Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (19): 5040-5049.doi: 10.12307/2026.407
Previous Articles Next Articles
Innovative application of kidney organoids in acute kidney injury
Huang Zhengbo1, Ou Min2, Li Guoshun2, Duan Fuhui2, Liu Jianqi2, Lou Juxiang2, Zhao Yanxiu2, Su Xiaoyan2
- 1Binzhou Medical University, Yantai 264003, Shandong Province, China; 2Fifth Affiliated Hospital of Dali University (Baoshan People's Hospital), Baoshan 678000, Yunnan Province, China
-
Received:2025-08-25Accepted:2025-12-03Online:2026-07-08Published:2026-02-24 -
Contact:Zhao Yanxiu, Chief physician, Fifth Affiliated Hospital of Dali University (Baoshan People's Hospital), Baoshan 678000, Yunnan Province, China; Co-corresponding author: Su Xiaoyan, Associate chief physician, Fifth Affiliated Hospital of Dali University (Baoshan People's Hospital), Baoshan 678000, Yunnan Province, China -
About author:Huang Zhengbo, Binzhou Medical University, Yantai 264003, Shandong Province, China; Ou Min, MS, Pharmacist, Fifth Affiliated Hospital of Dali University (Baoshan People's Hospital), Baoshan 678000, Yunnan Province, China -
Supported by:Medical Research Joint Special Key Project of Baoshan City, No. 2023bskjylzd002 (to ZYX)
CLC Number:
Cite this article
Huang Zhengbo, Ou Min, Li Guoshun, Duan Fuhui, Liu Jianqi, Lou Juxiang, Zhao Yanxiu, Su Xiaoyan. Innovative application of kidney organoids in acute kidney injury[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 5040-5049.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
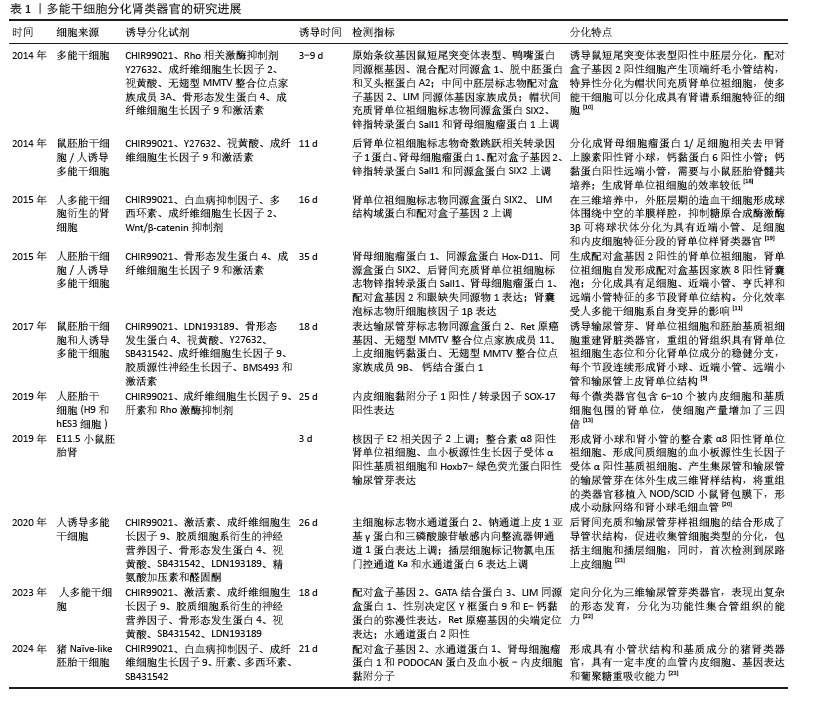
2.1 肾类器官构建的创新性技术 2.1.1 干细胞分化策略 (1)多能干细胞分化策略:人肾类器官不仅用于药代动力学和毒性机制研究,还应用于肾脏疾病研究的体外模型构建。源自人多能干细胞的肾脏类器官为开发临床前研究的肾脏模型和免疫相容性肾脏组织提供了新的机会[8]。自第1代肾类器官成功构建以来,已经开发了许多改进方案来诱导多能干细胞形成肾类器官,这些方案依赖于“定向分化”过程,以特定的生长因子和中间递质引导干细胞形成复杂的肾样结构。肾类器官能够表达来自不同肾细胞阶段的标记物,包括远端小管、近端小管、肾小球和足细胞[9]。多能干细胞可定向分化为不同的细胞,使其能够生成包括肾脏在内的各种人体组织类器官模型,多能干细胞包括人诱导多能干细胞和人胚胎干细胞。有学者根据以往对人胚胎干细胞的培养分化经验[10],提出了成纤维细胞生长因子2、激活素 A、视黄酸以及含或不含CHIR99021小分子诱导剂的两种分化方案,CHIR99021小分子诱导剂可以诱导干细胞分化成输尿管芽等肾祖细胞群体,形成含近端/远端肾小管、足细胞和血管内皮细胞的类肾结构。MORIZANE等[11]提出了一种培养肾脏类器官的优越方案,标志着该领域的重大进步,该方案联合使用CHIR99021与骨形态发生蛋白4拮抗剂Noggin,通过精准分化,将悬浮培养的肾单位祖细胞发育成三维肾类器官,形成肾单位样结构。TAKASATO等[12]用诱导多能干细胞分化而成的肾脏器官组织建立了肾小管样结构,主要由连接到肾小管结构的荚膜细胞组成。有研究通过悬浮培养方法产生大量肾脏微类器官,此方法具有简单、经济和高效的优势,具体表现在诱导多能干细胞和人胚胎干细胞分化形成的肾脏微类器官结构中包含的肾小管细胞计数增加了30-40倍,这种培养形式可将每百万个细胞的成本降低75%。与标准类器官数据集相比,微类器官数据集包含更高比例的肾单位细胞和更低比例的基质细胞[13]。 多能干细胞诱导的肾类器官在急性肾损伤和慢性肾损伤发生发展研究中发挥了重要作用。严重和/或反复肾损伤可导致肾小管萎缩和间质纤维化,阻碍了肾小管细胞的自我修复[14]。低重复剂量顺铂给药导致的肾类器官累积损伤表明,顺铂诱导可模拟急性肾损伤到慢性肾损伤的转变过程[15]。范可尼贫血D2组蛋白的表达缺失与肾纤维化显著相关,使用脱氧核糖核酸连接酶Ⅳ抑制剂SCR7治疗暴露于重复顺铂剂量的肾类器官可挽救由范可尼贫血D2组蛋白缺失引发的肾小管萎缩和肌成纤维细胞活化,促进近端小管从内源性修复到不完全修复的有限转变,表明靶向范可尼贫血D2组蛋白/DNA修复酶RAD51同源物通路可以阻止急性肾损伤到慢性肾损伤转变的疾病进展[16]。用白细胞介素1β、转化生长因子β或氯化钴激活人胚胎干细胞诱导的肾类器官细胞中,孤儿核受体COUP-TFII增加可以增强基质纤维化[17]。以上研究表明,类器官方案已经得到了很好的优化(表1)[5,10-11,13,18-23]。 "

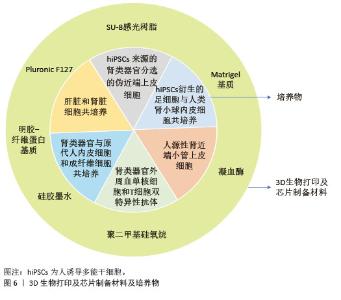
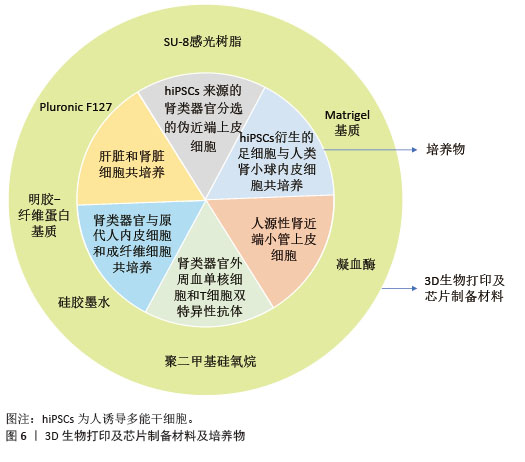
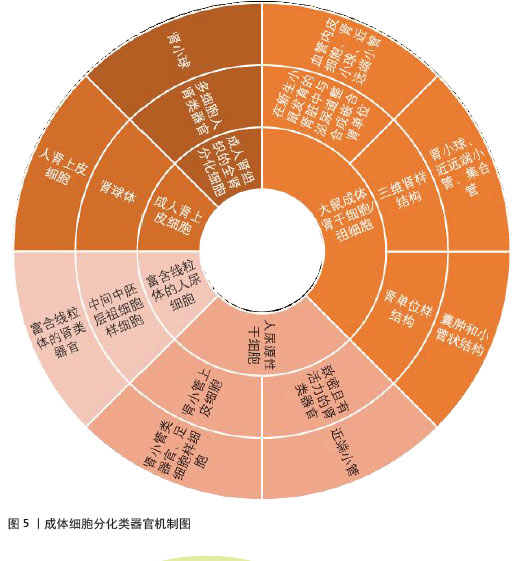
(2)成体干细胞分化策略:肾类器官也可以由成体干细胞建立,这种类型的类器官被称为“肾小管类器官”,仅由上皮细胞组成[24],模拟构成成熟肾单位的不同上皮亚群的结构和功能(图5)。成体干细胞衍生的类器官由于不需要像干细胞一样要很长的分化时间和额外的分化诱导因子从而使类器官的生成过程更加简化。来自成人肾组织的全肾分化细胞诱导的3D肾脏类器官形态接近正常肾小球的形态,显著表达肾小球足细胞标志物足突蛋白、突触极蛋白和足细胞相关去甲肾上腺素以及肾小管上皮水通道蛋白1和集合管水通道蛋白3,用阿司匹林、青霉素G或顺铂处理类器官可形成急性近端肾小管损伤[25]。从肾活检、肾切除获得的肾组织可用于生成特异性类器官、高通量药物肾毒性测试、药物筛选或其他后续应用。例如,感染SARS-CoV-2病毒的成体肾球体以及原代人肾单层培养物对肾细胞表现出特异性营养作用且感染的小管细胞中细胞损伤标志物的表达增加[26]。从成年大鼠肾近端小管S3段获得的大鼠肾干/祖细胞可以体外培养获得具有三维肾单位样结构的类器官[27]。肾单位样结构包含肾脏的各种亚结构,包括肾小球、近端小管、Henle袢和远端小管。肾单位样结构暴露于顺铂中可导致细胞死亡,并且在第14-20天类器官表面积的增加速率减慢,体外模拟的顺铂诱导的细胞死亡与体内急性肾损伤相似[28]。hNPSR/3D培养(支持Six2-GFP阳性肾单位祖细胞的高纯度长期体外扩增,包含骨调素1、成纤维细胞生长因子2、肝素、Y27632、白血病抑制因子和CHIR99021)使肾单位祖细胞系具有体外和体内肾形成潜力。将hNPSR培养的人肾单位祖细胞与脊髓一起移植到小鼠网膜时,它们能够在体内分化为肾单位的主要部分,将mCherry标记的小鼠肾单位祖细胞聚集体移植到HH18期鸡胚的侧板中胚层,在雏鸡胚胎中发现了完整的mCherry阳性管状结构,表明肾单位祖细胞系和小鼠肾单位祖细胞长期培养可具有肾形成潜力,且不同发育阶段的肾单位祖细胞的肾源性仍然是完整的[29]。将小鼠和大鼠的肾单位祖细胞注射到新生小鼠发育中的肾脏,可产生与宿主泌尿道整合的嵌合肾单位。单细胞RNA测序证实,这些嵌合肾单位表现出与宿主肾单位相似的排泄和重吸收功能以及顺铂诱导的急性和慢性期肾损伤[30]。在特定培养基中人尿细胞重编程的可扩展中间中胚层祖细胞产生富含线粒体的肾脏类器官,发现这些类器官可在雏鸡绒毛膜尿囊膜中进一步成熟,并且可由顺铂、庆大霉素和毛喉素造成典型的肾损伤,发生解剖结构异常和线粒体稳态改变[31]。 尿液干细胞起源于肾小球壁层干细胞群,可生成致密且有活力的类器官,在形态上与肾类器官非常接近。作为肾组织的替代品,尿液干细胞具有采集过程无痛、高度可扩增性、自我更新能力、肾脏起源以及多向分化潜能等特点,能够生成肾小管,在急性肾损伤患者和小鼠模型中表现出归巢和再生的作用。尿液干细胞可降低受损类器官中肾损伤分子1表达,提高顺铂诱导的人诱导多能干细胞分化肾类器官内小管细胞的存活率,减少细胞毒性[32]。肾内分泌产物促红细胞生成素、肾小球标志物肾小球足细胞裂隙膜蛋白和突触极蛋白在肾特异性细胞外基质的类器官中均呈阳性。肾毒性实验显示阿司匹林、青霉素G和顺铂对含有细胞外基质的尿液干细胞诱导的类器官产生药物毒性[33]。尿液干细胞被肾特异性细胞外基质诱导后分化为肾小管上皮细胞。3D类器官中细胞色素P450家族成员2E1和肾损伤分子1水平在丙酮和顺铂刺激下显著增加[34]。 "

