Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (19): 3077-3084.doi: 10.3969/j.issn.2095-4344.0261
Previous Articles Next Articles
Advances in diagnosis of periprosthetic infection after arthroplasty
Hu Xue-feng, Zuo Qiang, Wei Lei
- Department of Orthopedics, First Affiliated Hospital of Baotou Medical College, Baotou 014010, Inner Mongolia Autonomous Region, China
-
Online:2018-07-08Published:2018-07-08 -
Contact:Wei Lei, Master, Chief physician, Department of Orthopedics, First Affiliated Hospital of Baotou Medical College, Baotou 014010, Inner Mongolia Autonomous Region, China -
About author:Hu Xue-feng, Master, Attending physician, Department of Orthopedics, First Affiliated Hospital of Baotou Medical College, Baotou 014010, Inner Mongolia Autonomous Region, China
CLC Number:
Cite this article
Hu Xue-feng, Zuo Qiang, Wei Lei. Advances in diagnosis of periprosthetic infection after arthroplasty[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(19): 3077-3084.
share this article

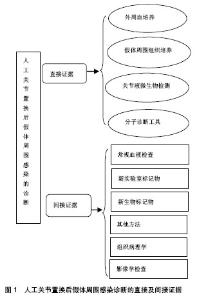
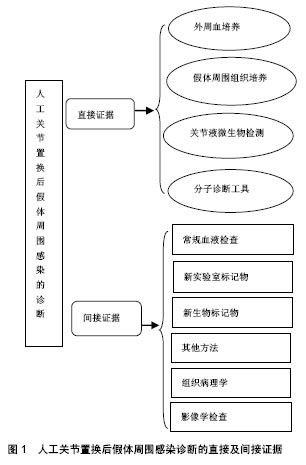
2.1.2 假体周围组织培养 假体周围组织培养对于假体周围感染的诊断是一项十分有价值的诊断方法。但是,在低毒力微生物存在的情况下,这一方法则表现出较低的敏感性和较大的波动性。有研究发现假体置换术后感染的微生物常常通过形成生物膜定植于假体表面,而且假体周围组织内的细菌长期保持静止状态,以抵御机体免疫系统的攻击,抵制抗生素对细菌的作用,从而限制了常规细菌培养技术的使用。常规假体周围组织取样进行培养往往不能取到假体表面的微生物。Peel等[3]应用贝叶斯潜伏类建模接种组织到血培养瓶中进行培养,与常规琼脂和肉汤培养物的灵敏度相比提高47%, 准确度达92%,而且培养时间短。Tunney等[4]在诊断假体周围感染时首次应用超声裂解液培养法,之后众多学者进行超声裂解液培养,证实相较传统方法其敏感性和特异性高,肯定了超声裂解液培养对于诊断假体周围感染的意义,也进一步证明了这一方法的可靠性。超声比传统组织培养更敏感,特异性更高,特别是对于以前服用抗生素的患者[5]。假体周围组织培养的取材关键是需要外科翻修手术来获取,这就限制了其使用,无法在术前诊断中发挥其作用。目前植入物超声处理,因为检测费用和临床易用性限制了该技术的使用,但是,未来可能极大地改变和拓宽临床微生物检测的手段。 2.1.3 关节液微生物检测 关节液微生物检测是传统诊断假体周围感染的金标准。关节液获取方便、安全,可以在术前诊断中充分发挥其作用。很多学者认为关节液培养有明显优势,关节穿刺相对便捷,而组织活检更加复杂,且不同的观察者之间对结果的判断差异很大[6]。而另外一些研究结果却得出相反的结论,认为其敏感度比组织活检低[7]。由于关节液中较低的细菌浓度和抗生素治疗等原因可能导致检出率低。张宁等[8]系统评价关节液和组织培养对假体周围感染的诊断价值,认为组织培养对关节置换术后感染诊断的敏感度上明显优于关节液培养。目前经过研究者的不断改进,发现利用BACTEC Plus/F瓶关节液细菌培养检测率明显提高,而且不易污 染[9]。Font-Vizcarra等[10]研究显示,利用关节液直接接种在血液培养瓶中培养与假体周围组织和拭子样品细菌培养结果相比,具有更高的灵敏度和特异性,而且在急性感染中比在慢性感染中更容易检出。Geller等[11]也认为关节液利用血液培养瓶系统准确性达83.7%,增加了下肢假体周围感染中病原体的鉴定成功率。不过,尽管这些新方法对于假体周围感染的诊断具有重大参考价值,但仍存在短板。Squire等[12]在其研究中提出,关节液培养所需时间长,有可能错失患者的最佳治疗时间窗。 2.1.4 分子诊断工具 (1)PCR:在诊断假体周围感染有以下特点:①检测迅速:PCR能将微量的病原菌脱氧核糖核酸大幅扩增,即可直接进行检测从而缩短了时间,赢得最佳治疗时间;②不受抗生素影响,检出率高;③假阳性率高:在PCR分析期间的每个步骤都可以引起污染,例如临床样品收集、脱氧核糖核酸提取等。目前16S核糖体核糖核酸PCR(16S rRNA PCR)应用最为广泛[13]。然而16S核糖体核糖核酸扩增呈现极限,缺乏敏感性或特异性。特性PCR显示其独特优点,通过特异性靶点基因来鉴定细菌。Ghebremedhin等[14]利用特性靶点hsp60、rpoB、sodA和tuf基因序列进行葡萄球菌种的遗传分类和鉴定,得到满意的结果。植入物的超声处理越来越多地被许多中心采用。分子诊断方法与超声处理组合,具有更高的灵敏度,更快的周转时间,并且不受先前的抗微生物治疗的影响。有研究在对超声波裂解液进行,PCR-电喷雾质谱法和组织培养法相比发现,PCR-电喷雾质谱法更敏感,尤其适用于接受抗菌药物的病例,在接受抗生素的假体周围感染受试者中,PCR-电喷雾质谱法的敏感性为85.7%,而组织培养法的敏感性为65.7%,但是其特异性低[15]。多重PCR的引入其检测敏感性和特异性大幅度提高,尤其结合超声处理,有研究报道其敏感性高达96%,而特异性甚至达100%[5]。Prieto-Borja 等[16]证实利用多重PCR(Unyvero i60®)具有较高的敏感性,而Hischebeth等[17]利用Unyvero i60®检测经超声处理的样本,分析获得的PCR结果显示,灵敏度仅为66.7%,而特异性高达100%。此外,实时PCR的定量分析对诊断假体周围感染有一定参考价值,虽然可早期诊断由金黄色葡萄球菌引起的假体周围感染,但该方法检测耐甲氧西林凝固酶阴性葡萄球菌的灵敏度和高成本可能限制其在该领域的使用[18]。 PCR技术虽显示出巨大潜力,但是仍然存在一些不容忽视的缺陷,如各种样本的检出率和各项研究结果差异性较大。这些技术复杂并且对结果解释至关重要,不适于临床微生物实验室中每天通常处理的绝大多数常规样品。而且需要有经过训练的专业技术人员的严格操作,以避免任何污染[19]。诊断成本和实用性限制其应用,在国内很少有医疗机构把其作为常规诊断手段。 (2)其他试验方法:近年来,基质辅助激光解吸电离飞行时间质谱已广泛应用,由于其快速、可靠、易于使用,丰富了临床微生物检测手段[20]。基质辅助激光解吸电离飞行时间质谱技术是对完整细菌和细菌提取物进行软激光电离,并在分析质谱的母体分子中碎片离子质量差异的基础上确定了分子结构。Peel等[21]应用基质辅助激光解吸电离飞行时间质谱技术分析了178例假体周围感染和82例无菌性失败的病例,鉴定出123个假体周围感染和74个无菌性失败,阳性率分别为69%和90%。