Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (31): 5048-5055.doi: 10.3969/j.issn.2095-4344.0569
Previous Articles Next Articles
Application of antibiotic-loaded cement spacers in two-stage revision for periprosthetic joint infection after total knee arthroplasty
Yu Qiang, Tian Jing
- Department of Orthopedics, Zhujiang Hospital of Southern Medical University, Guangzhou 510282, Guangdong Province, China
-
Online:2018-11-08Published:2018-11-08 -
Contact:Tian Jing, Master, Professor, Associate chief physician, Master’s supervisor, Department of Orthopedics, Zhujiang Hospital of Southern Medical University, Guangzhou 510282, Guangdong Province, China -
About author:Yu Qiang, Master candidate, Department of Orthopedics, Zhujiang Hospital of Southern Medical University, Guangzhou 510282, Guangdong Province, China
CLC Number:
Cite this article
Yu Qiang, Tian Jing. Application of antibiotic-loaded cement spacers in two-stage revision for periprosthetic joint infection after total knee arthroplasty[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(31): 5048-5055.
share this article
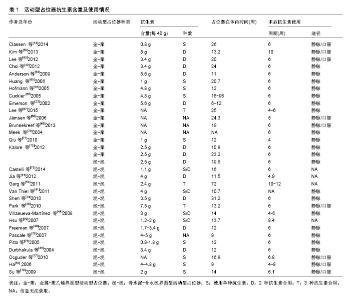
2.1 假体周围感染及诊断标准 全膝关节置换术后假体周围感染在发生率上虽然不及假体松动等并发症,但其所造成的后果严重影响患者生活质量和人工关节假体生存期,被认为是全膝关节置换术后最具灾难性的并发症之一[8-10]。假体周围感染不仅导致患者多次住院、反复手术,增加其经济负担,还严重影响关节功能,甚至有可能造成肢体永久性畸形[11]。造成膝关节假体周围感染的高危因素主要有高龄、营养不良、肥胖、糖尿病、HIV感染、存在远处感染灶以及既往发生过膝关节假体感染等[12],其他可以造成手术切口愈合的因素同样也会增加膝关节假体周围感染的风险,如切口缺血坏死、血肿及化脓感染等[11-12]。 诊断膝关节假体周围感染应结合病史、体格检查、影像学及实验室检查等多方面的结果。目前关于膝关节假体周围感染的诊断标准观点不尽相同。最被广泛接受的诊断标准为:①至少从受累关节2处不同部位取出的组织或关节液培养出相同病原菌;②假体与外界存在窦道相连通或假体周围存在化脓灶[13]。另外也有观点认为以下6条符合3条者即可诊断为假体周围感染:①C-反应蛋白升高;②血沉水平升高;③关节液中白细胞升高;④关节液中分叶核中性粒细胞百分比升高;⑤组织学分析阳性;⑥关节液或假体周围组织中培养出一种细 菌[14-15]。值得注意的是,对于未明确诊断前就已经应用抗生素的患者来说,出现假阴性结果也很常见。 2.2 治疗晚期慢性全膝关节置换后假体周围感染的“金标准” 二期翻修膝关节假体周围感染的基本治疗手段大致可分为7种:①仅使用抗生素抑制治疗;②一期翻修(在一次手术中取出感染的假体,彻底清创后置入新的假体);③二期翻修;④清创、滑膜切除;⑤关节切除的成形手术;⑥关节融合手术;⑦截肢手术。医生可根据每种方法的具体适应证、禁忌证以及患者的具体情况进行选择。 一期翻修于20世纪80年代到90年代期间在欧洲广泛应用[16],优点在于手术次数少、住院时间短、花费少、瘢痕少、术后膝关节功能恢复较好等[17-18],但一期翻修由于是在未明确完全控制感染的情况下进行的,因此存在感染灶未完全清除的风险,疗效也存在争议。二期翻修是目前最被广泛接受和推荐的治疗膝关节假体周围感染的方法,被誉为治疗晚期慢性全膝关节置换术后假体周围感染的“金标准”。二期翻修的过程主要为:第一次手术时移除已感染假体,彻底清创并置入抗生素骨水泥占位器,患者根据假体细菌培养结果接受静脉或口服敏感抗生素治疗,待感染根除后进行第2次手术移除占位器并置入新的膝关节假体。二期翻修优点在于治愈率较高,可达85%-95%[19]。而缺点则在于需多次手术、住院时间长、花费多,如果2次手术间隔过长,还有可能造成软组织挛缩、术后功能恢复欠佳。目前对于2次手术之间抗生素的使用时间还存在争议,可从6个周到6个月不等[11-12,20]。2次手术间隔时间过短则感染控制效果差、感染复发率高,时间过长则增加患者住院时间、增加治疗费用,术后功能恢复欠佳。因此第2次手术时机的选择要根据病原体培养结果以及患者实际情况而定。 2.3 抗生素骨水泥占位器 20世纪90年代以前,二期翻修存在的主要问题是软组织挛缩,膝关节僵硬及活动受限,第2次手术假体置入困难,术后功能差。为解决这些问题,Borden等[21]于1987年首次将静态型抗生素骨水泥占位器应用于膝关节二期翻修,一定程度上解决了软组织挛缩等问题,但却仍存在术后功能欠佳、第二次手术时暴露困难等问题[19,22-23],且这种占位器容易脱位导致严重的骨缺损问题[24]。因此活动型占位器应运而生。相比于静态型占位器,活动型占位器能使患者在2次手术期间也能活动,理论上可以改善术后活动度,促进术后康复。目前关于静态型占位器和活动型占位器孰优孰劣尚未有定论,如何选择以保证最佳感染控制率和术后功能恢复效果仍存在争议。 2.3.1 静态型占位器 静态型占位器主要特点在于2次手术期间使膝关节保持伸直状态或者仅允许膝关节有极微小的活动度。其抗生素骨水泥配方无统一标准,通常为万古霉素(1-4 g/40 g)和庆大霉素(2.4-4.8 g/40 g)混合或万古霉素(1-4 g/40 g)和妥布霉素(2.4-4.8 g/40 g)混合。 一些学者具体阐述了静态型占位器的优点:Faschingbauer等[25]认为静态型占位器可以有效缓解已感染软组织的充血反应,使感染得到更有效地控制,因此对于假体周围感染严重并且有广泛软组织肿胀的患者,使用静态型占位器尤为合适[26-27]。另外,相比于活动型占位器,静态型占位器花费更少[28]。当然,静态型占位器也存在不足。其静态的特点容易导致关节僵硬、二次手术暴露困难、翻修术后膝关节活动度差、功能恢复欠佳等。Emerson等[23]将二期翻修时使用静态型占位器和活动型占位器的患者进行对比研究,发现使用活动型占位器的患者在二期翻修术后平均活动度(Range of motion,ROM)显著高于使用静态型占位器的患者(分别为107.8°和93.7°)。