Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (32): 8427-8435.doi: 10.12307/2026.396
Previous Articles Next Articles
Quercetin-loaded hydrogel materials for treatment of infected bone defects
Liu Bangding1, Tang Yongliang1, Li Ni2, Ren Bo1
- 1Department of Orthopedics II, Xi'an Central Hospital, Xi'an 710004, Shaanxi Province, China; 2Health Examination Center, Xi'an Chest Hospital, Xi'an 710100, Shaanxi Province, China
-
Accepted:2025-11-10Online:2026-11-18Published:2026-04-27 -
Contact:Ren Bo, Attending physician, Department of Orthopedics II, Xi'an Central Hospital, Xi'an 710004, Shaanxi Province, China -
About author:Liu Bangding, Associate chief physician, Department of Orthopedics II, Xi'an Central Hospital, Xi'an 710004, Shaanxi Province, China
CLC Number:
Cite this article
Liu Bangding, Tang Yongliang, Li Ni, Ren Bo. Quercetin-loaded hydrogel materials for treatment of infected bone defects[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8427-8435.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
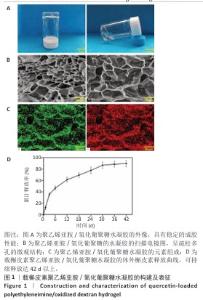
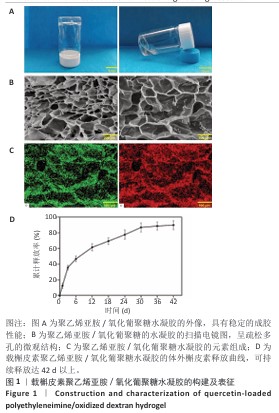
2.1 载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶的构建及表征 如图1A所示,聚乙烯亚胺/氧化葡聚糖水凝胶具有稳定的成胶性能;如图1B所示,扫描电镜图像显示聚乙烯亚胺/氧化葡聚糖水凝胶为高度多孔的表面结构,孔径在120-220 μm之间,该孔径范围有利于细胞黏附,同时也便于细胞营养物质及代谢废物的输送[17]。能谱分析表面扫描结果表明,聚乙烯亚胺/氧化葡聚糖水凝胶主要由C和O元素组成(图1C),C、O元素在材料表面均匀分布,此结果与聚乙烯亚胺/氧化葡聚糖水凝胶的分子组成一致,未检测到其他杂质元素。 载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶的体外槲皮素释放曲线如图1D所示。第1,3天的槲皮素总释放率分别为(12.6±1.6)%和(37.7±3.3)%,表现出较快的释放速度;在接下来的12 d内,槲皮素释放量逐渐增加到(68.9±5.4)%,显示出更好的缓释效果;第36天,槲皮素释放量为(87.5±6.6)%,此后释药量很少。负载在聚乙烯亚胺/氧化葡聚糖水凝胶基质中的槲皮素主要通过药物自由扩散和材料降解2种方式释放[18]:在早期阶段,槲皮素的释放主要依靠自由扩散,在此期间,位于表层的槲皮素更容易以较快的速度释放出来,能够在短时间内迅速达到有效药物浓度,为抗菌过程创造了有利条件,可高效发挥抗菌作用[19];到了后期,水凝胶的降解过程成为影响槲皮素释放的关键因素,随着水凝胶逐步降解,原本负载于水凝胶内部的槲皮素得以释放,然而随着材料持续降解,槲皮素的含量不断减少,最终致使药物释放量以及释放速度均出现下降。 "
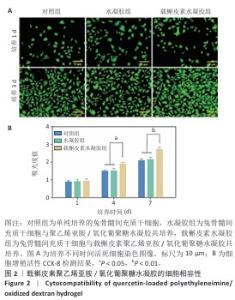
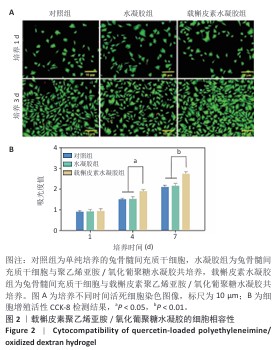
2.2 载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶的细胞相容性 如图2A所示,随着培养时间的延长,各组活细胞数量均增多,培养第1天,3组活细胞数量无明显差异;培养第3天,载槲皮素水凝胶组活细胞数量明显多余其他2组,3组死细胞数量无明显差异,说明载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶无细胞毒性,并且可以促进兔骨髓间充质干细胞的增殖。 各组细胞增殖活力检测结果见图2B。培养第4,7天,载槲皮素水凝胶组细胞增殖速度都明显快于其他两组(P < 0.05,P < 0.01),对照组和水凝胶组细胞增殖速度比较无明显差异(P > 0.05),说明载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶可促进兔骨髓间充质干细胞的增殖。以上结果均显示载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶具有良好的细胞相容性。"
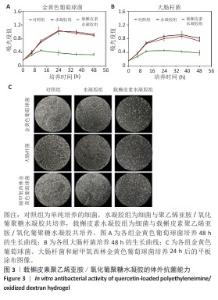
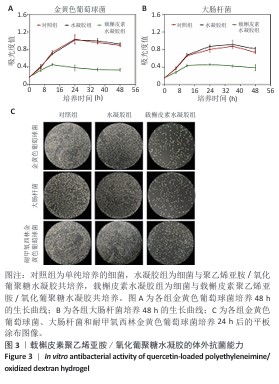