有研究将PCR技术与质谱分析联用,发现对于假体周围感染诊断的特异性为94%,这比应用超声裂解液进行细菌培养的特异性低(99%)[22]。其他研究方法还有:使用16S核糖体核糖核酸荧光原位杂交的分子荧光染色结合共聚焦激光扫描成像显微镜鉴定生物膜细菌[23],单独通过扫描电子显微镜,透射电子显微镜以及通过免疫荧光显微镜和扫描共聚焦激光显微镜对假体表面附着的细菌生物膜的视觉检测[24]。这些技术为假体周围感染生物膜的诊断提供了新的见解。还有通过微量热法来测量培养物中复制微生物的热量,也不失为一种快速,简单的筛选方法[25],以及定量反转录PCR检测滑液或手术样品中的细菌信使核糖核酸,作为假体周围感染诊断的潜在手段[26]。但是这些技术不作为常规诊断手段,多数为试验室研究应用。 2.2 间接证据 2.2.1 常规血液检查 常规血液检查包括白细胞计数、红细胞沉降率、C-反应蛋白和降钙素原,见表1。美国骨科协会指南强烈推荐将红细胞沉降率和C-反应蛋白作为诊断髋、膝关节假体周围感染的主要评估指标。美国骨科协会指南与肌肉骨骼感染学会共同指出:当血清红细胞沉降率>30 mm/h,C-反应蛋白>10 mg/L时,结合临床则有可能考虑假体周围感染[1,27]。然而,许多研究表明这些诊断手段的准确性有限,都没有足够的灵敏度或特异性来诊断或排除假体周围感染[28]。指南也指出红细胞沉降率和C-反应蛋白是炎症反应的非特异性标记物,在任何一种炎症性病变存在时都会有明显升高,因此它们在诊断假体周围感染时特异性很低。需要强调的是,红细胞沉降率或C-反应蛋白的正常值不排除假体周围感染,尤其是低毒感染病例,例如:痤疮丙酸杆菌这种微生物导致的感染并不会引起红细胞沉降率和C-反应蛋白的升高。此外,红细胞沉降率和C-反应蛋白在手术后正常也会增加。因此,如果关节置换术后患者红细胞沉降率和/或C-反应蛋白出现异常,建议进行关节穿刺抽取关节液进行细菌培养、关节液白细胞计数和分类检查综合判断。关节液的检查比血液更有帮助,因假体周围滑液是原发感染灶。美国骨科协会指出关节液中C-反应蛋白浓度达到3.7 mg/L时即有诊断意义[29],而在同一组病例中血浆C-反应蛋白浓度为16.5 mg/L才有诊断意义。关节液C-反应蛋白浓度诊断假体周围感染的敏感性是84%,特异性为97%,而准确性为96%,这几个方面均优于以血浆C-反应蛋白浓度为标准的诊断价值。 2.2.2 新实验室标记物 新的实验室标记物包括白细胞介素、Toll样受体、肿瘤坏死因子α、粒细胞集落刺激因子和γ-干扰素等,见表1。其中,白细胞介素的应用价值更广。在白细胞介素家族中,白细胞介素6又是最被看好的炎性细胞因子,它在与β-防御素2、β-防御素3和导管素37等抗菌肽联合使用时能提高假体周围感染诊断准确度[30]。Randau等[31]认为滑膜白细胞介素6、血清白细胞介素6、C-反应蛋白和降钙素原中前两者诊断精度最高(88%,83%)。Elgeidi等[32]研究表明,与红细胞沉降率、C-反应蛋白和白细胞计数相比,白细胞介素6是诊断假体周围感染的最准确的实验室标志物,当血清白细胞介素6>10.4 μg/L时,其准确度为92.5%,灵敏度为100%,特异性为90.9%,阳性预测值为79%,阴性预测值为100%;并且C-反应蛋白联合白细胞介素6(白细胞介素6阈值>10.4 ng/L,C-反应蛋白阈值> 18 mg/L)是诊断假体周围感染的最佳组合(灵敏度为100%,阴性预测值为100%)。Matsen Ko等[33]也认为白细胞介素6是最具诊断价值的血清学标志物,在假体周围感染患者的血清中白细胞介素6水平持续升高> 10 ng/L,在无菌性松动的患者中不升高。Frangiamore等[34]研究肩关节置换术后假体周围感染,发现在滑膜组织中联合白细胞介素6、肿瘤坏死因子α和白细胞介素2比单独的任何细胞因子更有诊断价值(灵敏度为0.80,特异性为0.93)。相反,Grosso等[35]报道,特别是对于肩关节,白细胞介素6具有非常差的灵敏度(0.12),但是特异性高(0.93),这使得其对于诊断肩关节假体周围感染无效。最近Ahmad等[36]荟萃分析指出,在诊断假体周围感染的实验室指标中把灵敏度和特异性的阈值设置为>90%时,只有血清白细胞介素6满足,同时指出最有效排除假体周围感染的诊断测试是血清白细胞介素6,也发现在无菌性松动的情况下白细胞介素6是正常的,翻修手术后其回归正常水平。 Toll样受体是参与非特异性免疫的一类重要蛋白质分子,当微生物侵入机体时,Toll样受体可以识别它们并激活机体免疫细胞免疫应答。在Toll样受体中,Toll样受体4和Toll样受体2主要表达于单核-巨噬细胞表面,识别广谱的革兰阳性和革兰阴性细菌,并诱导主要炎症反应。Galliera等[37]研究发现在假体周围感染患者中Toll样受体2显着增加而不是Toll样受体4,发现Toll样受体2受体升高与C-反应蛋白、白细胞介素6 和白细胞介素1β呈强正相关性。同时,Pajarinen等[38]发现无菌性松动磨损颗粒也可直接被某些Toll样受体识别。 2.2.3 新生物标记物 在2014年,Deirmengian等[39]提出假体周围感染诊断进入生物标志物时代,认为他们提出的生物标志物对于假体周围感染具有优良诊断性能,见表1。该优点归纳为:①诊断准确性和复杂的肌肉骨骼感染学会标准相似;②简便易行,实用性强,任何人可以运用诊断,不同人员对结果判断差异细微;③不受系统性炎症性疾病、抗生素及生物膜影响;④不易受标本污染的影响。Deirmengian和他的团队发现5个具有完美敏感度、特异度和曲线下面积的生物标记物,这几种标志物可以完全有效的区分无菌和感染,包括人α-防御素1-3、中性粒细胞弹性蛋白酶、抑菌/细菌通透性增强蛋白、中性粒细胞明胶酶相关蛋白、乳铁蛋白,见表1。这些标志物具有100%的灵敏度和特异性。尤其α-防御素成为目前的研究热点,认为滑膜液α-防御素在诊断假体周围感染时比目前的常规诊手段更有效,这也得到众多学者的支持。其中循证医学证据水平一级有Bonanzinga等[40]的研究报道,二级水平有3篇文 献[41-43],三级水平也有3篇文献[44-46]。Deirmengian等[45]还认为α-防御素适合包括革兰阳性/阴性、细菌/真菌、高毒力/低毒力等任何生物体类型感染导致的假体周围感染,都具有极佳的诊断价值。 2.2.4 其他方法 Parvizi等[47]的研究发现,白细胞酯酶与红细胞沉降率、C-反应蛋白、滑膜白细胞计数以及滑膜中性粒细胞分类比例之间有很好的相关性。白细胞酯酶比色法诊断假体周围感染,具有高达81%的敏感性和100%的特异性,而且该方法还有费用低廉,能够立即得到结果的优点,见表1。Colvin等[48]和Wetters等[49]也研究利用白细胞酯酶试剂条诊断假体周围感染,均发现白细胞酯酶是诊断假体周围感染的快速、便宜和敏感的工具。然而,它们的效用受到滑液中的血液或碎屑的限制,部分样品不能检测。Deirmengian等[42]研究发现白细胞酯酶测试条易受血液的干扰,其灵敏度为69%,特异性100%。Shafafy等[50]认为因关节穿刺的吸出物内存在红细胞,可能降低白细胞酯酶检测在诊断肩关节假体周围感染中的准确性。其他学者利用血清的脂多糖结合蛋白展开研究,见表1。Friedrich等[51]发现与无菌性松动患者相比,假体周围感染患者的脂多糖结合蛋白血清水平升高,阈值>7 μg/L时特异性为66%,灵敏度为71%,但是相比血清中的C-反应蛋白水平无明显优势,单独使用脂多糖结合蛋白不能区分假体周围感染和无菌性松动。另外还有可溶性尿激酶纤溶酶原激活物受体,有研究发现与未感染相比,血清可溶性尿激酶纤溶酶原激活物受体在假体周围感染患者中显着增加,并且与C-反应蛋白、白细胞介素6、白细胞介素1和肿瘤坏死因子α以及趋化因子2具有显著的正相关性,目前还没有得到可靠的证实,可能在将来假体周围感染的诊断中发挥重要作用[52]。