Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (32): 8536-8543.doi: 10.12307/2026.470
Previous Articles Next Articles
Material selection and manufacture method of non-porous adherent cell microcarriers
Sun Jingshan, Deng Jingqian, Qi Liya, Zhao Xiaohuan, Hou Dandan
- SINOPEC (Beijing) Research Institute of Chemical Industry Co., Ltd., SINOPEC Key Laboratory of Medical and Sanitary Materials Research and Application, Beijing 100013, China
-
Accepted:2026-03-09Online:2026-11-18Published:2026-04-29 -
Contact:Hou Dandan PhD, Researcher, SINOPEC (Beijing) Research Institute of Chemical Industry Co., Ltd., SINOPEC Key Laboratory of Medical and Sanitary Materials Research and Application, Beijing 100013, China -
About author:Sun Jingshan, MS, Assistant engineer, SINOPEC (Beijing) Research Institute of Chemical Industry Co., Ltd., SINOPEC Key Laboratory of Medical and Sanitary Materials Research and Application, Beijing 100013, China -
Supported by:SINOPEC Funding Project, Nos. KL223009 and 225058 (to HDD)
CLC Number:
Cite this article
Sun Jingshan, Deng Jingqian, Qi Liya, Zhao Xiaohuan, Hou Dandan. Material selection and manufacture method of non-porous adherent cell microcarriers[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8536-8543.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
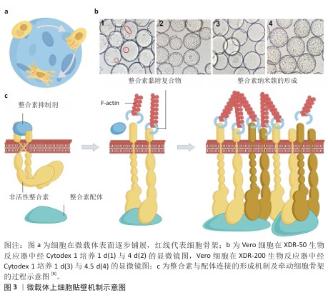
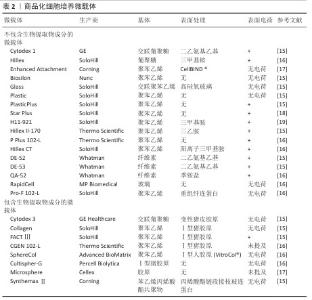
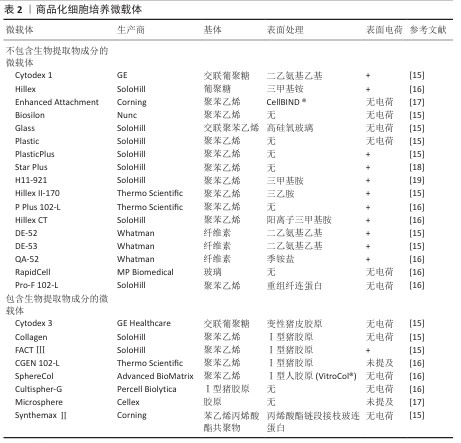
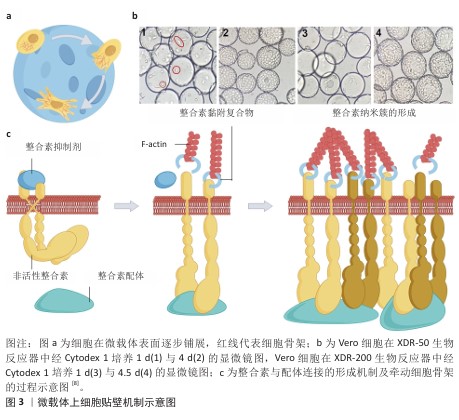
2.2 微载体上细胞贴壁的有利条件 整合素家族是调控细胞与细胞外基质黏附过程的关键蛋白,整合素形成的多种跨膜蛋白复合物通过连接细胞外基质与细胞骨架将细胞固定,整合素介导的连接形成后,细胞随之铺展,形成伪足[7](图3)。 血清含有多种整合素的配体蛋白,纤连蛋白与玻连蛋白是其中重要的两种[8]。KLEBE等[9]研究了50余种材料表面的纤连蛋白吸附与细胞增殖的关系,经纤连蛋白溶液浸润后的阳离子聚合物或疏水表面有利于细胞黏附与增殖。ISHIHARA等[10]报道了纤连蛋白在两性离子聚合物和阴离子聚合物表面上的吸附非常困难,容易吸附在阳离子聚合物和疏水表面上。 20世纪70-80年代,HIRTENSTEIN等[11]与MAROUDAS等[12]的研究均指出,微载体的电荷密度是调控细胞贴壁的关键,后续的多项研究以微载体的离子交换能力表征微载体电荷密度。细胞贴壁与增殖需微载体的离子交换能力达阈值,不同材质的微载体该阈值不同。表面接枝二乙氨基乙基的交联葡聚糖微球(Sephadex A-50)离子交换能力超过0.9 meq/g后,微球显现出支持细胞黏附和增殖的能力,当离子交换能力超过3.5 meq/g后则显现出细胞毒性,不能够支持细胞增殖[1]。GE公司在做出一系列不同离子交换能力的表面接枝二乙氨基乙基交联葡聚糖微球后,开发了Cytodex 1微载体,该载体的离子交换能力为1.5 meq/g,能够广谱地支持细胞在其表面贴壁与增殖。伯胺接枝的聚丙烯酰胺微球载体的离子交换能力为0.58 meq/g时完全无法支持细胞黏附,达到1.8-2 meq/g后该载体支持BHK细胞、MDCK细胞、CEF细胞、MRC-5细胞黏附和增殖[13-14]。根据上述研究,具有适量正电荷的微载体有利于细胞黏附,市售产品的表面电荷情况见表2。 除纤连蛋白外,玻连蛋白也是调控细胞贴壁的重要蛋白[20]。由于微载体细胞培养技术开发始于1967年,而利于玻连蛋白吸附的表面条件在近10年才有部分学者研究,因此,微载体表面性质研究的模型蛋白是玻连蛋白。LI等[21]的研究报道了材料表面氨基、羟基、羧基、甲基对玻连蛋白吸附的影响,为后续的生物材料开发提供了参考。 除微载体吸附纤连蛋白与玻连蛋白外,在微载体表面接枝整合素配体也是一种促进细胞黏附的策略。整合素配体还包括胶原蛋白、层粘连蛋白、软骨粘连蛋白、血小板反应蛋白、硫酸乙酰肝素、硫酸软骨素等多种蛋白[22]。GE公司开发的Cytodex 3微载体为在葡聚糖微球表面接枝变性的Ⅰ型胶原,能够支持多种细胞的黏附和生长,然而胶原会使部分细胞分化,因此需注意使用范围。胶原对大鼠脂肪间充质干细胞的分化没有影响,然而对于干细胞药物,一方面需严格限制异种成分的使用,另一方面使用异种成分增加了后续干细胞药物质量检测的难度,因此对于干细胞药物制造,如必须采用含有胶原的微载体,则需明确胶原是否为重组人源化胶原蛋白。 "