2.3 载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶的体外抗菌能力 在微生物的分类体系中,金黄色葡萄球菌作为革兰阳性菌的典型代表,大肠杆菌是革兰阴性菌的标志性微生物[10,20],这两类细菌在临床上是引发骨髓炎的常见病原菌[21]。近年来随着抗生素药物的广泛使用,耐甲氧西林金黄色葡萄球菌引发的骨髓炎也逐渐增多,这为治疗带来了诸多困难[22]。鉴于此,此次实验着重分析载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶针对金黄色葡萄球菌、大肠杆菌和耐甲氧西林金黄色葡萄球菌这3种细菌的杀伤能力展开深入验证。 如图3A,B所示,在培养的起始阶段,由于载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶中槲皮素的释放量相对有限,尚不足以将细菌完全消灭,然而随着时间的推移,药物持续、稳定地释放并不断作用于细菌,逐步破坏细菌的结构与功能,载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶的抗菌能力逐渐增强,而单独的聚乙烯亚胺/氧化葡聚糖水凝胶无明显的抗菌性能。结果表明,载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶表现出极为优异的抗菌能力,有效抑制了金黄色葡萄球菌与大肠杆菌的生长与繁殖。 细菌涂布实验结果见图3C。相较于对照组、水凝胶组,载槲皮素水凝胶组平板上形成的金黄色葡萄球菌、大肠杆菌、耐甲氧西林金黄色葡萄球菌菌落数量明显减少,这一结果直接且有力地证明了载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶强大的抗菌性能。 "
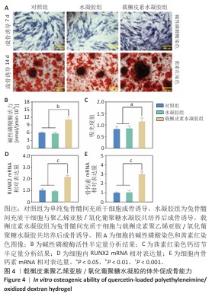
2.4 载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶的体外促成骨能力 成骨细胞会借助基质囊泡分泌碱性磷酸酶,这种酶能够引导钙离子在胶原纤维中沉积,因而被视作成骨细胞早期分化的标志[23]。各组兔骨髓间充质干细胞碱性磷酸酶染色结果见图4A,载槲皮素水凝胶组产生的紫色产物显著增多,表明该组中碱性磷酸酶含量表达较多。各组兔骨髓间充质干细胞碱性磷酸酶活性检测结果见图4B,载槲皮素水凝胶组碱性磷酸酶活性高于对照组、水凝胶组(P < 0.01),这一检测结果与碱性磷酸酶染色结果相符。 矿化实验的核心目的是借助茜素红染色对细胞外钙离子沉积情况进行精准鉴定,进而深入评价载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶释放槲皮素后对钙基质沉积产生的具体影响[24]。各组兔骨髓间充质干细胞茜素红染色结果见图4A,载槲皮素水凝胶组细胞外钙基质沉积量显著增加,呈现出更为密集的染色区域,直观地反映出该组在促进钙基质沉积方面的积极作用。钙结节定量分析结果显示,载槲皮素水凝胶组钙结节形成量多于对照组、水凝胶组(P < 0.05),见图4C,有力证实了载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶中槲皮素的释放对成骨过程的积极意义。 在成骨过程中,RUNX2能够精准地调控成骨分化相关蛋白的生成,通过这种调控方式对骨髓间充质干细胞的成骨分化进程施加关键影响[25]。骨钙素同样是成骨研究中的重要指标,骨钙素是由成骨细胞分泌至细胞外的基质成分,是成骨细胞分化和成熟的标志性物质。RT-qPCR检测结果显示,载槲皮素水凝胶组RUNX2、骨钙素 mRNA表达量均高于对照组、水凝胶组(P < 0.001),见图4D,E。"
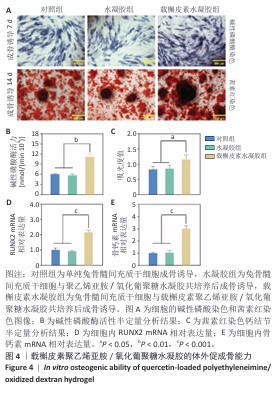
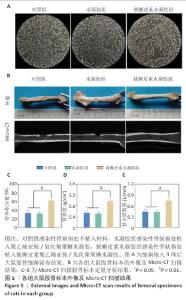
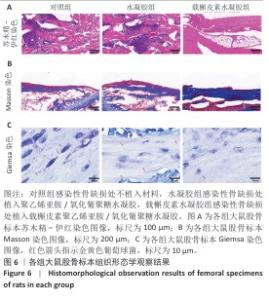
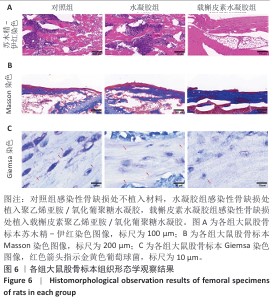
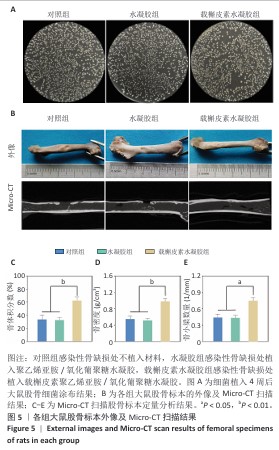
2.5 载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶大鼠感染性股骨缺损实验结果 2.5.1 实验动物数量分析 对照组(n=9)、水凝胶组(n=9)、载槲皮素水凝胶组(n=9)大鼠全部进入结果分析。 2.5.2 感染性股骨缺损模型制备成功 在细菌植入4周后,随机将3只大鼠股骨取出进行细菌涂布,结果如图5A所示。3只大鼠股骨均培养出细菌,表明感染性股骨缺损模型制备成功。 2.5.3 股骨标本Micro-CT扫描 各组大鼠股骨标本的外观形态及Micro-CT扫描图像,如图5B所示。载槲皮素水凝胶组股骨缺损部位呈现出良好的修复趋势,大量新生骨组织密集地填充于缺损区域,使得原本的骨缺损基本实现愈合,有力证明了载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶促进骨再生方面的能力;对照组和水凝胶组股骨缺损部位的修复情况明显滞后,仅能观察到少量新生骨,骨缺损依然较为显著。 各组大鼠股骨标本Micro-CT扫描定量分析结果,如图5C-E所示。载槲皮素水凝胶组新生骨的骨体积分数、骨密度、骨小梁数量均高于对照组、水凝胶组(P < 0.05,P < 0.01)。结果进一步证实了载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶在促进骨缺损修复过程中能够更有效地增加新生骨的体积、提升骨密度以及促进骨小梁的生成。 2.5.4 股骨标本组织形态学观察 苏木精-伊红染色与Masson染色能够细致地呈现股骨组织学形态特征,为深入了解骨组织的修复情况提供重要依据[26]。苏木精-伊红染色结果显示,对照组与水凝胶组可见大量炎性细胞浸润,组织纤维排列紊乱,这意味着组织的正常结构和生理功能受到了炎症干扰;载槲皮素水凝胶组仅见少量炎性细胞浸润,组织纤维排列较规则(图6A),表明载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶在抑制炎症反应方面发挥了积极作用。 Masson染色结果显示,对照组与水凝胶组仅见少量新生骨形成,载槲皮素水凝胶组可见大量新生骨形成(图6B),表明载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶能够有效促进骨组织的再生与修复。 利用Giemsa染色对股骨标本中的细菌进行检测,对照组和水凝胶组可观察到大量细菌,载槲皮素水凝胶组几乎没有细菌(图6C),说明载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶具有良好的抗菌性能。 综合以上染色结果,载槲皮素聚乙烯亚胺/氧化葡聚糖水凝胶中槲皮素的持续释放发挥了重要作用:一方面,槲皮素能够有效地清除感染部位的细菌,显著降低感染程度,为骨组织的修复创造一个无菌的成骨微环境;另一方面,槲皮素还具有显著促进骨修复的功效,通过刺激成骨细胞的增殖和活性加速新生骨的形成,从而推动骨缺损部位的修复进程。 "
