Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (25): 3956-3963.doi: 10.12307/2023.459
Previous Articles Next Articles
Preparation and characterization of nano-hydroxyapatite/aspirin/polyvinyl alcohol/gelatin/sodium alginate hydrogel scaffolds
Li Shaoping1, Yang Yuqing1, Xiao Wenyundeng1, Yin Lulu2, Liu Libo2, Liu Ying2, Sun Yifan2, Chen Zhiyu1
- 1Department of Prosthodontics, School of Stomatology · Hospital of Stomatology, Hebei Medical University, Hebei Provincial Key Laboratory of Stomatology, Hebei Provincial Clinical Research Center for Oral Diseases, Shijiazhuang 050017, Hebei Province, China; 2School of Stomatology, Hebei Medical University, Shijiazhuang 050017, Hebei Province, China
-
Received:2022-06-15Accepted:2022-07-18Online:2023-09-08Published:2023-01-17 -
Contact:Chen Zhiyu, PhD, Department of Prosthodontics, School of Stomatology · Hospital of Stomatology, Hebei Medical University, Hebei Provincial Key Laboratory of Stomatology, Hebei Provincial Clinical Research Center for Oral Diseases, Shijiazhuang 050017, Hebei Province, China -
About author:Li Shaoping, Master candidate, Department of Prosthodontics, School of Stomatology · Hospital of Stomatology, Hebei Medical University, Hebei Provincial Key Laboratory of Stomatology, Hebei Provincial Clinical Research Center for Oral Diseases, Shijiazhuang 050017, Hebei Province, China -
