Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (32): 8502-8511.doi: 10.12307/2026.461
Previous Articles Next Articles
Application of self-healing hydrogels for sports injury prevention and rehabilitation#br#
Meng Yihao1, Zhang Shuai2
- 1Xuzhou University of Technology, Xuzhou 221018, Jiangsu Province, China; 2Hebei Normal University, Shijiazhuang 050024, Hebei Province, China
-
Accepted:2026-02-08Online:2026-11-18Published:2026-04-29 -
Contact:Zhang Shuai, PhD, Lecturer, Hebei Normal University, Shijiazhuang 050024, Hebei Province, China -
About author:Meng Yihao, PhD, Lecturer, Xuzhou University of Technology, Xuzhou 221018, Jiangsu Province, China -
Supported by:National Social Science Fund (General Project), No. 22BTY082 (to ZS)
CLC Number:
Cite this article
Meng Yihao, Zhang Shuai. Application of self-healing hydrogels for sports injury prevention and rehabilitation#br#[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8502-8511.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1 自愈水凝胶研究发展历程及主要贡献 见表2。 2007年前后,研究人员首次提出在材料内部构建微血管网络以输送修复剂的理念,这一创新被视为自愈材料研究的起点。随后,这一概念被拓展至生物医用水凝胶领域,通过引入可逆交联结构,使水凝胶在断裂后实现自主修复,初步研究验证了此类水凝胶在运动相关的软骨和韧带修复中的可行性与应用潜力。 2013-2016年间,研究重点转向材料力学性能的优化与功能整合,研究者通过引入纳米纤维网络、金属离子配位和多响应交联机制显著提升了水凝胶的韧性和环境适应性,同时探索水凝胶在药物控释和3D打印组织工程中的潜力。随着智能康复和可穿戴技术的快速发展,自愈水凝胶开始与导电网络、传感器和AI算法结合,不仅可用于运动损伤的修复,还能实现实时监测与个性化康复,这一领域已从单一的修复功能,演变为集“修复-监测-康复”于一体的多功能平台,为运动医学材料研究奠定了坚实基础,并开辟了广阔的应用前景。"

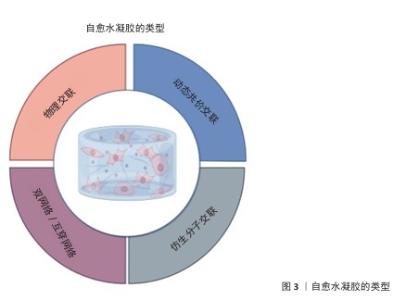
2.2 自愈水凝胶的基本原理与分类 2.2.1 自愈机制 在损伤出现以后,自愈水凝胶利用动态的分子相互作用达成化学键重建,做到结构与性能的修复[48-49]。根据交联方式的不同,自愈机制主要包括以下两类:①物理交联机制:依赖于非共价作用力,如氢键、疏水作用、静电相互作用和离子键等[50-51],这些作用力具有可逆特性,使水凝胶在受损后能够通过分子链的重新排列与结合实现自愈。物理交联机制的优点是反应速度快、无需外部刺激,但其力学性能通常较弱,并且愈合后的强度往往低于初始强度[52]。②化学交联机制:基于动态共价键,如席夫碱键、二硫键、Diels–Alder反应等[51-54],这类键可以赋予较高的力学强度,还具有可反转的属性,在特定条件下可出现断裂并重新构成,进而给予水凝胶更持久、稳定的自我修复能力[54]。化学交联机制常与物理交联协同使用,以实现快速愈合与高力学性能的平衡(图3)。 “水凝胶的自主自愈”概念尤为值得关注,自主自愈是指材料在无需外部刺激的情况下能够超越预设条件,通过自我修复或恢复实现功能再生的一类材料体系[55]。所谓“预定义条件”,通常指的是材料制备时的环境参数或实际应用中的特定条件(如温度、pH值、湿度)[56]。与刺激响应型水凝胶相比,自主自愈水凝胶可在更宽泛的环境里保持修复能力,这对运动医学中复杂又不可控的环境意义非凡。 "

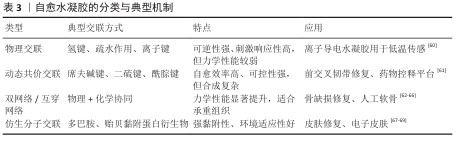
2.2.2 材料学特性 自愈水凝胶的性能除受交联机制影响外,还取决于其物理化学特性,常用的评价指标包括力学性能和自愈效果。其中,评估水凝胶自愈能力的最常见方法是通过宏观观察:将水凝胶分割为两部分后重新黏合,观察它们在界面处是否能够愈合并支撑自身质量[57]。仅仅简单观察,无法量化材料的愈合效率,研究者采用拉伸与压缩测试来评估愈合后样品力学性能。愈合效率(HE)常用以下公式表示: 其中P表示水凝胶的断裂强度、断裂伸长率或模量等力学参数。例如,在单轴拉伸实验中,比较愈合前后的最大断裂伸长率(λb,0与λb),即可获得定量的愈合效率[17],这一指标为不同水凝胶体系的性能比较提供了参考。 水合性与透气性:自愈水凝胶存有高占比的水分子,可以维持较好的水合性能及透气特性,这一特性使它在修复运动损伤期间可模仿天然细胞外基质的环境,有利于营养物质跟代谢产物相互交换[58]。 生物相容性与可降解性:作为运动损伤修复及康复应用的关键要点,自愈水凝胶必须体现出良好的生物相容性,防止产生免疫排斥及炎症反应,材料降解的速度需跟组织再生的过程相契合,以达到逐步替换与安全摄取[59]。 水凝胶自愈特性的评定不只是依赖宏观观察,还需运用多种力学测试与流变学手段开展系统量化,由于研究方法跟实验条件有差别,不同水凝胶体系之间的横向比较还是困难重重,创建标准化测试体系将成为推进自愈水凝胶在运动医学应用的重要阶段(表3)。 "
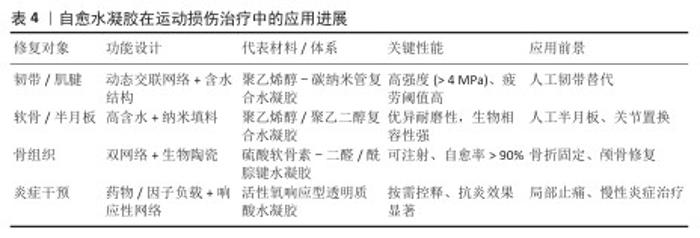
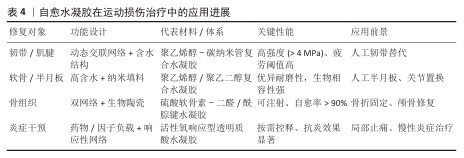
2.3 自愈水凝胶在运动损伤治疗中的应用 运动损伤多累及软组织(如韧带、肌腱、软骨、半月板)及骨组织,常伴随炎症与疼痛[62-66]。自愈水凝胶凭着高含水性、良好的生物相容性、可操控的力学性能以及动态修复能力,逐渐成为对运动损伤修复有潜力的材料,该节将从软组织修复、骨组织修复及炎症与镇痛干预三方面进行阐述(表4)。 部分研究表明,光热照射、自愈敷料以及抗菌治疗等多功能举措在运动损伤康复里有协同功效[70]。在介孔聚多巴胺孔隙中对金纳米颗粒进行原位负载,制备纳米复合物,然后引入聚乙烯醇-羧甲基壳聚糖-硼砂水凝胶中,制成了具备光热效应、抗菌活性与自我愈合性能的水凝胶[71],这类材料在体外表现出极佳的流变学特性以及抗菌效果,还在运动相关组织损伤模型中展示出了良好的原位适应性与光热治疗潜能,为运动损伤的综合康复给出了新举措。 "
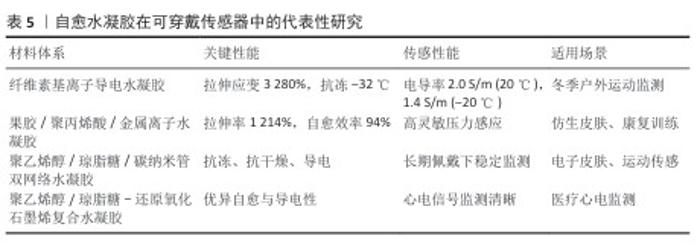
2.3.1 软组织修复 (1)韧带/肌腱修复:韧带和肌腱损伤在运动人群中的发生率较高,传统治疗手段多依赖手术缝合或移植物替代,但常存在愈合周期长、力学性能不足及二次损伤风险高等问题[72]。自愈水凝胶能够在损伤后快速重建三维网络结构,既能恢复整体力学性能,又可作为支架材料为成纤维细胞或成骨细胞的黏附、迁移与增殖提供良好微环境[56]。基于席夫碱键交联的自愈水凝胶已被证实能够模拟韧带和肌腱的力学特征,通过生长因子的负载促进组织再生,与传统不可降解植入物相比更具优势[73]。 天然韧带/肌腱组织兼具跨尺度的各向异性结构与高含水量,同时维持优异的强度与柔韧性,这为人工替代材料提出了挑战[74]。近期研究通过冷冻铸造辅助压缩退火与盐析处理制备出具有分级各向异性结构的聚乙烯醇基杂化水凝胶,该水凝胶的含水量高达79.5%,在掺杂碳纳米管后该材料表现出4.5 MPa的拉伸强度与1 467 J·m-2的疲劳阈值,同时具备优异的压力敏感性和耐用性,为人工韧带修复提供了新方案[75]。 针对膝关节前交叉韧带所面临的高损伤风险,研究者进一步提出了结构有序-无序转变的构建策略。在聚乙烯醇分子链中经由溶胀-盐析诱导结构重新组合制备了高性能水凝胶纤维,所得材料表现出优异的力学性能,断裂伸长率高达257%,拉伸强度达到190.04 MPa,弹性模量为137.31 MPa,显著优于天然韧带的力学水平(30-50 MPa);同时,该材料还具备良好的生物相容性和液体环境中的稳定性[75]。该研究策略为构建兼具高强度与高韧性的人工韧带材料提供了新的思路与技术路径。 研究者还探索了将双网络水凝胶与无机纳米颗粒复合的策略,以构建具备层次化各向异性结构的仿生材料。例如,将高纵横比的介孔二氧化硅颗粒引入藻酸盐-聚丙烯酰胺双网络水凝胶中,并结合离子交联与拉伸固定工艺实现了多尺度各向异性结构的构筑,该杂化水凝胶在溶胀状态中依然能维持7.2 MPa拉伸模量、1.3 MPa的强度以及1.4 MJ·m-3的韧性,明显胜过传统双网络水凝胶,这种结构性能兼备的设计为人工肌腱及韧带的再生提供了新的材料策略[76]。 (2)软骨修复:膝关节软骨及半月板损伤是导致骨关节炎及运动功能受限的主要因素[77-78]。 鉴于软骨组织没有血管、神经,自身修复能力极为受限,开发功能性替代材料的需求迫切[79]。自愈水凝胶凭借自身的高含水性、良好的力学韧性以及可控制的孔隙结构,可在一定程度上模仿天然软骨基质,既可以承受运动中反复出现的加载应力,又可推动营养物质渗透以及软骨细胞的黏附与增殖[80]。复合纳米颗粒或胶原纤维构成的自愈水凝胶,在动物模型中展现出良好的生物相容性与力学性能,已逐步接近天然半月板的水平,为关节软组织再生给出了新的途径[81]。 自愈水凝胶作为高度水合的三维网络材料,可通过动态共价键与非共价键的重排实现断裂后的自我修复,这一特性尤其适配软骨工程中细胞与药物的递送[82]。相关探究表明,自愈水凝胶不仅可充当药物、生长因子或细胞的缓释载体,促进软骨生长,还可当作自愈黏合剂,对软骨修复手术中的组织黏合起到辅助作用[83]。导电型自愈水凝胶还可感应与传导电信号,以此来与软骨的天然电生理特性相吻合,这为它在疾病诊断与组织修复方面的应用开创了新途径[84]。 从材料开发角度看,聚乙烯醇水凝胶凭借出色的生物相容性和低摩擦的特点,被广泛用在人工关节软骨的替换[85],它的力学性能存在不足仍是拦路虎,研究人员将聚乙二醇引入到水凝胶的网络中,采用物理共混和有机溶剂脱水工艺改善材料的力学与摩擦学特性,实验结果显示,经过有机溶剂脱水处理的聚乙烯醇/聚乙二醇复合水凝胶硬度、耐磨性以及稳定性均得到显著提升,磨损率降至未处理组的27.4%,同时维持出色的自愈能力与细胞相容性,为人工软骨修复带来了新型候选材料[86]。基因激活型自愈水凝胶还为软骨再生给出了新的策略,例如,向透明质酸基水凝胶中添加矿化丝素蛋白且搭载上miR-140-5p纳米颗粒,建立可连续释放关键软骨生成调控因子的支架,这种水凝胶在体内外实验期间均表现出良好的基因递送效率以及软骨再生能力,说明该水凝胶在关节软骨缺损治疗中有潜在临床应用价值[87]。 除去关节软骨以外,自愈水凝胶同样被探索来修复气管软骨缺损,研究者利用希夫碱反应来构建转化生长因子β1/核苷素复合自愈水凝胶,在兔长节段气管缺损模型中显示出较好的注射性、自愈性以及细胞相容性,有效减轻了吻合口狭窄现象,推动了软骨再生,呈现了该材料在复杂组织修复上的应用潜力[88]。 此外,基于Fe3+-羧基离子键和氢键相互作用的聚丙烯酸Fe3+水凝胶实现了在无外部刺激下的自主愈合,材料拉伸强度达120 kPa、断裂伸长率可达750%、愈合效率约为82%,显示出作为软骨组织工程支架的可行性[89]。另一项研究通过设计新型氨基甲硫酰基单体氨基乙基氨基甲硫酰基丙烯酰胺,在温和条件下合成了聚(丙烯酸/氨基乙基氨基甲硫酰基丙烯酰胺)共聚物自愈水凝胶,能够在3 s内快速愈合,并且具备8.48-107.9 kPa的力学强度[90]。