这样的结果与其他大部分研究者所得出结论如出一辙[29-31]。然而,Fehring等[22]也研究了2种占位器对术后功能恢复的影响,发现两者对术后平均ROM的影响差异并无显著性意义(分别为98°和105°);Choi等[32]也得出类似的结论(静态型和活动型分别为97°和100°)。这可能与影响术后ROM因素有关,除了占位器,术后持续康复锻炼及理疗方案等因素同样能够对ROM起到很大的作用。因此,关于静态型和活动型占位器对术后ROM的影响还需进行更多随机对照研究来得出更可靠的结论。 很多学者们不仅关注静态型占位器与活动型占位器对术后ROM的影响,也同样通过回顾文献的方法评估了两者在感染控制、术后功能恢复及并发症方面的差异。2013年Voleti等[33]总结了39项关于静态型和活动型占位器的对比研究并进行系统综述。39项研究证据级别均为Ⅲ级或Ⅳ级,共纳入1 526例膝关节置换术后假体周围感染患者,其中654例使用静态型占位器,872例使用活动型占位器。评价指标包括术后感染复发率、术后ROM、功能评分(特种外科医院膝关节评分和膝关节学会评分)、切口相关并发症和占位器相关并发症。结果显示,除了在术后平均ROM评估中活动型占位器明显优于静态型占位器外(101°和91°,P=0.000 2),其余指标(术后感染复发率、功能评分、并发症)两者差异无显著性意义。同年Pivec等[34]做了类似的研究,纳入的48篇文献包含共1 637例患者,其中688例患者使用静态型占位器,其余949例患者使用活动型占位器。评价指标同样为复发率、膝关节学会评分、ROM及术后并发症发生率。与Voleti等[33]所得结论类似,静态型占位器组和活动型占位器组在感染控制率、功能评分等方面差异并无显著性意义。此外Pivec还指出发生率最高的并发症是术口延迟愈合、假体无菌性松动、深静脉血栓和髌骨损伤,但2种占位器在并发症发生率上差异并没有显著性意义。然而有的学者得出的结论略有不同,2014年Guild等[35]纳入47项研究将2种占位器进行统计分析,共包含 2 011例进行二期翻修的患者(924例使用静态型占位器,1 087例使用活动型占位器)。结果显示,相比于静态型占位器组,活动型占位器组有更高的术后ROM、更低的术后复发率和更低的骨丢失发生率。另外,2016年Lee等[36]首次将目光转向二次翻修对患者心理状态的影响,发现使用2种占位器进行翻修术后患者的目测类比评分明显高于术前(P < 0.000 1),但2种占位器对目测类比评分的影响差异仍无显著性意义。众多关于静态型和活动型占位器的对分析所得出结论不全一致,甚至大相径庭。究其原因,可能是因为大部分对比研究证据级别仅为Ⅲ级或Ⅳ级,仅极少数研究为随机对照研究,因此基于这些文献所进行的系统综述或者Meta分析缺乏统计学严密性;另外,手术医生的手术技艺、随访时间、抗生素使用等多方面差异导致偏倚较大,所得出的结论说服力也大打折扣。因此还需进行更多的高质量的随机对照研究,充分对比2种占位器的感染控制率、复发率、术后功能恢复、并发症发生率及患者满意度等方面,为临床选择占位器提供更可靠的理论依据。 综合静态型占位器的优点和可能存在的劣势,有学者认为这种占位器可能更适合应用于膝关节韧带不稳、伸肌装置缺陷、已有严重骨缺损的患者[6,37-38]。 2.3.2 活动型占位器 活动型占位器最先应用于髋关节手术,随后逐渐应用于膝关节二期翻修中[39]。如前所述,活动型占位器与静态型占位器最大的区别在于理论上可以提供更好的活动度,利于术后功能恢复。这个特点也成为这种占位器的优势所在:即使在2次手术间隔期,患者膝关节仍保持一定活动度,这样避免了关节僵硬,同时防止瘢痕形成,更加利于二次手术的暴露[22-23, 40-41]。与静态型占位器一样,在维持下肢长度避免肌肉挛缩的同时,抗生素骨水泥的应用可以增加膝关节腔抗生素浓度,提高感染清除率。表1总结了近20年活动型占位器骨水泥中抗生素含量及使用情况。然而,活动型占位器并不是完美的。Fink等[42]提出活动型占位器容易磨损并产生碎屑颗粒进入假体周围组织中,并且他们用X射线荧光光谱分析及X射线衍射等手段证实了这一观点。因此在翻修术时,这些碎屑有可能成为第三小体颗粒的来源,影响手术效果,甚至造成翻修失败。Singh等[43]也证实活动型占位器所产生的第三小体颗粒能够刺激滑膜组织产生免疫调节反应并使血管周围淋巴细胞数量显著增加,因此他建议在二次翻修手术时应将滑膜彻底切除以避免严重的炎性反应。另外,很多研究也提出活动型占位器易发生破裂,影响手术效果[5,44]。 活动型占位器的分类标准不一。按制作细节的区别分可分为:①预制且已商业化的活动型占位器;②术中使用商业化模具定制的活动型占位器;③术中使用非商业化模具定制的活动型占位器及其他方法[29,45-48]。商业化预制的活动型占位器有购买方便、简化手术流程等优点,但是尺寸和抗生素剂量选择上有限。术中术者手工制作的活动型占位器有个性化定制的优点,但如何制作更加贴合的关节面且维持术后膝关节稳定仍是难题[49]。目前更加被普遍接受和应用的分类方法则是按关节面材料进行分类:①骨水泥-骨水泥界面型(cement-on- cement);②骨水泥-聚乙烯界面型(cement-on- polyethylene);③金属-聚乙烯界面型(metal-on- polyethylene)。 骨水泥-骨水泥界面型活动型占位器:骨水泥-骨水泥界面型活动型占位器最早由Mcpherson等[50]投入使用。顾名思义,这种占位器的股骨部分和胫骨部分都由抗生素骨水泥构成,可以由手术医生术中通过模具制成,也有商业化预制好的可供选择。对于术中制作的类型,模具也有多种类型。术者可以选择与患者原假体同样型号的试模假体来制作个性化定制模具,来实现更加贴合和稳定的效果。模具选择好后,将混有万古霉素 (3 g/40 g)、妥布霉素(0.5 g/40 g)和庆大霉素(0.5 g/40 g)的骨水泥倒入模具中,骨水泥固化后取出占位器等待置入。王利等[51]则使用了这种方法,利用科室自制模具术中制作了活动型占位器,并在此基础上做了加大立柱、做胫骨延长杆等改进,分散了应力,减少了再翻修时丢失的骨量,取得了较好的效果。Ha[52]则发明了另外一种制作模具的方法,一期手术时将患者已感染的假体取出并进行高温灭菌(股骨部分和胫骨部分),随后将骨水泥涂抹在假体表面制成模具,这样既保证了最终制成的占位器与原假体尺寸一致,又降低了成本。而对于预制好已投入市场的活动型占位器,目前较成熟的是“Spacer-Kr”(Tecres, Sommacampagna, Italy),这种预制的活动型抗生素骨水泥占位器有4种型号(小号,中号,大号和特大号)可供医生术中选择。而另外一种占位器“Interspace Knee spacer”(Exatech,Gainesville, FL.USA)是已经被美国FDA批准应用于假体周围感染二期翻修的活动型占位器[26],但是其抗生素用量 (0.8 g-1.7 g/40 g)显著低于建议用量(3.6 g/40 g),效果受争议。 Garg等[53]在原有方法基础上改进了这种活动型占位器。他们将占位器股骨部分和胫骨部分中间分别插入克氏针,并使用抗生素骨水泥包裹克氏针,最终分别置入股骨髓腔和胫骨髓腔。他们主要从2个方面陈述了这种方法的优势。