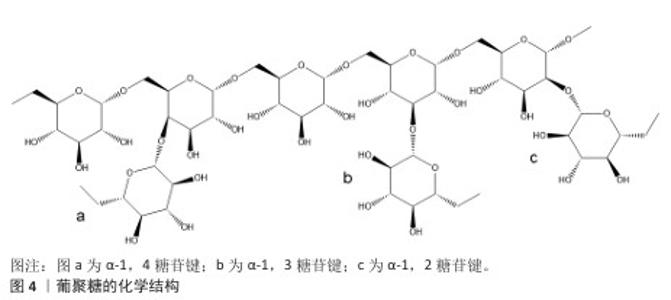
2.3 细胞培养微载体基质材料 2.3.1 葡聚糖 葡聚糖微载体在显微镜下透明,便于观察细胞生长状态以及粗略估计细胞密度,是制备细胞培养微载体的理想材料。葡聚糖的化学结构见图4,其是一种以α-1,6糖苷键为主链,α-1,2、α-1,3、α-1,4为支链的葡聚糖,不同牌号的葡聚糖各糖苷键比例不一,不同支化度的葡聚糖在水溶液中的溶解度也不同。市售的葡聚糖牌号众多,DíAZ-MONTES[23]研究了80余个牌号葡聚糖的制备方法、分子质量、糖苷键种类等信息,为葡聚糖微球制备提供了重要参考。 Cytodex 1是交联的表面修饰二乙氨基乙基的葡聚糖凝胶微球,GE公司售卖的Cytodex 1为干粉,即为脱水后的葡聚糖凝胶,其溶胀系数为18。葡聚糖的枝化度直接影响了交联过程的难易程度,枝化度过高的葡聚糖不溶于水,不适宜制备凝胶微球[24]。作者认为葡聚糖的枝化度同时影响了葡聚糖微球的溶胀系数及机械强度,如制备葡聚糖微球,需谨慎地选择原料来源。 葡聚糖侧基为羟基,无法提供细胞黏附所需的电荷条件,因此需在葡聚糖微载体表面修饰带有正电荷的小分子或胶原。由于VAN WEZEL[1]在1967年的研究以蛋白纯化柱层析的离子交换凝胶充当微载体(Sephadex A-50-DEAE),同时二乙氨乙基化葡聚糖为一种成熟的商品,因此后续交联葡聚糖微球多以二乙氨乙基为提供正电荷的小分子。该文2.2章节详细说明了适合细胞贴壁与增殖的葡聚糖微载体电荷密度,VAN WEZEL[1]验证了过多正电荷修饰的葡聚糖微球具有细胞毒性,由此说明葡聚糖微载体的电荷密度分布范围较窄。 "
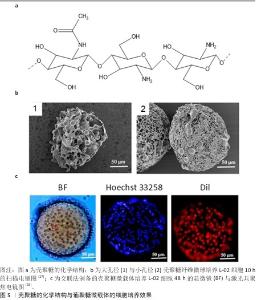
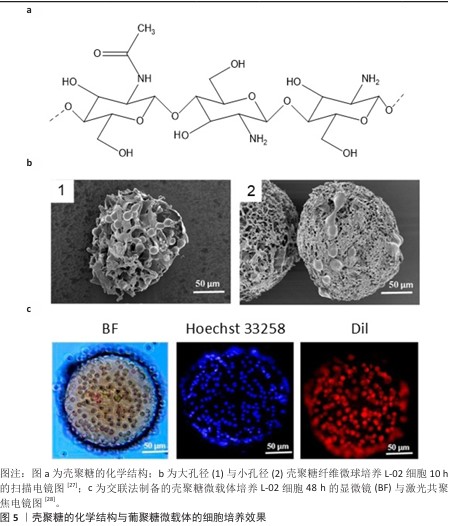
2.3.2 壳聚糖 壳聚糖即脱乙酰化的甲壳素(几丁质),结构式如图5a所示。从脱乙酰化程度上分类,50%-70%为低脱乙酰度壳聚糖,71%-85%为中脱乙酰度壳聚糖,86%-95%为高脱乙酰度壳聚糖[25]。随着脱乙酰度增高,壳聚糖氨基增多,在酸性水溶液中的溶解度增高。 壳聚糖的侧氨基能够提供正电荷,壳聚糖微载体的电荷密度取决于其脱乙酰化程度[26],由于质子化后的壳聚糖可溶于稀酸水溶液,因此,从壳聚糖在稀醋酸中的溶解情况能间接地表明载体电荷密度。 由于具有羟基、氨基以及醚键,壳聚糖可通过氢键自组装为纤维。壳聚糖纤维球是多孔球,细胞直径约为10 μm,过大的孔径会使细胞纵向生长进入孔隙内部,不利于收获细胞[27](图5b)。通过交联制备的壳聚糖微球则为实心球[28](图5c)。 "
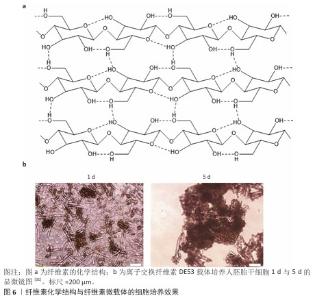
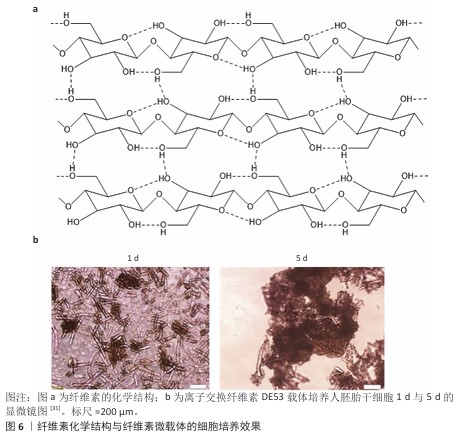
2.3.3 明胶 胶原由具有免疫原性的两端(N端与C端,N-terminal,C-terminal)和具有一定机械强度的三股肽螺旋中间段组成,三股肽螺旋由3个及以上的重复肽段构成,胶原变性后三股肽螺旋解螺旋形成明胶[29]。 由于暴露的肽序列更多,明胶相比胶原更有利于整合素的结合。从明胶的肽序列来看,不同物种、不同型的明胶肽序列不同,Ⅰ型和Ⅳ型胶原是整合素配体,商品化的微球载体多使用Ⅰ型胶原变性制得的明胶作为原料。 2.3.4 纤维素 纤维素是通过β-1,4葡萄糖苷键连接的一种线性高分子聚合物。大量的分子内与分子间氢键使纤维素形成超分子结构(图6a),因此纤维素难以在普通溶剂中溶解[30],这一性质使得纤维素微载体的制备成本较高。 离子交换纤维素DE52、DE53、QA52均为Whatman公司生产的柱层析填料,尚无公司售卖纤维素实心细胞培养微载体。CHEN等[31]以DE52、DE53、QA52作为载体培养人胚胎干细胞;REUVENY等[32]以这些载体培养BHK和MDCK细胞。CHEN等[31]的工作对比了多种电荷密度纤维素微载体的人胚胎干细胞培养效果,可估算纤维素微载体表面适宜细胞黏附和增殖的电荷密度约为1.09 meq/g,DE53载体的人胚胎干细胞培养效果见图6b。"

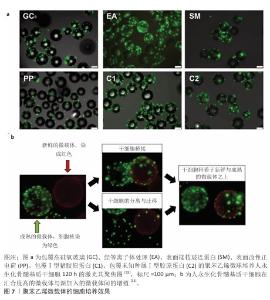
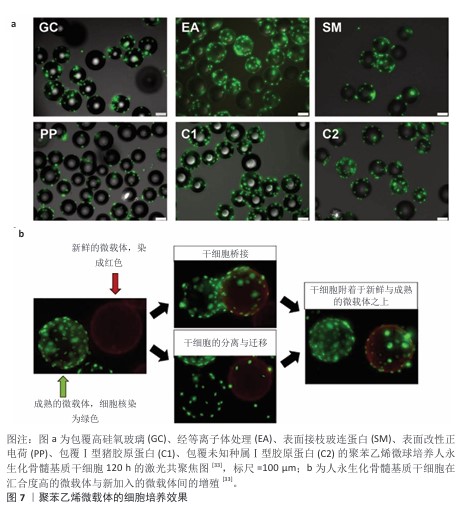
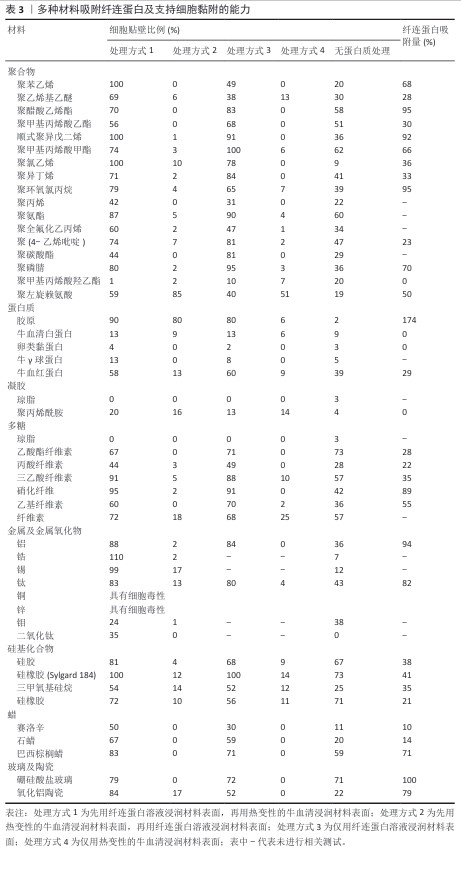