该类材料可作为人工软骨生成的模板,为关节替代物开发提供了新的路径。 自愈水凝胶在软骨修复领域展现出多样化的发展趋势:从高性能聚乙烯醇/聚乙二醇复合材料到基因激活水凝胶,再到离子交联与新型单体设计体系,它们在承载能力、耐磨性及再生效能方面不断提升,这些研究成果为关节软骨与半月板修复提供了新的材料基础,同时为运动损伤后骨关节炎的干预和临床转化奠定了坚实的理论和技术基础。 2.3.2 骨组织修复 骨折与骨缺损是运动人群常见且影响功能恢复的重大问题[91]。传统金属内固定虽可提供即时力学支撑,但易引发应力遮挡、二次取出手术等并发症[92-93]。自愈水凝胶具备可注射、原位成胶、可塑填充与损伤后网络自重建等优势,既能适配不规则骨缺损,又可通过功能化设计实现成骨促愈与免疫调控,为骨组织工程提供了新的材料平台[94]。自愈水凝胶的核心思路包括:①力学-生物双优化:引入生物陶瓷(如羟基磷灰石)或金属离子,增强支撑与矿化诱导;②动态交联网络:实现注射、自愈与长期稳定性之间的平衡;③微环境调控:通过生长因子/基因/免疫调控单元介入,促进血管化与成骨分化。 传统硫酸软骨素水凝胶强度与可调性不足且“可注射-自愈-稳定性”难以兼顾[95-96],通过狄尔斯-阿尔德“点击”反应化学+动态酰腙键的双重交联,可在同一体系内实现:①动态酰腙键:赋予可注射与快速自愈;② Diels-Alder(DA)共价网络:提供体内稳定性与力学可调控,该类水凝胶表现出可调黏弹/流变特性、更合理的溶胀/降解行为与优异的组织黏附性,包封大鼠间充质干细胞可提高细胞活力并降低凋亡;负载骨形态发生蛋白 4后用于颅骨缺损模型,观察到新骨形成显著增强,显示该材料作为颅骨组织工程支架的潜力[95]。 对临界尺寸骨缺损,仅依赖自体骨存在供体不足、二次损伤与慢性炎症等局限,自愈水凝胶通过支架-细胞-因子的组合策略有望替代/增强移植物疗效:①双重递送增强:将动态席夫碱网络(自愈组件)+硼砂离子物理网络(增强力学)构成的可注射自愈水凝胶作为载体,同时递送脱矿骨基质粉末与缺氧预处理的骨髓间充质干细胞,该策略一方面改善脱矿骨基质粉末的可操作性,另一方面显著提高骨髓间充质干细胞的血管内皮生长因子表达;在颅骨缺损模型中,水凝胶/脱矿骨基质/骨髓间充质干细胞复合物在骨形成效果上优于任一单一或双组分组合[65]。②多糖/羟基磷灰石复合:针对软多糖基体界面失配导致的分散与力学劣化问题,以透明质酸为生物矿化模板制备杂化颗粒,进一步氧化羟基磷灰石以参与席夫碱交联,获得复合水凝胶,提高氧化透明质酸/羟基磷灰石杂化颗粒浓度可缩短凝胶时间、提升储能模量,在3D培养中支持L929细胞的黏附与增殖,呈现良好可注射性与自愈性,适用于细胞封装与骨缺损修复[97]。 骨再生离不开免疫稳态重建与血管新生[98]。面向运动性骨缺损的快速修复,材料需要在早期诱导巨噬细胞向促再生表型极化、同时促进血管生成与成骨分化。以单步法在黑磷纳米片表面构建单宁酸-Mg2+螯合网络,提高黑鳞的稳定性与骨免疫调控能力,并增强黑鳞与甲基丙烯酸化丝素蛋白基体的界面结合,所得丝素甲基丙烯酸酯-黑磷/单宁酸-镁复合水凝胶显著诱导巨噬细胞M2极化,促进血管生成与成骨分化;体内证实该材料通过免疫微环境重塑→促血管化→促成骨的级联效应提升骨再生质量[99]。 骨为分层复合结构,材料需兼具宏观支撑与微观生物活性[100]。采用互穿聚合物网络或“动态共价+物理”双网络可在提升抗疲劳与断裂韧性的同时,配合骨形态发生蛋白2/骨形态发生蛋白4、碱性成纤维细胞生长因子、转化生长因子β3等因子实现时序递送[101]:①血管化优先→成骨推进:以碱性成纤维细胞生长因子快速释放促进早期微血管形成,随后转化生长因子β3持续释放诱导募集的内源性干细胞向成骨分化。在牙槽骨缺损模型中,此类介孔-多因子-自愈水凝胶在微型计算机断层扫描(骨体积分数、骨小梁数量、骨小梁厚度)与组织学染色中均显示优效的骨再生表型,提示其对复杂生物过程的高适配性与临床可转化潜力[64]。②力学-愈合二元优化:通过动态键(席夫碱/酰腙/金属配位)+强化网络实现“可注射/自愈/稳定性/韧性”协同[59]。 以矿化模板/表面改性解决“刚-柔界面”错配,确保无机填料均匀分散与应力传递:①微环境控制:集成免疫调控+血管生成+成骨诱导的级联设计,匹配运动场景下的高应力与再生时间窗。②标准化评价:除常规模量(储能模量、压缩/拉伸强度、疲劳阈值)与流变外,应纳入动态载荷/循环疲劳、应力遮挡与整合度的功能性评估,并在大动物模型与运动负荷模拟中验证长期稳定性与安全性[102]。 自愈水凝胶为骨组织修复提供了从材料结构-功能到免疫与血管化微环境的系统化解决方案:在实现可注射与自愈的同时,利用双网络/互穿网络与无机–有机复合实现力学与生物性能的双重提升;通过多因子时序递送与免疫重塑加速骨-血管双向耦合的再生进程。未来,亟需围绕运动场景的真实机械环境与临床转化路径(可规模化制备、批次稳定性、法规与长期随访)建立统一标准,以推动自愈水凝胶在运动医学骨缺损与骨折愈合中的临床应用。 2.3.3 其他相关应用 运动损伤后急/慢性炎症、氧化应激与组织微环境失衡常相互交织并影响修复质量,自愈水凝胶凭借可注射、原位自修复、可编程释放与组织黏附等特性,成为实现局部抗炎、抗氧化、免疫调控与血管再生耦合干预的有力平台[103]。 药物控释与局部抗炎:自愈水凝胶通过可调孔隙-动态交联网络实现药物在炎症微环境中的按需释放,以pH值/活性氧响应、金属配位、席夫碱/酰腙等动态键作为“阀门”,实现对非类固醇抗炎药、糖皮质激素、小分子抗炎剂的持续与触发式释放,在关节腔或损伤局部延长暴露时间并降低全身不良反应,结合组织黏附与原位成胶,提升在高活动部位中的驻留稳定性与给药效率[59]。 神经炎症/脊髓损伤:将Tc肽(趋化因子受体4、趋化因子受体7)负载于自愈水凝胶,抑制炎症级联并促进神经修复,转录组与体外实验显示材料通过腺嘌呤核苷酸转位酶1/视神经萎缩蛋白轴增强线粒体自噬,降低活性氧与NLRP3炎性小体表达,减少Ⅰ型小胶质细胞活化,进而促进运动神经元再生、轴突桥接与运动功能恢复[104]。 糖尿病足溃疡与“运动性”伤口:基于透明质酸的自愈水凝胶可贴合不规则伤口、维持湿性环境、断裂后自修复,适应反复拉伸/压缩的运动情境;作为多功能载体同步承载抗菌剂/生长因子/细胞外泌体,显著提升慢性炎症环境下的愈合质量[105]。 抗氧化与广谱抗炎组合:以苯硼酸-氧化透明质酸与儿茶酚化壳聚糖/瓜尔豆胶构建动态交联网络,并负载姜黄素脂质体,该体系在30 s内快速凝胶化,具组织黏附与自愈能力,具有较高清除效率,可持续释放姜黄素≥10 d,在不规则缺损的完全填充与抗炎修复中表现优良[106]。 中枢退行性疾病帕金森病:利用氧化单宁酸-金纳米交联剂构建的壳聚糖自愈水凝胶,具34 G(约80 μm)细针可注射性、导电与抗炎抗氧化性能,能支持神经干细胞长期增殖与神经元分化;脑内注射后改善帕金森病大鼠运动功能与电生理异常,增加酪氨酸羟化酶阳性神经元密度,提示该材料作为生物活性可注射植入物的潜力[107]。该类材料面向运动应用的要点:可在高活动组织实现稳定驻留与自修复;在“炎症-氧化应激-疼痛”三联问题上实现多机制协同;满足康复阶段长时程、低频次给药需求。 促血管生成与微环境调控:组织再生依赖早期免疫稳态重建与血管新生。自愈水凝胶可通过材料学与生物学双维设计构建“免疫-血管-成骨/成软骨”的级联微环境[108]。因子/离子介入在水凝胶中引入生长因子或Cu2+/Mg2+等促血管生成单元,配合细胞基质仿生网络刚度调控,促进内皮细胞迁移管腔化、改善局部灌注,并通过机械信号影响成纤维/间充质细胞的黏附、迁移与谱系分化[109]。以羟丁基壳聚糖-氧化魔芋葡甘露聚糖可逆席夫碱网络承载骨髓间充质干细胞外泌体,材料具高应变(350%)后的G′快速恢复能力,可显著降低外泌体在网络破坏条件下的泄漏,并在可拉伸部位的全层皮肤伤口中提升血管生成、胶原沉积与重塑,可作为运动相关高活动部位的先进敷料[110]。 智能多功能网络(光/电响应+高韧性+自愈):借鉴荧光蛋白/生物膜层级结构,利用聚电解质-表面活性剂水相自组装构建含2-脲基-4-吡啶酮疏水核的分层胶束网络[101],形成高韧性、强自愈与电响应兼具的智能水凝胶;2-脲基-4(1H)-嘧啶酮二聚体聚集/氢键网络限制分子内运动并增强荧光发射,掺入的磺酸基团调控胶束尺度以获得更佳电响应与力学性能,为可穿戴/植入式炎症监测-干预一体化提供材料基础[111]。该类材料面向运动应用的要点:兼顾可拉伸部位的力学稳定与长驻留;将免疫调控-血管化-组织重建进行程序化整合;支持监测-给药-力学保护的闭环康复范式。 自愈水凝胶在局部抗炎/抗氧化、免疫稳态重建、血管新生与组织重塑方面展现出系统化解决方案:从触发式控释到微环境级联调控,兼顾了运动损伤复杂生境中的力学-生物-时间三维约束。 2.4 自愈水凝胶在运动监测与康复设备中的应用 随着运动医学向智能化、个性化和远程化方向发展,自愈水凝胶不仅在损伤修复方面具有潜力,还逐渐成为运动监测与康复设备的关键功能材料。自愈水凝胶的柔软性、自愈性和生物相容性使其能够与可穿戴电子、传感平台以及智能康复系统深度融合(表5)。 2.4.1 可穿戴传感器 自愈水凝胶凭借高柔韧性、导电性及动态自修复能力,被广泛应用于可穿戴应变和压力传感器的开发,用于实时监测人体运动姿态和关节受力。在高频率的弯曲、拉伸或压缩条件下,传统柔性传感器容易因微裂纹累积而失效,而自愈水凝胶依靠动态交联网络能够自动修复结构损伤,从而显著延长器件的服役寿命并保持稳定的监测性能[44]。 运动姿态与关节应力监测:基于自愈导电水凝胶的应变传感器可精确检测膝关节、肘关节等关键部位的角度变化,并实时反馈运动模式;同时,结合柔性压力传感器,能够动态监测下肢落地冲击力和关节负荷,为运动损伤风险的早期预测提供量化数据[112-113]。 与柔性电子和惯性测量单元的集成:通过与惯性测量单元、可拉伸电极及无线通信模块的集成,自愈水凝胶传感器可构建轻量化、低能耗的运动监测系统,实现全身动态数据的实时采集与远程传输,为个性化运动训练和康复评估提供支持。近年来,多种新型自愈水凝胶被开发用于可穿戴传感器:①抗冻与超拉伸水凝胶:离子导电纤维素基水凝胶展现出3 280%的超高拉伸应变、-32 ℃抗冻性能以及在低温条件下仍保持高电导率(20 ℃为2.0 S/m,-20 ℃为1.4 S/m),该类水凝胶兼具优异的粘附性和自愈能力,所制备的传感器能够在宽温区间下实现对人体运动的快速响应;②类皮肤自愈水凝胶:基于果胶、聚丙烯酸与金属离子间氢键及双配位作用构建的水凝胶具有1 214%的拉伸性和94%的自愈效率(2 h内完成),无需外部刺激即可愈合,由其构建的电容型传感器可检测多种人体动作并模拟皮肤的感知功能[114];③快速成胶与可逆响应水凝胶:通过二官能化聚乙二醇与壳聚糖的席夫碱反应,几秒内即可实现凝胶化,所得水凝胶具有多孔结构、优异的延展性(断裂应变88.2%,断裂应力12.1 kPa) 及pH值响应特性,可用作柔性应变传感器以监测人体大幅或精细动作[60];④双网络导电水凝胶:在聚乙烯醇/琼脂糖/硼砂网络中引入甘油、丁烷四羧酸和聚丙烯酸钠,制备的双网络兼具自愈、抗冻(-20 ℃下仍柔韧)和抗干燥特性,掺杂碳纳米管后赋予导电性,可作为电子皮肤的应变传感介质,实现长时间皮肤附着下的人体运动监测[69];⑤环境耐受型水凝胶:基于聚乙烯醇-氯化锂体系的一步法自愈水凝胶,表现出良好的自愈性、黏附性和紫外线屏蔽能力,室温与-18 ℃下存放30 d仍稳定,该材料的高离子电导率(24.29 S/m,室温)及近100%紫外线屏蔽性能使其适合冬季户外运动的柔性传感器[115];⑥形状记忆自愈水凝胶:通过动态静电作用与亚胺键双交联制备的水凝胶,兼具高拉伸性(556%)、断裂强度(63 kPa)与形状记忆特性(愈合效率99%),可在pH值或金属离子刺激下实现快速形状固定,用于电阻应变和电容压力传感器,灵敏检测如手指弯曲、吞咽及脉搏等生理信号[116];⑦石墨烯复合导电自愈水凝胶:靠碱性热还原途径在聚合物溶液中进行原位合成,增进分散性,赋予聚乙烯醇/琼脂糖双网络水凝胶良好的导电性与自愈能力,该体系可充当心电图的电极,供应清晰波形以及低噪声信号,同时拥有可实现打印的性能,增加了它在运动监测和柔性电子范畴的应用前景;⑧互穿聚合物网络水凝胶:在吉兰胶-瓜尔豆胶体系里添加硼酸酯和氢键交联制得的水凝胶,展现出不错的拉伸性能,伸长率为216%,展现出突出的自我修复性与导电性,可产出稳定且可重复呈现的电信号,用于开展高可靠性的运动监测[117]。 自愈水凝胶在可穿戴传感器当中呈现出明显优势:拥有高拉伸的性能与抗冻/抗干燥能力,适应各式各样的运动环境;依靠动态网络实现微裂纹的修复,提高器件的存活时长;兼备导电性、形状记忆、光热响应等不同功能,推动电子皮肤跟柔性传感系统的进步。 "