首先约1/3的膝关节假体周围感染最终会蔓延到髓腔导致骨髓炎,将外层包裹抗生素骨水泥的克氏针插入髓腔可以有效控制髓腔感染,加快清除感染的进 度[54];另一方面克氏针为活动型占位器提供了支撑,能够避免发生占位器脱位,进而防止骨丢失等并发症。 金属-聚乙烯界面型活动型占位器:最早投入使用的活动型占位器就是金属-聚乙烯界面型占位器。Hoffman团队[55]将翻修过程中第一次手术取出的假体股骨部分在重新进行高温灭菌,并在同次手术中重新置入股骨,而胫骨部分则是全新的聚乙烯衬垫。按照4.8 g/ 40 g的用量将妥布霉素混入骨水泥用以固定活动型占位器的胫骨和股骨部分。1995年该团队发表了关于这种占位器的回顾性研究。26例使用这种占位器的患者,除了1例因肺炎死亡外,其余25例都成功进行了二期翻修。在进行了平均31个月的随访后,约72%患者预后良好,术后ROM显著增加(约30°),无术后复发且无深静脉血栓、术口愈合不良等并发症发生。10年后该团队就同一问题再次发表了一篇回顾性研究[56]。该项平均随访时间为73个月的研究中却发现,在50例使用这种活动型占位器的患者中有6例感染复发,预后较差。2次研究结果的差别可能主要归结于随访时间的差异及纳入样本量的不同。另外一种广泛应用的金属-聚乙烯界面活动型占位器是PROSTALAC(prosthesis with antibiotic-loaded acrylic cement,DePuy Synthes,Warsaw,IN),这种预制活动型占位器于1987年投入市场。该间隔期的胫骨部分为聚乙烯衬垫,股骨部分主要由抗生素骨水泥制成,但股骨两侧髁表面覆盖有金属外壳。这种占位器接触面更加光滑,理论上可以减少碎屑的产生。 骨水泥-聚乙烯界面型活动型占位器:这种活动型占位器应用较少,也鲜有文献报道。Evans[57]将聚乙烯材质的股骨部分包裹混有妥布霉素(4.8 g/40 g)和万古霉素(4 g/40 g)的抗生素骨水泥并使用模具塑形,胫骨部分则由聚乙烯构成,制成骨水泥-聚乙烯界面型活动型占位器。在他们的回顾性研究中,31例使用这种占位器进行膝关节翻修的患者在经过至少2年的随访后,只有2例患者出现术后感染复发,总感染控制率为93.5%。 目前有较多研究将静态型占位器和活动型占位器进行比较,且结论大都一致地肯定了活动型占位器在对术后ROM优势,但较少研究将几种不同的活动型占位器进行比较。Spivey等[58]的Meta分析针对4种不同活动型占位器的临床效果进行比较:金属-聚乙烯界面型,术中手工制作的骨水泥-骨水泥界面型,厂家预制的骨水泥-骨水泥界面型,术中使用模具制作的骨水泥-骨水泥界面型。该研究总共纳入34篇文献,包含1 016个活动型占位器,评价指标包括膝关节评分、ROM、感染清除率和术后并发症发生率。结果显示相比于其他活动型占位器,金属-聚乙烯界面型占位器组有更高的ROM(P < 0.03)和更低的并发症发生率(P < 0.043)。由于该Meta分析所纳入研究均为回顾性研究,结论可靠性因可能存在的偏倚而说服力降低。 几种不同的活动型占位器主要区别在于股骨部分与胫骨部分的接触面材料不同,有观点认为全抗生素接触面的活动型占位器(骨水泥-骨水泥型)在感染清除率方面可能要优于含金属或聚乙烯的占位器(金属-骨水泥型、金属-聚乙烯型)。主要有以下2点依据:①金属和聚乙烯属于生物惰性材料,为菌膜提供了良好的附着位点。在关节腔内,漂浮的细菌在假体表面附着,产生由多糖、DNA、蛋白质等物质构成的聚合物[59],从而形成菌膜并对抗生素及机体的免疫系统产生抵抗作用[60]。菌膜表面的细菌会脱离菌膜再次变为漂浮状态,因此形成了漂浮细菌-菌膜-漂浮细菌的恶性循环,导致了长期的假体周围感染或高复发率[61]。而金属和聚乙烯由于其本身材料特性更易于菌膜附着,因此有可能造成感染清除效果欠佳和术后感染复发率增加[60];②由于金属和聚乙烯材料的存在,相比于全抗生素骨水泥的占位器,金 属-骨水泥型和金属-聚乙烯型占位器的有效释放抗生素面积较小,因此可能会影响关节腔内抗生素浓度。有研究表明关节腔内骨水泥的抗生素释放量与释放面积直接相关[57]。但这些观点目前仍停留在理论层面,其准确性还需更多随机对照研究及循证医学的验证。 "

| [1] Lima AL, Oliveira PR, Carvalho VC, et al. Periprosthetic joint infections. Interdiscip Perspect Infect Dis. 2013;2013:542796.[2] Kotelnicki J, Mitts K. Surgical treatments for patients with an infected total knee arthroplasty. JAAPA. 2009;22(11):40, 43-46.[3] Toms AD, Davidson D, Masri BA, et al. The management of peri-prosthetic infection in total joint arthroplasty. J Bone Joint Surg Br. 2006;88(2):149-155.[4] Stammers J, Kahane S, Ranawat V, et al. Outcomes of infected revision knee arthroplasty managed by two-stage revision in a tertiary referral centre. Knee. 2015;22(1):56-62.[5] Cui Q, Mihalko WM, Shields JS, et al. Antibiotic-impregnated cement spacers for the treatment of infection associated with total hip or knee arthroplasty. J Bone Joint Surg Am. 2007;89(4): 871-882.[6] Anagnostakos K. Therapeutic Use of Antibiotic-loaded Bone Cement in the Treatment of Hip and Knee Joint Infections. J Bone Jt Infect. 2017;2(1):29-37.[7] Antoci V, Phillips MJ, Antoci VJ, et al. The treatment of recurrent chronic infected knee arthroplasty with a 2-stage procedure. J Arthroplasty. 2009;24(1):113-159.[8] Kim JK, Lee DY, Kang DW, et al. Efficacy of antifungal- impregnated cement spacer against chronic fungal periprosthetic joint infections after total knee arthroplasty. Knee. 2018.[9] George J, Miller EM, Curtis GL, et al. Success of Two-Stage Reimplantation in Patients Requiring an Interim Spacer Exchange. J Arthroplasty. 