2.3.6 其他材料 除上述材料外,多种材料具有制备微培养载体的潜质。LEBER等[33]研究了多种材料吸附纤连蛋白及支持细胞黏附的能力,以细胞在皮氏培养皿中的黏附细胞数量为100%,结果如表3所示[9]。除胶原纤维外,所有材料在先用热变性的牛血清浸润表面,再用纤连蛋白溶液浸润表面后均丧失了黏附细胞的能力,由于整合素是介导细胞贴壁的关键膜蛋白,而纤连蛋白是整合素的重要配体,当先用热变形的牛血清浸润材料后,材料表面已经吸附了过量的变性蛋白,无法吸附纤连蛋白,因此细胞贴壁比例低;同时,这一现象也说明仅有胶原纤维对纤连蛋白有特异性吸附。 除材料能否支持细胞黏附与生长外,需特别注意应用场景对微载体制备材料的要求。材料密度以微载体在培养基中的沉降速度(cm/min)来标定更为合理。以下落速度标定可以较易推算出能使微载体充分接触培养基的搅拌速度或流量,细胞在微载体上能承受的搅拌速度或流量有上限,以Vero细胞为例,一般Vero细胞在Cytodex 1上培养所能承受的搅拌速度在60 r/min以内[34],如果微载体密度过大,则在60 r/min下搅拌不充分,增大搅拌速度会让Vero细胞承受过高的剪切力,可能因此发生细胞脱落。由于多数微载体需经高压蒸汽灭菌处理,因此微载体在高温条件下的形貌稳定性也需重点考虑。 "
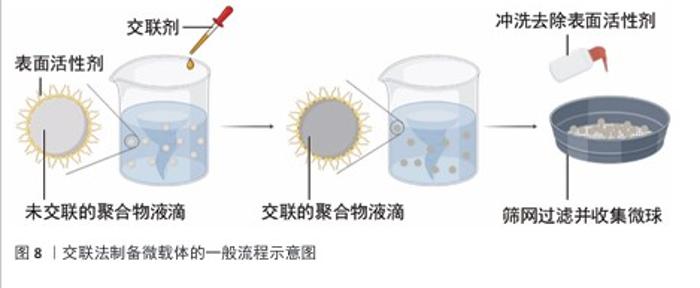
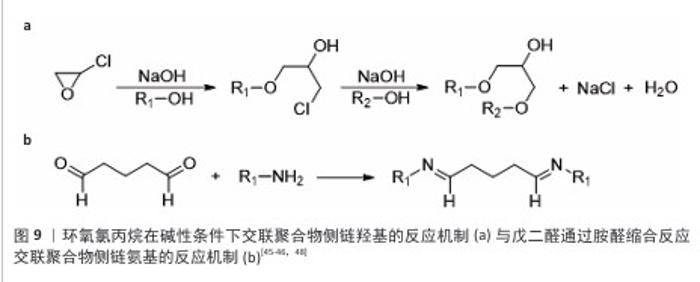
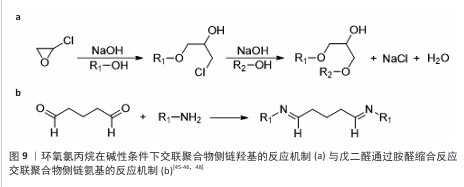
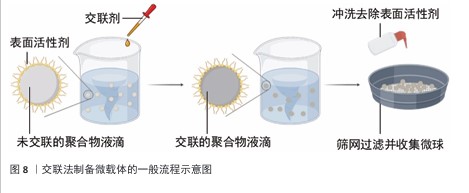
2.4 微载体的制备方法 2.4.1 交联法 交联聚合物液滴是制备微球载体的有效方法,交联法制备微载体的一般流程示意图见图8,该节按照如下顺序阐明交联聚合物液滴法:聚合物的溶解,散体系的构建,聚合物液滴的交联。 前述内容详述了壳聚糖溶于稀酸水溶液的原因,具体为1%-2%的乙酸水溶液[28]。此外,FANG等[35]报道了一种能够溶解不同脱乙酰度壳聚糖的碱性离子液体,溶解机制是:KOH与NaOH破坏C=O与活泼氢的氢键;LiOH破坏-NH2与O6之间的氢键,并将该溶解方法用于壳聚糖微球的制备[36-37]。由于肽序列的亲水侧基充分暴露,胶原能够溶于50 ℃的水中,较易获得分散相[38]。 相较于葡聚糖、壳聚糖及胶原,纤维素的溶解更加困难。纤维素具有3种溶剂体系[39-40],分别为非衍生化溶剂、衍生化溶剂以及纤维素衍生物-直接溶解体系。非衍生化溶剂通过溶剂与纤维素的物理相互作用破坏纤维素内部氢键,进而溶解纤维素。衍生化溶剂通过将纤维素转换成不稳定的纤维素衍生物,在溶解后通过加入水、改变pH值或温度而引起不稳定的衍生物裂解形成纤维素微球。纤维素衍生物-直接溶解体系为溶于有机溶剂的纤维素稳定衍生物,该溶解体系再经凝聚或蒸发将该衍生物转化为纤维素。 将聚合物水相滴入含有表面活性剂的连续相可制备具有一定尺寸的聚合物乳液。油水分散体系中的液滴尺寸可通过调控表面活性剂的含量及种类、油水比例、油相转速等方法调控。壳聚糖、葡聚糖、胶原乳液的表面活性剂多使用Span 80与Tween 60[31,36,41-42]。纤维素的溶剂体系种类多,因此分散体系构建复杂程度更大。由交联法制备的纤维球粒径为10-100 μm[35-36,43],由于细胞直径约为10 μm,细胞培养微载体的粒径应为100-400 μm,这一尺寸要求限制了纤维素乳液的液滴尺寸,即限制了分散体系的选用。CAO等[44]提供了一种粒径约为110 μm的纤维素透明微球制备方法,采用NaOH/尿素/H2O作为溶剂体系溶解纤维素,以液体石蜡作为连续相,以Span 80作为表面活性剂,以环氧氯丙烷作为交联剂。壳聚糖、葡聚糖、明胶、纤维素都具有特定的官能团侧基,因此选择适当的交联剂共价连接液滴中的分子链是可行的。 环氧氯丙烷可与侧链羟基在碱性条件下发生图9a中的反应[45-46]。葡聚糖可溶于碱性水溶液,壳聚糖可溶于碱性离子溶液,纤维素可溶于NaOH/尿素/H2O溶剂体系,因此环氧氯丙烷可用于交联葡聚糖和壳聚糖及纤维素。氨基需为壳聚糖微载体提供适宜细胞黏附的电荷条件,然而,氨基具有比羟基更强的亲核性,也可与环氧氯丙烷反应,因此,环氧氯丙烷交联壳聚糖液滴前需用甲醛保护侧氨基,在交联后再脱去保护暴露氨基[47]。明胶具有包括羟基在内的多种侧基,作者认为环氧氯丙烷会破坏整合素的结合位点,如RGD(精氨酰-甘氨酰-天冬氨酸)肽序列,因此环氧氯丙烷并不适合作为制备明胶微载体的交联剂。 戊二醛通过胺醛缩合反应交联聚合物侧链氨基[48](图9b),也可与侧链羟基反应形成缩醛结构[49]。然而,由于席夫碱反应消耗氨基、屏蔽电荷,戊二醛不适合用于壳聚糖微载体的制备。HUANG等[28]报道了一种戊二醛交联的壳聚糖微球,该微球需修饰胶原以创造适宜细胞黏附的表面条件。FARRIS等[49]研究了戊二醛交联胶原的反应机制,在高pH值下(即接近赖氨酸的pKa),交联反应主要由席夫碱形成控制,在低pH值下(即赖氨酸的氨基被质子化时),交联反应涉及羟基脯氨酸和羟基赖氨酸的羟基基团,导致缩醛结构的形成。 除化学交联外,离子交联也可制备微球载体,然而离子交联暂无商业化的产品,该文未归纳。 2.4.2 相反转法 相反转法的原理为:在构建材料的液滴分散体系后,改变分散相的理化性质使材料的溶解度改变,进而获得和分散相液滴尺寸近似的微球。 壳聚糖可通过升高pH值实现在水中的溶解度转变,它是相反转法制备微载体的理想材料[27]。纤维素具有衍生及非衍生溶剂体系,溶剂溶解纤维素的原理使纤维素成为相反转法制备微载体的理想材料(衍生及非衍生溶剂体系溶解纤维素的机制见2.4.1)。然而,由于相反转法制备的纤维素微球大于500 μm[50-51],对于细胞培养微载体而言尺寸较大,因此相反转法并不适用于纤维素微球载体的制备。葡聚糖及胶原由于在水中的溶解度高,采用相反转法制备微球载体的报道较少。 2.4.3 聚合法 聚合法是聚苯乙烯微载体的制备方法,聚合法生产聚苯乙烯微载体可用苯乙烯与另一种单体共聚,如Corning公司的 SynthemaxⅡ为非交联的苯乙烯丙烯酸酯共聚物;此外,商品化的聚苯乙烯微球载体分为交联和非交联的聚苯乙烯微载体。 ARIFIN等[52]报道了一种均聚苯乙烯细胞微球载体的制备方法,该方法以聚乙烯醇作为表面活性剂,以氯仿溶解苯乙烯作为油相构建分散体系,而后聚合并用筛网得到150-200 μm的非交联聚苯乙烯微球,再经紫外/臭氧处理使微球表面带羧基,最后通过羧基与胶原的氨基通过缩合反应将胶原包覆在非交联聚苯乙烯微球表面,制得非交联聚苯乙烯微载体。"
