Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (32): 8496-8501.doi: 10.12307/2026.873
Previous Articles Next Articles
Hydrogel-based drug delivery systems for rheumatoid arthritis treatment
Li Minghui, Qie Haoyu, Pan Min, Bi Ruijie, Lyu Xiaomeng, Zhang Haoya, Han Yifei
- Binzhou Polytechnic College, Binzhou 256603, Shandong Province, China
-
Accepted:2026-01-26Online:2026-11-18Published:2026-04-28 -
Contact:Li Minghui, MS, Teaching assistant, Binzhou Polytechnic College, Binzhou 256603, Shandong Province, China -
About author:Li Minghui, MS, Teaching assistant, Binzhou Polytechnic College, Binzhou 256603, Shandong Province, China
CLC Number:
Cite this article
Li Minghui, Qie Haoyu, Pan Min, Bi Ruijie, Lyu Xiaomeng, Zhang Haoya, Han Yifei. Hydrogel-based drug delivery systems for rheumatoid arthritis treatment[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8496-8501.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
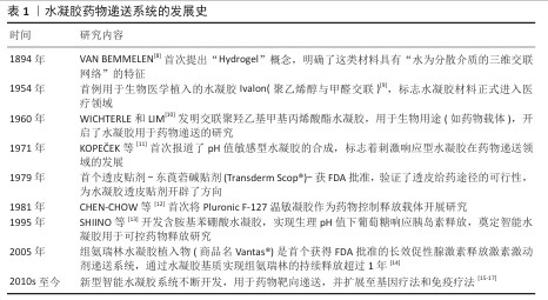
2.1 水凝胶药物递送系统的发展史 见表1。 2.2 常见类风湿性关节炎治疗药物 作为一种慢性的自身免疫性疾病,类风湿性关节炎的典型特征是关节部位持续性炎症反应,常表现为疼痛、肿胀及活动受限,长期炎症甚至引起关节结构破坏,导致畸形和残疾。当前类风湿性关节炎的临床干预手段主要包括药物治疗、物理治疗与手术治疗[18]。药物治疗作为主要治疗方法,根据作用机制可分为非类固醇抗炎药、糖皮质激素、改善病情抗风湿药和生物制剂四类。近年来,中国在新型类风湿性关节炎治疗药物研发领域取得瞩目成就,如硫酸艾玛昔替尼片和泰它西普。硫酸艾玛昔替尼片(艾速达?)是国内首个用于类风湿性关节炎治疗的高选择性Janus激酶1抑制剂,该药物精准抑制Janus激酶1激酶活性,阻断Janus激酶/信号传导和转录激活蛋白信号通路,从而减少炎症因子(如白细胞介素6、白细胞介素23)的产生,发挥抗炎和免疫调节作用[19]。作为新型B细胞双靶点制剂,泰它西普可同时抑制B细胞活化关键因子——B淋巴细胞刺激因子和增殖诱导配体,从而调控B细胞分化和自身抗体产生,恢复免疫耐受[20]。常见类风湿性关节炎治疗药物见表2。 "

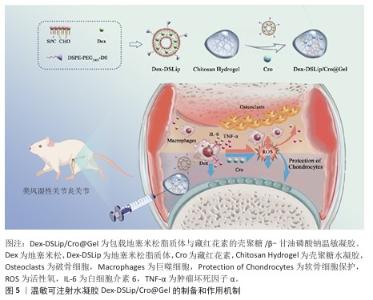
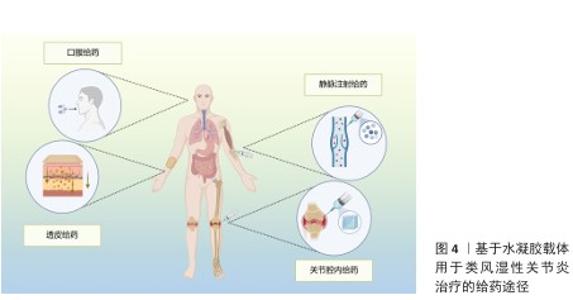
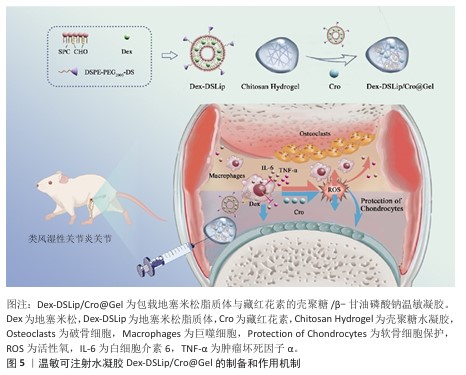
2.3.1 口服给药 口服给药因患者依从性高而成为临床常用给药方式,但多数药物(尤其是蛋白质类药物)易在胃肠道酸性环境及酶解作用下失活,难以抵达靶部位发挥作用。水凝胶载体可通过保护作用及pH值响应性设计克服这一局限[37-38]。CARRILLO-CONDE等[23]开发了基于聚(甲基丙烯酸)和乙二醇的pH值响应性水凝胶微粒,用于递送抗肿瘤坏死因子α抗体,该系统在胃部酸性环境中释放较少的药物,避免药物在胃内失活,而在小肠中性环境中体系快速释放药物,保护了药物活性;体外实验表明,该系统在pH值=3.2环境中前90 min内药物释放量为10%-50%,而在pH值为中性(7.4)环境中的药物释放量明显增加。JIANG等[39]报道了青藤碱-甘草酸通过静电相互作用自组装形成的口服纳米水凝胶(S-G Hydrogel)体系,该纳米水凝胶在胃酸微环境中可稳定维持凝胶状态并实现药物缓慢释放,不仅显著提升青藤碱的生物利用度,还能有效降低药物的胃肠道毒性;进入肠道后,该体系仍保持缓释特性,可避免血药浓度出现峰谷波动;在动物实验中,该纳米水凝胶通过抑制中性粒细胞过度活化、促进中性粒细胞凋亡进程及阻断中性粒细胞胞外陷阱生成,显著减轻胶原诱导性关节炎及佐剂性关节炎模型小鼠的关节炎症反应、软骨侵蚀及骨破坏程度,疗效优于单一成分给药,并且无明显肝肾毒性。 2.3.2 经皮给药 经皮给药能够避免首过代谢,同时提高患者的依从性,然而,皮肤的角质层是药物渗透的主要屏障,影响药物吸收。水凝胶系统可作为胶凝剂,延长药物与皮肤的接触时间,增强渗透性,从而提高治疗效果[40-41]。KHAN等[42]开发了一种基于Eudragit? L100(EL100)和Carbopol? 934的pH值响应性纳米粒-原位水凝胶系统,用于经皮递送布洛芬,该系统能够实现药物在炎症部位的靶向释放,显著提高局部药物浓度并减少全身不良反应,体外实验表明药物在72 h内呈pH值依赖性持续释放;体内实验显示,该系统在急性及慢性类风湿性关节炎模型中的治疗效果显著优于市售布洛芬乳膏,并且无明显皮肤刺激性。