Supported by:Hebei Provincial Talents Project, No. A202102010 (to CZY); Hebei Provincial Government Funded Clinical Medical Talents Training Project, No. 361029 (to CZY)
CLC Number:
Cite this article
Li Shaoping, Yang Yuqing, Xiao Wenyundeng, Yin Lulu, Liu Libo, Liu Ying, Sun Yifan, Chen Zhiyu. Preparation and characterization of nano-hydroxyapatite/aspirin/polyvinyl alcohol/gelatin/sodium alginate hydrogel scaffolds[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(25): 3956-3963.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
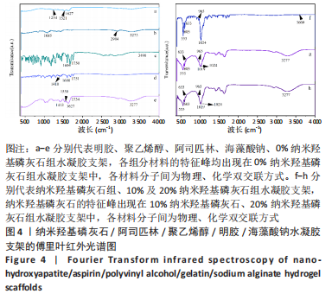
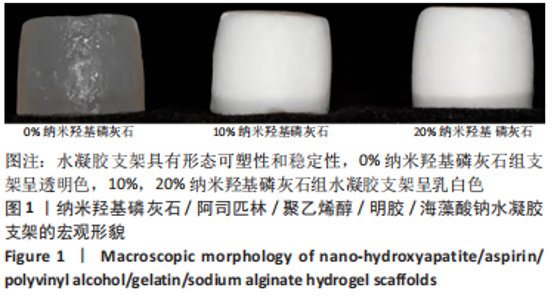
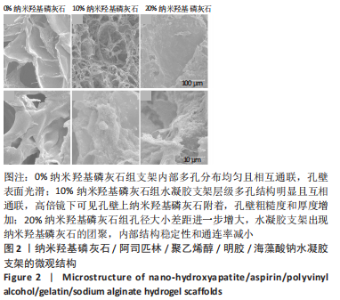
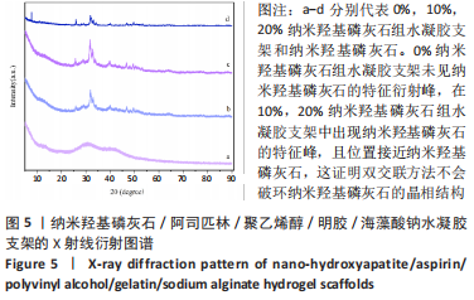
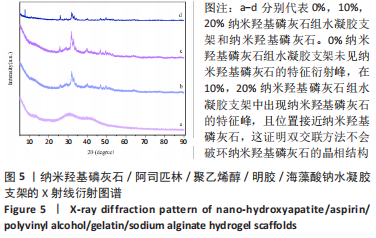
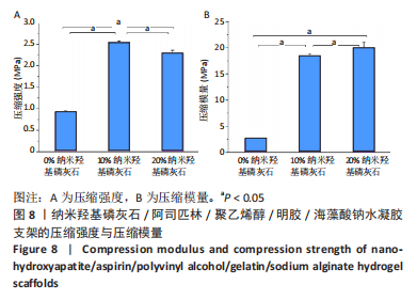
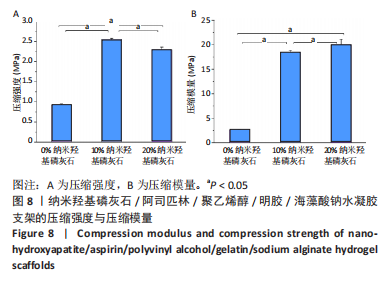
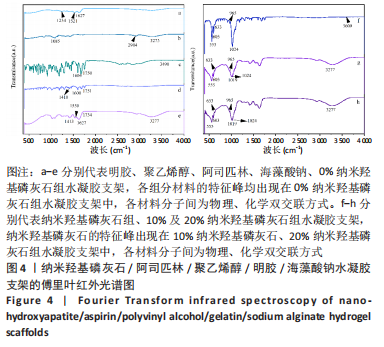
2.3 水凝胶支架的傅里叶红外光谱分析 明胶的特征吸收峰酰胺Ⅰ带(C=O的伸缩振动峰)、酰胺Ⅱ带(N-H的弯曲振动峰)、酰胺Ⅲ带(C-N的伸缩振动峰)分别位于1 627,1 521,1 234 cm-1。聚乙烯醇的特征吸收峰位于3 273,2 904,1 085 cm-1处,分别为O-H、C-O伸缩振动和C-H的伸缩振动峰。海藻酸钠位于1 751 cm-1处的特征吸收峰为C=O的伸缩振动峰,在1 600 cm-1处为COO-的反对称收缩峰、1 410 cm-1处为对称收缩峰。