| [1] VAN WEZEL AL. Growth of Cell-strains and Primary Cells on Micro-carriers in Homogeneous Culture. Nature. 1967; 216(5110):64-65. [2] SILVA COUTO P, ROTONDI MC, BERSENEV A, et al. Expansion of human mesenchymal stem/stromal cells (hMSCs) in bioreactors using microcarriers: lessons learnt and what the future holds. Biotechnol Adv. 2020;45: 107636. [3] MERED B, ALBRECHT P, HOPPS HE. Cell growth optimization in microcarrier culture. In Vitro. 1980;16(10):859-865. [4] DERAKHTI S, SAFIABADI-TALI SH, AMOABEDINY G, et al. Attachment and detachment strategies in microcarrier-based cell culture technology: A comprehensive review. Mater Sci Eng C Mater Biol Appl. 2019;103:109782. [5] BAI QF, TANG H, CHEN Y, et al. Modular strategy with autologous bioreactor: a potential way for organ engineering. Int J Extrem Manuf. 2025; 7(2):22001. [6] PAIVA WA, ALAKWE SD, MARFAI J, et al. From Bioreactor to Bulk Rheology: Achieving Scalable Production of Highly Concentrated Circular DNA. Adv Mater. 2024; 36(35):e2405490. [7] KANCHANAWONG P, CALDERWOOD DA. Organization, dynamics and mechanoregulation of integrin-mediated cell–ECM adhesions. Nat Rev Mol Cell Biol. 2023;24(2):142-161. [8] YANG J, GUERTIN P, JIA G, et al. Large-scale microcarrier culture of HEK293T cells and Vero cells in single-use bioreactors. AMB Express. 2019;9(1):70. [9] KLEBE RJ, BENTLEY KL, SCHOEN RC. Adhesive substrates for fibronectin. J Cell Physiol. 1981; 109(3):481-488. [10] ISHIHARA K, MITERA K, INOUE Y, et al. Effects of molecular interactions at various polymer brush surfaces on fibronectin adsorption induced cell adhesion. Colloids Surf B Biointerfaces. 2020;194:111205. [11] HIRTENSTEIN M, CLARK J, LINDGREN G, et al. Microcarriers for Animal Cell Culture: A Brief Review of Theory and Practice. Dev Biol Stand. 1980;46:109-116. [12] MAROUDAS NG. Adhesion and spreading of cells on charged surfaces. J Theor Biol. 1975; 49(2):417-424. [13] REUVENY S, MIZRAHI A, KOTLER M, et al. Factors affecting cell attachment, spreading, and growth on derivatized microcarriers. I. Establishment of working system and effect of the type of the amino-charged groups. Biotechnol Bioeng. 