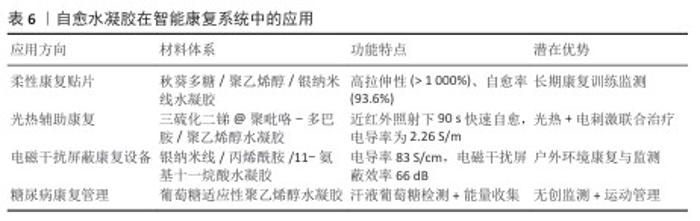
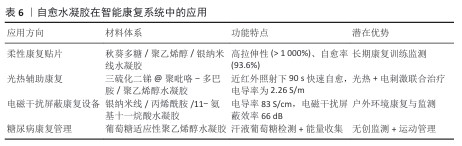
2.4.2 智能康复系统 自愈水凝胶在智能康复设备中的应用逐步拓展,尤其在柔性康复贴片、康复训练使用的辅具和数字化康复平台中体现出关键意义,主要的优势就在于自愈性、导电性、可注射性和生物相容性,能于运动环境中维持长期稳定运行(表6)[118]。 基于自愈水凝胶的柔性贴片能紧密地贴合皮肤表面,实现对肌肉收缩跟关节运动的实时监测,还具备药物逐步释放与电刺激治疗功能,与传统材料易产生性能衰减的状况不一样,一旦有微损伤,自愈水凝胶贴片就可迅速恢复导电性与力学性能,由此可支持长时间佩戴及使用[119]。 当水凝胶传感器与虚拟现实技术结合时,能够将患者的运动状态实时可视化并反馈到虚拟训练环境中,这种沉浸式的康复模式既提高了患者的主动参与性,也能通过实时运动参数校正动作偏差,从而显著提升康复训练的效果和依从性[119]。 基于自愈水凝胶的可穿戴设备可通过无线通信接入远程医疗平台,医生与康复师能够实时监测患者的运动功能恢复情况,及时调整康复处方与训练方案,这种模式推动了运动康复向数字化、远程化和个性化方向发展[120]。 近年来,多种创新型自愈水凝胶体系被用于智能康复设备。利用聚乙烯醇、秋葵多糖、硼砂及银纳米线构建的聚乙烯醇/秋葵多糖/硼砂导电水凝胶(夹层结构传感器),具有优异拉伸性(约1 073.7%)和快速自愈能力(5 min内愈合效率达93.6%), 其传感器表现出高灵敏度(GF=6.34)、快速响应(约20 ms)和良好的工作稳定性,可广泛用于运动康复监测[121]。 光热辅助自愈水凝胶:通过将多巴胺修饰聚吡咯涂层的Sb2S3纳米棒引入聚乙烯醇基体中,开发了光热辅助自愈水凝胶,材料的力学性能显著(拉伸强度1.25 MPa,伸长率620%),并具备优异的导电性(2.26 S/m),在近红外照射下该体系可在90 s内实现快速自愈,同时兼具应变传感、心电信号采集和能量收集功能,展现了“监测-治疗-能量回收”一体化的发展方向[122]。 电磁干扰屏蔽复合水凝胶:通过在三维银纳米线网络中引入聚丙烯酰胺和功能化单体制备的复合水凝胶,具有高电导率(83 S/cm)、超高拉伸应变(> 800%)和优异电磁干扰屏蔽性能(66 dB,X波段),自愈效率达90%,在愈合后仍能稳定监测人体运动,该研究为智能可穿戴电子与电磁干扰屏蔽的融合提供了新的思路[123]。 多功能黏附水凝胶:以精氨酸衍生物单体、离子液体和丙烯酸共聚合成,此水凝胶不仅可对多种运动开展监测,还表现出抗菌、形状记忆及荧光书写的相关特性,扩大了它在健康监测以及信息传递中的应用潜能[124]。 葡萄糖适应性自愈水凝胶:为适应糖尿病康复人群无创、实时监测的要求,研究人员开发出基于葡萄糖适应性聚乙烯醇水凝胶的摩擦电生物传感器,此体系借助葡萄糖氧化酶的固定化和β -环糊精包合作用,在汗液葡萄糖环境下调控导电性并转变为电信号,实现高灵敏状态下的无痛监测,此水凝胶可自行开展高血糖水平检测且依靠运动过程里的能量收集供电,极大拓宽了糖尿病患者运动康复过程里的管理手段[125]。 2.4.3 多功能集成化发展 随着运动医学与康复工程的不断发展,单一功能的传感或治疗已难以满足临床对精准化、个性化和长期化干预的需求。自愈水凝胶凭借其结构可调性和动态修复特性,为实现监测-治疗-康复一体化提供了新的契机。近年来,研究逐渐从单一性能优化转向多功能集成化设计,探索在同一体系中同时实现传感、药物递送、抗炎修复及能量管理等功能[126-127]。 在水凝胶网络中嵌入药物负载单元及导电网络,能同步实现运动应力监测以及药物控释[128]。基于pH值响应机制的自愈水凝胶,能在炎症微环境里“按需”释放非类固醇抗炎药物,同时监测局部受力变化,兼具损伤监测和精准治疗的两项功能,该智能反馈型材料为运动损伤的个性化康复想出了新思路[107]。在柔性储能范畴,作为电解质和隔膜采用水凝胶的固态超级电容器受关注,一项研究采用热聚合策略合成了聚丙烯酸-铁水凝胶,其Fe3+-羧酸可逆配位键让材料获得了优异的自愈性能与力学强度,还具备离子电导率(0.076 S/cm)与出色的循环稳定性,给予该材料的一体化间充质干细胞以二氧化锰碳布复合电极为基础,呈现出优异的电容保持率,还能借助汗液电解质达成自充电,这为自修复型、可穿戴能源纺织品的开发提供了关键指引[129]。 通过引入能量收集单元(如摩擦电纳米发电机TENG、光热转换颗粒),水凝胶不仅可作为应变传感器采集运动信号,还能将机械能转化为电能,用于驱动药物释放或电刺激治疗。掺杂近红外响应颗粒的水凝胶在光照下能够局部升温并触发药物释放,实现能量收集-监测-治疗的多功能耦合[130]。近期研究报道了一种基于β-环糊精接枝纤维素与纳米聚苯胺的导电水凝胶,该体系无需二次封装即可构建全凝胶超级电容器,表现出卓越的比电容(1 003.50 mF/cm2)、高能量密度(89.20 μWh/cm2) 和优异循环稳定性(2 000次循环后电容保持率90%),该装置可驱动发光二极管闪光灯,展现出在绿色储能与智能医疗领域的应用潜力[131]。 处于实际康复环境中,水凝胶材料需同时拥有抗冻、抵御干燥与紫外防护性能,以契合复杂的运动场景,普鲁士蓝与聚(3,4 - 乙撑二氧噻吩)/聚丙烯酰胺复合水凝胶器件不仅呈现出良好的力学性能,还能在-30 ℃低温环境下维持柔韧性,因光热响应特性,在太阳光照射期间可将赝电容回升到原始水平的287.1%,此研究证明了光热增强水凝胶赝电容器在低温环境下开展康复训练与进行能量存储的可行性[132]。 未来多功能集成水凝胶不应只是进行功能叠加,而应做到能编程、按时间顺序推进的多因子释放与响应,在康复起步的早期,可着重进行抗炎镇痛;中期主要推动血管新生与组织修复;后期需强化负荷监测以及运动反馈,将大数据跟人工智能结合,可构建起个体化康复闭环,借助自愈水凝胶电解质的柔性准固态锌离子电池,经过弯曲和切割愈合后还能保持高容量(> 120 mAhg-1)以及稳定的循环特性,这类器件不仅表现出自愈与能量存储的双重作用,还拥有应用在可穿戴康复电子范畴的潜力。 多功能自愈水凝胶正突破传统“单一功能材料”所设的局限,渐渐发展成将监测、治疗、能量管理和康复训练整合在一起的综合平台,这些进展体现出自愈水凝胶将在运动医学、柔性电子与智能康复交叉领域起到越来越关键的功效。 "
| [1] DING DY, PRENTICE HA, REYES C, et al. Patient and Operative Risk Factors for Subsequent Knee Arthroplasty After Primary Anterior Cruciate Ligament Reconstruction: A Cohort Study of 52,222 Patients. Am J Sports Med. 2025;53(10): 2370-2378. [2] OHSAWA T, KIMURA M, CHIKUDA H. Patient-reported evaluation on giving way is important for return to preinjury activity level after Anterior Cruciate Ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2020;29(4):1128-1136. [3] DAHLSTRÖM Ö, JACOBSSON J, TIMPKA T. Overcoming the organization-practice barrier in sports injury prevention: A nonhierarchical organizational model. Scand J Med Sci Sports. 2015;25(4):e414-422. [4] ACKERMAN IN, BOHENSKY MA, KEMP J L, et al. Likelihood of knee replacement surgery up to 15 years after sports injury: A population-level data linkage study. J Sci Med Sport. 2019;22(6): 629-634. [5] WEBER J, SZYMSKI D, HUBER L, et al. Knee joint dislocations-Current epidemiology and treatment in Germany. Knee Surg Sports Traumatol Arthrosc. 2024;33(6):2114-2121. [6] CHAKRAVARTHY J, RAMISETTY N, PIMPALNERKAR A, et al. Surgical repair of complete proximal hamstring tendon ruptures in water skiers and bull riders: a report of four cases and review of the literature. Br J Sports Med. 2005;39(8):569-572. [7] ISANI A. Prevention and treatment of ligamentous sports injuries to the hand. Sports Med. 1990; 9(1):48-61. [8] KANE P, FREDERICK R, TUCKER B, et al. Surgical restoration/repair of articular cartilage injuries in athletes. Physician Sportsmed. 2013;41(2):75-86. [9] KIM YH, SO WY. Effects of Rehabilitation in Bankart Lesion in Non-athletes: A Report of Three Cases. Open Med (Wars). 2019;14(1): 369-375. [10] SHELBOURNE KD, KLOOTWYK TE, CARR DR. Low-velocity knee disclocation associated with sports injury. Oper Tech Sports Med. 2003;11(3): 226-234. [11] CAI J, WANG J, YE K, et al. Dual-layer aligned-random nanofibrous scaffolds for improving gradient microstructure of tendon-to-bone healing in a rabbit extra-articular model. Int J Nanomedicine. 2018;13:3481-3492. [12] YANG G, LIN R, LI H, et al. Implantable wireless suture sensor for in situ tendon and ligament strain monitoring. Sci Adv. 2025;11(9):eadt3811. [13] TAYLOR DL, IN HET PANHUIS M. Self‐Healing Hydrogels. Adv Mater. 2016;28(41):9060-9093. [14] LEI J, LI X, WANG S, et al. Facile Fabrication of Biocompatible Gelatin-Based Self-Healing Hydrogels. ACS Appl Polym Mater. 