2018.[10] Lee YS, Chen AF. Two-Stage Reimplantation in Infected Total Knee Arthroplasty. Knee Surg Relat Res. 2018;30(2):107-114.[11] Zimmerli W. Infection and musculoskeletal conditions: Prosthetic- joint-associated infections. Best Pract Res Clin Rheumatol. 2006; 20(6):1045-1063.[12] Pozo JLD, Patel R. Infection Associated with Prosthetic Joints. New Eng J Med. 2009,361(8):787.[13] Osmon DR, Berbari EF, Berendt AR, et al. Executive summary: diagnosis and management of prosthetic joint infection: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis. 2013;56(1):1-10.[14] Della VC, Parvizi J, Bauer TW, et al. American Academy of Orthopaedic Surgeons clinical practice guideline on: the diagnosis of periprosthetic joint infections of the hip and knee. J Bone Joint Surg Am. 2011;93(14):1355-1357.[15] Parvizi J, Zmistowski B, Berbari EF, et al. New definition for periprosthetic joint infection: from the Workgroup of the Musculoskeletal Infection Society. Clin Orthop Relat Res. 2011;469(11):2992-2994.[16] Buchholz HW, Elson RA, Heinert K. Antibiotic-loaded acrylic cement: current concepts. Clin Orthop Relat Res. 1984;190(190): 96.[17] Singer J, Merz A, Frommelt L, et al. High rate of infection control with one-stage revision of septic knee prostheses excluding MRSA and MRSE. Clin Orthop Relat Res. 2012;470(5): 1461-1471.[18] Oussedik SI, Dodd MB, Haddad FS. Outcomes of revision total hip replacement for infection after grading according to a standard protocol. J Bone Joint Surg Br. 2010;92(9):1222-1226.[19] Hanssen AD. Managing the infected knee: as good as it gets. J Arthroplasty. 2002;17(4 Suppl 1):98-101.[20] Frommelt L. Principles of systemic antimicrobial therapy in foreign material associated infection in bone tissue, with special focus on periprosthetic infection. Injury. 2006;37 Suppl 2:S87-S94.[21] Borden LS, Gearen PF. Infected total knee arthroplasty. A protocol for management. J Arthroplasty. 1987;2(1):27-36.[22] Fehring TK, Odum S, Calton TF, et al. Articulating versus static spacers in revision total knee arthroplasty for sepsis. The Ranawat Award. Clin Orthop Relat Res. 2000;(380):9-16.[23] Emerson RJ, Muncie M, Tarbox TR, et al. Comparison of a static with a mobile spacer in total knee infection. Clin Orthop Relat Res. 2002;(404):132-138.[24] 张晓岗,曹力. 人工全髋关节置换术后假体周围感染的处理[J]. 中华关节外科杂志(电子版),2014,8(1):101-104.[25] Faschingbauer M, Bieger R, Reichel H, et al. Complications associated with 133 static, antibiotic-laden spacers after TKA. Knee Surg Sports Traumatol Arthrosc. 2016;24(10):3096-3099.[26] Jacobs C, Christensen CP, Berend ME. Static and mobile antibiotic-impregnated cement spacers for the management of prosthetic joint infection. J Am Acad Orthop Surg. 2009;17(6): 356-368.[27] Mazzucchelli L, Rosso F, Marmotti A, et al. The use of spacers (static and mobile) in infection knee arthroplasty. Curr Rev Musculoskelet Med. 2015;8(4):373-382.[28] Antoci V, Phillips MJ, Antoci VJ, et al. Using an antibiotic-impregnated cement rod-spacer in the treatment of infected total knee arthroplasty. Am J Orthop (Belle Mead NJ). 2009;38(1):31-33.