Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (19): 4926-4933.doi: 10.12307/2026.192
Previous Articles Next Articles
Non-coding RNA-activated by DNA damage promotes proliferation and inhibits apoptosis of induced pluripotent stem cell-derived cardiomyocytes
Huan Kanghui, Jiang Yujian, Bian Weihua
- Department of Pharmacy, Binzhou Medical University, Yantai 264003, Shandong Province, China
-
Received:2025-06-20Accepted:2025-09-13Online:2026-07-08Published:2026-02-14 -
Contact:Bian Weihua, PhD, Professor, Master’s supervisor, Department of Pharmacy, Binzhou Medical University, Yantai 264003, Shandong Province, China -
About author:Huan Kanghui, Master candidate, Department of Pharmacy, Binzhou Medical University, Yantai 264003, Shandong Province, China -
Supported by:National Natural Science Foundation of China, No. 82270305 (to BWH)
CLC Number:
Cite this article
Huan Kanghui, Jiang Yujian, Bian Weihua. Non-coding RNA-activated by DNA damage promotes proliferation and inhibits apoptosis of induced pluripotent stem cell-derived cardiomyocytes[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 4926-4933.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
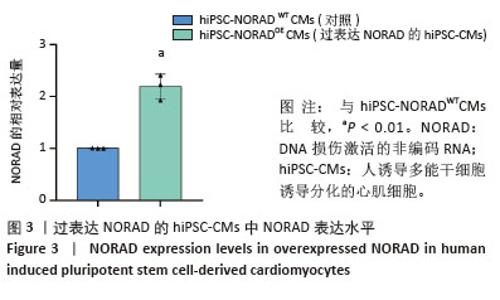
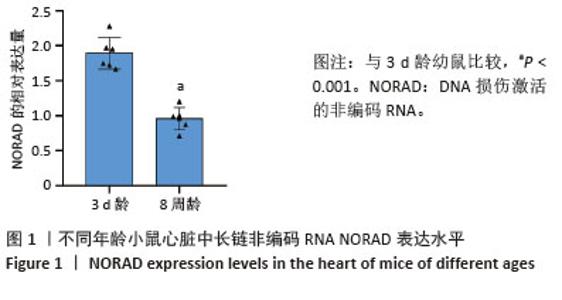
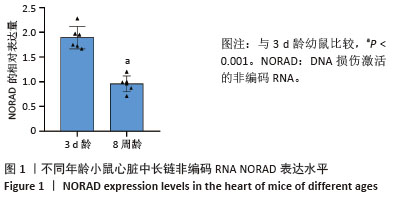
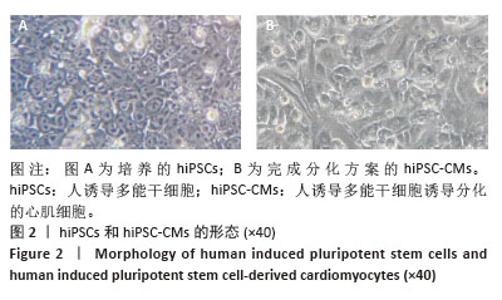
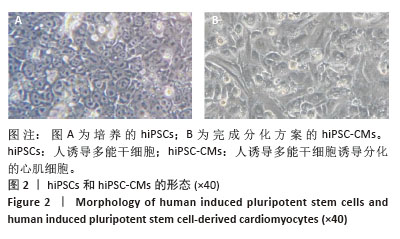
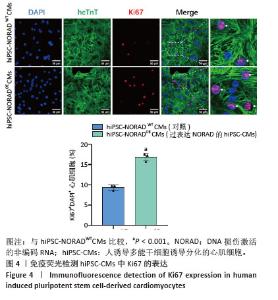
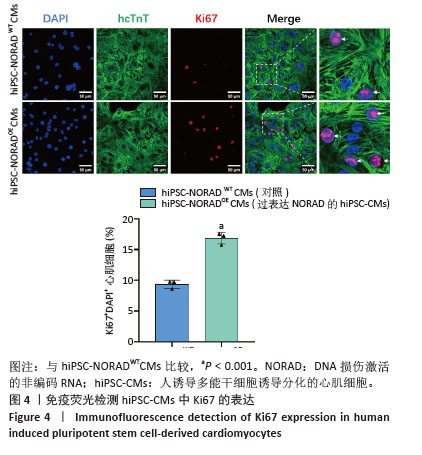
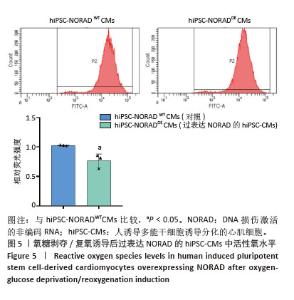
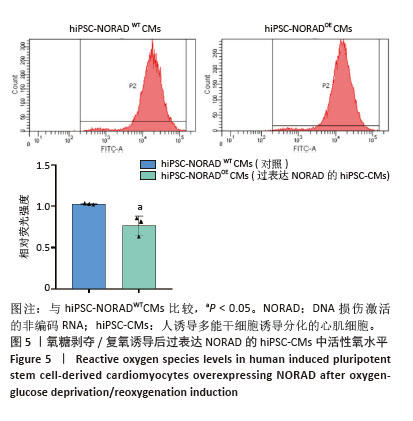
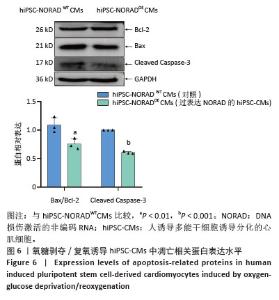
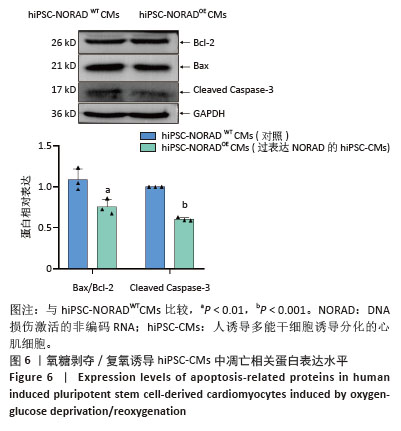
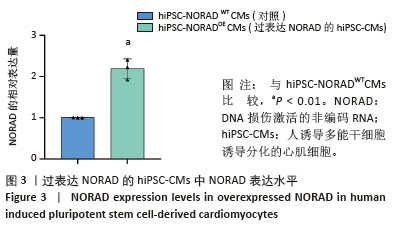
2.3 过表达NORAD的hiPSC-CMs构建及验证 为了构建长链非编码RNA NORAD过表达的hiPSC-CMs。将NORAD亚克隆到含有心肌特异性αMHC启动子的质粒载体中,以此构建在心肌细胞中特异性过表达NORAD的质粒:Lenti-αMHC-NORAD。通过利用Lenti-αMHC-NORAD质粒进行慢病毒包装,感染人诱导多能干细胞,经抗生素筛选,获得导入NORAD基因的稳定细胞株,诱导分化获得过表达NORAD的hiPSC-CMs(hiPSC-NORADOECMs),同时以只导入载体质粒 hiPSCs分化的hiPSC-CMs(hiPSC-NORADWTCMs)作为对照。RT-qPCR结果显示:hiPSC-NORADOECMs中NORAD表达水平为hiPSC-NORADWTCMs的2.2倍,见图3。"
| [1] CHORADIA A, BAI K, SONI S, et al. Beyond hot flashes: Exploring the role of estrogen therapy in postmenopausal women for myocardial infarction prevention and recovery. Biomol Biomed. 2024;24(1):4-13. [2] SALARI N, MORDDARVANJOGHI F, ABDOLMALEKI A, et al. The global prevalence of myocardial infarction: a systematic review and meta-analysis. BMC Cardiovasc Disord. 2023;23(1):206. [3] HU D, LI R, LI Y, et al. Inflammation-Targeted Nanomedicines Alleviate Oxidative Stress and Reprogram Macrophages Polarization for Myocardial Infarction Treatment. Adv Sci (Weinh). 2024;11(21):e2308910. [4] MAGADUM A, SINGH N, KURIAN AA, et al. Pkm2 Regulates Cardiomyocyte Cell Cycle and Promotes Cardiac Regeneration. Circulation. 2020;141(15): 1249-1265. [5] BAE J, SALAMON RJ, BRANDT EB, et al. Malonate Promotes Adult Cardiomyocyte Proliferation and Heart Regeneration. Circulation. 