此外,KHAN等[43]还开发了一种基于聚己内酯-聚乙二醇-聚己内酯三嵌段共聚物的反向纳米胶束系统,递送大分子药物英夫利昔单抗,并进一步载入含桉树油的Carbopol-934水凝胶中以增强皮肤渗透,药物在72 h内呈pH值依赖性持续释放;在完全弗氏佐剂诱导的类风湿性关节炎小鼠模型中,该制剂显著减轻关节肿胀、降低炎症,改善疼痛阈值和肌肉功能,并且无明显皮肤刺激性。GHARAT等[44]开发了一种边缘活化型乙醇体凝胶系统,用于经皮联合递送姜黄素与环孢素治疗类风湿性关节炎,该系统采用乙醇注入法制备了姜黄素与环孢素纳米粒子,并进一步载入Carbopol? Ultrez 10NF凝胶中,形成稳定、透明的半固体剂型,体外实验显示该系统的释放符合Hixson-Crowell 模型,细胞实验证实其显著抑制促炎因子(白细胞介素6、肿瘤坏死因子α等)、上调抗炎因子白细胞介素10;在完全弗氏佐剂诱导类风湿性关节炎大鼠模型中,28 d内可有效减轻关节肿胀、改善血液指标,疗效优于市售甲氨蝶呤凝胶,并且无皮肤刺激,可降低环孢素肾毒性,展现优异得生物相容性。 尽管经皮给药在类风湿性关节炎治疗中展现一定的潜力,但仍存在药物渗透效率低、难以靶向深层组织以及剂量控制困难等局限性。近年来,微针透皮递送系统因独特的物理穿孔机制成为研究热点,该系统通过微米级针体可逆性穿透角质层屏障,在无痛前提下形成药物扩散通道,显著提升生物大分子的透皮效率;同时,其针体长度可精准调控(通常200-600 μm),实现药物向真皮层及浅表皮下组织的靶向递送,促进药物经毛细血管/淋巴管循环富集于关节滑膜等炎症部位;此外,基于可溶性或涂层型微针的设计,该系统可通过针体材料降解动力学或表面载药量精确控制药物释放速率与总量,有效规避全身毒性风险[45-49]。LI等[48]将雷公藤甲素载入透明质酸-聚乙烯吡咯烷酮可溶性微针系统,借助微针的物理穿刺作用与基质快速溶解特性的双重机制,实现药物在关节局部的持续释放,在佐剂性关节炎大鼠模型中,该系统可显著降低关节肿胀程度、减轻骨侵蚀并下调炎症因子水平,并且肝肾毒性明显低于传统给药方式,表明中药单体的经皮递送策略能够在保证疗效的同时规避口服给药带来的毒副作用。HUA等[50]将小檗碱与青藤碱共同包封于活性氧响应型聚乳酸羟基乙酸共聚物-酮缩硫醇-聚乙二醇微球中,再将其嵌入可溶性微针,微针插入皮肤后,微球可在炎症部位高活性氧微环境中触发级联释药过程,通过联合抑制 M1型巨噬细胞极化与血管内皮迁移使胶原诱导性关节炎小鼠的滑膜增生、软骨破坏及骨侵蚀得到显著改善,该结果验证了“抗炎+抗血管新生”双靶点类风湿性关节炎治疗策略经皮实施的可行性。LIU等[51]报道了一种内置气泡驱动的活性微针系统,在微针基质中预置碳酸氢钠/柠檬酸组分,当微针刺入皮肤后可瞬间产生CO?微气泡,形成局部“微泵”效应,促使甲氨蝶呤在皮下的主动渗透与快速释放,在数分钟内实现深层渗透并快速富集于炎症关节;在佐剂性关节炎大鼠模型中,仅需贴敷90 s即能达到接近皮下注射的治疗效果,并且皮肤屏障可在2 h内完全恢复。 微针透皮递送系统不仅可用于递送传统化学药物,还可有效递送抗体、基因治疗药物及核酸类药物等生物大分子,从而拓展类风湿性关节炎的治疗选择。有研究构建了基于明胶甲基丙烯酰化的可溶性微针贴片,通过微针介导的经皮给药模式实现双特异性纳米抗体(白细胞介素6受体-肿瘤坏死因子α抗体)的协同递送,该抗体可精准靶向肿瘤坏死因子α 和白细胞介素6受体以干预类风湿性关节炎的进展,该微针可显著提升纳米抗体的生物利用度并延长半衰期,有效中和肿瘤坏死因子α诱导的细胞毒性,可抑制核因子κB与Janus激酶-信号传导和转录激活蛋白3炎症通路的激活;在胶原诱导性关节炎大鼠模型中,该微针系统在缓解关节炎症及促进骨与软骨再生方面疗效显著,与注射给药的效果相当,同时规避了频繁注射带来的临床不便与不良反应风险[51-52]。侵袭性成纤维样滑膜细胞的异常增殖会加速类风湿性关节炎的进展,HUA等[53]利用透明质酸可溶性微针系统协同递送促凋亡基因 PUMA质粒与雷公藤红素,以调控成纤维样滑膜细胞和巨噬细胞,通过转染成纤维样滑膜细胞上调p53凋亡调控因子(PUMA)表达,诱导成纤维样滑膜细胞高效凋亡,雷公藤红素可通过阻断核因子κB通路激活抑制巨噬细胞的炎症反应;在胶原诱导性关节炎小鼠模型中,该微针可清除成纤维样滑膜细胞、抑制M1型巨噬细胞活化、减轻滑膜增生,并减少骨和软骨侵蚀。 2.3.3 关节内给药 关节内注射可直接将药物递送至病变关节,减少全身不良反应,然而,游离药物在关节内的保留时间较短,限制了其疗效。水凝胶系统能够延长药物在关节内的留存时间,从而提升药效[54]。此外,通过化学手段对水凝胶进行改性,使它能够清除类风湿性关节炎进展中产生的炎症递质(如一氧化氮),可使水凝胶具备对复杂类风湿性关节炎微环境的调节能力,进一步提升治疗效果。利用水凝胶进行关节内给药,可实现多种药物共递送至病灶部位,达到更好的类风湿性关节炎治疗效果。例如,以壳聚糖/β-甘油磷酸钠温敏凝胶做为载体,包载地塞米松脂质体与藏红花素,从而制备温敏可注射水凝胶Dex-DSLip/Cro@Gel(图5),对佐剂性关节炎大鼠单次注射该水凝胶7 d后,大鼠踝部肿瘤坏死因子α和白细胞介素6水平降低70%、骨侵蚀程度减少近 50%,有效阻断了炎症-骨破坏恶性循环[55]。除了化学药物,水凝胶凭借优异的生物相容性、可降解性以及独特的三维网络结构,成为多肽和酶类生物分子的理想递送载体。针对中性粒细胞胞外诱捕网这一关键靶点,WANG团队[56]利用氧化透明质酸和羧甲基壳聚糖构建水凝胶用于脱氧核糖核酸酶Ⅰ的递送,通过持续缓释脱氧核糖核酸酶Ⅰ降解中性粒细胞胞外诱捕网,阻断其对巨噬细胞M1型极化的刺激作用。