| [1] GUO J, SHU X, YU S, et al. Injectable hydrogel microsphere-bomb for MRSA-infected chronic osteomyelitis. J Control Release. 2024;376:337-353. [2] MISTRY S, ROY S, BHOWMICK D, et al. Investigation of Antibiotic-Releasing Biodegradable Composite Bone Cements for Treating Experimental Chronic Maxillofacial Bone Infection. J Biomed Mater Res A. 2025;113(1): 7858-7866. [3] LI S, LIU Z, GAO X, et al. Preparation and properties of a 3D printed nHA/PLA bone tissue engineering scaffold loaded with a β-CD-CHX combined dECM hydrogel. RSC Adv. 2024;14(14):9848-9859. [4] HU Y, HUANG D, LI Y, et al. Investigation on characterization of novel anti-bacterial chitosan/gelatin composite membranes loaded with quercetin via electrophoretic deposition. J Biomater Sci Polym Ed. 2023;34(6):734-752. [5] WU Z, KAEWMANEE R, ZHAO M, et al. Quercetin-loaded porous biocomposite of polyimide and molybdenum disulfide nanosheets with antibacterial capability for boosting osteoblastic differentiation and bone-bonding. Biomater Adv. 2023;154:213585-213598. [6] HU X, LEI X, LIN W, et al. Quercetin promotes osteogenic differentiation of bone marrow mesenchymal stem cells by modulating the miR-214-3p/Wnt3a/β-catenin signaling pathway. Exp Cell Res. 2025;444(2):4386-4399. [7] SHEN D, FENG Y, ZHANG X, et al. Antiosteoporosis Studies of 20 Medicine Food Homology Plants Containing Quercetin, Rutin, and Kaempferol: TCM Characteristics, In Vivo and In Vitro Activities, Potential Mechanisms, and Food Functions. Evid Based Complement Alternat Med. 2022;2022: 5902293. [8] JAFARBEGLOU M, MEIMANDI-PARIZI A, DERAKHSHANDEH A, et al. Silk fibroin/chitosan thiourea hydrogel scaffold with vancomycin and quercetin-loaded PLGA nanoparticles for treating chronic MRSA osteomyelitis in rats. Int J Pharm. 2024;666:4826-4844. [9] YU J, CHU X, CAI Y, et al. Preparation and characterization of antimicrobial nano-hydroxyapatite composites. Mater Sci Eng C Mater Biol Appl. 2014; 37:54-59. [10] LI Z, REN K, CHEN J, et al. Bioactive hydrogel formulations for regeneration of pathological bone defects. J Control Release. 2025;380:686-714. [11] WANG Y, LV H, REN S, et al. Biological Functions of Macromolecular Protein Hydrogels in Constructing Osteogenic Microenvironment. ACS Biomater Sci Eng. 2024;10(9):5513-5536. [12] DENG QS, GAO Y, RUI BY, et al. Double-network hydrogel enhanced by SS31-loaded mesoporous polydopamine nanoparticles: Symphonic collaboration of near-infrared photothermal antibacterial effect and mitochondrial maintenance for full-thickness wound healing in diabetes mellitus. Bioact Mater. 