还文献报道在人β-防御素2和β-防御素3、导管素37等方面的研究[30]。 2.2.5 组织病理学 组织病理学检测是诊断假体周围感染不可缺少的环节。术中冰冻切片和石蜡切片对假体周围感染都有辅助诊断价值,诊断标准为每高倍镜下≥5个中性粒细胞,见表1。根据Morawietz和Krenn分 类[53]:Ⅰ型为磨损颗粒诱导,Ⅱ型为感染型,Ⅲ型为组合型和Ⅳ型为不确定型。Janz等[54]认为组织病理学检测可高达87%的灵敏度和100%的特异性。Claassen等[55]回顾性分析56全膝关节置换患者,发现利用关节镜假体周围组织活检的灵敏度和特异性均能达到88%。Kwiecien等[56]同样认为术中冷冻切片具有高达98.8%的灵敏度和94.1%特异性,冷冻切片和石蜡切片之间的差异率低。Fink等[7]利用活检组织细菌学培养结合组织病理学检查,结果显示在诊断髋关节假体周围感染方面,比关节液抽吸检测和C-反应蛋白有更高的诊断价值,具有91%的准确度和82%的灵敏度,特异性为98%。 然而,Zimmerli等[57]对其诊断的准确性有所质疑,因不同观察者间差异性和样品之间甚至在个体组织切片的渗透程度变化很大。George等[58]认为冷冻切片虽然有较高的特异性94%,而灵敏度只有50%,其排除感染的效用较小,评估二期假体再置入时指导意义不大。国内学者Tang等[59]回顾性分析52例假体周围感染,发现组织学分析的敏感性较低为55.0%,而特异性高达89.7%。Banke等[60]认为关节液的检测(滑膜白细胞介素6、C-反应蛋白和白细胞酯酶,新的生物标志物)优于滑膜组织活检,相较组织病理学检测结果更快、实用性强,对患者安全性高、成本低。针对其缺点近来有新的研究,Gravius等[61]应用基质辅助激光解吸电离飞行时间质谱技术检测与嗜中性粒细胞浸润区域相关的肽,代替嗜中性粒细胞计数,发现假体周围感染患者存在大量嗜中性粒细胞相关的特异性峰。Kölbel等[62]利用CD15特异性单克隆抗体免疫组化法鉴定中性粒细胞,发现使用CD15定量软件分析灵敏度为83%,特异性为86%,准确性为84.6%,曲线下面积为为0.878。自动化程序相比人工显微镜下观察快速且省时省力。 2.2.6 影像学检查 见表1。普通X射线检查是诊断假体周围感染的常规和基础手段,用以排除包括假体断裂和松动等其他原因引起的关节疼痛[63]。通过普通X射线检查可在骨水泥表面观察到假体错位、骨膜反应、水泥裂缝、经皮窦道等[64-65]。动态系列X射线片检查对假体周围感染诊断及治疗具有重要的参考价值。6-12个月内假体移位大于2 mm高度怀疑感染,但是其特异性低,无法区分感染性和无菌性骨质溶解,无法在急性假体周围感染和早期诊断中作出判断。 超声检查应用广泛、方便快速、不受金属伪影的影响、无辐射、价格低廉、尤其适合髋关节置换患者。超声引导的关节抽吸和引流精确性高,但是受限于灵敏度和特异性,目前超声在诊断关节置换术后假体周围感染方面诊断价值有限。 计算机断层扫描较X射线片更为敏感,对识别皮下脓肿、关节积液、窦道、骨质侵蚀和假体松动具有明显优 势[65]。与超声检查类似,计算机断层扫描扫描可指导关节抽吸和引流。主要缺点是存在金属伪影,影响图像质量。计算机断层扫描诊断假体周围感染灵敏度和特异性低。 磁共振成像检查能提供更好的分辨率,与计算机断层扫描相比进一步缩减伪影,成像质量更高[66]。随着假体植入物是非铁磁化,磁共振成像的使用已逐渐增加,但是其特异性低。 核成像诊断骨科感染已被广泛利用,据报道具有很好的诊断性能,但其各研究数据不统一。有研究认为 99标记的二膦酸盐在骨矿物质基质表面上的摄取增加;然而,在无菌性炎症、骨重建、骨折和无菌性松动的情况下也引起骨成像改变[66]。相反,另外一些研究认为99Tcm标记骨扫描在诊断假体周围感染时有较高的准确度和特异性[67-68],且具有高的阴性预测价值:即如果扫描结果是正常的,则实际上排除了松动。体外标记的白细胞扫描使用铟111氧喹诺酮或99Tc-六甲基亚丙基胺肟,当白细胞扫描再联合硫胶体骨髓扫描诊断假体周围感染的准确性更高[66],该方法具有88%-98%的总体精度,是当前首选的诊断方法[64]。最近发表的几项关于白细胞闪烁扫描的研究提示当正确地获取图像以及正确地解释扫描时,诊断准确度>93%[69-70]。其次,使用单克隆抗体或抗体片段的抗粒细胞闪烁扫描术可能是另一种有吸引力的检测假体周围感染的方法。Pakos等[71]的一项关于抗粒细胞闪烁扫描诊断性能的meta分析报告的敏感性和特异性分别为83%和80%,但对中性粒细胞减少患者无效。如果使用抗生素则会降低该方法在临床中的可用性[72]。Tcm 单光子发射计算机断层扫描能够更好地定位放射性示踪剂摄取。当与白细胞扫描结合时,单光子发射计算机断层扫描的特异性显著增加[73]。荧光脱氧葡萄糖正电子发射断层扫描提供高分辨率成像,具有优异的假体周围感染诊断灵敏度[74]。Parvizi等[29]采用对全髋关节置换术后髋关节疼痛的患者以18F-fluoro-2-deoxyglucose标记的正电子发射断层扫描检测,以鉴别是细菌性还是无菌性病变,结果发现荧光脱氧葡萄糖正电子发射断层扫描对假体周围感染诊断的敏感性和特异性分别为85%和93%。Del Arco等[75]也报道荧光脱氧葡萄糖正电子发射断层扫描在诊断髋关节或膝关节假体感染的敏感性和特异性分别为82.1%和86.6%,同时也得到多数学者的认 同[66]。其他研究方法包括使用99Tc-白细胞介素8、111-生物素闪烁扫描、99Tc-环丙沙星、99Tc-重组膜联蛋白闪烁照相术、放射性标记泛素的合成片段、99Tc-标记的噬菌体和胸苷激酶等扫描方法。这些方法目前还没有得到可靠的证实,可能在将来假体周围感染的诊断中发挥作用[66]。 "

| [1] Parvizi J,Zmistowski B,Berbari EF, et al. New definition for periprosthetic joint infection: from the Workgroup of theMusculoskeletal Infection Society. Clin Orthop Relat Res. 2011; 469(11): 2992-2994.[2] 袁俊,冯建民. 人工关节置换术后假体周围感染的病原学诊断研究进展[J].中华关节外科杂志:电子版,2016,10(4):432-435.[3] Peel TN,Dylla BL,Hughes JG,et al.Improved diagnosis ofprosthetic joint infection by culturing periprosthetic tissuespecimens in blood culture bottles. MBio. 2016;7(1):e01776-15. [4] Tunney MM,Patrick S,Gorman SP,et al. Improved detection ofinfection in hip replacements. A currently underestimated problem. J Bone Joint Surg Br. 1998; 80(4): 568-572.[5] Rak M, Kav?I? M, Trebše R, et al. Detection of bacteria with molecular methods in prosthetic joint infection: sonication fluid better than periprosthetic tissue. Acta Orthop. 