2019;1(6): 1350-1358. [15] KIRCHMAJER DM, GORKIN III R, IN HET PANHUIS M. An overview of the suitability of hydrogel-forming polymers for extrusion-based 3D-printing. J Mater Chem B. 2015;3(20):4105-4117. [16] BROCHU ABW, CRAIG SL, REICHERT WM. Self-healing biomaterials. J Biomed Mater Res A. 2011; 96(2):492-506. [17] FANG Y, WANG CF, ZHANG ZH, et al. Robust self-healing hydrogels assisted by cross-linked nanofiber networks. Sci Rep. 2013;3:2811. [18] TOOHEY KS, SOTTOS NR, LEWIS JA, et al. Self-healing materials with microvascular networks. Nat Mater. 2007;6(8):581-585. [19] AHN BK, LEE DW, ISRAELACHVILI JN, et al. Surface-initiated self-healing of polymers in aqueous media. Nat Mater. 2014;13(9):867-872. [20] KROGSGAARD M, HANSEN MR, BIRKEDAL H. Metals & polymers in the mix: fine-tuning the mechanical properties & color of self-healing mussel-inspired hydrogels. J Mater Chem B. 2014;2(47):8292-8297. [21] KROGSGAARD M, BEHRENS MA, PEDERSEN JS, et al. Self-healing mussel-inspired multi-pH-responsive hydrogels. Biomacromolecules. 2013; 14(2):297-301. [22] ROTTENSTEINER U, SARKER B, HEUSINGER D, et al. In vitro and in vivo Biocompatibility of Alginate Dialdehyde/Gelatin Hydrogels with and without Nanoscaled Bioactive Glass for Bone Tissue Engineering Applications. Materials (Basel). 2014;7(3):1957-1974. [23] FERRIS CJ, GILMORE KG, WALLACE GG, et al. Biofabrication: an overview of the approaches used for printing of living cells. Appl Microbiol Biotechnol. 2013;97(10):4243-4258. [24] KITAMURA N, YASUDA K, OGAWA M, et al. Induction of spontaneous hyaline cartilage regeneration using a double-network gel: efficacy of a novel therapeutic strategy for an articular cartilage defect. Am J Sports Med. 2011; 39(6):1160-1169. [25] MERINO S, MARTÍN C, KOSTARELOS K, et al. Nanocomposite Hydrogels: 3D Polymer-Nanoparticle Synergies for On-Demand Drug Delivery. ACS Nano. 2015;9(5):4686-4697. [26] LI L, YAN B, YANG J, et al. Novel mussel-inspired injectable self-healing hydrogel with anti-biofouling property. Adv Mater. 2015;27(7): 1294-1299. [27] APPEL EA, TIBBITT MW, WEBBER MJ, et al. Self-assembled hydrogels utilizing polymer-nanoparticle interactions. Nat Commun. 2015; 6:6295. [28] VASHIST A, VASHIST A, GUPTA YK, et al. Recent advances in hydrogel based drug delivery systems for the human body. J Mater Chem B. 2014;2(2):147-166. [29] SALEHI SM, PROFIO GD, FONTANANOVA E, et al. Membrane distillation by novel hydrogel composite membranes. J Membr Sci. 2016;504: 220-229. [30] SU T, LIU Y, HE H, et al. Strong Bioinspired Polymer Hydrogel with Tunable Stiffness and Toughness for Mimicking the Extracellular Matrix. ACS Macro Lett. 2016;5(11):1217-1221. [31] XIAO L, ZHOU Y, ZHANG C, et al. Improving impact toughness of Fe–20Mn–9Al-1.5C–2Ni–3Cr low-density steel by optimizing grain boundaries via multi-stage heat treatment without compromising high strength and ductility. J Mater Res Technol. 2024;29:2396-2404. [32] GONG JP. Materials science. Materials both tough and soft. Science. 2014;344(6180):161-162. [33] LUO F, SUN TL, NAKAJIMA T, et al. Oppositely charged polyelectrolytes form tough, self-healing, and rebuildable hydrogels. Adv Mater. 2015;27(17):2722-2727. [34] LUO F, SUN TL, NAKAJIMA T, et al. Free Reprocessability of Tough and Self-Healing Hydrogels Based on Polyion Complex. ACS Macro Lett. 2015;4(9):961-964. [35] ZHAO X. Multi-scale multi-mechanism design of tough hydrogels: building dissipation into stretchy networks. Soft Matter. 2014;10(5):672-687. [36] EJEROMEDOGHENE O, KUMI M, AKOR E, et al. The application of machine learning in 3D/4D printed stimuli-responsive hydrogels. Adv Colloid Interface Sci. 2025;336:103360. [37] HASHEMI B, ASSADPOUR E, WANG Y, et al. Application of oleogels, hydrogels and bigels as novel edible inks for 3D/4D printing of food products. Adv Colloid Interface Sci. 2025;343:103578. [38] ALLI YA, ANUAR H, BAMISAYE A, et al. The appealing prospect of hydrogel in 3D/4D printing technology: Overview and opportunities. Polymer. 2024;315:127823. [39] PERRUCHOUD D, PISOTTA I, CARDA S, et al. Biomimetic rehabilitation engineering: the importance of somatosensory feedback for brain-machine interfaces. J Neural Eng. 2016;13(4): 041001. [40] ISLAM MRR, ISLAM R, SANO H, et al. Emerging trends of injectable hydrogels for vital pulp therapy: A comprehensive review. Int Endod J. 2025;58(10):1490-1528. [41] XU J, HSU SH. Self-healing hydrogel as an injectable implant: translation in brain diseases. J Biomed Sci. 2023;30(1):43. [42] KARVINEN J, KELLOMÄKI M. Characterization of self-healing hydrogels for biomedical applications. Eur Polym J. 2022;181:111641. [43] QIN T, LIAO W, YU L, et al. Recent progress in conductive self-healing hydrogels for flexible sensors. J Polym Sci. 2022;60(18):2607-2634. [41] TANG H, KANG B, LI Y, et al. Self-Healing Hydrogels as Flexible Sensor for Human Motion Monitoring. ChemistrySelect. 2021;6(40):11130-11136. [45] ZHANG N, ZHAO G, GAO F, et al. Wearable Flexible Sensors for Human Motion Detection with Self-Healing, Tough Guar Gum-Hydrogels of GO-P4VPBA/PDA Janus Nanosheets. ACS Appl Polym Mater. 