阿司匹林的特征吸收峰为O-H、C=O和C=C的伸缩振动峰,分别位于3 490,1 750,1 605 cm-1处。在0%纳米羟基磷灰石组水凝胶支架的红外光谱中,O-H峰位移到3 277 cm-1处,宽度明显增加,1 627 cm-1处(酰胺Ⅰ带)的峰明显增宽,1 521 cm-1处(酰胺Ⅱ带)的峰蓝移至1 550 cm-1处以及阿司匹林、海藻酸钠在指纹区的微弱小峰在水凝胶支架中的消失证实了聚乙烯醇、明胶、海藻酸钠、阿司匹林之间相互作用并形成氢键[13,20];海藻酸钠中C=O吸收峰从1 751 cm-1处移动至1 734 cm-1处变成微弱的小峰,证明了海藻酸钠与明胶之间强烈的静电引力[21-22];海藻酸钠中COO-由1 400 cm-1移动至1 413 cm-1处,证明Ca2+与COO-化学交联形成蛋壳结构[23],见图4。纳米羟基磷灰石的特征吸收峰为PO43-的伸缩振动峰,位于1 024 cm-1和965cm-1处,以及PO43-的变形振动峰,位于603,555 cm-1处,-OH伸缩振动峰为3 600 cm-1处、弯曲振动峰为633 cm-1处。水凝胶支架中加入纳米羟基磷灰石后,在603,555 cm-1处出现了纳米羟基磷灰石特征峰,965 cm-1处特征峰消失、1 024 cm-1处峰值红移至1 019 cm-1处且峰增宽这可能由于纳米羟基磷灰石中的PO43-(1 024 cm-1)与聚乙烯醇发生相互作用形成氢键[24],见图4。因此聚乙烯醇、海藻酸钠、明胶、阿司匹林、纳米羟基磷灰石之间存在着物理和化学双交联的形式,但未出现成分的丢失,加入纳米羟基磷灰石可以使支架更加紧密、提高机械性能、持续缓慢释放药物,使支架能够适应和调节骨缺损修复的微环境。"
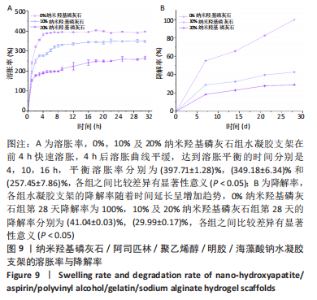
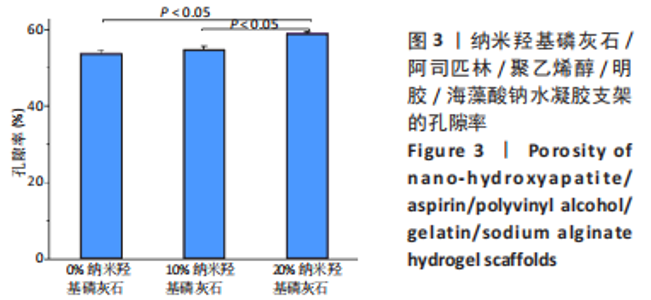
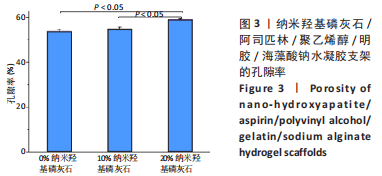
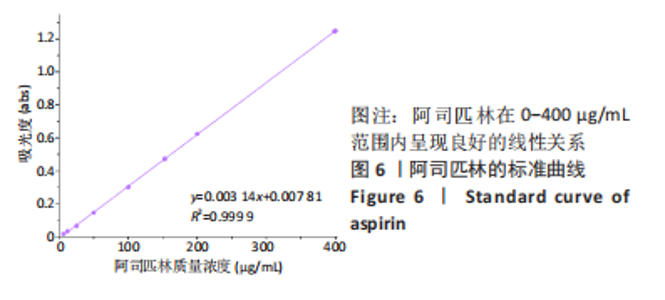
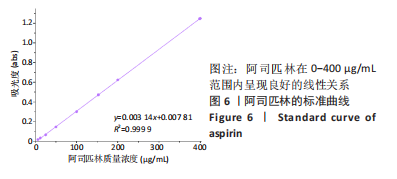
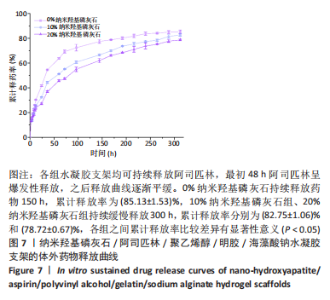
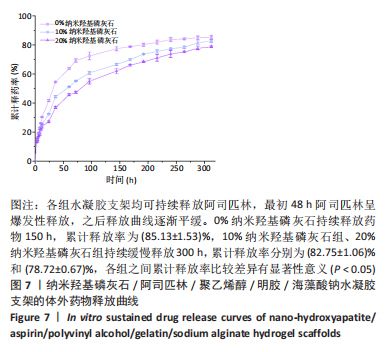
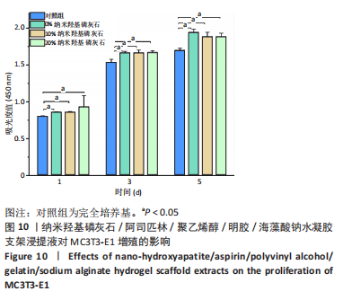
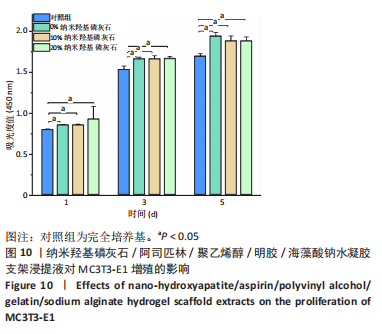
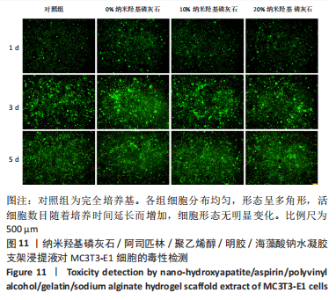
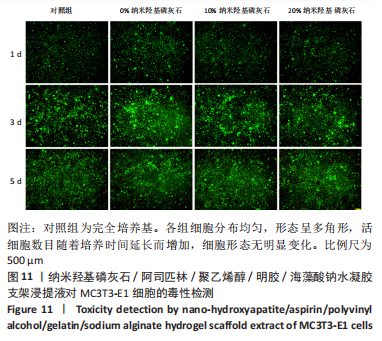
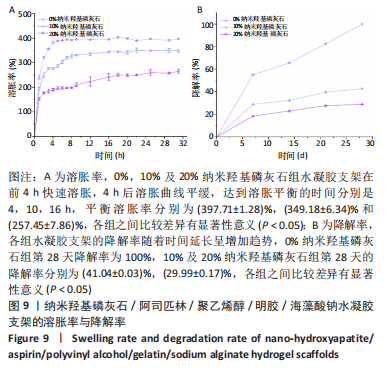