3D生物打印是通过打印连续的薄层材料将数字图像“转换”为三维实体的技术。它利用计算机辅助将沉积细胞与支持性生物材料一起进行精心设计,这种方法可以精确和特异性地构建复杂的多细胞结构[36]。2016年研究人员报道了一种生物打印方法[37],用于在体外创建3D人肾近端小管,这些小管完全嵌入细胞外基质中,安置在可灌注的组织芯片中维持2个多月,其卷曲的管状结构被近端小管上皮细胞包围,并通过开放的管腔积极灌注,这些芯片上的工程3D近端小管与有或没有灌注的2D对照生长细胞相比,表现出显著增强的上皮形态和功能特性,为按需编程制造高级人体肾脏组织模型提供了新的途径。 应用微流体技术设计的“芯片上的器官”具有许多优点,包括延长寿命、生物相容性、更高的气体渗透性和敏感性以及低成本。通过微流体培养系统,探讨细胞外基质、培养基组成、流体剪切应力以及与人内皮细胞共培养对肾类器官体外发育的影响。最初芯片肾脏的研究使用一种以活体人肾脏上皮细胞的微流体装置模拟人肾近端小管,它比传统Transwell培养系统能更好地模拟体内对顺铂毒性和磷酸甘油酸磷酸酶外排转运活性的反应系统,可将上皮单层暴露在模拟活体肾小管的流体剪切应力下,使上皮细胞极化和初级纤毛形成增强[38]。MUSAH等[39]将人诱导多能干细胞衍生的足细胞与一层人肾小球内皮细胞共培养在器官芯片微流体装置中,人诱导多能干细胞衍生的足细胞会产生肾小球基底膜胶原蛋白,并模拟肾小球毛细血管壁的正常组织-组织界面和分子过滤特性,形成具有成熟足细胞功能的人肾小球体外模型。发育中的肾脏类器官在血流下表现出增强的可灌注血管,促进成体转录因子碱核蛋白2、神经元PAS结构域蛋白2、鼻咽毛囊综合征蛋白Ⅰ的表达,体外肾小球血管形成和成熟反映了体内发育的早期阶段,与在外源干扰梯度中培养的类器官观察到的随机散发性生长不同,在未受干扰的培养基条件下观察到连接密度降低,平均血管长度增加,表明血管朝着内源血管内皮生长因子受体梯度生长,使血管及时到达肾小球样腔室,而不是包裹这些体外肾类器官内的鲍曼囊样结构[40]。一项使用芯片培养人源性肾近端小管上皮细胞的研究表明,与二维培养相比,芯片培养的细胞改善了极化紧密连接、顶端刷状缘纤毛的形成以及基底膜上八聚体结合转录因子2转运体的表达[41]。 芯片肾脏可在肾毒性药物应用中发挥重要作用。它能够将肾毒性药物应用于上皮顶端或基底外侧表面。在芯片上培养表达有机阴离子转运蛋白1转运体的永生化近端小管上皮细胞,可实现肾脏药物的高通量筛选和与高含量检测平台的兼容性[42]。有学者开发了一种基于人诱导多能干细胞衍生肾脏类器官的近端芯片肾小管模型,该模型可用于评估矢量跨细胞转运和肾毒性,与肾近端肾小管上皮细胞相比,近端芯片肾小管增强了关键SLC蛋白(如有机阴离子转运蛋白1/3、八聚体结合转录因子2和单向外排转运蛋白)的表达和活性,在应用阿德福韦、瑞舒伐他汀、二甲双胍、AA1和顺铂等化合物处理时,模拟药物生理转运和肾毒性方面的熟练程度尤其明显,突出了该芯片在药物筛选和毒性评估方面的潜力[43]。微流体模型的另一重要应用是它能够同时培养不同的细胞类型,用于评估不同细胞和类器官之间的相互作用。例如,有研究将人诱导多能干细胞分化的肾类器官移植到贴壁的细胞外基质上,引入循环外周血单核细胞和T细胞双特异性抗体共同构建免疫浸润肾类器官芯片模型[44]。研究表明,与单层培养系统相比,三维培养的肾脏近端管状上皮细胞显示出药物转运蛋白的表达增强和更高的预测性能[45],使用器官芯片与肝脏和肾脏共培养物还可以检测药物代谢物诱导的肾毒性[46]。 2.1.3 基因编辑在人肾类器官中的应用 过去10年引入了CRISPR/Cas基因编辑系统,这是对人类基因组理解和控制的一个重大飞跃[47]。CRISPR-Cas9复合物改编自细菌中的基因组编辑系统,作为对抗感染病毒的免疫系统,通过生成小RNA片段(向导RNA)来编辑人类基因组,向导RNA识别特定的脱氧核糖核酸序列之后,Cas9内切酶在目标位置执行双链断裂,重新修复细胞原生脱氧核糖核酸,由此可通过基因编辑改变不同的脱氧核糖核酸序列(基因敲入、基因敲除、用定制的脱氧核糖核酸序列取代现有序列或产生基因融合)[48],为准确促进靶基因组位点的修饰、纠正突变、调节转录和促进表观基因组的变化提供了可能性[49]。目前,CRISPR/Cas基因组编辑已成功用于研究人诱导多能干细胞衍生类器官和组织体细胞3D类器官中的基因功能、器官发育和构建人类疾病模型48]。CRISPR技术在急性肾损伤中用于生成肾脏谱系特异性荧光报告多能诱导干细胞系,在分化为肾脏类器官后能够放大和分类特定的肾脏细胞类型[50],促进包括细胞隔离、延时成像、方案优化和谱系追踪方法的一系列应用[51]。CRISPR/Cas9介导的肾脏谱系荧光标记,可以在活细胞中监测肾单位祖细胞、足细胞的分化和成熟标记。悬浮培养的类器官在具有微绒毛、紧密连接和足细胞的肾皮质结构中表达肾脏生物学和脉管系统的关键标志物[52]。这些技术有助于追踪特定细胞类型的不同损伤机制,并允许分选不同的细胞群,以便在单细胞水平上进行高分辨率基因组、蛋白质组、表观基因组和代谢组学表征。 HMOX1是已知的氧化应激标志物。最近,研究人员使用CRISPR技术将荧光报告基因(mCherry)插入HMOX1 基因座,生成了多能诱导干细胞HMOX1报告基因系,将其分化为肾脏类器官,并使用该基因系筛选盲编码药物化合物中推定的肾毒性物质[53]。缺乏HNF4A的类器官显示转运相关基因、内吞相关基因和刷状缘相关基因表达下降,类器官近端小管根尖管腔内刷状缘结构紊乱。通过CRISPR介导的转录激活诱导HNF4A或HNF4G表达,在肾类器官分化过程中驱动选定靶基因的表达增加[54]。COVID-19住院患者中,急性肾损伤与生存率低显著相关,急性肾损伤是COVID-19的主要并发症[55]。SARS-CoV-2可感染多能诱导干细胞来源肾类器官中的近端肾小管细胞、足细胞和基质细胞,刺激Ⅰ型胶原沉积,促进类器官纤维化[56]。增强近端小管细胞成熟度的新型肾类器官单细胞RNA测序显示广泛的病毒进入因子表达,血管紧张素转换酶2敲除的多能诱导干细胞生成类器官病毒滴度显著降低,表明SARS-CoV-2进入类器官依赖于血管紧张素转换酶2的表达[57]。过表达AP001007可保护人胚胎干细胞系H1分化的肾脏类器官免受脂多糖损伤[58]。在多能诱导干细胞细胞中过表达促红细胞生成素诱导分化肾类器官,将其皮下植入免疫缺陷小鼠1个月后,促红细胞生成素mRNA水平升高,并显著增加内皮细胞数量,血细胞比容显著升高,表明经促红细胞生成素支架/基质附着区域脱氧核糖核酸载体修饰的肾脏类器官可以长期稳定地递送促红细胞生成素[59]。 2.2 类器官在急性肾损伤研究中的创新应用 2.2.1 药物毒性模拟与机制解析 由于许多在动物中认为安全有效的治疗方法在临床试验中具有肾毒性或治疗效果不显著,所以将动物研究转化为临床应用是长久以来研究人员面临的一大难题。因此,在体外发展以人细胞为主要的研究工具可能是药物开发现有方法的重要补充。与体内的细胞相比,在传统的二维细胞培养中生长的近端小管细胞往往缺乏或迅速失去关键的表型和功能,如细胞极性、顶端刷状缘、基底膜蛋白沉积和重要受体介导的转运。用50,100 μmol/L环孢菌素A处理后,在2D塑料培养皿上生长的近端肾小管上皮细胞的细胞活力降低了40%和60%[37],这些因素阻碍了对体内肾毒性的准确纵向预测。3D基质胶环境中生长的近端小管细胞的管状网络形成高度分化的小管,这些小管对已知的肾毒素反应更敏感[60]。溶质转运蛋白家族22、有机阴离子转运蛋白1/3、八聚体结合转录因子2在类器官近端小管中表达增加,2D培养细胞在缺乏细胞外基质和液体流动诱导的情况下,转运蛋白表达有限,而这种药物转运蛋白在实验模型中的表达对于药物肾毒性相关研究是必不可少的[61]。因此,迫切需要患者特异性的体外模型,以模拟人肾脏组织结构。目前诱导肾类器官肾毒性的常用药物是顺铂,它在抗肿瘤治疗的同时可诱导急性肾损伤的发生。源自肾组织干细胞的肾脏类器官可作为急性肾小管损伤模型肾毒性组织病理学评估的有用工具,肾干细胞是来自肾近端小管S3段的成年大鼠肾干/祖细胞样细胞系,表现出很强的自我更新能力、延长增殖和再生潜力[62]。GUPTA等[16]利用多能诱导干细胞衍生的肾类器官,重点研究顺铂诱导的肾损伤从内在细胞修复向不完全细胞修复过渡的病理机制和特征,利用肾类器官系统,他们证明单次暴露于顺铂导致完全修复,这与体内模型一致,涉及近端小管去分化和进入细胞周期。PD-1作为抗肿瘤药物使用临床剂量即可诱导肾类器官产生炎症反应,造成肾损伤,但损伤程度和顺铂诱导的急性肾损伤相比程度较轻,细胞存活率较高,同时,与免疫细胞外周血单核细胞共培养后可加重上述反应[63]。 除了常用的顺铂诱导的急性肾损伤,庆大霉素、阿霉素及嘌呤霉素氨基核苷等药物同样具有致急性肾损伤发生的作用。它们的主要作用机制是以炎症的级联反应诱发肾类器官脱氧核糖核酸损伤及线粒体功能障碍,导致急性肾损伤的发生。