2021; 143(20):1973-1986. [6] ZHANG H, PEI L, OUYANG Z, et al. AP-1 activation mediates post-natal cardiomyocyte maturation. Cardiovasc Res. 2023;119(2):536-550. [7] GIACCA M. Cardiac Regeneration After Myocardial Infarction: an Approachable Goal. Curr Cardiol Rep. 2020;22(10):122. [8] KINO T, KHAN M, MOHSIN S. The Regulatory Role of T Cell Responses in Cardiac Remodeling Following Myocardial Infarction. Int J Mol Sci. 2020;21(14):5013. [9] YAN W, XIA Y, ZHAO H, et al. Stem cell-based therapy in cardiac repair after myocardial infarction: Promise, challenges, and future directions. J Mol Cell Cardiol. 2024;188:1-14. [10] KARAKIKES I, AMEEN M, TERMGLINCHAN V, et al. Human induced pluripotent stem cell-derived cardiomyocytes: insights into molecular, cellular, and functional phenotypes. Circ Res. 2015;117(1):80-88. [11] MUMMERY CL, ZHANG J, NG ES, et al. Differentiation of human embryonic stem cells and induced pluripotent stem cells to cardiomyocytes: a methods overview. Circ Res. 2012;111(3):344-358. [12] LIN Y, GIL CH, YODER MC. Differentiation, Evaluation, and Application of Human Induced Pluripotent Stem Cell-Derived Endothelial Cells. Arterioscler Thromb Vasc Biol. 2017;37(11):2014-2025. [13] PATSCH C, CHALLET-MEYLAN L, THOMA EC, et al. Generation of vascular endothelial and smooth muscle cells from human pluripotent stem cells. Nat Cell Biol. 2015;17(8):994-1003. [14] ENGLE SJ, BLAHA L, KLEIMAN RJ. Best Practices for Translational Disease Modeling Using Human iPSC-Derived Neurons. Neuron. 2018; 100(4):783-797. [15] POURRIER M, FEDIDA D. The Emergence of Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes (hiPSC-CMs) as a Platform to Model Arrhythmogenic Diseases. Int J Mol Sci. 2020;21(2):657. [16] SHARMA A, BURRIDGE PW, MCKEITHAN WL, et al. High-throughput screening of tyrosine kinase inhibitor cardiotoxicity with human induced pluripotent stem cells. Sci Transl Med. 2017;9(377):eaaf2584. [17] SEIBERTZ F, SUTANTO H, DÜLK R, et al. Electrophysiological and calcium-handling development during long-term culture of human-induced pluripotent stem cell-derived cardiomyocytes. Basic Res Cardiol. 2023; 118(1):14. [18] BHATTACHARYA S, BURRIDGE PW, KROPP EM, et al. High efficiency differentiation of human pluripotent stem cells to cardiomyocytes and characterization by flow cytometry. J Vis Exp. 2014;(91):52010. [19] DEUSE T, HU X, GRAVINA A, et al. Hypoimmunogenic derivatives of induced pluripotent stem cells evade immune rejection in fully immunocompetent allogeneic recipients. Nat Biotechnol. 2019; 37(3):252-258. [20] BIAN W, CHEN W, NGUYEN T, et al. miR-199a Overexpression Enhances the Potency of Human Induced-Pluripotent Stem-Cell-Derived Cardiomyocytes for Myocardial Repair. Front Pharmacol. 