2023;27:409-428. [13] ZHANG H, YUAN S, ZHENG B, et al. Lubricating and Dual-Responsive Injectable Hydrogels Formulated From ZIF-8 Facilitate Osteoarthritis Treatment by Remodeling the Microenvironment. Small. 2025;21(3): e2407885. [14] HE X, LIU W, LIU Y, et al. Nano artificial periosteum PLGA/MgO/Quercetin accelerates repair of bone defects through promoting osteogenic - angiogenic coupling effect via Wnt/β-catenin pathway. Mater Today Bio. 2022;16:10348-10357. [15] DING W, ZHOU J, ZENG Y, et al. Preparation of oxidized sodium alginate with different molecular weights and its application for crosslinking collagen fiber. Carbohydr Polym. 2017;157:1650-1656. [16] KAN YC, GUO R, XU Y, et al. Investigating the in vitro antibacterial efficacy of composite bone cement incorporating natural product-based monomers and gentamicin. J Orthop Surg Res. 2024;19(1):169-181. [17] XU Y, XU C, YANG K, et al. Copper Ion-Modified Germanium Phosphorus Nanosheets Integrated with an Electroactive and Biodegradable Hydrogel for Neuro-Vascularized Bone Regeneration. Adv Healthc Mater. 2023;12(27):1151-1164. [18] ZHANG Q, ZHOU X, DU H, et al. Bifunctional Hydrogel-Integrated 3D Printed Scaffold for Repairing Infected Bone Defects. ACS Biomater Sci Eng. 2023;9(8):458345-458396. [19] JIA X, GU M, DAI J, et al. Quercetin attenuates Pseudomonas aeruginosa-induced acute lung inflammation by inhibiting PI3K/AKT/NF-κB signaling pathway. Inflammopharmacology. 2024;32(2):1059-1076. [20] OU Z, WEI J, LEI J, et al. Biodegradable Janus sonozyme with continuous reactive oxygen species regulation for treating infected critical-sized bone defects. Nat Commun. 2024;15(1):10525. [21] LI J, LEUNG SSY, CHUNG YL, et al. Hydrogel delivery of DNase I and liposomal vancomycin to eradicate fracture-related methicillin-resistant staphylococcus aureus infection and support osteoporotic fracture healing. Acta Biomater. 2023;164:223-239. [22] XU Y, XU C, XIE M, et al. Ultrasound Activated Piezoelectric Catalysis and Neurogenic Activity for Effective Therapy of MRSA Infected Bone Defects by Phase/Defect-Engineered Barium Strontium Titanate. Small Methods. 2025:e2402174. doi: 10.1002/smtd.202402174. [23] BOSE S, CHAUDHARI VS, KUSHRAM P. 3D printed scaffolds with quercetin and vitamin D3 nanocarriers: In vitro cellular evaluation. J Biomed Mater Res A. 2024;112(12):2110-2123. [24] YANG S, ZHU Y, JI C, et al. A five-in-one novel MOF-modified injectable hydrogel with thermo-sensitive and adhesive properties for promoting alveolar bone repair in periodontitis: Antibacterial, hemostasis, immune reprogramming, pro-osteo-/angiogenesis and recruitment. Bioact Mater. 