有研究从成年大鼠肾干细胞中提取的肾类器官模型来评估含有红曲成分保健品的潜在肾小管毒性[64]。使用红曲CholesteHelp营养补充剂可诱导器官组织出现明显的肾小管损伤,表现为上皮细胞变薄、结构破坏以及凋亡的裂解半胱天冬氨酸蛋白酶3阳性肾小管细胞增多,这与使用顺铂处理的器官组织相似。分化21 d后,用庆大霉素处理48 h的人类多能干细胞构建的肾脏类器官显示肾小管中肾损伤分子1的剂量依赖性上调,表明近端肾小管细胞受损[11]。在暴露于2.5或5 μg/mL阿霉素24 h后,可诱导人类多能干细胞分化的肾微类器官急性肾损伤[13]。这些发现强调了肾脏类器官作为评估药物和其他化学物质肾毒性强大作用的有效性,为肾脏病理学和潜在的治疗干预提供了重要见解。2型志贺毒素对3D人近端肾小管上皮球状体或微型肾球状体造成严重损伤,促进炎性因子的表达,与O-GlcNAc转移酶抑制剂OSMI-1共同处理的条件下,3D结构得到了很好的维护。O-GlcNAcylation是一种翻译后修饰,在志贺毒素诱导宿主细胞内质网应激后急剧增加。2型志贺毒素处理的多能诱导干细胞衍生肾类器官显示出明显不同的形态,没有明显的肾单位样分割,急性肾功能衰竭指标肾损伤分子1显著增加。O-GlcNAcylation减少可抑制3D人小肾球状体和多能诱导干细胞衍生肾类器官中由2型志贺毒素介导的细胞毒性和促炎反应[65]。 商陆皂苷甲诱导小鼠急性肾损伤并伴有病理改变和细胞凋亡增强,损伤多能诱导干细胞诱导的肾类器官足细胞和近端小管内皮细胞的方式与体内相似,类器官中肾损伤生物标志物和炎症细胞因子肾损伤分子1、β2-微球蛋白和胱抑素C水平增加。建议使用肾类器官作为评估中药肾毒性的可靠平台[66]。商陆皂苷甲诱导的肾毒性人肾类器官模型可诱导内皮炎症,导致线粒体损伤并通过将线粒体脱氧核糖核酸转移到细胞质中激活干扰素基因刺激因子,从而形成炎症级联反应并破坏具有间质变化的肾内皮细胞[67]。嘌呤霉素氨基核苷是一种肾毒素,可用于诱导急性肾小球损伤和肾小球疾病模型。在整个分化过程中,人诱导多能干细胞诱导的肾脏类器官维持高增殖率,在用嘌呤霉素氨基核苷处理的类器官切片中Ki67阳性细胞显著减少。嘌呤霉素氨基核苷还可诱导肾脏类器官的脱氧核糖核酸损伤,转录组分析揭示了其诱导炎症和急性免疫反应,激活肾素-醛固酮-血管紧张素系统。肾素-醛固酮-血管紧张素系统在维持血压和体液稳态方面起关键作用,对肾脏正常发育至关重要,表明嘌呤霉素氨基核苷和其他肾毒性药物可能的区别是具有调控血压和维持体液稳态的作用[68]。 用人多能干细胞系H9诱导肾脏类器官,肾类器官含有肾实质成分并表达Toll样受体4,Toll样受体4是脂多糖的基本受体,当被脂多糖激活时,介导肾脏发生损伤,因此肾脏类器官适用于脂多糖诱导的肾损伤研究。脂多糖可诱导足细胞和近端肾小管细胞凋亡,增加氧化应激反应,肾脏类器官可作为有效评估脂多糖相关急性肾损伤的合适平台[69]。肾脏类器官系统具有显著的优势,它可成功地模拟由各种发挥肾细胞特异性作用的肾毒性药物诱导的急性肾损伤(表2)[11,13,16,31-32,34,58,62,64-65,67-75]。"

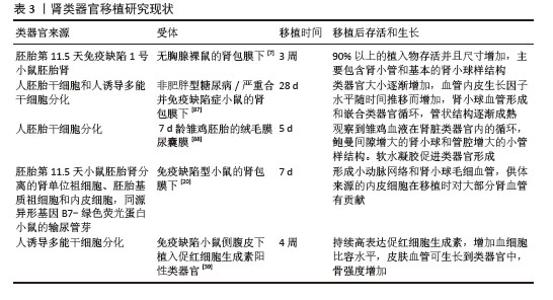
2.2.2 高通量药物筛选与治疗靶点发现 高通量筛选是用于发现小分子药物设计活动起始化合物的策略之一。多年来,高通量筛选的重点已经从数量优先转向试验质量优先[76]。毒素诱导的人诱导多能干细胞衍生肾类器官急性肾损伤模型由于小尺寸和遗传可塑性的特征而适用于高通量药物的筛选,可深入了解治疗干预反应的个体间差异。因此,选择在实验环境中可复制疾病表型的可靠模型是必不可少的,对表型筛选的成功至关重要[77]。虽然肾类器官通过药物筛选显示出巨大的治疗发展潜力,但与2D细胞培养模型不同,类器官的3D结构复杂性阻碍了类器官用于大规模药物筛选,因此,利用这些复杂的体外模型进行高通量药物筛选需要建立新的筛选通道,开发新的药物评估表型筛选方法。有研究通过全自动3D图像采集解决了肾脏类器官技术的局限性,并辅以机器学习方法自动分析肾单位节段特异性上皮形态,这种方法可以识别潜在的肾毒性和肾保护剂[73]。一项快速生成高度可重复的3D共培养球状体模型更好地展示了肾小球的特殊物理和分子结构。成熟的胶原蛋白IVα3和层粘连蛋白α5显著上调,在透射电镜和扫描电镜图像中看到的叉指足突进一步表明3D共培养球状体模型中足细胞的肾小球样成熟,可以看到原始血管形成,这表明存在足够的可溶性信号(例如血管内皮生长因子)来启动毛细血管形成。另外,球状体培养的性质非常适合更高通量的筛选。使用In cell分析仪进行更高通量的药物筛选证明了3D共培养球状体模型的快速性和可扩展性,3D共培养球状体模型测定的动态范围更大[78]。 2.2.3 再生医学与肾脏移植功能重建 再生医学的目的是通过增强自身的修复能力或移植体外功能性器官来治愈受损器官[79]。随着再生医学的快速发展,类器官已经成为有价值的药物开发研究平台,可克服传统模型的不足。在肾功能衰竭的实验动物模型中,人胎儿肾单位祖细胞已被证明参与肾组织的修复,由人诱导多能干细胞产生的肾单位祖细胞可用于开发针对肾脏疾病的再生医学研究[80]。将人诱导多能干细胞分化生成的肾单位祖细胞移植到缺血再灌注损伤小鼠急性肾损伤模型的亚囊中,发现移植后可显著抑制宿主小鼠血尿素氮和血清肌酐的升高,减轻肾小管坏死、肾小管扩张等组织病理学改变,抑制间质纤维化[81]。将人诱导多能干细胞来源的肾祖细胞尾静脉注射于顺铂诱导的急性肾损伤小鼠模型体内,小鼠肾功能显著改善,血尿素氮水平降低55%,肾组织学(细胞肿胀、脱落物沉积、刷状缘丧失和细胞坏死)显著改善[82]。通过共培养由人诱导多能干细胞分别诱导的肾单位祖细胞和输尿管芽细胞在体外生成人肾类器官,其中肾单位祖细胞来源的肾小球和肾小管以及输尿管芽来源的集合管相互连接,当移植到免疫缺陷小鼠肾囊下间隙时,这些人诱导多能干细胞衍生的肾脏类器官整合到宿主小鼠的血管中[83]。GOTO等[84]将野生型小鼠胚胎干细胞注射到无肾型Sall1(?/?)大鼠囊胚中,成功在宿主大鼠中特异性生成小鼠肾脏,小鼠多能干细胞可以有效地分化成大鼠后肾间充质,从而允许在Sall1突变大鼠中产生小鼠多能干细胞衍生的肾脏。虽然移植到宿主肾脏中会改变源自人诱导多能干细胞的肾脏类器官的肾单位隔室结构,但并未诱导细胞完全成熟。来自宿主小鼠肾脏的内皮细胞广泛浸润到移植的肾脏类器官中并形成血管网络。CMC11诱导多能干细胞分化的类器官移植到NOD-SCID小鼠体内,小鼠在移植后存活数月,小鼠内皮细胞血管化,移植物间质充满了波形蛋白阳性间充质细胞,观察到软骨形成、膀胱形成和间质扩张。转录谱显示,肾类器官移植后长期维持诱导转录组重编程,显著抑制细胞周期相关基因和上调细胞外基质组织[85]。有研究开发了一种细胞移植方法,通过该方法将多能诱导干细胞衍生的肾单位祖细胞移植到子宫内小鼠胚胎的肾脏发育区域(即器官生成生态位)[73],其中移植的肾单位祖细胞和宿主肾单位祖细胞共同促进了嵌合帽状间充质,该间充质与宿主小鼠输尿管芽相连,证明移植的小鼠或大鼠肾单位祖细胞在体外和体内成功再生成熟肾单位。啮齿动物之间再生的种间嵌合肾在体内表现出产尿能力。将人胚胎干细胞衍生的肾类器官移植到具有成熟的人类适应性免疫系统的人源化小鼠体内,移植的HLA错配肾类器官在移植30 d后引发了强烈的同种异体免疫反应,其特征是免疫细胞密集浸润和记忆T细胞表型增强,表明肾类器官可能作为人类同种异体免疫研究的宝贵工具[79]。尽管肾脏类器官在移植后表现出更好的肾单位样结构,但肾小管刷状缘、过滤屏障和毛细血管腔的结构不如成熟肾脏组织有序,需要进一步改善肾脏类器官的分化[86]。肾类器官移植研究现状,见表3[7,20,59,87-89]。 "

| [1] PICKKERS P, DARMON M, HOSTE E, et al. Acute kidney injury in the critically ill: an updated review on pathophysiology and management. Intensive Care Med. 2021;47(8):835-850. [2] WANG JJ, ZHENG Y, LI YL, et al. Emerging role of mesenchymal stem cell-derived exosomes in the repair of acute kidney injury. World J Stem Cells. 2025;17(3):103360. [3] UCHINO S, KELLUM JA, BELLOMO R, et al. Acute renal failure in critically ill patients: a multinational, multicenter study. JAMA. 2005;294(7):813-818. [4] NISHINAKAMURA R. Human kidney organoids: progress and remaining challenges. Nat Rev Nephrol. 