[29] Hsu YC, Cheng HC, Ng TP, et al. Antibiotic-loaded cement articulating spacer for 2-stage reimplantation in infected total knee arthroplasty: a simple and economic method. J Arthroplasty. 2007;22(7):1060-1066.[30] Park SJ, Song EK, Seon JK, et al. Comparison of static and mobile antibiotic-impregnated cement spacers for the treatment of infected total knee arthroplasty. Int Orthop. 2010;34(8):1181-1186.[31] Classen T, von Knoch M, Wernsmann M, et al. Functional interest of an articulating spacer in two-stage infected total knee arthroplasty revision. Orthop Traumatol Surg Res. 2014;100(4): 409-412.[32] Choi HR, Malchau H, Bedair H. Are prosthetic spacers safe to use in 2-stage treatment for infected total knee arthroplasty? J Arthroplasty. 2012;27(8):1474-1479.[33] Voleti PB, Baldwin KD, Lee GC. Use of static or articulating spacers for infection following total knee arthroplasty: a systematic literature review. J Bone Joint Surg Am. 2013;95(17):1594-1599.[34] Pivec R, Naziri Q, Issa K, et al. Systematic review comparing static and articulating spacers used for revision of infected total knee arthroplasty. J Arthroplasty. 2014;29(3):553-557.[35] Guild GR, Wu B, Scuderi GR. Articulating vs. Static antibiotic impregnated spacers in revision total knee arthroplasty for sepsis. A systematic review. J Arthroplasty. 2014;29(3):558-563.[36] Lee C, Gondalia S. Do articulating spacers or static spacers have more effect on the mental health status of patients undergoing two-stage revision total knee arthroplasty for periprosthetic joint infection? Curr Orthop Prac. 2016;27:1.[37] Macmull S, Bartlett W, Miles J, et al. Custom-made hinged spacers in revision knee surgery for patients with infection, bone loss and instability. Knee. 2010;17(6):403-406.[38] Kotwal SY, Farid YR, Patil SS, et al. Intramedullary rod and cement static spacer construct in chronically infected total knee arthroplasty. J Arthroplasty. 2012;27(2):253-259.[39] Anagnostakos K, Fink B. Antibiotic-loaded cement spacers - lessons learned from the past 20 years. Expert Rev Med Devices. 2018;15(3):231-245.[40] Freeman MG, Fehring TK, Odum SM, et al. Functional advantage of articulating versus static spacers in 2-stage revision for total knee arthroplasty infection. J Arthroplasty. 2007;22(8):1116-1121.[41] Van Thiel GS, Berend KR, Klein GR, et al. Intraoperative molds to create an articulating spacer for the infected knee arthroplasty. Clin Orthop Relat Res. 2011;469(4):994-1001.[42] Fink B, Rechtenbach A, Buchner H, et al. Articulating spacers used in two-stage revision of infected hip and knee prostheses abrade with time. Clin Orthop Relat Res. 2011;469(4):1095-1102.[43] Singh G, Deutloff N, Maertens N, et al. Articulating polymethylmethacrylate (PMMA) spacers may have an immunomodulating effect on synovial tissue. Bone Joint J. 2016; 98-B(8):1062-1068.[44] Villanueva-Martinez M, Rios-Luna A, Pereiro J, et al. Hand-made articulating spacers in two-stage revision for infected total knee arthroplasty: good outcome in 30 patients. Acta Orthop. 2008; 79(5):674-682.