1983;25(2):469-480. [14] GARCIA AJ, DUCHEYNE P, BOETTIGER D. Cell Adhesion Strength Increases Linearly with Adsorbed Fibronectin Surface Density. Tissue Eng. 1997;3(2):197-206. [15] SART S, AGATHOS SN, LI Y. Engineering Stem Cell Fate with Biochemical and Biomechanical Properties of Microcarriers. Biotechnol Prog. 2013;29(6):1354-1366. [16] TAVASSOLI H, ALHOSSEINI SN, TAY A, et al. Large-scale production of stem cells utilizing microcarriers: A biomaterials engineering perspective from academic research to commercialized products. Biomaterials. 2018;181:333-346. [17] RAFIQ QA, COOPMAN K, NIENOW AW, et al. Systematic microcarrier screening and agitated culture conditions improves human mesenchymal stem cell yield in bioreactors. Biotechnol J. 2016;11(4): 473-486. [18] JOSSEN V, VAN DEN BOS C, EIBL R, et al. Manufacturing human mesenchymal stem cells at clinical scale: process and regulatory challenges. Appl Microbiol Biotechnol. 2018; 102(9):3981-3994. [19] NIE Y, BERGENDAHL V, HEI DJ, et al. Scalable culture and cryopreservation of human embryonic stem cells on microcarriers. Biotechnol Prog. 2009;25(1):20-31. [20] HAYMAN EG, PIERSCHBACHER MD, SUZUKI S, et al. Vitronectin--a major cell attachment-promoting protein in fetal bovine serum. Exp Cell Res. 1985;160(2):245-258. [21] LI T, HAO L, LI J, et al. Insight into vitronectin structural evolution on material surface chemistries: The mediation for cell adhesion. Bioact Mater. 2020;5(4):1044-1052. [22] LIDDINGTON RC. Structural aspects of integrins. Adv Exp Med Biol. 2014;819: 111-126. [23] DÍAZ-MONTES E. Dextran: Sources, Structures, and Properties. Polysaccharides. 2021;2(3): 554-565. [24] CÔTÉ GL, LEATHERS TD. Insoluble glucans from planktonic and biofilm cultures of mutants of Leuconostoc mesenteroides NRRL B-1355. Appl Microbiol Biotechnol. 2009;82(1): 149-154. [25] LIZARDI-MENDOZA J, ARGÜELLES MONAL WM, GOYCOOLEA VALENCIA FM. Chapter 1 - Chemical Characteristics and Functional Properties of Chitosan//BAUTISTA-BAÑOS S, ROMANAZZI G, JIMÉNEZ-APARICIO A. Chitosan in the Preservation of Agricultural Commodities. San Diego; Academic Press. 2016:3-31. [26] DING L, HUANG Y, CAI X, et al. Impact of pH, ionic strength and chitosan charge density on chitosan/casein complexation and phase behavior. Carbohydr Polym. 2019;208:133-141. [27] HUANG L, XIAO L, POUDEL AJ, et al. Porous Chitosan Microspheres as Microcarriers for 3D Cell Culture. Carbohydr Polym. 