2019;15(10):613-624. [5] TAGUCHI A, NISHINAKAMURA R. Higher-Order Kidney Organogenesis from Pluripotent Stem Cells. Cell Stem Cell. 2017;21(6):730-746.e6. [6] BUZHOR E, HARARI-STEINBERG O, OMER D, et al. Kidney spheroids recapitulate tubular organoids leading to enhanced tubulogenic potency of human kidney-derived cells. Tissue Eng Part A. 2011;17(17-18):2305-2319. [7] XINARIS C, BENEDETTI V, RIZZO P, et al. In vivo maturation of functional renal organoids formed from embryonic cell suspensions. J Am Soc Nephrol. 2012;23(11):1857-1868. [8] RIZKI-SAFITRI A, GUPTA N, HIRATSUKA K, et al. Live functional assays reveal longitudinal maturation of transepithelial transport in kidney organoids. Front Cell Dev Biol. 2022;10:978888. [9] BEJOY J, QIAN ES, WOODARD LE. Tissue Culture Models of AKI: From Tubule Cells to Human Kidney Organoids. J Am Soc Nephrol. 2022;33(3):487-501. [10] LAM AQ, FREEDMAN BS, MORIZANE R, et al. Rapid and efficient differentiation of human pluripotent stem cells into intermediate mesoderm that forms tubules expressing kidney proximal tubular markers. J Am Soc Nephrol. 2014;25(6):1211-1225. [11] MORIZANE R, LAM AQ, FREEDMAN BS, et al. Nephron organoids derived from human pluripotent stem cells model kidney development and injury. Nat Biotechnol. 2015;33(11):1193-1200. [12] TAKASATO M, ER PX, CHIU HS, et al. Kidney organoids from human iPS cells contain multiple lineages and model human nephrogenesis. Nature. 2016;536(7615):238. [13] KUMAR SV, ER PX, LAWLOR KT, et al. Kidney micro-organoids in suspension culture as a scalable source of human pluripotent stem cell-derived kidney cells. Development. 2019;146(5): dev172361. [14] FERENBACH DA, BONVENTRE JV. Mechanisms of maladaptive repair after AKI leading to accelerated kidney ageing and CKD. Nat Rev Nephrol. 2015; 11(5):264-276. [15] DIGBY JLM, VANICHAPOL T, PRZEPIORSKI A, et al. Evaluation of cisplatin-induced injury in human kidney organoids. Am J Physiol Renal Physiol. 2020;318(4):F971-F978. [16] GUPTA N, MATSUMOTO T, HIRATSUKA K, et al. Modeling injury and repair in kidney organoids reveals that homologous recombination governs tubular intrinsic repair. Sci Transl Med. 2022;14(634):eabj4772. [17] LI L, GALICHON P, XIAO X, et al. Orphan nuclear receptor COUP-TFII enhances myofibroblast glycolysis leading to kidney fibrosis. EMBO Rep. 2021;22(6):e51169. [18] TAGUCHI A, KAKU Y, OHMORI T, et al. Redefining the in vivo origin of metanephric nephron progenitors enables generation of complex kidney structures from pluripotent stem cells. Cell Stem Cell. 2014;14(1):53-67. [19] FREEDMAN BS, BROOKS CR, LAM AQ, et al. Modelling kidney disease with CRISPR-mutant kidney organoids derived from human pluripotent epiblast spheroids. Nat Commun. 2015;6:8715. [20] MURAKAMI Y, NAGANUMA H, TANIGAWA S, et al. Reconstitution of the embryonic kidney identifies a donor cell contribution to the renal vasculature upon transplantation. Sci Rep. 2019;9(1):1172. [21] UCHIMURA K, WU H, YOSHIMURA Y, et al. Human Pluripotent Stem Cell-Derived Kidney Organoids with Improved Collecting Duct Maturation and Injury Modeling. Cell Rep. 2020;33(11):108514. [22] SHI M, FU P, BONVENTRE JV, et al. Directed differentiation of ureteric bud and collecting duct organoids from human pluripotent stem cells. Nat Protoc. 2023;18(8):2485-2508. [23] LI M, GUO X, CHENG L, et al. Porcine Kidney Organoids Derived from Naïve-like Embryonic Stem Cells. Int J Mol Sci. 2024;25(1):682. [24] CECCOTTI E, SEMNANI A, BUSSOLATI B, et al. Human kidney organoids for modeling the development of different diseases. Curr Top Dev Biol. 2025;163:364-393. [25] DING B, SUN G, LIU S, et al. Three-Dimensional Renal Organoids from Whole Kidney Cells: Generation, Optimization, and Potential Application in Nephrotoxicology In Vitro. Cell Transplant. 2020;29:963689719897066. [26] OMER D, PLENICEANU O, GNATEK Y, et al. Human Kidney Spheroids and Monolayers Provide Insights into SARS-CoV-2 Renal Interactions. J Am Soc Nephrol. 2021;32(9):2242-2254. [27] KITAMURA S, SAKURAI H, MAKINO H. Single adult kidney stem/progenitor cells reconstitute three-dimensional nephron structures in vitro. Stem Cells. 