[45] Durbhakula SM, Czajka J, Fuchs MD, et al. Antibiotic-loaded articulating cement spacer in the 2-stage exchange of infected total knee arthroplasty. J Arthroplasty. 2004;19(6):768-774.[46] Coe PO, Jain NP, Johnson DS. Custom fitted articulating cement spacers for use in two-stage revision knee replacement. Ann R Coll Surg Engl. 2014;96(8):624-625.[47] Marothi DP, Sharma V, Sheth AN, et al. Management of Infected Total Knee Arthroplasty by a New Innovative Customized Articulating Knee Spacer: An Early Experience. J Orthop Case Rep. 2016;6(4):20-22.[48] Shaikh AA, Ha CW, Park YG, et al. Two-stage approach to primary TKA in infected arthritic knees using intraoperatively molded articulating cement spacers. Clin Orthop Relat Res. 2014; 472(7):2201-2207.[49] Burnett RS, Kelly MA, Hanssen A, et al. Technique and timing of two-stage exchange for infection in TKA. Clin Orthop Relat Res. 2007;464:164-178.[50] Mcpherson EJ, Lewonowski K, Dorr LD. Techniques in arthroplasty. Use of an articulated PMMA spacer in the infected total knee arthroplasty. J Arthroplasty. 1995;10(1):87-89.[51] 王利,哈巴西•卡肯,殷剑,等. 自制活动型抗生素骨水泥间隔器:在全膝关节表面置换后感染翻修中的应用[J]. 中国组织工程研究,2015, 19(13):2017-2022.[52] Ha CW. A technique for intraoperative construction of antibiotic spacers. Clin Orthop Relat Res. 2006;445:204-209.[53] Garg P, Ranjan R, Bandyopadhyay U, et al. Antibiotic- impregnated articulating cement spacer for infected total knee arthroplasty. Indian J Orthop. 2011;45(6):535-540.[54] Hanssen AD, Spangehl MJ. Practical applications of antibiotic-loaded bone cement for treatment of infected joint replacements. Clin Orthop Relat Res. 2004;(427):79-85.[55] Hofmann AA, Kane KR, Tkach TK, et al. Treatment of infected total knee arthroplasty using an articulating spacer. Clin Orthop Relat Res. 1995;(321):45-54.[56] Hofmann AA, Goldberg T, Tanner AM, et al. Treatment of infected total knee arthroplasty using an articulating spacer: 2- to 12-year experience. Clin Orthop Relat Res. 2005;(430):125-131.[57] Evans RP. Successful treatment of total hip and knee infection with articulating antibiotic components: a modified treatment method. Clin Orthop Relat Res. 2004;(427):37-46.[58] Spivey JC, Guild GR, Scuderi GR. Use of Articulating Spacer Technique in Revision Total Knee Arthroplasty Complicated by Sepsis: A Systematic Meta-Analysis. Orthopedics. 2017;40(4): 212-220.[59] Mcconoughey SJ, Howlin R, Granger JF, et al. Biofilms in periprosthetic orthopedic infections. Future Microbiol. 2014; 9(8):987-1007.[60] Antony S, Farran Y. Prosthetic Joint and Orthopedic Device Related Infections. The Role of Biofilm in the Pathogenesis and Treatment. Infect Disord Drug Targets. 2016;16(1):22-27.[61] Jia YT, Zhang Y, Ding C, et al. Antibiotic-loaded articulating cement spacers in two-stage revision for infected total knee arthroplasty: individual antibiotic treatment and early results of 21 cases. Chin J Traumatol. 2012;15(4):212-221.[62] Kim YS, Bae KC, Cho CH, et al. Two-stage revision using a modified articulating spacer in infected total knee arthroplasty. Knee Surge Relat Res. 2013;25(4):180-185.[63] Lee JK, Choi CH. Two-stage reimplantation in infected total knee arthroplasty using a re-sterilized tibial polyethylene insert and femoral component. J Arthroplasty. 