LEE等[57]将TAP2 (Toll样受体4拮抗肽)交联于透明质酸水凝胶中以延长肽段在关节的滞留时间,持续阻断核因子κB核转位。该团队进一步将环状噬菌体展示衍生抑制肽与透明质酸共价偶联后嵌入温敏聚乙二醇基水凝胶,在胶原诱导性关节炎大鼠中单次给药后,衍生抑制肽关节滞留时间较游离肽延长约20倍,6周内肿瘤坏死因子α和白细胞介素6水平持续降低,软骨厚度恢复至正常水平的91% [27]。 在利用水凝胶进行类风湿性关节炎的治疗时,应充分考虑到治疗分子在病灶部位的可控释放动力学。类风湿性关节炎关节内的病理进程呈现显著的空间异质性和时间动态性(炎症的急性爆发与缓解交替),因此,设计具有“精准调控能力”的水凝胶以实现治疗分子的按需、精准释放,成为了提升类风湿性关节炎治疗效果的关键方向。LIU等[58]将介孔聚多巴胺纳米粒嵌入透明质酸-聚乙二醇网络,构建具有智能响应特性的给药系统。纳米粒不仅具备活性氧清除能力,更可作为“智能阀门”响应关节氧化应激水平,当急性期氧化应激升高时加速释放甲氨蝶呤,应激降低时转为缓慢释放,实现按需释药。SINGH等[59]通过将地塞米松-脂质纳米粒包载于基质金属蛋白酶响应型水凝胶中,构建了具有“双屏障” 特性的智能给药系统,该策略一方面凭借CD44靶向能力增强对滑膜组织的特异性富集,另一方面通过对关节局部基质金属蛋白酶2/9水平的动态感知,实现智能释药,即在炎症急性发作期,高表达的基质金属蛋白酶2/9可触发水凝胶网络降解,促使地塞米松-脂质纳米粒快速释放以遏制炎症进展;当炎症缓解、基质金属蛋白酶水平降低时,地塞米松-脂质纳米粒的释放速率则随之减缓,避免药物过量暴露。 2.3.4 其他创新应用 类风湿性关节炎的治疗亟需同步解决免疫失衡与组织破坏两大核心难题,而水凝胶递送间充质干细胞的策略受到关注。水凝胶三维网络可模拟干细胞生态位,屏蔽损伤,实现间充质干细胞的精准定植,提升干细胞疗效[60]。已有研究将MnCoO纳米酶嵌入明胶-聚赖氨酸水凝胶,在氧化应激时,纳米酶高效分解局部H?O?供氧并清除活性氧;应激降低后,水凝胶通过可逆亚胺键平稳释放骨髓间充质干细胞,维持成骨微环境[61]。SHI等[62]将间充质干细胞包埋于海藻酸钙水凝胶微球,构建“免疫耐受舱”,微球以Ca2?交联,在炎症高峰期稳定释放间充质干细胞,间充质干细胞通过上调白细胞介素10并激活CD39/CD73-腺苷通路,使树突状细胞稳定为(耐受性树突状细胞,并不断扩增调节性T细胞;随着炎症指标回落,水凝胶微球失去高钙微环境而逐步降解,实现急慢性期协同治疗。 "
| [1] DIAZ-GONZALEZ F, HERNÁNDEZ-HERNÁNDEZ MV. Rheumatoid arthritis. Med Clin (Barc). 2023; 161(12):533-542. [2] ALMUTAIRI K, NOSSENT J, PREEN D, et al. The global prevalence of rheumatoid arthritis: a meta-analysis based on a systematic review. Rheumatol Int. 202;41(5):863-877. [3] MUELLER AL, PAYANDEH Z, MOHAMMADKHANI N, et al. Recent advances in understanding the pathogenesis of rheumatoid arthritis: new treatment strategies. Cells. 2021;10(11):3011. [4] DERKSEN V, HUIZINGA TWJ, VAN DER WOUDE D. The role of autoantibodies in the pathophysiology of rheumatoid arthritis. Semin Immunopathol. 2017;39(4):437-446. [5] MCINNES IB, SCHETT G. Cytokines in the pathogenesis of rheumatoid arthritis. Nat Rev Immunol. 2007;7(6):429-442. [6] 国家皮肤与免疫疾病临床医学研究中心(北京协和医院),中国医师协会风湿免疫专科医师分会,中国康复医学会风湿免疫病康复专业委员会,等.2024中国类风湿关节炎诊疗指南[J].中华内科杂志,2024,63(11):1059-1077. [7] BAIG MMFA, WONG LK, ZIA AW, et al. Development of biomedical hydrogels for rheumatoid arthritis treatment. Asian J Pharm Sci. 2024;19(1):100887. [8] VAN BEMMELEN JM. Das Hydrogel und das krystallinische Hydrat des Kupferoxyds. Diese Zeitschr. 1894;5(1):466-483. [9] DODGE HW JR, GRINDLAY JH, CRAIG WM, et al. Use of polyvinyl sponge in neurosurgery. J Neurosurg. 1954;11(3):258-261. [10] WICHTERLE O, LÍM D. Hydrophilic Gels for Biological Use. Nature. 1960;185:117-118. [11] KOPEČEK J, VACÍK J, LÍM D. Permeability of membranes containing ionogenic groups. J Polym Sci A1. 