2018;202: 611-620. [28] HUANG L, JIANG Y, CHEN X, et al. Supramolecular Responsive Chitosan Microcarriers for Cell Detachment Triggered by Adamantane. Polymers (Basel). 2023; 15(19):4024. [29] GELSE K, PÖSCHL E, AIGNER T. Collagens--structure, function, and biosynthesis. Adv Drug Deliv Rev. 2003;55(12):1531-1546. [30] ACHARYA S, LIYANAGE S, PARAJULI P, et al. Utilization of Cellulose to Its Full Potential: A Review on Cellulose Dissolution, Regeneration, and Applications. Polymers (Basel). 2021; 13(24):4344. [31] CHEN AK, CHEN X, CHOO AB, et al. Critical microcarrier properties affecting the expansion of undifferentiated human embryonic stem cells. Stem Cell Res. 2011;7(2):97-111. [32] REUVENY S, SILBERSTEIN L, SHAHAR A, et al. DE-52 and DE-53 cellulose microcarriers. I. Growth of primary and established anchorage-dependent cells. In Vitro. 1982;18(2):92-98. [33] LEBER J, BAREKZAI J, BLUMENSTOCK M, et al. Microcarrier choice and bead-to-bead transfer for human mesenchymal stem cells in serum-containing and chemically defined media. Process Biochemi. 2017;59(pt.b):255-265. [34] MENDONÇA RZ, PEREIRA CA. High density VERO cell culture on microcarriers in a cell bioreactor. Bioprocess Biosyst Eng. 1995; 12(6):279-282. [35] FANG Y, ZHANG R, DUAN B, et al. Recyclable Universal Solvents for Chitin to Chitosan with Various Degrees of Acetylation and Construction of Robust Hydrogels. ACS Sustainable Chem Eng. 2017;5(3):2725-2733. [36] LIANG X, DUAN J, XU Q, et al. Ampholytic microspheres constructed from chitosan and carrageenan in alkali/urea aqueous solution for purification of various wastewater. Chem Eng J. 2017;317:766-776. [37] LIANG X, GAO M, XIE H, et al. Controllable Wrinkling Patterns on Chitosan Microspheres Generated from Self-Assembling Metal Nanoparticles. ACS Appl Mater Interfaces. 2019;11(25):22824-22833. [38] LATORRE ME, LIFSCHITZ AL, PURSLOW PP. New recommendations for measuring collagen solubility. Meat Sci. 2016;118:78-81. [39] MULYADI A, ZHANG Z, DENG Y. Fluorine-Free Oil Absorbents Made from Cellulose Nanofibril Aerogels. ACS Appl Mater Interfaces. 2016;8(4):2732-2740. [40] GERICKE M, TRYGG J, FARDIM P. Functional cellulose beads: preparation, characterization, and applications. Chem Rev. 2013;113(7): 4812-4836. [41] VOLODKIN DV, LARIONOVA NI, SUKHORUKOV GB. Protein encapsulation via porous CaCO3 microparticles templating. Biomacromolecules. 