2022;4(5):3394-3407. [46] GE G, YUAN W, ZHAO W, et al. Highly stretchable and autonomously healable epidermal sensor based on multi-functional hydrogel frameworks. J Mater Chem A. 2019;7(11):5949-5956. [47] HUANG X, GE G, SHE M, et al. Self-healing hydrogel with multiple dynamic interactions for multifunctional epidermal sensor. Appl Surf Sci. 2022;598:153803. [48] LI W, CHEN Z, XU C, et al. An omnidirectionally self-healable supramolecular hydrogel supercapacitor via cooperative effect of a dynamically dual-cross-linked network. J Energy Storage. 2025; 132:117983. [49] OLLIER RC, WEBBER MJ. Strain-Stiffening Mechanoresponse in Dynamic-Covalent Cellulose Hydrogels. Biomacromolecules. 2024;25(7): 4406-4419. [50] GINTING M, MASMUR I, PASARIBU SP, et al. A simple one-pot fabrication of silver loaded semi-interpenetrating polymer network (IPN) hydrogels with self-healing and bactericidal abilities. RSC Adv. 2019;9(67):39515-39522. [51] CUI W, PI MH, LI YS, et al. Multimechanism Physical Cross-Linking Results in Tough and Self-Healing Hydrogels for Various Applications. ACS Appl Energy Mater. 2020;2(8):3378-3389. [52] KHATTAK S, ULLAH I, XIE H, et al. Self-healing hydrogels as injectable implants: Advances in translational wound healing. Coord Chem Rev. 2024;509:215790. [53] CAI L, XIONG X, QIAO M, et al. Aggregation-induced emission luminogen based self-healing hydrogels fluorescent sensors for α-amylase. Polym Chem. 2022;13(6):819-828. [54] LI W, LIU H, MI Y, et al. Robust and conductive hydrogel based on mussel adhesive chemistry for remote monitoring of body signals. Friction. 2020;10(1):80-93. [55] HEIDARIAN P, KAYNAK A, PAULINO M, et al. Dynamic nanocellulose hydrogels: Recent advancements and future outlook. Carbohydr Polym. 2021;270:118357. [56] HEIDARIAN P, KOUZANI AZ, KAYNAK A, et al. Dynamic Mussel-Inspired Chitin Nanocomposite Hydrogels for Wearable Strain Sensors. Polymers (Basel). 2020;12(6):1416. [57] DENG CC, BROOKS WLA, ABBOUD KA, et al. Boronic Acid-Based Hydrogels Undergo Self-Healing at Neutral and Acidic pH. ACS Macro Lett. 2015;4(2):220-224. [58] FU J. Hydrogel electronics: New horizons of flexible, wearable, and implantable devices. J Polym Sci. 2022;60(18):2605-2606. [59] XIE T, VOGT BD. A Virtual Special Issue on Self-Healing Materials. ACS Appl Mater Interfaces. 2020;12(44):49277-49280. [60] XU Q, HOU M, WANG L, et al. A spider silk-inspired, transparent, anti-freezing ionic conductive hydrogel as a flexible sensor device. J Mater Chem B. 2025;13(16):4842-4854. [61] MENG Q, ZHANG N, XIAO M, et al. Hydrogel-based treatment of ACL injuries: mechanisms, applications and prospects. Eur Polym J. 2025;238:114222. [62] LI W, WU Y, ZHANG X, et al. Self-healing hydrogels for bone defect repair. RSC Adv. 2023; 13(25):16773-16788. [63] YU F, GENG D, KUANG Z, et al. Sequentially releasing self-healing hydrogel fabricated with TGFβ3-microspheres and bFGF to facilitate rat alveolar bone defect repair. Asian J Pharm Sci. 2022;17(3):425-434. [64] LI D, YANG Z, ZHAO X, et al. A bone regeneration strategy via dual delivery of demineralized bone matrix powder and hypoxia-pretreated bone marrow stromal cells using an injectable self-healing hydrogel. J Mater Chem B. 2021;9(2):479-493. [65] EUFRÁSIO-DA-SILVA T, EREZUMA I, DOLATSHAHI-PIROUZ A, et al. Enhancing regenerative medicine with self-healing hydrogels: A solution for tissue repair and advanced cyborganic healthcare devices. Biomater Adv. 2024;161:213869. [66] WU S, ZHANG Z, XU R, et al. A spray-filming, tissue-adhesive, and bioactive polysaccharide self-healing hydrogel for skin regeneration. Mater Des. 2022;217:110669. [67] DAI S, WANG S, YAN H, et al. Stretchable and self-healable hydrogel-based capacitance pressure and strain sensor for electronic skin systems. Mater Res Express. 2019;6(8):0850b9. [68] SEO J, OH S, CHOI G, et al. Multifunctional Double-Network Self-Healable Hydrogel and Its Application to Highly Reliable Strain Sensors. ACS Appl Polym Mater. 2022;4(9):6495-6504. [69] HUSSAIN I, MA X, LUO Y, et al. Fabrication and characterization of glycogen-based elastic, self-healable, and conductive hydrogels as a wearable strain-sensor for flexible e-skin. Polymer. 2020;210:122961. [70] DANG X, FU Y, WANG X. Versatile Biomass-Based Injectable Photothermal Hydrogel for Integrated Regenerative Wound Healing and Skin Bioelectronics. Adv Funct Mater. 2024;34(42): 2405745. [71] ZHOU Y, XU B, ZHOU P, et al. Gold@mesoporous polydopamine nanoparticles modified self-healing hydrogel for sport-injuring therapy. Int J Biol Macromol. 2023;253:127441. [72] MIGLIORINI F, PINTORE A, OLIVA F, et al. Allografts as alternative to autografts in primary posterior cruciate ligament reconstruction: a systematic review and meta-analysis. Knee Surg Sports Traumatol Arthrosc. 2022;31(7):2852-2860. [73] GUO H, HUANG S, YANG X, et al. Injectable and Self-Healing Hydrogels with Double-Dynamic Bond Tunable Mechanical, Gel–Sol Transition and Drug Delivery Properties for Promoting Periodontium Regeneration in Periodontitis. ACS Appl Mater Interfaces. 2021;13(51): 61638-61652. [74] CHEN Y, SUN X, LUO L, et al. Super Strong and Tough PVA Hydrogel Fibers Based on an Ordered-to-Disordered Structural Construction Strategy Targeting Artificial Ligaments. Adv Funct Mater. 