1971;9:2801-2815. [12] CHEN-CHOW PC, FRANK SG. In vitro release of lidocaine from Pluronic F-127 gels. Int J Pharm. 1981;8(2):89-99. [13] SHIINO D, MURATA Y, KUBO A, et al. Amine containing phenylboronic acid gel for glucose-responsive insulin release under physiological pH. J Control Release. 1995;37(3):269-276. [14] DINEEN MK, TIERNEY DS, KUZMA P, et al. An evaluation of the pharmacokinetics and pharmacodynamics of the histrelin implant for the palliative treatment of prostate cancer. J Clin Pharmacol. 2005;45(11):1245-1249. [15] CHENG R, FENG F, MENG F, et al. Glutathione-responsive nano-vehicles as a promising platform for targeted intracellular drug and gene delivery. J Control Release. 2011;152(1):2-12. [16] DUNN SS, TIAN S, BLAKE S, et al. Reductively responsive siRNA-conjugated hydrogel nanoparticles for gene silencing. J Am Chem Soc. 2012;134(17):7423-7430. [17] SHI K, FU W, FARHADI SABET Z, et al. Hydrogel-Mediated Jamming of Exosome Communications That Counter Tumor Adaption in the Tumor Immune Microenvironment. ACS Nano. 2024; 18(48):33042-33057. [18] PRASAD P, VERMA S, SURBHI, et al. Rheumatoid arthritis: advances in treatment strategies. Mol Cell Biochem. 2023;478(1):69-88. [19] LIU J, JIANG Y, ZHANG S, et al. Ivarmacitinib, a selective Janus kinase 1 inhibitor, in patients with moderate-to-severe active rheumatoid arthritis and inadequate response to conventional synthetic DMARDs: results from a phase III randomised clinical trial. Ann Rheum Dis. 2025;84(2):188-200. [20] WANG L, XU D, FANG J, et al. Telitacicept, a Human Recombinant Fusion Protein Targeting and Neutralizing B Lymphocyte Stimulator (BlyS) and a Proliferation-Inducing Ligand (APRIL), in Rheumatoid Arthritis (RA) Patients with an Inadequate Response to Methotrexate (MTX): A Randomized, Double-Blind, Phase 3 Study. Arthritis Rheumatol. 2023;75(suppl 9).doi:10.1002/art.42355. [21] GRAHAM GG, SCOTT KF. Limitations of drug concentrations used in cell culture studies for understanding clinical responses of NSAIDs. Inflammopharmacol. 2021;29(5):1261-1278. [22] KOETTING MC, GUIDO JF, GUPTA M, et al. pH-responsive and enzymatically-responsive hydrogel microparticles for the oral delivery of therapeutic proteins: Effects of protein size, crosslinking density, and hydrogel degradation on protein delivery. J Control Release. 