修复骨缺损尚无统一标准,一般认为腔隙型的包容性骨缺损可考虑此方法。王金良等[15]认为对于截骨后胫骨缺损深度 <5 mm,且<胫骨平台面积50%者,可选择只采用骨水泥填充。当骨质缺损较大且为非包容性时,需要联合应用加强螺丝钉以增强生物学强度,从关节线和骨水泥界面分散负荷,增强骨水泥充填的强度。Kharbanda等[16]对54例胫骨平台骨缺损采用骨水泥加螺钉修复,并进行了5-10年的随访,术后关节功能恢复满意。 应用骨水泥+螺钉时,术中需完全显露骨缺损处以充分评估骨缺损面积和形态,当缺损为楔形时,需要把缺损修整为阶梯型后以减少应力。清除缺损处硬化骨并钻孔后间隔5-10 mm拧入螺钉(通常5 mm或6.5 mm),放置假体试模,调整螺钉高度,要求低于截骨平面以使螺钉头与胫骨假体无直接接触,填充骨水泥并加压填充修复缺损。骨水泥结合螺钉用于处理骨缺损是较常见的策略,具有较高效价比。与植骨相比,可早期下地完全负重活动,且可避免植骨不愈合及骨吸收的风险。对于胫骨平台骨缺损,中短期效果可靠。此方法也存在着一定的不足,骨水泥弹性模量较人体组织低,抗应力作用较差,不利于维持骨量。骨水泥亦具有潜在的骨断裂、松动,以及固化时体积缩小导致假体下沉等缺点。 同种异体骨移植:年轻患者可能面临多次翻修,对骨量的保持要求更高,植骨是较好的办法。同种异体骨包括结构性骨与颗粒骨。同种异体颗粒骨经过简单的塑形即可用于骨囊肿和小面积包容性骨缺损的填充。异体结构性植骨则常用于AORI ⅡB和Ⅲ型骨缺损,具有较易恢复骨量、相对节约手术时间等优点,通过修整后直接植入宿主骨,是修复骨缺损的常用材料[17]。胡忠洲等[18]对16例初次全膝关节置换中Rand C型骨缺损患者使用自体骨与异体骨混合植骨,可为胫骨假体提供良好的初始稳定性。但此法存在着较多的缺点,早期不能对假体形成较好的支持,延长术后负重的时间;存在着异体骨和宿主骨愈合不佳风险,导致假体不稳;植骨吸收、排异的发生以及疾病的传染也是潜在风险。因此,对于骨愈合不佳风险较高的老年患者,不建议使用异体结构 骨[19],一般在自体骨骨量不足的情况下采用,经济允许或可用结构性金属替代[20]。 金属垫块:金属垫块是处理初次全膝关节置换中等大小骨缺损( AORI Ⅰ,Ⅱ型) 、重建关节线的一种有效方法,其具有良好的负荷传导能力,可以对假体提供稳定的支撑[21],对抗扭力的性能也优于骨水泥[22]。安装金属垫块需先在骨缺损侧矩形截骨,使骨缺损处呈阶梯状并显露新鲜骨质。然后用螺钉组合金属垫块试模与胫骨假体试模,安放于胫骨平台。张志强等[23]在初次全膝关节置换术中运用组合式金属垫块处理胫骨AORI Ⅱ型骨缺损10例,平均随访18个月,取得较好的临床效果。Lee等[24]对46例伴胫骨平台骨缺损使用矩形金属垫块修补后行初次全膝关节置换,平均随访78.6个月,结果均为优良。Tsukada等[25]报道初次全膝关节置换术中因内侧胫骨平台缺损使用金属垫块修补,共33例,对比无骨缺损行全膝关节置换132例,随访时间3-6年,术后膝关节功能评分及假体的生存率差异无显著性意义。 金属垫块的缺点:主要因其与骨的弹性模量存在差别,可引起应力遮挡,导致骨量丢失[26-27];若金属垫块的位置偏外,可对侧副韧带造成慢性切割。且较多的病例中出现影像学上非进展性透亮线,金属垫块存在微动的可能,翻修的难度也较大。 2.3.2 膝关节置换翻修 骨水泥+螺钉:骨水泥联合螺钉技术多用于全膝关节置换翻修术,修复轻-中度骨缺损疗效较为肯定,15年的随访时间无失败的证据[28]。Ritter[29]对57例胫骨骨缺损平均深度为9 mm,25%出现骨-水泥界面非进展性透亮线,但无假体失败者。7年随访报道显示,骨-水泥界面或骨水泥-假体界面出现的透亮线均未增宽[30]。研究推测,透亮线或许在手术时就已经出现,原因可能是骨水泥与宿主的坏死骨粘合不良[31]。 金属垫块:在全膝关节置换翻修术中,骨缺损的类型多、程度较严重,金属垫块的利用也较多。Sculco等[32]认为金属垫块最多可代替5-25 mm 以内的骨缺损;对于较大范围的骨缺损,矩形金属垫块比楔形金属垫块能更稳定地分散应力[33]。Heesterbeek等[34]报道32例Ⅰ、Ⅱ型骨缺损患者在2年随访的随机对照试验中,比较单纯骨水泥与合并打压植骨技术两种方法对术后假体微动及临床效果,2组关节功能差异无显著性意义,尽管都有>1 mm或>1°微动,但未发现假体松动的证据。Kosse等[35]更长随访(6.5年)发现2组患者的临床疗效及假体松动情况差异无显著性意义,2例在术后1年内出现了股骨侧假体的进展性移位。 Gofton等[36]强调,在严重骨缺损的全膝关节置换翻修术中,仅使用金属垫块不能满足重建需求,但金属垫块可减少全膝关节置换翻修术中异体骨移植的需求,且可提供稳定及功能良好的关节。 结构性同种异体骨重建:异体结构骨主要用于AORI ⅡB或Ⅲ型骨缺损的重建,多因缺损面积过大而无法使用假体垫块填充或经济较困难者[37],此种技术用于不同人种疗效接[38]。Clatworthy等[39]对52例全膝关节置换翻修伴随重度非包容性的胫骨平台骨缺损行同种异体骨假体复合物移植术,至少5年的随访,75%术后HSS评分提高20分以上,移植骨10年生存率为72%,疗效肯定。Backstein等[40]对61例全膝关节置换翻修术伴胫骨、股骨的骨缺损回顾,平均随访5.4年,13例失败,3例出现骨吸收。有学者利用冰冻异体股骨头,对全膝关节置换翻修术患者伴严重骨缺损的回顾研究,平均随访107个月,平均骨愈合时间为7个月,未发现骨塌陷、疾病的传染、应力性骨折等。 钽金属袖套与锥形块:钽金属具有出色的生物兼容性和安全性[41],定制结构可用于多种类型包括胫骨、及股骨的骨缺损,适用于股骨和胫骨侧的包容性-腔隙性骨缺损和腔隙性-节段性干骺端骨缺损(图2)。金属采用多孔表面设计、非骨水泥固定,具有很好的弹性模量,与人的骨质接近,可以较好地传导压力。Girerd等[42]对于51例2个中心的回顾性研究,随访2年,证实钽金属锥形块在膝关节翻修置换术中可提供持久稳定的结构重建,疗效可靠。 