2016;87(4):339-345. [6] Cross MC, Kransdorf MJ, Chivers FS, et al. Utility of percutaneous joint aspiration and synovial biopsy in identifying culture-positive infected hip arthroplasty. Skeletal Radiol. 2014;43: 165-168.[7] Fink B, Gebhard A, Fuerst M, et al. High diagnostic value of synovial biopsy in periprosthetic joint infection of the hip. Clin Orthop Relat Res. 2013;471: 956-964.[8] 张宁,赵翔,周鑫叠,等.关节液与组织培养在髋膝关节置换术后感染诊断价值的Meta分析[J]. 中华关节外科杂志:电子版,2014,8(1):72-76.[9] Durmaz G, Us T, Aydinli A, et al. Optimum detection times for bacteria and yeast species with the BACTEC 9120 aerobic blood culture system: evaluation for a 5-year period in a Turkish university hospital. J Clin Microbiol. 2003;41(2):819-821.[10] Font-Vizcarra L, García S, Martínez-Pastor JC, et al. Blood culture flasks for culturing synovial fluid in prosthetic joint infections. Clin Orthop Relat Res. 2010;468(8):2238-2243.[11] Geller JA, MacCallum KP, Murtaugh TS, et al. Prospective Comparison of Blood Culture Bottles and Conventional Swabs for Microbial Identification of Suspected Periprosthetic Joint Infection.J Arthroplasty. 2016;31(8):1779-1783.[12] Squire MW,Della Valle CJ,Parvizi J. Preoperative diagnosis of periprosthetic joint infection: role of aspiration. AJR Am J Roentgenol. 2011; 196(4): 875-879.[13] Bémer P,Plouzeau C,Tande D,et al. Evaluation of 16S rRNAgene PCR sensitivity and specificity for diagnosis of prosthetic joint infection: a prospective multicenter cross- sectional study. J Clin Microbiol. 2014; 52(10): 3583-3589.[14] Ghebremedhin B, Layer F, König W,et al. Genetic classification and distinguishing of Staphylococcus species based on different partial gap, 16S rRNA, hsp60, rpoB,sodA, and tuf gene sequences. J Clin Microbiol. 2008;46(3):1019-1025.[15] Greenwood-Quaintance KE,Uhl JR,Hanssen AD,et al. Diagnosis of Prosthetic Joint Infection by Use of PCR-Electrospray Ionization Mass Spectrometry. J Clin Microbiology. 2014; 52(2):642-649.[16] Prieto-Borja L, Rodriguez-Sevilla G, Auñon A, et al. Evaluation of a commercial multiplex PCR (Unyvero i60®) designed for the diagnosis of bone and joint infections using prosthetic-joint sonication. Enferm Infecc Microbiol Clin. 2016,pii: S0213-005X(16)30287-7. [17] Hischebeth GT, Randau TM, Buhr JK, et al. Unyvero i60 implant and tissue infection (ITI) multiplex PCR system in diagnosing periprosthetic joint infection. J Microbiol Methods. 2016;121:27-32.[18] Lourtet-Hascoëtt J, Bicart-See A, Félicé MP,et al. Is Xpert MRSA/SA SSTI real-time PCR a reliable tool for fast detection of methicillin- resistant coagulase-negative staphylococci in periprosthetic joint infections? Diagn Microbiol Infect Dis. 2015;83(1):59-62.[19] Larsen LH, Lange J, Xu Y, et al. Optimizing culture methods for diagnosis of prosthetic joint infections: a summary of modifications and improvements reported since 1995. J Med Microbiol. 2012;61(Pt 3): 309-316.[20] Bizzini A, Greub G. Matrix-assisted laser desorption ionization time-of-flight mass spectrometry, a revolution in clinical microbial identification. Clin Microbiol Infect. 2010;16(11):1614-1619.[21] Peel TN, Cole NC, Dylla BL,et al. Matrix-assisted laser desorption ionization time of flight mass spectrometry and diagnostic testing forprosthetic joint infection in the clinical microbiology laboratory. Diagn Microbiol Infect Dis. 2015;81(3):163-168.