2021;12:673621. [21] CAI H, HAN XJ, LUO ZR, et al. Pretreatment with Notoginsenoside R1 enhances the efficacy of neonatal rat mesenchymal stem cell transplantation in model of myocardial infarction through regulating PI3K/Akt/FoxO1 signaling pathways. Stem Cell Res Ther. 2024;15(1):419. [22] MATTICK JS, AMARAL PP, CARNINCI P, et al. Long non-coding RNAs: definitions, functions, challenges and recommendations. Nat Rev Mol Cell Biol. 2023;24(6):430-447. [23] LI K, WANG Z. lncRNA NEAT1: Key player in neurodegenerative diseases. Ageing Res Rev. 2023;86:101878. [24] BRIDGES MC, DAULAGALA AC, KOURTIDIS A. LNCcation: lncRNA localization and function. J Cell Biol. 2021;220(2):e202009045. [25] NI H, GE Y, ZHUGE Y, et al. LncRNA MIR181A1HG Deficiency Attenuates Vascular Inflammation and Atherosclerosis. Circ Res. 2025;136(8):862-883. [26] YANG R, YANG B, LIU W, et al. Emerging role of non-coding RNAs in neuroinflammation mediated by microglia and astrocytes. J Neuroinflammation. 2023;20(1):173. [27] VIERECK J, KUMARSWAMY R, FOINQUINOS A, et al. Long noncoding RNA Chast promotes cardiac remodeling. Sci Transl Med. 2016;8(326): 326ra22. [28] SOGHLI N, YOUSEFI T, ABOLGHASEMI M, et al. NORAD, a critical long non-coding RNA in human cancers. Life Sci. 2021;264:118665. [29] ALVES-VALE C, CAPELA AM, TAVARES-MARCOS C, et al. Expression of NORAD correlates with breast cancer aggressiveness and protects breast cancer cells from chemotherapy. Mol Ther Nucleic Acids. 2023; 33:910-924. [30] SUN DS, GUAN CH, WANG WN, et al. LncRNA NORAD promotes proliferation, migration and angiogenesis of hepatocellular carcinoma cells through targeting miR-211-5p/FOXD1/VEGF-A axis. Microvasc Res. 2021;134:104120. [31] ZHAO W, WANG L, XU F. LncRNA NORAD stimulates proliferation and migration of renal cancer via activating the miR-144-3p/MYCN axis. Eur Rev Med Pharmacol Sci. 2020;24(20):10426-10432. [32] ZHANG Y, LI Y. Long non-coding RNA NORAD contributes to the proliferation, invasion and EMT progression of prostate cancer via the miR-30a-5p/RAB11A/WNT/β-catenin pathway. Cancer Cell Int. 2020;20(1):571. [33] BIAN W, JING X, YANG Z, et al. Downregulation of LncRNA NORAD promotes Ox-LDL-induced vascular endothelial cell injury and atherosclerosis. Aging (Albany NY). 2020;12(7):6385-6400. [34] ZHANG X, KAN X, SHEN J, et al. Increased long non-coding RNA NORAD reflects serious cardiovascular stenosis, aggravated inflammation status, and higher lipid level in coronary heart disease. J Clin Lab Anal. 2022; 36(11):e24717. [35] HAN Y, TIAN H, GAO X. NORAD regulates proliferation and apoptosis in cardiomyocytes under high-glucose treatment through miRNA-150-5p/ZEB1 axis. Eur Rev Med Pharmacol Sci. 2020;24(21):11259-11265. [36] REED GW, ROSSI JE, CANNON CP. Acute myocardial infarction. Lancet. 2017;389(10065):197-210. [37] BUSSMANN WD, LÖHNER J, KALTENBACH M. Orally administered isosorbide dinitrate in patients with and without left ventricular failure due to acute myocardial infarction. Am J Cardiol. 1977;39(1):91-96. [38] 覃可乐.成纤维细胞来源外泌体通过激活AMPK通路促进hiPSC-CMs成熟机制研究[D].长沙:中南大学,2023. [39] PARIZADEH SM, JAFARZADEH-ESFEHANI R, GHANDEHARI M, et al. Stem cell therapy: A novel approach for myocardial infarction. J Cell Physiol. 