主要缺点包括:价格昂贵,假体需要定制;表面设计具有一定的细菌黏附作用,具有感染的风险。Brown等[43]利用钽金属骨小梁锥形块对行初次及翻修全膝关节置换术中的骨缺损进行修补,缺损部位包括胫骨平台、股骨及胫骨股骨同时缺损,共83例,随访时间平均为40个月,结果10例因感染、假体周围骨折及假体无菌性松动需行再次手术;结构特殊,再次翻修时难以取出假体。但目前研究提示中期疗效稳定,翻修率较低。Fedorka等[44]在回顾79例金属袖套修补缺损,平均6年随访,由于假体松动而翻修者占6.8%。Kamath等[45]报道63例全膝关节置换翻修患者随访5-9年,95.4%的病例无需翻修。有研究报道了157例股骨端锥形钽金属修补AORI Ⅲ型骨缺损进行5年的随访,6例无菌性松动,生存率为96%。 组配式金属延长杆:延长杆主要用于传导应力,增加假体的稳定性(图3)。延长杆的类型根据放置的部位分为胫骨侧与股骨侧,又根据材料分为骨水泥型、混合型及非骨水泥型,目前相关研究提示骨水泥型与非骨水泥型的延长杆的效果对比差异无显著性意义[34,46]。使用金属垫块时,通常需要使用延长杆。全膝关节置换翻修术中胫骨或股骨骨缺损,均应使用延长杆以加强假体稳定性。Nelson等[47]统计了130例全膝关节置换翻修术,其中67例使用股骨侧延长杆,63例不使用,术后4年随访发现延长杆组的手术失败率明显降低(9% vs. 44%)。Lachiewicz等[48]报道了58例AORIⅡB和Ⅲ型的骨缺损,全膝关节置换翻修术使用30 mm的胫骨侧延长杆者,平 均随访5年无翻修或假体松动。Nadorf等[49]体外实验提示全膝关节置换翻修术中使用延长杆可有效减少假体的移位,越长效果越显著。 不同处理策略的比较:目前国内外尚无对于膝关节置换伴骨缺损不同重建策略的随机对照试验,因此不同缺损的最优选择方案尚未明确。Berend等[50]对骨水泥联合螺钉技术重建胫骨骨缺损5-20 mm的初次全膝关节置换患者进行20年的长期随访,对比无骨缺损行全膝关节置换患者无显著功能差异,且较使用金属垫块的性价比高。Beckmann等[51]对AORIⅡ型、Ⅲ型骨缺损,对比利用异体骨移植(476例)及钽金属锥形块(223例)修复骨缺损的全膝关节置换翻修术的临床证据(包括单臂试验、回顾性研究)进行系统评价,中期随访,金属锥形块具有更低的无菌松动率,但远期疗效尚难以定论。 2.3.3 髌骨的骨缺损 髌骨的缺损常见于全膝关节置换翻修术,最常见原因为感染。髌骨与人工关节满意的解剖关系对关节置换的临床效果也起着重要的影响。髌骨骨缺损的处理方式主要包括聚乙烯联合钢丝髌骨表面置入、双凸面髌骨假体表面置换、骨移植、海鸥型截骨、钽金属置换等。目前对于髌骨骨缺损重建的方式仍存在着较大的争议,亦缺少相关的分型[52-56]。 当缺损涉及髌骨的中心及边缘骨皮质时,可以考虑使用圆顶型的全聚乙烯双凸面髌骨假体,Maheshwer等[57]报道了20例全膝关节置换翻修术中行全聚乙烯双凸面髌骨假体置入重建髌骨缺损,平均髌骨厚度为 6.5 mm,平均随访35个月,未出现髌骨骨折,KSS评分及关节活动度明显改善,仅3例出现屈曲时髌前疼痛及爬楼梯困难。 当髌骨的厚度<8 mm时,镶嵌式假体结合钢丝可用于髌骨骨皮质边缘不完整,优势在于能保留伸膝装置,髌股关节与初次全膝关节置换接近,但骨质太薄时,骨折的风险较高。Seo等[58]利用聚乙烯镶嵌式假体结合钢丝固定,对9例全膝关节置换翻修术严重髌骨缺损,髌骨平均厚度5.6 mm进行重建,随访10.3个月,1例在术后1周发生髌骨骨折,余功能良好。海鸥式截骨是指通过对髌骨骨缺损仅剩的骨壳截骨修整,转变成为海鸥型(Ⅴ型)髌骨,维持髌骨活动轨迹处于股骨滑车中心位置。Gililland等[59]对行海鸥式截骨者进行2年随访,9/10有骨愈合的影像学证据,患者的膝关节功能、关节活动度,生活质量均得到改善。 多孔钽金属髌骨假体置换的优势在于无需骨髌骨边缘皮质的支持,其设计依赖于软组织及骨质向假体内生长,此技术可用于严重的髌骨骨缺损。Nasser等[60]报道11例平均随访32个月,其中10例发现骨长入证据。部分学者认为,在全膝关节置换翻修术中,若髌骨在初次全膝关节置换时未进行置换,且骨缺损小、未影响髌骨运动轨迹同时骨质量较好时,不应行髌骨置换;若初次全膝关节置换已行置换,但无松动证据、无感染、髌骨假体与翻修假体匹配时,则不应该再次行髌骨置换[61]。 "
| [1] Glyn-Jones S, Palmer AJ, Agricola R, et al. Osteoarthritis. Lancet. 2015;386(9991):376-387.[2] Kurtz S, Ong K, Lau E, et al. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780-785.[3] Krackow KA, Mihalko WM. The effects of severe femoral bone loss on the flexion extension joint space in revision total knee arthroplasty: a cadaveric analysis and clinical consequences. Orthopedics. 2001;24(2):121-126.[4] Panegrossi G, Ceretti M, Papalia M, et al. Bone loss management in total knee revision surgery. Int Orthop. 2014;38(2):419-427.[5] Harris AI, Poddar S, Gitelis S, et al. Arthroplasty with a composite of an allograft and a prosthesis for knees with severe deficiency of bone. J Bone Joint Surg Am. 1995;77(3):373-386.