[22] Melendez DP, UhI JR, Greenwood-Quaintance KE, et al. Detection of prosthetic joint infection by use of PCR-electrospray ionization mass spectrometry applied tosynovial fluid. J Clin Microbiol. 2014,52(6): 2202-2205. [23] Nistico L, Hall-Stoodley L, Stoodley P. Imaging bacteria and biofilms on hardware and periprosthetic tissue in orthopedic infections. Methods Mol Biol. 2014;1147:105-126.[24] Hogdall D, Hvolris JJ, Christensen L. Improved detection methods for infected hip joint prostheses. APMIS.2010;118(11):815-823.[25] Trampuz A, Steinhuber A, Wittwer M, et al. Rapid diagnosis of experimental meningitis by bacterial heat production in cerebrospinal fluid. BMC Infect Dis. 2007;7:116.[26] Corvec S, Portillo ME, Pasticci BM,et al. Epidemiology and new developments in the diagnosis of prosthetic joint infection. Int J Artif Organs. 2012;35(10):923-934.[27] Parvizi J, Della Valle CJ. AAOS clinical practice guideline: Diagnosis and treatment of periprosthetic joint infections of the hip and knee. J Am Acad Orthop Surg. 2010;18:771-772.[28] Saltzman MD, Marecek GS, Edwards SL, et al. Infection after shoulder surgery. J Am Acad Orthop Surg. 2011;19:208-218.[29] Parvizi J, Adeli B, Zmistowski B, et al. Management of periprosthetic joint infection: the current knowledge: AAOS exhibit selection.J Bone Joint Surg Am. 2012;94(14):e104. [30] Chen A, Fei J, Deirmengian C. Diagnosis ofperiprosthetic infection: novel developments. J Knee Surg. 2014; 27(4): 259-265.[31] Randau TM, Friedrich MJ, Wimmer MD, et al. Interleukin-6 in serum and in synovial fluid enhances the differentiation between periprosthetic joint infection and aseptic loosening. PLoS One. 2014; 9(2):e89045.[32] Elgeidi A, Elganainy AE, Abou Elkhier N,et al. Interleukin-6 and other inflammatory markers in diagnosis of periprosthetic joint infection. Int Orthop. 2014;38(12):2591-2595.[33] Matsen Ko L, Parvizi J.Diagnosis of Periprosthetic Infection: Novel Developments. Orthop Clin North Am. 2016;47(1):1-9.[34] Frangiamore SJ, Saleh A, Grosso MJ,et al. Neer Award 2015: Analysis of cytokine profiles inthe diagnosis of periprosthetic joint infections of theshoulder. J Shoulder Elbow Surg. 2016. pii: S1058-2746(16)30244-0. [35] Grosso MJ, Frangiamore SJ, Saleh A,et al. Poor utility of seruminterleukin-6 levels to predict indolent periprosthetic shoulderinfections. J Shoulder Elbow Surg .2014;23(9):1277-1281.[36] Ahmad SS, Shaker A, Saffarini M,et al. Accuracy of diagnostictests for prosthetic joint infection: a systematic review. Knee Surg Sports Traumatol Arthrosc. 2016;24(10):3064-3074. [37] Galliera E, Drago L, Vassena C, et al. Toll-Like Receptor 2 in Serum: a Potential Diagnostic Marker of Prosthetic Joint Infection? J Clin Microbiol. 2014; 52(2): 620-623.[38] Pajarinen J, Jamsen E, Konttinen YT, et al. Innate Immune Reactions in Septic and Aseptic Osteolysis Around Hip Implants. J Long Term Eff Med Implants. 2014; 24(4): 283-296.[39] Deirmengian C, Kardos K, Kilmartin P,et al. Diagnosing Periprosthetic Joint Infection Has the Era of the Biomarker Arrived?.Clin Orthop Relat Res. 2014;472:3254-3262.