2025;35(8):2415737. [75] HAN S, WU Q, ZHU J, et al. Tough hydrogel with high water content and ordered fibrous structures as an artificial human ligament. Mater Horiz. 2023; 10(3):1012-1019. [76] CHOI S, CHOI Y, KIM J. Anisotropic Hybrid Hydrogels with Superior Mechanical Properties Reminiscent of Tendons or Ligaments. Adv Funct Mater. 2019;29(38):1904342. [77] WRIGHT RW, HUSTON LJ, NWOSU SK, et al. Meniscal and Articular Cartilage Predictors of Clinical Outcome After Revision Anterior Cruciate Ligament Reconstruction. Am J Sports Med. 2016; 44(7):1671-1679. [78] HARIS A, TAN VBC. Stress response envelopes of intact tibiofemoral joint and knee osteoarthritis. Proc Inst Mech Eng H. 2020;234(10):1151-1161. [79] LI L, YANG L, ZHANG K, et al. Three-dimensional finite-element analysis of aggravating medial meniscus tears on knee osteoarthritis. J Orthop Translat. 2020;20:47-55. [80] CHEN M, FENG Z, GUO W, et al. PCL-MECM-Based Hydrogel Hybrid Scaffolds and Meniscal Fibrochondrocytes Promote Whole Meniscus Regeneration in a Rabbit Meniscectomy Model. ACS Appl Mater Interfaces. 2019;11(44): 41626-41639. [81] ZHAO H, ZHANG Y, LIU Y, et al. In Situ Forming Cellulose Nanofibril-Reinforced Hyaluronic Acid Hydrogel for Cartilage Regeneration. Biomacromolecules. 2021;22(12):5097-5107. [82] BABALUEI M, MOJARAB Y, MOTTAGHITALAB F, et al. Injectable multifunctional hydrogels based on dopamine grafted onto tragacanth/carboxymethyl chitosan/silk fibroin containing graphene oxide@vancomycin and pirfenidone for infected wound healing. Chem Eng J. 2025; 519:164945. [83] GAN D, XU T, XING W, et al. Mussel-inspired dopamine oligomer intercalated tough and resilient gelatin methacryloyl (GelMA) hydrogels for cartilage regeneration. J Mater Chem B. 2019; 7(10):1716-1725. [84] CHEN Z, ZHANG T, CHEN CT, et al. Mechanically and electrically biocompatible hydrogel ionotronic fibers for fabricating structurally stable implants and enabling noncontact physioelectrical modulation. Mater Horiz. 2022;9(6):1735-1749. [85] LI W, QIAO K, ZHENG Y, et al. Preparation, mechanical properties, fatigue and tribological behavior of double crosslinked high strength hydrogel. J Mech Behav Biomed Mater. 2022;126: 105009. [86] YE Z, LU H, CHAI G, et al. Glycerol-modified poly(vinyl alcohol)/poly(ethylene glycol) self-healing hydrogel for artificial cartilage. Polym Int. 2022;72(1):27-38. [87] ZHU W, WANG H, FENG B, et al. Self-Healing Hyaluronic Acid-based Hydrogel with miRNA140-5p Loaded MON-PEI Nanoparticles for Chondrocyte Regeneration: Schiff Base Self-Assembly Approach. Adv Sci (Weinh). 2025;12(1): e2406479. [88] GUO L, LIU X, WANG Y, et al. Enhancing long-segmental tracheal restoration: A self-repairing hydrogel loaded with chondrocytokines for sutureless anastomosis and cartilage regeneration. Mater Today Bio. 2024;28:101208. [89] KANG M, CHENG Y, HU Y, et al. Self-healing poly(acrylic acid) hydrogels fabricated by hydrogen bonding and Fe3 ion cross-linking for cartilage tissue engineering. Front Mater Sci. 2023;17(3). doi:10.1007/s11706-023-0655-7. [90] GHOBASHY MM, EL-SATTAR NEAA. Radiation Synthesis of Rapidly Self‐Healing Hydrogel Derived from Poly(acrylic acid) with Good Mechanical Strength. Macromol Chem Phys. 2020;221(19). doi:10.1002/macp.202000218. [91] KHAN MUA, ASLAM MA, ABDULLAH MFB, et al. Recent advances of bone tissue engineering: carbohydrate and ceramic materials, fundamental properties and advanced biofabrication strategies-a comprehensive review. Biomed Mater. 2024;19(5):052005. [92] COUTO M, KUROWSKI M, MOREIRA A, et al. Mechanisms of exercise-induced bronchoconstriction in athletes: Current perspectives and future challenges. Allergy. 2017;73(1):8-16. [93] VAN LAARHOVEN SN, VLES GF, VAN HAAREN EH, et al. Tapered, fluted, modular, titanium stems in Vancouver B periprosthetic femoral fractures: an analysis of 87 consecutive revisions. Hip Int. 2020;31(4):555-561. [94] EREZUMA I, LUKIN I, DESIMONE M, et al. Progress in self-healing hydrogels and their applications in bone tissue engineering. Biomater Adv. 2023; 146:213274. [95] LÜ S, BAI X, LIU H, et al. An injectable and self-healing hydrogel with covalent cross-linking in vivo for cranial bone repair. J Mater Chem B. 2017;5(20):3739-3748. [96] LI M, ZHOU Y, LI X, et al. Highly stretchable, injectable hydrogels with cyclic endurance and shape-stability in dynamic mechanical environments, by microunit reformation. J Mater Chem B. 2023;11(13):3001-3013. [97] TAN Y, MA L, CHEN X, et al. Injectable hyaluronic acid/hydroxyapatite composite hydrogels as cell carriers for bone repair. Int J Biol Macromol. 2022; 216:547-557. [98] HU ZC, LU JQ, ZHANG TW, et al. Piezoresistive MXene/Silk fibroin nanocomposite hydrogel for accelerating bone regeneration by Re-establishing electrical microenvironment. Bioact Mater. 2023; 22:1-17. [99] HUANG J, TAN QC, BAI H, et al. Harnessing immunomodulation for efficient bone Regeneration: Bioengineered black phosphorus-incorporated Self-Healing hydrogel. Chem Eng J. 2023;470:144117. [100] XUE J, FENG C, XIA L, et al. Assembly Preparation of Multilayered Biomaterials with High Mechanical Strength and Bone-Forming Bioactivity. Chem Mater. 2018;30(14):4646-4657. [101] ES-HAGHI S, WEISS RA. Finite strain damage-elastoplasticity in double-network hydrogels. Polymer. 