[6] Mulhall KJ, Ghomrawi HM, Engh GA, et al. Radiographic prediction of intraoperative bone loss in knee arthroplasty revision. Clin Orthop Relat Res. 2006;446:51-58.[7] Reish TG, Clarke HD, Scuderi GR, et al. Use of multi-detector computed tomography for the detection of periprosthetic osteolysis in total knee arthroplasty. J Knee Surg.2006;19(4): 259-264.[8] Rand JA. Bone deficiency in total knee arthroplasty. Use of metal wedge augmentation. Clin Orthop Relat Res. 1991;(271):63-71.[9] Bargar WL, Murzic WJ, Taylor JK, et al. Management of bone loss in revision total hip arthroplasty using custom cementless femoral components. J Arthroplasty. 1993;8(3):245-252.[10] Engh GA, Parks NL. The management of bone defects in revision total knee arthroplasty. Instr Course Lect. 1997;46: 227-236.[11] Engh GA, Ammeen DJ. Classification and preoperative radiographic evaluation: knee. Orthop Clin North Am. 1998;29(2): 205-217.[12] Sugita T, Aizawa T, Sasaki A, et al. Autologous morselised bone grafting for medial tibial defects in total knee arthroplasty. J Orthop Surg (Hong Kong). 2015;23(2):185-189.[13] Sugita T, Aizawa T, Miyatake N, et al. Preliminary results of managing large medial tibial defects in primary total knee arthroplasty: autogenous morcellised bone graft. Int Orthop. 2017;41(5):931-937.[14] 马喜洪,马建军,邵芳,等.自体打压植骨法在胫骨平台骨缺损膝关节置换术中的应用[J].临床骨科杂志, 2013,16(1):18-19.[15] 王金良,孙京涛,李玲,等.骨水泥联合螺钉修复全膝关节置换术中胫骨平台内侧骨缺损[J].中医正骨, 2015,27(12):55-56.[16] Kharbanda Y, Sharma M. Autograft reconstructions for bone defects in primary total knee replacement in severe varus knees. Indian J Orthop. 2014;48(3):313-318.[17] 左健,康建敏,潘乐. 同种异体骨移植用于骨缺损修复的应用现状[J]. 中国组织工程研究, 2012,16(18):3395-3398.[18] 胡忠洲,王韶进,刘文广,等. 自体混合植骨治疗初次全膝关节置换术中RandC型骨缺损的疗效[J]. 山东大学学报(医学版), 2012,50(11): 96-99.[19] Hoell S, Sieweke A, Gosheger G, et al. Eradication rates, risk factors, and implant selection in two-stage revision knee arthroplasty: a mid-term follow-up study. J Orthop Surg Res, 2016,11(1):93.[20] Vasso M, Beaufils P, Cerciello S, et al. Bone loss following knee arthroplasty: potential treatment options. Arch Orthop Trauma Surg, 2014,134(4):543-553.[21] Hockman DE, Ammeen D, Engh GA. Augments and allografts in revision total knee arthroplasty: usage and outcome using one modular revision prosthesis. J Arthroplasty.2005;20(1):35-41.[22] Frehill B, Crocombe AD, Agarwal Y, et al. Finite element assessment of block-augmented total knee arthroplasty. Comput Methods Biomech Biomed Engin. 2015;18(15):1726-1736.[23] 张志强,贾永社,李栋,等.髁限制性膝关节假体治疗严重膝内翻畸形合并胫骨平台内侧骨缺损[J].