[40] Bonanzinga T, Zahar A, Dütsch M, et al. How Reliable Is the Alpha-defensin Immunoassay Test for Diagnosing Periprosthetic Joint Infection? A Prospective Study.Clin Orthop Relat Res. 2016. [Epub ahead of print][41] Wyatt MC, Beswick AD, Kunutsor SK,et al. The Alpha-Defensin Immunoassay and Leukocyte Esterase Colorimetric Strip Test for the Diagnosis of Periprosthetic Infection: A Systematic Review and Meta-Analysis. J Bone Joint Surg Am. 2016;98(12):992-1000. [42] Deirmengian C, Kardos K, Kilmartin P, et al. The alpha-defensin test for periprosthetic joint infection outperforms the leukocyte esterase test strip.Clin Orthop Relat Res. 2015;473(1):198-203. [43] Deirmengian C, Kardos K, Kilmartin P, et al. Combined measurement of synovial fluid α-Defensin and C-reactive protein levels: highly accurate fordiagnosing periprosthetic joint infection. J Bone Joint Surg Am. 2014;96(17):1439-1445. [44] Bingham J, Clarke H, Spangehl M,et al. The alpha defensin-1 biomarker assay can be used to evaluate the potentially infected total jointarthroplasty. Clin Orthop Relat Res. 2014;472(12):4006-4009. [45] Deirmengian C, Kardos K, Kilmartin P, et al. The Alpha-defensin Test for Periprosthetic Joint Infection Responds to a Wide Spectrum of Organisms. Clin Orthop Relat Res. 2015;473(7):2229-2235. [46] Shahi A, Parvizi J, Kazarian GS,et al. The Alpha-defensin Test for Periprosthetic Joint Infections Is Not Affected by Prior Antibiotic Administration.Clin Orthop Relat Res. 2016;474(7):1610-1615.[47] Parvizi J, Jacovides C, Antoci V, et al. Diagnosis of periprosthetic joint infection: the utility of a simple yet unappreciated enzyme.J Bone Joint Surg Am. 2011;93(24):2242-2248.[48] Colvin OC, Kransdorf MJ, Roberts CC, et al.Leukocyte esterase analysis in the diagnosis of joint infection: can we make a diagnosis using a simpleurine dipstick? Skeletal Radiol. 2015;44(5):673-677.[49] Wetters NG, Berend KR, Lombardi AV,et al. Leukocyte esterase reagent strips for the rapid diagnosis of periprosthetic joint infection.J Arthroplasty. 2012;27(8 Suppl):8-11.[50] Shafafy R, McClatchie W, Chettiar K,et al.Use of leucocyte esterase reagent strips in the diagnosis or exclusion of prosthetic jointinfection. Bone Joint J. 2015;97-B(9):1232-1236.[51] Friedrich MJ, Randau TM, Wimmer MD, et al. Lipopolysaccharide- binding protein: a valuable biomarker inthe differentiation between periprosthetic jointinfection and aseptic loosening?Int Orthop. 2014; 38(10):2201-2207.[52] Galliera E, Drago L, Marazzi MG, et al. Soluble urokinase-type plasminogen activator receptor (suPAR) as new biomarker of the prosthetic jointinfection: correlation with inflammatory cytokines. Clin Chim Acta. 2015;441:23-28.[53] Morawietz L, Schröder JH, Dynybil C,et al. Proposal for a histopathological consensus classification of the periprosthetic interface membrane. J Clin Pathol. 2006;59:591-597. doi: 10.1136/jcp.2005.027458. [54] Janz V, Wassilew GI, Hasart O,et al. Evaluation of sonicate fluid cultures in comparison to histological analysis of theperiprosthetic membrane for the detection of periprosthetic joint infection. Int Orthop. 2013;37(5):931-936.[55] Claassen L, Ettinger S, Pastor MF,et al. The value of arthroscopic neosynovium biopsies to diagnose periprosthetic knee joint low-grade infection. Arch Orthop Trauma Surg. 2016;136(12):1753-1759. [56] Kwiecien G, George J, Klika AK,et al. Intraoperative Frozen Section Histology: Matched for Musculoskeletal Infection Society Criteria. J Arthroplasty. 2016. pii: S0883-5403(16)30286-8. [57] Zimmerli W, Trampuz A, Ochsner PE. Prosthetic-joint infections. N Engl J Med. 2004;351(16):1645-1654.