2012;27(9):1701-1706.[64] Anderson JA, Sculco PK, Heitkemper S, et al. An articulating spacer to treat and mobilize patients with infected total knee arthroplasty. J Arthroplasty. 2009;24(4):631-635.[65] Huang HT, Su JY, Chen SK. The results of articulating spacer technique for infected total knee arthroplasty. J Arthroplasty. 2006;21(8):1163-1168.[66] Cuckler JM. The infected total knee: management options. J Arthroplasty. 2005;20(2):33-36.[67] Lee BJ, Kyung HS, Yoon SD. Two-Stage Revision for Infected Total Knee Arthroplasty: Based on Autoclaving the Recycled Femoral Component and Intraoperative Molding Using Antibiotic-Impregnated Cement on the Tibial Side. Clin Orthop Surg.2015;7(3):310-317.[68] Jämsen E, Sheng P, Halonen P, et al. Spacer prostheses in two-stage revision of infected knee arthroplasty. Int Orthop. 2006;30(4):257-261.[69] Brunnekreef J, Hannink G, Malefijt MW. Recovery of knee mobility after a static or mobile spacer in total knee infection. Acta Orthopaedica Belgica. 2013;79(1):83-89.[70] Meek RM, Dunlop D, Garbuz DS, et al. Patient satisfaction and functional status after aseptic versus septic revision total knee arthroplasty using the PROSTALAC articulating spacer. J Arthroplasty. 2004;19(7):874-879.[71] Qiu XS, Xu S, Mphil D, et al. Application of an articulating spacer in two‐stage revision for severe infection after total knee arthroplasty. Orthop Surg. 2010;2(4):299-304.[72] Kalore NV, Maheshwari A, Sharma A, et al. Is there a preferred articulating spacer technique for infected knee arthroplasty? A preliminary study. Clin Orthop Relat Res. 2012;470(1):228-235.[73] Castelli CC, Gotti V, Ferrari R. Two-stage treatment of infected total knee arthroplasty: two to thirteen year experience using an articulating preformed spacer. Int Orthop. 2014;38(2):405-412.[74] Shen H, Zhang X, Jiang Y, et al. Intraoperatively-made cement-on-cement antibiotic-loaded articulating spacer for infected total knee arthroplasty. Knee. 2010;17(6):407-411.[75] Pascale V, Pascale W. Custom-made Articulating Spacer in Two-stage Revision Total Knee Arthroplasty. An Early Follow-up of 14 Cases of at Least 1 Year After Surgery. Hss J. 2007;3(2): 159-163.[76] Pitto RP, Castelli CC, Ferrari R, et al. Pre-formed articulating knee spacer in two-stage revision for the infected total knee arthroplasty. Int Orthop. 2005;29(5):305-308.[77] Ocguder A, Firat A, Tecimel O, et al. Two-stage total infected knee arthroplasty treatment with articulating cement spacer. Arch Orthop Trauma Surg. 2010;130(6):719-725.[78] Su YP, Lee OK, Chen WM, et al. A Facile Technique to Make Articulating Spacers for Infected Total Knee Arthroplasty. J Chin Med Assoc. 2009;72(3):138-145. |
| [1] | Zhang Yu, Tian Shaoqi, Zeng Guobo, Hu Chuan. Risk factors for myocardial infarction following primary total joint arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1340-1345. |
| [2] | Li Dadi, Zhu Liang, Zheng Li, Zhao Fengchao. Correlation of total knee arthroplasty efficacy with satisfaction and personality characteristics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1346-1350. |