实用骨科杂志, 2012,18(10):894-897.[24] Lee JK, Choi CH. Management of tibial bone defects with metal augmentation in primary total knee replacement: a minimum five-year review. J Bone Joint Surg Br. 2011;93(11):1493-1496.[25] Tsukada S, Wakui M, Matsueda M. Metal block augmentation for bone defects of the medial tibia during primary total knee arthroplasty. J Orthop Surg Res.2013;8:36.[26] Patel JV, Masonis JL, Guerin J, et al. The fate of augments to treat type-2 bone defects in revision knee arthroplasty. J Bone Joint Surg Br. 2004;86(2):195-199.[27] Stuchin SA. Allografting in total knee replacement arthroplasty. Semin Arthroplasty.1993;4(2):117-122.[28] Brooks PJ, Walker PS, Scott RD. Tibial component fixation in deficient tibial bone stock. Clin Orthop Relat Res. 1984;(184): 302-308.[29] Ritter MA. Screw and cement fixation of large defects in total knee arthroplasty. J Arthroplasty.1986;1(2):125-129.[30] Ritter MA, Harty LD. Medial screws and cement: a possible mechanical augmentation in total knee arthroplasty. J Arthroplasty. 2004;19(5):587-589.[31] Ritter MA, Harty LD, Davis KE, et al. Simultaneous bilateral, staged bilateral, and unilateral total knee arthroplasty. A survival analysis. J Bone Joint Surg Am. 2003;85-A(8):1532-1537.[32] Sculco PK, Abdel MP, Hanssen AD, et al. The management of bone loss in revision total knee arthroplasty: rebuild, reinforce, and augment. Bone Joint J. 2016;98-B(1 Suppl A):120-124.[33] Panni AS, Vasso M, Cerciello S. Modular augmentation in revision total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. 2013;21(12):2837-2843.[34] Heesterbeek PJ, Wymenga AB, van Hellemondt GG. No difference in implant micromotion between hybrid fixation and fully cemented revision total knee arthroplasty: a randomized controlled trial with radiostereometric analysis of patients with mild-to-moderate bone loss. J Bone Joint Surg Am. 2016;98(16): 1359-1369.[35] Kosse NM, van Hellemondt GG, Wymenga AB, et al. Comparable stability of cemented vs press-fit placed stems in revision total knee arthroplasty with mild to moderate bone loss: 6.5-year results from a randomized controlled trial with radiostereometric analysis. J Arthroplasty. 2017;32(1):197-201.[36] Gofton WT, Tsigaras H, Butler RA, et al. Revision total knee arthroplasty: fixation with modular stems. Clin Orthop Relat Res. 2002;(404):158-168.[37] 来志超,蒋超,葛怡宁,等.全膝关节置换翻修术的假体选择[J]. 中华骨与关节外科杂志, 2015,8(4):359-362.[38] Wang JW, Hsu CH, Huang CC, et al. Reconstruction using femoral head allograft in revision total knee replacement: an experience in Asian patients. Bone Joint J. 2013;95-B(5):643-648.