2016;103: 277-287. [102] SAMAN NM, AHMAD MH, BUNTAT Z. Application of Cold Plasma in Nanofillers Surface Modification for Enhancement of Insulation Characteristics of Polymer Nanocomposites: A Review. IEEE Access. 2021;9:80906-80930. [103] ZHU Y, LIU H, WU P, et al. Multifunctional injectable hydrogel system as a mild photothermal-assisted therapeutic platform for programmed regulation of inflammation and osteo-microenvironment for enhanced healing of diabetic bone defects in situ. Theranostics. 2024;14(18):7140-7198. [104] DONG X, ZHAO J, JIANG D, et al. Self-healing hydrogel reduces inflammation through ANT1/OPTN axis mediated mitophagy for spinal cord injury repair. Chem Eng J. 2024;492:152263. [105] CHHILLAR A, JAISWAL A. Hyaluronic Acid-Based Self-Healing Hydrogels for Diabetic Wound Healing. Adv Healthc Mater. 2025;14(4):e2404255. [106] WU C, NING X, LIU Q, et al. Sustained Release of Curcumin from Cur-LPs Loaded Adaptive Injectable Self-Healing Hydrogels. Polymers (Basel). 2024;16(24):3451. [107] XU J, CHEN TY, TAI CH, et al. Bioactive self-healing hydrogel based on tannic acid modified gold nano-crosslinker as an injectable brain implant for treating Parkinson’s disease. Biomater Res. 2023;27(1):8. [108] CHEN J, LUO J, FENG J, et al. Spatiotemporal controlled released hydrogels for multi-system regulated bone regeneration. J Control Release. 2024;372:846-861. [109] FRANGOGIANNIS NG. Fact and Fiction About Fibroblast to Endothelium Conversion. Circulation. 2020;142(17):1663-1666. [110] QIN D, ZHANG A, WANG N, et al. Hydroxybutyl chitosan/ oxidized glucomannan self-healing hydrogels as BMSCs-derived exosomes carriers for advanced stretchable wounds. Appl Mater Today. 2022;26:101342. [111] LIU T, WANG F, WU Q, et al. Fluorescent, electrically responsive and ultratough self-healing hydrogels via bioinspired all-in-one hierarchical micelles. Mater Horiz. 2021;8(11):3096-3104. [112] DIAO Q, PENG J, YANG W, et al. A versatile organohydrogel with ultra-stretchability, self-healing and self-adhesion for multifunctional flexible electronics. Colloids Surf A Physicochem Eng Asp. 2025;725:137707. [113] MIRICA KA. Unlocking the Potential of Wearable Sensors in Healthcare and Beyond. ACS Sens. 2024;9(2):533-534. [114] ZHAO X, WANG H, LUO J, et al. Ultrastretchable, Adhesive, Anti-freezing, Conductive, and Self-Healing Hydrogel for Wearable Devices. ACS Appl Polym Mater. 2022;4(3):1784-1793. [115] YANG Y, SUN H, SHI C, et al. Self-healing Hydrogel with Multiple Adhesion as Sensors for Winter Sports. J Colloid Interface Sci. 2023; 629:1021-1031. [116] CHEN Y, DAI S, ZHU H, et al. Self-healing hydrogel sensors with multiple shape memory properties for human motion monitoring. New J Chem. 2021;45(1):314-320. [117] XING R, HUANG R, SU R, et al. Facile fabrication of a printable conductive self-healing hydrogel for human motion and electrocardiogram monitoring. New J Chem. 2023;47(23): 11063-11070. [118] ZHAO Y, LI L, ZHANG J, et al. A High-Accuracy Facial Expression Recognition System Combining Triboelectric Hydrogel Sensors With Deep Learning. Adv Funct Mater. 2025;35(19): 02418265. [119] GAO W, YU C. Wearable and Implantable Devices for Healthcare. Adv Healthc Mater. 2021;10(17): e2101548. [120] FU Y, DANG X. Bio‐Inspired Highly Stretchable and Ultrafast Autonomous Self‐Healing Supramolecular Hydrogel for Multifunctional Durable Self‐Powered Wearable Devices. Small. 2025;21(7):e2408640. [121] MA Y, LIU K, LAO L, et al. A stretchable, self-healing, okra polysaccharide-based hydrogel for fast-response and ultra-sensitive strain sensors. Int J Biol Macromol. 2022;205:491-499. [122] XU L, CHEN Y, YU M, et al. NIR light-induced rapid self-healing hydrogel toward multifunctional applications in sensing. Nano Energy. 2023;107: 108119. [123] HUANG X, WANG L, SHEN Z, et al. Super-Stretchable and Self-Healing hydrogel with a Three-Dimensional silver nanowires network structure for wearable sensor and electromagnetic interference shielding. Chem Eng J. 2022;446:137136. [124] ZHANG H, HE J, PENG T, et al. Ultra-stretchable, Antifatigue, Adhesive, and Self-Healing Hydrogels Based on the Amino Acid Derivative and Ionic Liquid for Flexible Strain Sensors. ACS Appl Polym Mater. 2022;4(10):7575-7586. [125] KANOKPAKA P, CHANG YH, CHANG CC, et al. Enabling glucose adaptive self-healing hydrogel based triboelectric biosensor for tracking a human perspiration. Nano Energy. 2023;112:108513. [126] CHENG T, LI L, CHEN YL, et al. Stretchable and Self-Healing Interlocking All-in-One Supercapacitors Based on Multiple Cross-Linked Hydrogel Electrolytes. Adv Mater Interfaces. 2022;9(29): 2201137. [127] WANG J, LIU Z, ZHOU Y, et al. A multifunctional sensor for real-time monitoring and pro-healing of frostbite wounds. Acta Biomater. 2023;172: 330-342. [128] DAS P, GANGULY S, SARAVANAN A, et al. Naturally Derived Carbon Dots In Situ Confined Self-Healing and Breathable Hydrogel Monolith for Anomalous Diffusion-Driven Phytomedicine Release. ACS Appl Bio Mater. 2022;5(12): 5617-5633. [129] YANG X, CHENG X, LIAO S, et al. A Self-Healing and Sweat-Chargeable Hydrogel Electrolyte for All-in-One Flexible Supercapacitors. ACS Appl Mater Interfaces. 