2004;5(5):1962-1972. [42] RONG D, JIMIN C, CHONGSUN Y, et al. Zedoary turmeric oil gelatin microspheres for hepatical arterial embolization. Acta Pharm Sin B. 2000; 35:539-543. [43] KOTOULAS C, KIPARISSIDES C. A generalized population balance model for the prediction of particle size distribution in suspension polymerization reactors. Chem Eng Sci. 2006; 61(2):332-346. [44] CAO X, ZHU B, CHEN T, et al. Recombinant protein A immobilized on cross-linked cellulose microspheres for immunoglobulin G adsorption from human plasma. BioResources. 2016;11(2):4885-4898. [45] XIA HF, LIN DQ, WANG LP, et al. Preparation and Evaluation of Cellulose Adsorbents for Hydrophobic Charge Induction Chromatography. Ind Eng Chem Res. 2008; 47(23):9566-9572. [46] BURTON SC, HARDING DRK. Bifunctional etherification of a bead cellulose for ligand attachment with allyl bromide and allyl glycidyl ether. J Chromatogr A. 1997; 775(1):29-38. [47] KANG ML, KANG SG, JIANG HL, et al. In vivo induction of mucosal immune responses by intranasal administration of chitosan microspheres containing Bordetella bronchiseptica DNT. Eur J Pharm Biopharm. 2006;63(2):215-220. [48] KIM SY, CHO SM, LEE YM, et al. Thermo- and pH-responsive behaviors of graft copolymer and blend based on chitosan and N-isopropylacrylamide. J App Polym Sci. 2015;78(7):1381-1391. [49] FARRIS S, SONG J, HUANG Q. Alternative reaction mechanism for the cross-linking of gelatin with glutaraldehyde. J Agric Food Chem. 2010;58(2):998-1003. [50] TRYGG J, FARDIM P, GERICKE M, et al. Physicochemical design of the morphology and ultrastructure of cellulose beads. Carbohydr Polym. 2013;93(1):291-299. [51] SESCOUSSE R, GAVILLON R, BUDTOVA T. Wet and dry highly porous cellulose beads from cellulose–NaOH–water solutions: influence of the preparation conditions on beads shape and encapsulation of inorganic particles. J Mater Sci. 2011;46(3):759-765. [52] ARIFIN MA, MEL M, SWAN SY, et al. Optimization of ultraviolet/ozone (UVO(3)) process conditions for the preparation of gelatin coated polystyrene (PS) microcarriers. Prep Biochem Biotechnol. 2022;52(2): 181-196. |
| [1] | Yang Lixia, Diao Liqin, Li Hua, Feng Yachan, Liu Xin, Yu Yuexin, Dou Xixi, Gu Huifeng, Xu Lanju. Regulatory mechanism of recombinant type III humanized collagen protein improving photoaging skin in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1988-2000. |