[39] Clatworthy MG, Ballance J, Brick GW, et al. The use of structural allograft for uncontained defects in revision total knee arthroplasty. A minimum five-year review. J Bone Joint Surg Am. 2001;83-A(3): 404-411.[40] Backstein D, Safir O, Gross A. Management of bone loss: structural grafts in revision total knee arthroplasty. Clin Orthop Relat Res. 2006;446:104-112.[41] Black J. Biological performance of tantalum. Clin Mater. 1994; 16(3):167-173.[42] Girerd D, Parratte S, Lunebourg A, et al. Total knee arthroplasty revision with trabecular tantalum cones: Preliminary retrospective study of 51 patients from two centres with a minimal 2-year follow-up. Orthop Traumatol Surg Res. 2016;102(4):429-433.[43] Brown NM, Bell JA, Jung EK, et al. The use of trabecular metal cones in complex primary and revision total knee arthroplasty. J Arthroplasty. 2015;30(9 Suppl):90-93.[44] Fedorka CJ, Chen AF, Pagnotto MR, et al. Revision total knee arthroplasty with porous-coated metaphyseal sleeves provides radiographic ingrowth and stable fixation. Knee Surg Sports Traumatol Arthrosc. 2017.[45] Kamath AF, Lewallen DG, Hanssen AD. Porous tantalum metaphyseal cones for severe tibial bone loss in revision knee arthroplasty: a five to nine-year follow-up. J Bone Joint Surg Am. 2015;97(3):216-223.[46] van Hamersveld KT, Marang-van de Mheen PJ, Tsonaka R, et al. Fixation and clinical outcome of uncemented peri-apatite-coated versus cemented total knee arthroplasty : five-year follow-up of a randomised controlled trial using radiostereometric analysis (RSA).Bone Joint J. 2017;99-B(11):1467-1476.[47] Nelson CL, Vanushkina M, Irgit K, et al. Stemmed femoral implants show lower failure rates in revision total knee arthroplasty. Knee. 2015;22(5):429-434.[48] Lachiewicz PF, Soileau ES. A 30-mm cemented stem extension provides adequate fixation of the tibial component in revision knee arthroplasty. Clin Orthop Relat Res. 2015;473(1):185-189.[49] Nadorf J, Kinkel S, Gantz S, et al. Tibial revision knee arthroplasty with metaphyseal sleeves: The effect of stems on implant fixation and bone flexibility. PLoS One. 2017;12(5):e177285.[50] Berend ME, Ritter MA, Keating EM, et al. Use of screws and cement in primary TKA with up to 20 years follow-up. J Arthroplasty. 2014;29(6):1207-1210.[51] Beckmann NA, Mueller S, Gondan M, et al. Treatment of severe bone defects during revision total knee arthroplasty with structural allografts and porous metal cones-a systematic review. J Arthroplasty. 2015;30(2):249-253.