2019;234(10):16904-16912. [40] YAMANAKA S. Pluripotent Stem Cell-Based Cell Therapy-Promise and Challenges. Cell Stem Cell. 2020;27(4):523-531. [41] FAN C, FAST VG, TANG Y, et al. Cardiomyocytes from CCND2-overexpressing human induced-pluripotent stem cells repopulate the myocardial scar in mice: A 6-month study. J Mol Cell Cardiol. 2019;137:25-33. [42] TAO Z, LOO S, SU L, et al. Angiopoietin-1 enhanced myocyte mitosis, engraftment, and the reparability of hiPSC-CMs for treatment of myocardial infarction. Cardiovasc Res. 2021;117(6):1578-1591. [43] ABBAS N, PERBELLINI F, THUM T. Non-coding RNAs: emerging players in cardiomyocyte proliferation and cardiac regeneration. Basic Res Cardiol. 2020;115(5):52. [44] PICCOLI MT, GUPTA SK, THUM T. Noncoding RNAs as regulators of cardiomyocyte proliferation and death. J Mol Cell Cardiol. 2015; 89(Pt A):59-67. [45] BUGGER H, PFEIL K. Mitochondrial ROS in myocardial ischemia reperfusion and remodeling. Biochim Biophys Acta Mol Basis Dis. 2020;1866(7):165768. [46] SUN Q, MA H, ZHANG J, et al. A Self-Sustaining Antioxidant Strategy for Effective Treatment of Myocardial Infarction. Adv Sci (Weinh). 2023; 10(5):e2204999. [47] XIANG M, LU Y, XIN L, et al. Role of Oxidative Stress in Reperfusion following Myocardial Ischemia and Its Treatments. Oxid Med Cell Longev. 2021;2021:6614009. [48] ZHENG H, XU Y, LIEHN EA, et al. Vitamin C as Scavenger of Reactive Oxygen Species during Healing after Myocardial Infarction. Int J Mol Sci. 2024;25(6):3114. [49] 李恩,孙利强,刘宗芳,等.瑞舒伐他汀对大鼠心肌梗死后活性氧物质产生及骨膜蛋白和心肌营养素表达的影响[J].中国循环杂志, 2014,29(10):823-827. [50] LUO Z, XU X, SHO T, et al. ROS-induced autophagy regulates porcine trophectoderm cell apoptosis, proliferation, and differentiation. Am J Physiol Cell Physiol. 2019;316(2):C198-C209. [51] XIONG X, LIU J, HE Q, et al. Long non-coding RNA NORAD aggravates acute myocardial infarction by promoting fibrosis and apoptosis via miR-577/COBLL1 axis. Environ Toxicol. 2021;36(11):2256-2265. [52] LI C, ZHANG L, BU X, et al. LncRNA NORAD promotes the progression of myocardial infarction by targeting the miR-22-3p/PTEN axis. Acta Biochim Biophys Sin (Shanghai). 2022;54(4):463-473. |
| [1] | Wang Qisa, Lu Yuzheng, Han Xiufeng, Zhao Wenling, Shi Haitao, Xu Zhe. Cytocompatibility of 3D printed methyl acrylated hyaluronic acid/decellularized skin hydrogel scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1912-1920. |
| [2] | Sun Lei, Zhang Qi, Zhang Yu. Pro-osteoblastic effect of chlorogenic acid protein microsphere/polycaprolactone electrospinning membrane [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1877-1884. |
| [3] | Liu Yang, Liu Donghui , Xu Lei, Zhan Xu, Sun Haobo, Kang Kai. Role and trend of stimuli-responsive injectable hydrogels in precise myocardial infarction therapy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2072-2080. |