[58] George J, Kwiecien G, Klika AK,et al.Are Frozen Sections and MSIS Criteria Reliable at the Time of Reimplantation of Two-stageRevision Arthroplasty?Clin Orthop Relat Res. 2016;474(7):1619-1626. [59] Tang X, Wang Q, Wang H,et al. [Comparison and estimation of different diagnostic methods in detecting the presence of periprosthetic joint infection]. Zhonghua Wai Ke Za Zhi. 2016;54(4):251-257.[60] Banke IJ, Stade N, Prodinger PM,et al.[Synovial biomarkers for differential diagnosis of painful arthroplasty].Orthopade. 2015;44(12): 936-938, 940-941. [61] Gravius S, Randau TM, Casadonte R,et al. Investigation of neutrophilic peptides in periprosthetic tissue by matrix-assisted laser desorption ionisationtime-of-flight imaging mass spectrometry. Int Orthop. 2015;39(3):559-567.[62] Kölbel B, Wienert S, Dimitriadis J, et al. [CD15 focus score for diagnostics of periprosthetic joint infections : Neutrophilic granulocytesquantification mode andthe development of morphometric software (CD15 quantifier)]. Z Rheumatol. 2015;74(7):622-630.[63] Osmon DR, Berbari EF, Berendt AR, et al; Infectious Diseases Society of America. Executive summary: diagnosis and management of prosthetic joint infection: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis. 2013; 56(1): 1-10.[64] Brause BD. Infection with Prostheses in Bones and Joints.In: Mandell GL, Bennett JE, Dolin R, (eds). Mandell,Douglas and Bennett’s principles and practice of infectious diseases, 7th edition (vol 1). Philadelphia: Elsevier Inc; 2010: 1469-1474.[65] Sendi P, Zimmerli W. Diagnosis of periprosthetic joint infections in clinical practice. Int J Artif Organs. 2012;35(10): 913-922.[66] Gemmel F, Van den Wyngaert H, Love C,et al. Prosthetic joint infections: radionuclide state-of-the-art imaging. Eur J Nucl Med Mol Imaging. 2012; 39(5): 892-909.[67] Trevail C, Ravindranath-Reddy P, Sulkin T,et al. An evaluation of the role of nuclear medicine imaging in the diagnosis of periprostheticinfections of the hip. Clin Radiol. 2016;71(3):211-219. [68] Ouyang Z, Li H, Liu X, et al.Prosthesis infection: diagnosis after total joint arthroplasty with three-phase bonescintigraphy. Ann Nucl Med. 2014;28(10):994-1003.[69] Glaudemans AW, de Vries EF, Vermeulen LE, et al. A large retrospective single centre study to define best acquisition protocols and interpretation criteria for white blood cell scintigraphy with99mTc-HMPAO-labelled leukocytes in musculoskeletal infections. Eur J Nucl Med Mol Imaging. 2013;40: 1760.[70] Erba PA, Glaudemans AW, Veltman NC, et al. Image acquisition and interpretation criteria for99mTc-HMPAO-labelled white blood cell scintigraphy: results of a multicenter study. Eur J Nucl Med Mol Imaging. 2014; 41: 615.[71] Pakos EE, Trikalinos TA, Fotopoulos AD, et al. Prosthesis infection: diagnosis after total joint arthroplasty with antigranulocytescintigraphy with 99mTc-labeled monoclonal antibodies-a meta-analysis. Radiology. 2007; 242 (1): 101-108.[72] Al Mohajer M, Darouiche RO. Darouiche.The expanding horizon of prosthetic joint infections. J Appl Biomater Funct Mater. 2014;12(1): 1-12.[73] Filippi L, Schillaci O. Usefulness of hybrid SPECT/CT in 99mTc-HMPAO-labeled leukocyte scintigraphy for bone and joint infections. J Nucl Med. 2006; 47(12):1908-1913.[74] Zhuang H, Yang H, Alavi A. Critical role of 18F-labeled fluorodeoxyglucose PET in the management of patients with arthroplasty. Radiol Clin North Am. 2007; 45(4):711-718, vii.[75] Del Arco A, Bertrand ML. The diagnosis of periprosthetic infection. Open Orthop J. 2013;7:178-183. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zhang Yu, Tian Shaoqi, Zeng Guobo, Hu Chuan. Risk factors for myocardial infarction following primary total joint arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1340-1345. |