[52] Garcia RM, Kraay MJ, Conroy-Smith PA, et al. Management of the deficient patella in revision total knee arthroplasty. Clin Orthop Relat Res. 2008;466(11):2790-2797.[53] Masri BA, Meek RM, Greidanus NV, et al. Effect of retaining a patellar prosthesis on pain, functional, and satisfaction outcomes after revision total knee arthroplasty. J Arthroplasty. 2006;21(8): 1169-1174.[54] Ries MD, Cabalo A, Bozic KJ, et al. Porous tantalum patellar augmentation: the importance of residual bone stock. Clin Orthop Relat Res. 2006;452:166-170.[55] Busfield BT, Ries MD. Whole patellar allograft for total knee arthroplasty after previous patellectomy. Clin Orthop Relat Res. 2006;450:145-149.[56] Rorabeck CH, Mehin R, Barrack RL. Patellar options in revision total knee arthroplasty. Clin Orthop Relat Res. 2003;(416):84-92.[57] Maheshwer CB, Mitchell E, Kraay M, et al. Revision of the patella with deficient bone using a biconvex component. Clin Orthop Relat Res. 2005;440:126-130.[58] Seo JG, Moon YW, Lim SJ, et al. Augmentation with transcortical wiring of an onlay-type prosthesis for a deficient patella during revision total knee arthroplasty. Clin Orthop Surg. 2012;4(2): 163-166.[59] Gililland JM, Swann P, Pelt CE, et al. What Is the role for patelloplasty with Gullwing osteotomy in revision TKA? Clin Orthop Relat Res. 2016;474(1):101-106.[60] Nasser S, Poggie RA. Revision and salvage patellar arthroplasty using a porous tantalum implant. J Arthroplasty. 2004;19(5): 562-572.[61] Qiu YY, Yan CH, Chiu KY, et al. Review article: bone defect classifications in revision total knee arthroplasty. J Orthop Surg (Hong Kong). 2011;19(2):238-243.[62] Luo W, Huang L, Liu H, et al. Customized knee prosthesis in treatment of giant cell tumors of the proximal tibia: application of 3-dimensional printing technology in surgical design. Med Sci Monit. 2017;23:1691-1700. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zhang Yu, Tian Shaoqi, Zeng Guobo, Hu Chuan. Risk factors for myocardial infarction following primary total joint arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1340-1345. |
| [3] | Li Dadi, Zhu Liang, Zheng Li, Zhao Fengchao. Correlation of total knee arthroplasty efficacy with satisfaction and personality characteristics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1346-1350. |
| [4] | Wei Wei, Li Jian, Huang Linhai, Lan Mindong, Lu Xianwei, Huang Shaodong. Factors affecting fall fear in the first movement of elderly patients after total knee or hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1351-1355. |