| [4] | Yuan Xiaoshuang, Yang Xu, Yang Bo, Chen Xiaoxu, Tian Ting, Wang Feiqing, Li Yanju, Liu Yang, Yang Wenxiu. Effect of conditioned medium of diffuse large B-cell lymphoma cells on proliferation and apoptosis of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1632-1640. |
| [5] | Jin Dongsheng, Zhao Zhanghong, Zhu Ziyin, Zhang Sen, Sun Zuyan, Deng Jiang. Effects of icariin-loaded microsphere-three-dimensional scaffold on osteogenic differentiation of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1658-1668. |
| [6] | He Jiale, Huang Xi, Dong Hongfei, Chen Lang, Zhong Fangyu, Li Xianhui. Acellular dermal matrix combined with adipose-derived stem cell exosomes promotes burn wound healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1699-1710. |
| [7] | Liu Anting, Lu Jiangtao, Zhang Wenjie, He Ling, Tang Zongsheng, Chen Xiaoling. Regulation of AMP-activated protein kinase by platelet lysate inhibits cadmium-induced neuronal apoptosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1800-1807. |
| [8] | Jia Jinwen, Airefate·Ainiwaer, Zhang Juan. Effects of EP300 on autophagy and apoptosis related to allergic rhinitis in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1439-1449. |
| [9] | Huang Liuyan, Zhang Wenxi, Chen Shuwen, Yu Shimei, Dai Zhong, Zuo Changqing. Forskolin promotes C2C12 myoblast differentiation via regulating the ERK and Akt signaling pathways [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1114-1121. |
| [10] | Liu Yu, Lei Senlin, Zhou Jintao, Liu Hui, Li Xianhui. Mechanisms by which aerobic and resistance exercises improve obesity-related cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1171-1183. |
| [11] | Wang Zhengye, Liu Wanlin, Zhao Zhenqun. Advance in the mechanisms underlying miRNAs in steroid-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1207-1214. |
| [12] | Yang Xiao, Bai Yuehui, Zhao Tiantian, Wang Donghao, Zhao Chen, Yuan Shuo. Cartilage degeneration in temporomandibular joint osteoarthritis: mechanisms and regenerative challenges [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 926-935. |
| [13] | Bao Zhuoma, Hou Ziming, Jiang Lu, Li Weiyi, Zhang Zongxing, Liu Daozhong, Yuan Lin. Effect and mechanism by which Pterocarya hupehensis skan total flavonoids regulates the proliferation, migration and apoptosis of fibroblast-like synoviocytes [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 816-823. |
| [14] | Wang Zhengye, Liu Wanlin, Zhao Zhenqun. Mechanism by which vascular endothelial growth factor A targets regulation of angiogenesis in the treatment of steroid-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 671-679. |
| [15] | Zhang Jingyi, Ma Fang, Liu Honglin, Wang Jianjun, Xia Tongtong, Yang Jiaqi, Wang Yajing, Shen Jiangyong, Jiang Yideng. Regulatory role of ADAMTS8 in proliferation and apoptosis of hypertrophic scar fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7260-7266. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||