| [3] | Li Dadi, Zhu Liang, Zheng Li, Zhao Fengchao. Correlation of total knee arthroplasty efficacy with satisfaction and personality characteristics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1346-1350. |
| [4] | Wei Wei, Li Jian, Huang Linhai, Lan Mindong, Lu Xianwei, Huang Shaodong. Factors affecting fall fear in the first movement of elderly patients after total knee or hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1351-1355. |
| [5] | Wang Jinjun, Deng Zengfa, Liu Kang, He Zhiyong, Yu Xinping, Liang Jianji, Li Chen, Guo Zhouyang. Hemostatic effect and safety of intravenous drip of tranexamic acid combined with topical application of cocktail containing tranexamic acid in total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1356-1361. |
| [6] | Xiao Guoqing, Liu Xuanze, Yan Yuhao, Zhong Xihong. Influencing factors of knee flexion limitation after total knee arthroplasty with posterior stabilized prostheses [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1362-1367. |
| [7] | Huang Zexiao, Yang Mei, Lin Shiwei, He Heyu. Correlation between the level of serum n-3 polyunsaturated fatty acids and quadriceps weakness in the early stage after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1375-1380. |
| [8] | Zhang Chong, Liu Zhiang, Yao Shuaihui, Gao Junsheng, Jiang Yan, Zhang Lu. Safety and effectiveness of topical application of tranexamic acid to reduce drainage of elderly femoral neck fractures after total hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1381-1386. |
| [9] | Yuan Jiawei, Zhang Haitao, Jie Ke, Cao Houran, Zeng Yirong. Underlying targets and mechanism of Taohong Siwu Decoction in prosthetic joint infection on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1428-1433. |
| [10] | Chen Junming, Yue Chen, He Peilin, Zhang Juntao, Sun Moyuan, Liu Youwen. Hip arthroplasty versus proximal femoral nail antirotation for intertrochanteric fractures in older adults: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1452-1457. |
| [11] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [12] | Zhao Zhongyi, Li Yongzhen, Chen Feng, Ji Aiyu. Comparison of total knee arthroplasty and unicompartmental knee arthroplasty in treatment of traumatic osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 854-859. |
| [13] | Liu Shaohua, Zhou Guanming, Chen Xicong, Xiao Keming, Cai Jian, Liu Xiaofang. Influence of anterior cruciate ligament defect on the mid-term outcome of fixed-bearing unicompartmental knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 860-865. |
| [14] | Zhang Nianjun, Chen Ru. Analgesic effect of cocktail therapy combined with femoral nerve block on total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 866-872. |
| [15] | Yuan Jun, Yang Jiafu. Hemostatic effect of topical tranexamic acid infiltration in cementless total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 873-877. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||