2015;33(3):774-784. [28] KUROMI Y, KITAMURA S, ENDO T, et al. Cisplatin Induces Cell Death in Rat Adult Kidney Stem/ Progenitor Cell-Derived Kidney Organoids. Yonago Acta Med. 2023;66(1):153-158. [29] LI Z, ARAOKA T, WU J, et al. 3D Culture Supports Long-Term Expansion of Mouse and Human Nephrogenic Progenitors. Cell Stem Cell. 2016; 19(4):516-529. [30] MATSUI K, YAMANAKA S, CHEN S, et al. Long-term viable chimeric nephrons generated from progenitor cells are a reliable model in cisplatin-induced toxicity. Commun Biol. 2023;6(1):1097. [31] YUAN Y, CHEN H, OU S, et al. Generation of mitochondria-rich kidney organoids from expandable intermediate mesoderm progenitors reprogrammed from human urine cells under defined medium. Cell Biosci. 2022;12(1):174. [32] BEJOY J, WELCH RC, QIAN ES, et al. Urine-derived stem cells display homing, incorporation, and regeneration in human organoid and mouse models of acute kidney injury. Mol Ther. 2025; 33(7):3307-3320. [33] SUN G, DING B, WAN M, et al. Formation and optimization of three-dimensional organoids generated from urine-derived stem cells for renal function in vitro. Stem Cell Res Ther. 2020; 11(1):309. [34] GUO H, DENG N, DOU L, et al. 3-D Human Renal Tubular Organoids Generated from Urine-Derived Stem Cells for Nephrotoxicity Screening. ACS Biomater Sci Eng. 2020;6(12):6701-6709. [35] KADOTANI A, HAYASE G, YOSHINO D. Geometrically engineered organoid units and their assembly for pre-construction of organ structures. APL Bioeng. 2024;8(4):046112. [36] SONG SS, PARK HJ, KIM YK, et al. Revolutionizing biomedical research: The imperative need for heart-kidney-connected organoids. APL Bioeng. 2024;8(1):010902. [37] HOMAN KA, KOLESKY DB, SKYLAR-SCOTT MA, et al. Bioprinting of 3D Convoluted Renal Proximal Tubules on Perfusable Chips. Sci Rep. 2016;6: 34845. [38] JANG KJ, MEHR AP, HAMILTON GA, et al. Human kidney proximal tubule-on-a-chip for drug transport and nephrotoxicity assessment. Integr Biol (Camb). 2013;5(9):1119-1129. [39] MUSAH S, MAMMOTO A, FERRANTE TC, et al. Mature induced-pluripotent-stem-cell-derived human podocytes reconstitute kidney glomerular-capillary-wall function on a chip. Nat Biomed Eng. 2017;1:0069. [40] HOMAN KA, GUPTA N, KROLL KT, et al. Flow-enhanced vascularization and maturation of kidney organoids in vitro. Nat Methods. 2019; 16(3):255-262. [41] NIESKENS TTG, PERSSON M, KELLY EJ, et al. A Multicompartment Human Kidney Proximal Tubule-on-a-Chip Replicates Cell Polarization-Dependent Cisplatin Toxicity. Drug Metab Dispos. 2020;48(12):1303-1311. [42] VRIEND J, NIESKENS TTG, VORMANN MK, et al. Screening of Drug-Transporter Interactions in a 3D Microfluidic Renal Proximal Tubule on a Chip. AAPS J. 2018;20(5):87. [43] KROLL KT, MATA MM, HOMAN KA, et al. Immune-infiltrated kidney organoid-on-chip model for assessing T cell bispecific antibodies. Proc Natl Acad Sci U S A. 2023;120(35):e2305322120. [44] MA C, BANAN SADEGHIAN R, NEGORO R, et al. Efficient proximal tubule-on-chip model from hiPSC-derived kidney organoids for functional analysis of renal transporters. iScience. 2024; 27(9):110760. [45] CHOUCHA-SNOUBER L, ANINAT C, GRSICOM L, et al. Investigation of ifosfamide nephrotoxicity induced in a liver-kidney co-culture biochip. Biotechnol Bioeng. 2013;110(2):597-608. [46] ARAKAWA H, MATSUSHITA K, ISHIGURO N. Advanced in vitro evaluation of drug-induced kidney injury using microphysiological systems in drug discovery and development. Drug Metab Pharmacokinet. 2025;61:101056. [47] PODE-SHAKKED N, DEVARAJAN P. Human Stem Cell and Organoid Models to Advance Acute Kidney Injury Diagnostics and Therapeutics. Int J Mol Sci. 2022;23(13):7211. [48] HENDRIKS D, CLEVERS H, ARTEGIANI B. CRISPR-Cas Tools and Their Application in Genetic Engineering of Human Stem Cells and Organoids. Cell Stem Cell. 2020;27(5):705-731. [49] KOSTYUSHEV D, KOSTYUSHEVA A, BREZGIN S, et al. Gene Editing by Extracellular Vesicles. Int J Mol Sci. 2020;21(19):7362. [50] HOWDEN SE, VANSLAMBROUCK JM, WILSON SB, et al. Reporter-based fate mapping in human kidney organoids confirms nephron lineage relationships and reveals synchronous nephron formation. EMBO Rep. 2019;20(4):e47483. [51] VANSLAMBROUCK JM, WILSON SB, TAN KS, et al. A Toolbox to Characterize Human Induced Pluripotent Stem Cell-Derived Kidney Cell Types and Organoids. J Am Soc Nephrol. 