2024;41:239-256. [25] DAI G, XU C, HAN B, et al. Treatment of bone-cartilage defects with dual-layer tissue-engineered scaffolds loaded with icariin and quercetin. J Biomed Mater Res A. 2024;112(12):2170-2186. [26] HAN C, WU Z, GAO Y, et al. An Additive-Fabricated Biphasic Scaffold for Procedurally Promoting Bone Regeneration via Antioxidant and Osteogenesis. Biotechnol Bioeng. 2025;122(3):654-666. [27] MA L, CHENG Y, FENG X, et al. A Janus-ROS Healing System Promoting Infectious Bone Regeneration via Sono-Epigenetic Modulation. Adv Mater. 2024;36(2):7846-7867. [28] SZWED-GEORGIOU A, PŁOCIŃSKI P, KUPIKOWSKA-STOBBA B, et al. Bioactive Materials for Bone Regeneration: Biomolecules and Delivery Systems. ACS Biomater Sci Eng. 2023;9(9):5222-5254. [29] WU H, CHEN C, LI J, et al. Engineered Magneto-Piezoelectric Nanoparticles-Enhanced Scaffolds Disrupt Biofilms and Activate Oxidative Phosphorylation in Icam1(+) Macrophages for Infectious Bone Defect Regeneration. ACS Nano. 2024;18(52):35575-35594. [30] GHAVIMI MA, BANI SHAHABADI A, JAROLMASJED S, et al. Nanofibrous asymmetric collagen/curcumin membrane containing aspirin-loaded PLGA nanoparticles for guided bone regeneration. Sci Rep. 2020;10(1): 18200-18218. [31] QIAO L, LIANG Y, CHEN J, et al. Antibacterial conductive self-healing hydrogel wound dressing with dual dynamic bonds promotes infected wound healing. Bioact Mater. 2023;30:129-141. [32] EFSTATHIOU S, WEMYSS AM, PATIAS G, et al. Self-healing and mechanical performance of dynamic glycol chitosan hydrogel nanocomposites. J Mater Chem B. 2021;9(3):809-823. [33] REN Y, WEEKS J, XUE T, et al. Evidence of bisphosphonate-conjugated sitafloxacin eradication of established methicillin-resistant S. aureus infection with osseointegration in murine models of implant-associated osteomyelitis. Bone Res. 2023;11(1):1055-1067. [34] YANG SY, HU Y, ZHAO R, et al. Quercetin-loaded mesoporous nano-delivery system remodels osteoimmune microenvironment to regenerate alveolar bone in periodontitis via the miR-21a-5p/PDCD4/NF-κB pathway. J Nanobiotechnology. 2024;22(1):94. [35] ZHU H, CAI C, YU Y, et al. Quercetin-Loaded Bioglass Injectable Hydrogel Promotes m6A Alteration of Per1 to Alleviate Oxidative Stress for Periodontal Bone Defects. Adv Sci (Weinh). 2024;11(29):e2403412. [36] SUN S, WANG Q, ZHANG B, et al. Vancomycin-Loaded in situ Gelled Hydrogel as an Antibacterial System for Enhancing Repair of Infected Bone Defects. Int J Nanomedicine. 2024;19:10227-10245. [37] YANG F, SHI Z, HU Y, et al. Nanohybrid Hydrogel with Dual Functions: Controlled Low-Temperature Photothermal Antibacterial Activity and Promoted Regeneration for Treating MRSA-Infected Bone Defects. Adv Healthc Mater. 2025;14(11):e2500092. [38] WANG S, LEI H, MI Y, et al. Chitosan and hyaluronic acid based injectable dual network hydrogels - Mediating antimicrobial and inflammatory modulation to promote healing of infected bone defects. Int J Biol Macromol. 