| [3] | Wei Wei, Li Jian, Huang Linhai, Lan Mindong, Lu Xianwei, Huang Shaodong. Factors affecting fall fear in the first movement of elderly patients after total knee or hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1351-1355. |
| [4] | Wang Jinjun, Deng Zengfa, Liu Kang, He Zhiyong, Yu Xinping, Liang Jianji, Li Chen, Guo Zhouyang. Hemostatic effect and safety of intravenous drip of tranexamic acid combined with topical application of cocktail containing tranexamic acid in total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1356-1361. |
| [5] | Xiao Guoqing, Liu Xuanze, Yan Yuhao, Zhong Xihong. Influencing factors of knee flexion limitation after total knee arthroplasty with posterior stabilized prostheses [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1362-1367. |
| [6] | Huang Zexiao, Yang Mei, Lin Shiwei, He Heyu. Correlation between the level of serum n-3 polyunsaturated fatty acids and quadriceps weakness in the early stage after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1375-1380. |
| [7] | Zhang Chong, Liu Zhiang, Yao Shuaihui, Gao Junsheng, Jiang Yan, Zhang Lu. Safety and effectiveness of topical application of tranexamic acid to reduce drainage of elderly femoral neck fractures after total hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1381-1386. |
| [8] | Yuan Jiawei, Zhang Haitao, Jie Ke, Cao Houran, Zeng Yirong. Underlying targets and mechanism of Taohong Siwu Decoction in prosthetic joint infection on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1428-1433. |
| [9] | Chen Junming, Yue Chen, He Peilin, Zhang Juntao, Sun Moyuan, Liu Youwen. Hip arthroplasty versus proximal femoral nail antirotation for intertrochanteric fractures in older adults: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1452-1457. |
| [10] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Comparison of the short-term efficacy of extracorporeal shock wave therapy for middle-aged and elderly knee osteoarthritis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1471-1476. |
| [11] | Zhong Hehe, Sun Pengpeng, Sang Peng, Wu Shuhong, Liu Yi. Evaluation of knee stability after simulated reconstruction of the core ligament of the posterolateral complex [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 821-825. |
| [12] | Zhao Zhongyi, Li Yongzhen, Chen Feng, Ji Aiyu. Comparison of total knee arthroplasty and unicompartmental knee arthroplasty in treatment of traumatic osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 854-859. |
| [13] | Liu Shaohua, Zhou Guanming, Chen Xicong, Xiao Keming, Cai Jian, Liu Xiaofang. Influence of anterior cruciate ligament defect on the mid-term outcome of fixed-bearing unicompartmental knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 860-865. |
| [14] | Zhang Nianjun, Chen Ru. Analgesic effect of cocktail therapy combined with femoral nerve block on total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 866-872. |
| [15] | Yuan Jun, Yang Jiafu. Hemostatic effect of topical tranexamic acid infiltration in cementless total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 873-877. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||