2019;30(10):1811-1823. [52] BORESTRÖM C, JONEBRING A, GUO J, et al. A CRISP(e)R view on kidney organoids allows generation of an induced pluripotent stem cell-derived kidney model for drug discovery. Kidney Int. 2018;94(6):1099-1110. [53] LAWRENCE ML, ELHENDAWI M, MORLOCK M, et al. Human iPSC-derived renal organoids engineered to report oxidative stress can predict drug-induced toxicity. iScience. 2022;25(3):103884. [54] YOSHIMURA Y, MUTO Y, OMACHI K, et al. Elucidating the Proximal Tubule HNF4A Gene Regulatory Network in Human Kidney Organoids. J Am Soc Nephrol. 2023;34(10):1672-1686. [55] HIRSCH JS, NG JH, ROSS DW, et al. Acute kidney injury in patients hospitalized with COVID-19. Kidney Int. 2020;98(1):209-218. [56] JANSEN J, REIMER KC, NAGAI JS, et al. SARS-CoV-2 infects the human kidney and drives fibrosis in kidney organoids. Cell Stem Cell. 2022;29(2): 217-231.e8. [57] VANSLAMBROUCK JM, NEIL JA, RUDRARAJU R, et al. Kidney organoids reveal redundancy in viral entry pathways during ACE2-dependent SARS-CoV-2 infection. J Virol. 2024;98(3):e0180223. [58] LIN S, REN Z, LI L, et al. LncRNA AP001007 protects human renal tubular epithelial HK-2 cells and kidney organoids from LPS-induced injury. Sci Rep. 2024;14(1):28578. [59] DU Z, BAS-CRISTÓBAL MENÉNDEZ A, URBAN M, et al. Erythropoietin delivery through kidney organoids engineered with an episomal DNA vector. Stem Cell Res Ther. 2025;16(1):174. [60] SECKER PF, LUKS L, SCHLICHENMAIER N, et al. RPTEC/TERT1 cells form highly differentiated tubules when cultured in a 3D matrix. ALTEX. 2018;35(2):223-234. [61] ACEVES JO, HEJA S, KOBAYASHI K, et al. 3D proximal tubule-on-chip model derived from kidney organoids with improved drug uptake. Sci Rep. 2022;12(1):14997. [62] UENO S, KOKURA K, KUROMI Y, et al. Kidney organoid derived from renal tissue stem cells is a useful tool for histopathological assessment of nephrotoxicity in a cisplatin-induced acute renal tubular injury model. J Toxicol Pathol. 2022; 35(4):333-343. [63] 马瑞麟,岳亮,贠志敏,等.基于人肾类器官构建顺铂诱导急性肾损伤模型[J].中国药理学与毒理学杂志,2024,38(4):279-285. [64] NAKANOH H, TSUJI K, FUKUSHIMA K, et al. Supplement-Induced Acute Kidney Injury Reproduced in Kidney Organoids. Am J Nephrol. 2025;56(4):520-528. [65] LEE KS, LEE J, LEE P, et al. Inhibition of O-GlcNAcylation protects from Shiga toxin-mediated cell injury and lethality in host. EMBO Mol Med. 2022;14(1):e14678. [66] GU S, WU G, LU D, et al. Nephrotoxicity assessment of Esculentoside A using human-induced pluripotent stem cell-derived organoids. Phytother Res. 2024;38(10):4893-4903. [67] GU S, WU G, LU D, et al. Human kidney organoids model of Esculentoside A nephrotoxicity to investigate the role of epithelial-mesenchymal transition via STING signaling. Toxicol Lett. 2023; 373:172-183. [68] NGUYEN L, WRUCK W, ERICHSEN L, et al. The Nephrotoxin Puromycin Aminonucleoside Induces Injury in Kidney Organoids Differentiated from Induced Pluripotent Stem Cells. Cells. 2022;11(4):635. [69] ZHANG W, QI R, LI T, et al. Kidney Organoids as a Novel Platform to Evaluate Lipopolysaccharide-Induced Oxidative Stress and Apoptosis in Acute Kidney Injury. Front Med (Lausanne). 2021;8: 766073. [70] SHEN C, WANG Q, YE X, et al. Celastrol reduces cisplatin-induced nephrotoxicity by downregulating SNORD3A level in kidney organoids derived from human iPSCs. Front Pharmacol. 2025;16:1464525. [71] WU H, NING Y, SUN Z, et al. Both carvedilol and cimetidine alleviate cisplatin-induced nephrotoxicity via downregulating OCT2. Biochim Biophys Acta Mol Basis Dis. 2025;1871(5):167754. [72] SAHARA Y, FUKUI C, KUNIYOSHI Y, et al. Proximal tubule cell maturation rate and function are controlled by PPARα signaling in kidney organoids. Commun Biol. 2024;7(1):1532. [73] OISHI H, TABIBZADEH N, MORIZANE R. Advancing preclinical drug evaluation through automated 3D imaging for high-throughput screening with kidney organoids. Biofabrication. 