| [5] | Wang Jinjun, Deng Zengfa, Liu Kang, He Zhiyong, Yu Xinping, Liang Jianji, Li Chen, Guo Zhouyang. Hemostatic effect and safety of intravenous drip of tranexamic acid combined with topical application of cocktail containing tranexamic acid in total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1356-1361. |
| [6] | Xiao Guoqing, Liu Xuanze, Yan Yuhao, Zhong Xihong. Influencing factors of knee flexion limitation after total knee arthroplasty with posterior stabilized prostheses [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1362-1367. |
| [7] | Huang Zexiao, Yang Mei, Lin Shiwei, He Heyu. Correlation between the level of serum n-3 polyunsaturated fatty acids and quadriceps weakness in the early stage after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1375-1380. |
| [8] | Zhang Chong, Liu Zhiang, Yao Shuaihui, Gao Junsheng, Jiang Yan, Zhang Lu. Safety and effectiveness of topical application of tranexamic acid to reduce drainage of elderly femoral neck fractures after total hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1381-1386. |
| [9] | Yuan Jiawei, Zhang Haitao, Jie Ke, Cao Houran, Zeng Yirong. Underlying targets and mechanism of Taohong Siwu Decoction in prosthetic joint infection on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1428-1433. |
| [10] | Chen Junming, Yue Chen, He Peilin, Zhang Juntao, Sun Moyuan, Liu Youwen. Hip arthroplasty versus proximal femoral nail antirotation for intertrochanteric fractures in older adults: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1452-1457. |
| [11] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Comparison of the short-term efficacy of extracorporeal shock wave therapy for middle-aged and elderly knee osteoarthritis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1471-1476. |
| [12] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [13] | Zhong Hehe, Sun Pengpeng, Sang Peng, Wu Shuhong, Liu Yi. Evaluation of knee stability after simulated reconstruction of the core ligament of the posterolateral complex [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 821-825. |
| [14] | Zhao Zhongyi, Li Yongzhen, Chen Feng, Ji Aiyu. Comparison of total knee arthroplasty and unicompartmental knee arthroplasty in treatment of traumatic osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 854-859. |
| [15] | Liu Shaohua, Zhou Guanming, Chen Xicong, Xiao Keming, Cai Jian, Liu Xiaofang. Influence of anterior cruciate ligament defect on the mid-term outcome of fixed-bearing unicompartmental knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 860-865. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||