| [2] | Zhang Yibo, Li Jian, Wang Peng, Jiang Qing. Curcumin-loaded chitosan/beta-glycerophosphate sodium thermoresponsive hydrogel promotes tendon healing in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6779-6798. |
| [3] | Song Muze, Liu Chuyi, , Tang Qingjuan, Dai Yuankun, , Song Wenshan, Li Bafang, Wang Yuanyuan. Biocompatibility evaluation of polylactic acid/collagen electrospinning bilayer guided tissue regeneration membrane [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6880-6891. |
| [4] | Zhang Kaijing, Li Chunnian, Li Yizhuo, Xu Shifang, Liu Xinyue. A network meta-analysis of therapeutic effects of different bone repair materials on apical bone defects [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6946-6951. |
| [5] | Chen Ying, Sun Xuheng, Liu Qing, Xiao Cong, Jiang Hongjing, Lin Zhanyi. A serum-free culture medium for the early-stage formation of tissue-engineered vascular grafts [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5093-5102. |
| [6] | Tan Fengyi, Xie Jiamin, Pan Zhenfeng, Zhang Xinxu, Zheng Zetai, Zeng Zhiying, Zhou Yanfang. Effect and mechanism of collagen combined with microneedles in treatment of skin photoaging [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(2): 451-458. |
| [7] | Fang Yulu, Yi Bingcheng, Shen Yanbing, Tang Han, Zhang Yanzhong. Potential of corn husk fibers reinforced chitosan-based hydrogels in cartilage tissue engineering scaffold [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5493-5501. |
| [8] | Zhang Jian, Chen Miao, Li Weixin, Ye Yichao, Xu Huiyou, Ma Ke, Chen Xuyi, Sun Hongtao, Zhang Sai. Collagen/heparin sulfate scaffolds loaded with brain-derived neurotrophic factor promote neurological and locomotor function recovery in rats after traumatic brain injury [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5538-5544. |
| [9] | Liao Jian, Huang Xiaolin, Zhou Qian, Huo Hua, Qi Yuhan, Wu Chao, Shi Qianhui, Yang Tongjing, Liao Yunmao, Liang Xing. Calcined bone/chitosan composite promotes osteogenic differentiation of bone marrow mesenchymal stem cells in Sprague-Dawley rats [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 4941-4947. |
| [10] | Liu Xiaoyin, , Zhong Lin, Zheng Bo, Wei Pan, Dai Chen, Hu Liangcong, Wang Tiantian, Liang Xiaolong, Zhang Sai, Wang Xiaoli. Diffusion tensor imaging predicting locomotor function recovery with 3D printing scaffold after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(28): 4547-4554. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||