2024;16(3). doi: 10.1088/1758-5090/ad38df. [74] PENG K, XIE W, WANG T, et al. HIF-1α promotes kidney organoid vascularization and applications in disease modeling. Stem Cell Res Ther. 2023; 14(1):336. [75] 吴欢,嵇姞,鲁敏,等.基于肾脏类器官研究镁离子减轻顺铂诱导的急性肾损伤的作用和机制[J].复旦学报(医学版), 2024,51(4): 455-464+483. [76] BLAY V, TOLANI B, HO SP, et al. High-Throughput Screening: today’s biochemical and cell-based approaches. Drug Discov Today. 2020;25(10): 1807-1821. [77] VINCENT F, NUEDA A, LEE J, et al. Phenotypic drug discovery: recent successes, lessons learned and new directions. Nat Rev Drug Discov. 2022;21(12):899-914. [78] TUFFIN J, CHESOR M, KUZMUK V, et al. GlomSpheres as a 3D co-culture spheroid model of the kidney glomerulus for rapid drug-screening. Commun Biol. 2021;4(1):1351. [79] MON-WEI YU S, CHOI JY, KADY J, et al. Transplantation of human kidney organoids elicited a robust allogeneic response in a humanized mouse model. Kidney Int. 2025;107(6):1011-1016. [80] HARARI-STEINBERG O, METSUYANIM S, OMER D, et al. Identification of human nephron progenitors capable of generation of kidney structures and functional repair of chronic renal disease. EMBO Mol Med. 2013;5(10):1556-1568. [81] TOYOHARA T, MAE S, SUETA S, et al. Cell Therapy Using Human Induced Pluripotent Stem Cell-Derived Renal Progenitors Ameliorates Acute Kidney Injury in Mice. Stem Cells Transl Med. 2015;4(9):980-992. [82] IMBERTI B, TOMASONI S, CIAMPI O, et al. Renal progenitors derived from human iPSCs engraft and restore function in a mouse model of acute kidney injury. Sci Rep. 2015;5:8826. [83] OSAFUNE K. iPSC technology-based regenerative medicine for kidney diseases. Clin Exp Nephrol. 2021;25(6):574-584. [84] GOTO T, HARA H, SANBO M, et al. Generation of pluripotent stem cell-derived mouse kidneys in Sall1-targeted anephric rats. Nat Commun. 2019;10(1):451. [85] NAM SA, SEO E, KIM JW, et al. Graft immaturity and safety concerns in transplanted human kidney organoids. Exp Mol Med. 2019;51(11):1-13. [86] PAN B, FAN G. Stem cell-based treatment of kidney diseases. Exp Biol Med (Maywood). 2020; 245(10):902-910. [87] VAN DEN BERG CW, RITSMA L, AVRAMUT MC, et al. Renal Subcapsular Transplantation of PSC-Derived Kidney Organoids Induces Neo-vasculogenesis and Significant Glomerular and Tubular Maturation In Vivo. Stem Cell Reports. 2018;10(3):751-765. [88] GARRETA E, PRADO P, TARANTINO C, et al. Fine tuning the extracellular environment accelerates the derivation of kidney organoids from human pluripotent stem cells. Nat Mater. 2019;18(4):397-405. [89] IDE H, MIIKE K, OHMORI T, et al. Mouse embryonic kidney transplantation identifies maturation defects in the medulla. Sci Rep. 2024;14(1):30293. [90] WIRAJA C, MORI Y, ICHIMURA T, et al. Nephrotoxicity Assessment with Human Kidney Tubuloids using Spherical Nucleic Acid-Based mRNA Nanoflares. Nano Lett. 2021;21(13): 5850-5858. [91] WAKAI E, SUZUMURA Y, IKEMURA K, et al. An Integrated In Silico and In Vivo Approach to Identify Protective Effects of Palonosetron in Cisplatin-Induced Nephrotoxicity. Pharmaceuticals (Basel). 2020;13(12):480. [92] XIA S, WU M, CHEN S, et al. Long Term Culture of Human Kidney Proximal Tubule Epithelial Cells Maintains Lineage Functions and Serves as an Ex vivo Model for Coronavirus Associated Kidney Injury. Virol Sin. 2020;35(3):311-320. [93] CLERKIN S, SINGH K, DAVIS JL, et al. Tuneable gelatin methacryloyl (GelMA) hydrogels for the directed specification of renal cell types for hiPSC-derived kidney organoid maturation. Biomaterials. 2025;322:123349. [94] KIM D, LIM H, YOUN J, et al. Scalable production of uniform and mature organoids in a 3D geometrically-engineered permeable membrane. Nat Commun. 2024;15(1):9420. [95] MOHAMED T, SEQUEIRA-LOPEZ MLS. Development of the renal vasculature. Semin Cell Dev Biol. 2019;91:132-146. [96] LOW JH, LI P, CHEW EGY, et al. Generation of Human PSC-Derived Kidney Organoids with Patterned Nephron Segments and a De Novo Vascular Network. Cell Stem Cell. 2019;25(3): 373-387.e9. [97] GOUX CORREDERA I, AMATO G, MOYA-RULL D, et al. Unlocking the full potential of human pluripotent stem cell-derived kidney organoids through bioengineering. Kidney Int. 2025;108(1):38-47. [98] PRZEPIORSKI A, SANDER V, TRAN T, et al. A Simple Bioreactor-Based Method to Generate Kidney Organoids from Pluripotent Stem Cells. Stem Cell Reports. 2018;11(2):470-484. [99] VAN WINKLE AP, GATES ID, KALLOS MS. Mass transfer limitations in embryoid bodies during human embryonic stem cell differentiation. Cells Tissues Organs. 2012;196(1):34-47. |