2024;16(37):49337-49348. [130] WANG W, SUN C, JING L, et al. Multifunctional nanocomposite hydrogel dressing with low-temperature photothermal and controlled antibiotics release for combating bacterial infection. Mater Des. 2024;239:112812. [131] GONG Q, LI Y, LIU X, et al. A facile preparation of polyaniline/cellulose hydrogels for all-in-one flexible supercapacitor with remarkable enhanced performance. Carbohydr Polym. 2020; 245:116611. [132] LU Y, LI K, SONG YY, et al. Ultra-flexible all-in-one anti-freeze photothermally enhanced supercapacitors. Nano Res. 2024;17(8): 7221-7229. [133] YU H, LIU Y, YANG H, et al. An Injectable Self‐Healing Hydrogel Based on Chain‐Extended PEO‐PPO‐PEO Multiblock Copolymer. Macromol Rapid Commun. 2016;37(21): 1723-1728. [134] LIU S, RAO Z, WU R, et al. Fabrication of Microcapsules by the Combination of Biomass Porous Carbon and Polydopamine for Dual Self-Healing Hydrogels. J Agric Food Chem. 2019; 67(4):1061-1071. [135] SUN Q, XIAO L, NIE Y, et al. Fabrication of Janus-type nanocomposites from cellulose nanocrystals for self-healing hydrogels’ flexible sensors. Colloids Surf B Biointerfaces. 2022; 216:112554. [136] DOU Z, TANG H, CHEN K, et al. Highly elastic and self-healing nanostructured gelatin/clay colloidal gels with osteogenic capacity for minimally invasive and customized bone regeneration. Biofabrication. 2023;15(2):025001. |
| [1] | Li Guangmin, Wang Jiangna. Mechanical loading of the patellofemoral joint and its correlation with various body mass indices in older adults practicing Tai Chi exercises [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6182-6188. |
| [2] | Liu Mengling, Li Yongjie, Liu Hongju. Effect of increased stride length on knee kinematics and dynamics of asymmetric gait after anterior cruciate ligament reconstruction [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(33): 7109-7115. |
| [3] | Wei Zhiheng, Guan Tianmin, Liu Qing, Gong Jue, Xiang Xianxiang. Application of 3D printing accurate osteotomy guide combined with the revision of anterior cruciate ligament with abnormally increased posterior slope of tibial plateau [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(33): 7130-7136. |
| [4] | Gao Yuan, Xiong Zheyu, Zheng Wei, Chen Haonan, Chen Fangyuqing. Lower extremity biomechanical characterization during step-down test in patients with patellofemoral pain [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(32): 6913-6919. |
| [5] | Wang Changbing, Zhao Lilian, Fu Chuying, Li Yanjin. Modified double-bundle arthroscopic repair of the anterior cruciate ligament after Sherman type I injury [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1192-1198. |
| [6] | You Jing, Huang Wenqi, Zheng Wei, Lu Jieming, Guo Yanhua, Gao Yuan, Xiong Zheyu. Effects of kinesio taping on the biomechanical characteristics of the lower limbs during side-step cutting [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(27): 4383-4389. |
| [7] | Zhang Xuebin, Ao Wenjun, Jiang Xin, Song Chenglin. Improvement of delayed-onset muscle soreness by pre-exercise whole-body vibration at 50 Hz [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(32): 5085-5090. |
| [8] | Liu Kun, Yin Lulu, Ma Zheng, Huang Lihua, Zhu Ran, Fang Ping, Zhang Ye, Ma Yanhong. Kinesiology tape for treating shoulder pain [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(14): 2214-2221. |
| [9] | Tan Xinfang, Guo Yanxing, Qin Xiaofei, Zhang Binqing, Zhao Dongliang, Pan Kunkun, Li Yuzhuo, Chen Haoyu. Effect of uniaxial fatigue exercise on patellofemoral cartilage injury in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(14): 2184-2189. |
| [10] | Shao Yangyang, Zhang Junxia, Jiang Meijiao, Liu Zelong, Gao Kun, Yu Shuhan. Kinematics characteristics of lower limb joints of young men running wearing knee pads [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 832-837. |
| [11] | Tan Xinfang, Guo Yanxing, Qin Xiaofei, Zhang Binqing, Zhao Dongliang, Pan Kunkun, Li Yuzhuo, Chen Haoyu. Effect of uniaxial fatigue exercise on patellofemoral cartilage injury in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(在线): 1-6. |
| [12] | Wang Bo, Bai Shengchao, Li Wenbo, Wang Lin. A rabbit model of high-intensity jumping-induced tendon injury followed by post-exercise cold water immersion: inflammations and collagen remodeling in the patellar tendon [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(29): 4619-4625. |
| [13] | Bai Shengchao, Gao Yang, Wang Bo, Li Junping, Wang Ruiyuan. Dynamic changes of mitochondrial function of the skeletal muscle after acupuncture intervention in rats with heavy load exercise-induced injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3648-3653. |
| [14] | Wang Jiangna, Zheng Huifen, Sun Wei. Changes in dynamic stability, motor coordination and joint mechanics of the lower extremity during stair descent and performing phone task [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 837-843. |
| [15] | Zhao Yanxu, Zhang Zhao, Luo Dong, Li Manglai, Wang Jing, Fang Jie, Jiang Wen, Feng Yong, Peng Jiang, Xu Wenjing, Wang Yu, Wang Aiyuan, Zhang Minze. Risk factors for stress fracture of lower limbs and future research direction [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(9): 1422-1429. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||