2016;221:18-25. [23] CARRILLO-CONDE BR, BREWER E, LOWMAN A, et al. Complexation Hydrogels as Oral Delivery Vehicles of Therapeutic Antibodies: An in Vitro and ex Vivo Evaluation of Antibody Stability and Bioactivity. Ind Eng Chem Res. 2015;54(42): 10197-10205. [24] COLLINS KH, PFERDEHIRT L, SALEH LS, et al. Hydrogel Encapsulation of Genome-Engineered Stem Cells for Long-Term Self-Regulating Anti-Cytokine Therapy. Gels. 2023;9(2):169. [25] CHENG F, SU T, LIU Y, et al. Targeting Lymph Nodes for Systemic Immunosuppression Using Cell-Free-DNA-Scavenging And cGAS-Inhibiting Nanomedicine-In-Hydrogel for Rheumatoid Arthritis Immunotherapy. Adv Sci (Weinh). 2023; 10(26):e2302575. [26] XU Y, ZHAO M, CAO J, et al. Applications and recent advances in transdermal drug delivery systems for the treatment of rheumatoid arthritis. Acta Pharm Sin B. 2023;13(11): 4417-4441. [27] LEE S, CHOI S, KIM MS. Intra-articular hydrogel formulation prolongs the in vivo stability of Toll-like receptor antagonistic peptides for rheumatoid arthritis treatment. J Control Release. 2024;372:467-481. [28] COX N, MALLEN CD, SCOTT IC. Pharmacological pain management in patients with rheumatoid arthritis: a narrative literature review. BMC Med. 2025;23(1):54. [29] ARFEEN M, SRIVASTAVA A, SRIVASTAVA N, et al. Design, classification, and adverse effects of NSAIDs: A review on recent advancements. Bioorg Med Chem. 2024;112:117899. [30] ZHOU S, ZOU H, CHEN G, et al. Synthesis and biological activities of chemical drugs for the treatment of rheumatoid arthritis. Top Curr Chem. 2019;377(5):28. [31] DOUMEN M, PAZMINO S, BERTRAND D, et al. Glucocorticoids in rheumatoid arthritis: Balancing benefits and harm by leveraging the therapeutic window of opportunity. Joint Bone Spine. 2023; 90(3):105491. [32] HOFMAN ZLM, ROODENRIJS NMT, NIKIPHOROU E, et al. Difficult-to-treat rheumatoid arthritis: what have we learned and what do we still need to learn? Rheumatology. 2025;64(1):65-73. [33] NEGI S, TANDEL N, SHARMA P, et al. Aceclofenac and methotrexate combination therapy could influence Th1/Th17 axis to modulate rheumatoid-arthritis-induced inflammation. Drug Discov Today. 2023;28(8):103671. [34] MADRID-GARCÍA A, FREITES-NUNEZ D, MERINO-BARBANCHO B, et al. Mapping two decades of research in rheumatology-specific journals: a topic modeling analysis with BERTopic. Ther Adv Musculoskelet Dis. 2024;16:1759720X241308037. [35] ZHAO F, HU Z, LI G, et al. Angiogenesis in rheumatoid arthritis: Pathological characterization, pathogenic mechanisms, and nano-targeted therapeutic strategies. Bioact Mater. 