| [1] | Zhu Jing, Zhai Xiguo, Wu Qizhen, Wang Yupei, Lyu Ling, Hou Qinzheng. Development and application of human amniotic membrane in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1818-1827. |
| [2] | Liu Xingyu, Li Lijie. Secretome of stem cells from human exfoliated deciduous teeth: a new hotspot in tissue engineering and stem cell therapy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1858-1868. |
| [3] | Peng Tuanhui, Song Hongming, Yang Ling, Ding Xiaoge, Meng Pengjun. Effects of long-term endurance exercise on kl/FGF23 axis and calcium-phosphorus metabolism in naturally aging mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1089-1095. |
| [4] | Sun Yajie, Zhao Xinchen, Bo Shuangling. Spatiotemporal expression of bone morphologic protein 7 in mouse kidney development [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1156-1161. |
| [5] | Wang Jiangjing, Zhao Na, Hu Xiaona, Zhao Na . Safety of 3D printed titanium alloy bone trabecular cup prosthesis combined with modified Kidney Tonifying and Blood Activating Decoction for elderly hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 612-619. |
| [6] | Zhou Li, Li Rui, Chen Hao, Chen Jiaqi, Liu Yuhong, Wu Na. Constructing an in vitro model of ulcerative colitis in mice based on organoid technology [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6231-6238. |
| [7] | Wang Yan, Lyu Hao, Hu Zhimu, Zhou Yao, Liu Qiang, Yang Yuxiang, Yi Hairu, Wang Jiuxiang, Jiang Ting. Intervention with Compound Kidney-Invigorating Granules in a mouse model of osteoporosis: role of the TRIB3/beta-catenin axis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6142-6149. |
| [8] | Mou Jiancheng, Luo Jie, Liu Haotian, Yang Zhuotao, Mu Yuxiao, Qian Da, Meng Xuli. Application and prospects of precision-medicine-driven breast cancer organoids in therapeutic drug discovery [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 5033-5039. |
| [9] | Niu Qi, Chen Junji, Tu Haining, Mo Weibin, Zhong Yujin, Li Mingliang. Effect of swimming exercise combined with probiotic intervention on anti-inflammatory and apoptotic gene expression in renal tissue of type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(16): 4105-4114. |
| [10] | Wu Jiazhou, Qian Tao, Liu Zexian, Wu Yanbin, He Ying, Li Yazhou, Peng Jiang. Three-dimensional culture of stromal vascular fraction self-assembles into complex vascularized osteogenic organoids [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(11): 2681-2690. |
| [11] | Luo Wenbin, Li Ruoyun, Pan Chaofan, Luo Changjiang. Engineered exosomes for repairing tissue damage: application potential, excellent biological stability, and targeting specificity [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(1): 204-217. |
| [12] | Zhao Qianwei, Sun Guangyuan . Intestinal organoids: a bibliometric analysis of the latest trends in tissue/organ biology, disease modeling, and clinical applications [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(1): 238-247. |
| [13] | Yuan Weibo, Liu Chan, Yu Limei. Potential application of liver organoids in liver disease models and transplantation therapy [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(8): 1684-1692. |
| [14] | Li Jialin, Zhang Yaodong, Lou Yanru, Yu Yang, Yang Rui. Molecular mechanisms underlying role of mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1512-1522. |
| [15] |
Huang Xiaobin, Ge Jirong, Li Shengqiang, Xie Lihua, Huang Jingwen, He Yanyan, Xue Lipeng.
Mechanisms of different yin nourishing and kidney tonifying methods on osteoclastysis pathway in ovariectomized rats #br#
#br#
[J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1214-1219.
|
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||