2024;274(Pt 1):133124-133142. [39] SOHRABI M, HESARAKI S, SHAHREZAEE M, et al. The release behavior and in vitro osteogenesis of quercetin-loaded bioactive glass/hyaluronic acid/sodium alginate nanocomposite paste. Int J Biol Macromol. 2024;280(Pt 4): 136094-136110. [40] DURMAZ B, GUNES N, KOPARAL M, et al. Investigation of the effects of quercetin and xenograft on the healing of bone defects: An experimental study. J Oral Biol Craniofac Res. 2023;13(1):22-27. [41] YANG Y, ZHOU B, LI M, et al. GO/Cu Nanosheet-Integrated Hydrogel Platform as a Bioactive and Biocompatible Scaffold for Enhanced Calvarial Bone Regeneration. Int J Nanomedicine. 2024;19:8309-8336. [42] SHAO B, FU Y, LI B, et al. Icariin-loaded chitosan/β-glycerophosphate thermosensitive hydrogel enhanced infection control and bone regeneration in canine with infectious bone defects. J Biomater Appl. 2025;39(7): 696-713. [43] FANG B, QIU P, XIA C, et al. Extracellular matrix scaffold crosslinked with vancomycin for multifunctional antibacterial bone infection therapy. Biomaterials. 2021;268:120603-120631. |
| [1] | Fan Xiao, Li Huiyun, Dou Fuguo, Zhang Nan, Zhang Xinyan. Copper phosphate nanohydrogel dressing promotes skin wound healing in mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8413-8419. |
| [2] | Chen Shichao, Deng Yunyi, Zhao Renshengjie, Yu Ke, Li Guangwen. Antibacterial properties of photocrosslinkable hydrogel loaded with quercetin-silver nanoparticles for infected wounds [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8436-8442. |
| [3] | Li Minghui, Qie Haoyu, Pan Min, Bi Ruijie, Lyu Xiaomeng, Zhang Haoya, Han Yifei. Hydrogel-based drug delivery systems for rheumatoid arthritis treatment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8496-8501. |
| [4] | Jin Yuan, Zhou Jiabing. Applications and advances of hydrogels in bone tissue engineering repair related to sports injuries [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8512-8520. |
| [5] | Zheng Ying, Li Mengyao, Zheng Fanfan, He Zhao, Zhang Ning, Zou Jialun, Li Youlei, Gao Feng. Mechanism of action of extracellular vesicles loaded with biomaterials in repairing spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8544-8554. |
| [6] | Fang Yulu, Yi Bingcheng, Shen Yanbing, Tang Han, Zhang Yanzhong. Potential of corn husk fibers reinforced chitosan-based hydrogels in cartilage tissue engineering scaffold [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5493-5501. |
| [7] | Yu Xingge, Lin Kaili. Application of nanocomposite hydrogels in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5441-5446. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||