2025;50: 603-639. [36] SONG YJ, NAM SW, SUH CH, et al. Biosimilars in the treatment of rheumatoid arthritis: a pharmacokinetic overview. Expert Opin Drug Metab Toxicol. 2023;19(11):751-768. [37] BACK W, PARK JH. Hydrogel-Based Strategies for Managing Rheumatoid Arthritis: From Sustained Drug Release to Cell-Based Therapies. ACS Biomater Sci Eng. 2025;11(6):3262-3275. [38] ALTEN R, KRUGER K, RELLECKE J, et al. Examining patient preferences in the treatment of rheumatoid arthritis using a discrete-choice approach. Patient Prefer Adher. 2016;10: 2217-2228. [39] JIANG H, LU Q, HUANG X, et al. Sinomenine-glycyrrhizic acid self-assembly enhanced the anti-inflammatory effect of sinomenine in the treatment of rheumatoid arthritis. J Control Release. 2025;382:113718. [40] QINDEEL M, ULLAH MH, FAKHAR-UD-DIN, et al. Recent trends, challenges and future outlook of transdermal drug delivery systems for rheumatoid arthritis therapy. J Control Release. 2020;327: 595-615. [41] ZHAO YP, HAN JF, ZHANG FY, et al. Flexible nano-liposomes-based transdermal hydrogel for targeted delivery of dexamethasone for rheumatoid arthritis therapy. Drug Deliv. 2022; 29(1):2269-2282. [42] KHAN D, QINDEEL M, AHMED N, et al. Development of an intelligent, stimuli-responsive transdermal system for efficient delivery of ibuprofen against rheumatoid arthritis. Int J Pharm. 2021;610:121242. [43] KHAN D, AHMED N, MUHAMMAD A, et al. A macromolecule infliximab loaded reverse nanomicelles-based transdermal hydrogel: An innovative approach against rheumatoid arthritis. Biomater Adv. 2025;167:214093. [44] GHARAT S, MOMIN M, PANCHAL U, et al. Novel ethosomal gel formulation for enhanced transdermal delivery of curcumin and cyclosporine: a preclinical approach to rheumatoid arthritis management. Drug Deliv. 2025;32(1):2512620. [45] WENDONG Y, XINGXING Y, XIANZE X, et al. Nanoformulation-assisted microneedle transdermal drug delivery system: An innovative platform enhancing rheumatoid arthritis treatment. Biomed Pharmacother. 2024;178: 117219. [46] WANG X, YUE J, GUO S, et al. Dissolving microneedles: A transdermal drug delivery system for the treatment of rheumatoid arthritis. Int J Pharm. 2025;671:125206. [47] BALDE A, KIM SK, NAZEER RA. A review on microneedle patch as a delivery system for proteins/peptides and their applications in transdermal inflammation suppression. I Int J Biol Macromol. 2025;307(Pt 2):141963. [48] LI S, CHEN Q, ZHANG Y, et al. Hyaluronic acid dissolving microneedle patch-assisted acupoint transdermal delivery of triptolide for effective rheumatoid arthritis treatment. Sci Rep. 2024; 14(1):25256. [49] ZHAO W, ZHENG L, YANG J, et al. Dissolving microneedle patches-mediated percutaneous delivery of tetramethylpyrazine for rheumatoid arthritis treatment. Eur J Pharm Sci. 2023;184: 106409. [50] HUA P, YANG S, YU L, et al. Natural product-integrated microneedle patch for rheumatoid arthritis treatment through anti-inflammation and angiogenesis suppression. Biomater Sci. 2025;13(9):2462-2474. [51] LIU T, FU J, ZHENG Z, et al. Active microneedle patch equipped with spontaneous bubble generation for enhanced rheumatoid arthritis treatment. Theranostics. 2025;15(8): 3424-3438. [52] ZHANG X, CHEN J, HUANG N, et al. Microneedle-based sustained release delivery of TNF-α/IL-6R dual-specific fenobody alleviates inflammation and promotes bone regeneration in rheumatoid arthritis rat model. Mater Today Bio. 2025;33:101905. [53] HUA P, LIANG R, YANG S, et al. Microneedle-assisted dual delivery of PUMA gene and celastrol for synergistic therapy of rheumatoid arthritis through restoring synovial homeostasis. Bioact Mater. 2024;36:83-95. [54] BACK W, JEONG M, LE HT, et al. Immunosuppressive Cytokine-Tethered Hydrogel for Treating Rheumatoid Arthritis. Adv Healthc Mater. 2025;14(19):e2501613. [55] XU M, FU T, ZHANG C, et al. Prolonged, staged, and self-regulated methotrexate release coupled with ROS scavenging in an injectable hydrogel for rheumatoid arthritis therapy. J Control Release. 2024;375:60-73. [56] WANG N, MA J, SONG W, et al. An injectable hydrogel to disrupt neutrophil extracellular traps for treating rheumatoid arthritis. Drug Deliv. 2023;30(1):2173332. [57] LEE S, SEO J, KIM Y H, et al. Enhanced intra-articular therapy for rheumatoid arthritis using click-crosslinked hyaluronic acid hydrogels loaded with toll-like receptor antagonizing peptides. Acta Biomater. 2023;172:188-205. [58] LIU H, LIU Y, TIAN Z, et al. Coordinating Macrophage Targeting and Antioxidation by Injectable Nanocomposite Hydrogel for Enhanced Rheumatoid Arthritis Treatment. ACS Appl Mater Interfaces. 2024;16(29):37656-37668. [59] SINGH R, MALHOTRA H, JADHAV K, et al. Intelligently Actuating Dual-Barrier Hyaluronic Acid-Functionalized Inflammation-Responsive Nanohydrogel for Targeted Rheumatoid Arthritis Therapy. ACS Appl Mater Interfaces. 2025;17(28): 40012-40034. [60] ZHAO Y, LI Z, CHEN X, et al. MXenzyme-reinforced hydrogel enhances stem cell therapy through multidimensional regulation in rheumatoid arthritis management. Biomaterials. 2025;325:123560. [61] ZHAO Y, SONG S, WANG D, et al. Nanozyme-reinforced hydrogel as a H2O2-driven oxygenerator for enhancing prosthetic interface osseointegration in rheumatoid arthritis therapy. Nat Commun. 2022;13(1):6758. [62] SHI G, ZHOU Y, LIU W, et al. Bone-derived MSCs encapsulated in alginate hydrogel prevent collagen-induced arthritis in mice through the activation of adenosine A2A/2B receptors in tolerogenic dendritic cells. Acta Pharm Sin B. 2023;13(6):2778-2794. |
| [1] | Zheng Ying, Li Mengyao, Zheng Fanfan, He Zhao, Zhang Ning, Zou Jialun, Li Youlei, Gao Feng. Mechanism of action of extracellular vesicles loaded with biomaterials in repairing spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8544-8554. |
| [2] | Fan Xiao, Li Huiyun, Dou Fuguo, Zhang Nan, Zhang Xinyan. Copper phosphate nanohydrogel dressing promotes skin wound healing in mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8413-8419. |
| [3] | Liu Bangding, Tang Yongliang, Li Ni, Ren Bo. Quercetin-loaded hydrogel materials for treatment of infected bone defects [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8427-8435. |
| [4] | Chen Shichao, Deng Yunyi, Zhao Renshengjie, Yu Ke, Li Guangwen. Antibacterial properties of photocrosslinkable hydrogel loaded with quercetin-silver nanoparticles for infected wounds [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8436-8442. |
| [5] | Zhang Yiwei, Fang Ya, Sun Xin, Yang Han, Lin Haiyang, Chen Zhouhao, Zheng Yue, Fu Jingke, Wang Jinwu. Regulatory effects of optimized extraction processes for chlorella-derived peptides on key pathological links in rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8460-8470. |
| [6] | Ding Hao, Gao Yuan, Li Bin, Yu Rui, Wang Jianru, Sun Yudie, Wang Xuanyang, Zhang Wenping, Zhu Mingjun. Potential and application prospects of combined treatment of acute myocardial infarction with hydrogel cardiac patches and traditional Chinese medicine [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8479-8486. |
| [7] | Fang Yulu, Yi Bingcheng, Shen Yanbing, Tang Han, Zhang Yanzhong. Potential of corn husk fibers reinforced chitosan-based hydrogels in cartilage tissue engineering scaffold [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5493-5501. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||