2.7 各组水凝胶支架的溶胀性能分析 0%,10%,20%纳米羟基磷灰石组水凝胶支架在前4 h快速溶胀,4 h后溶胀曲线平缓,分别于4,10,16 h达到溶胀平衡,平衡溶胀率分别为(397.71±1.28)%,(349.18±6.34)%和(257.45±7.86)%,各组之间比较差异有显著性意义(P < 0.05),见图9A。随着纳米羟基磷灰石质量分数的增高,溶胀率依次降低,但各组支架均具有良好的溶胀性能。 2.8 各组水凝胶支架的降解性能分析 各组水凝胶支架的降解率随着时间延长,呈增加趋势,0%纳米羟基磷灰石组水凝胶支架第21天的降解率为(82.33±0.77)%,第28天支架结构崩解,降解率为100%;加入纳米羟基磷灰石后水凝胶支架降解速率显著下降,10%,20%纳米羟基磷灰石组第28天的降解率分别为(41.04±0.03)%,(29.99±0.17)%,各组之间比较差异有显著性意义(P < 0.05),见图9B。结果表明,水凝胶支架加入纳米羟基磷灰石可显著降低降解速率,并与骨组织再生速度相匹配。"
| [1] ESPOSITO M, GRUSOVIN MG, WORTHINGTON HV, et al. Interventions for replacing missing teeth: bone augmentation techniques for dental implant treatment. Cochrane Database Syst Rev. 2006;25(1):CD003607. [2] SCHMITZ JP, HOLLINGER JO. The critical size defect as an experimental model for craniomandibulofacial nonunions. Clin Orthop Relat Res. 1986;(205):299-308. [3] HEGDE R, PRASAD K, SHROFF K. Maxillary sinus augmentation using sinus membrane elevation without grafts - A Systematic Review. J Indian Prosthodont Soc. 2016;16(4):317-322. [4] OGUNDIPE OK, GBOLAHAN OO. Immediate iliac bone graft reconstruction of post ablative defect of benign mandibular pathology - a systematic review. Plast Aesthet Res. 2017;4(8):137-143. [5] ELAKKIYA S, RAMESH AS, PRABHU K. Systematic analysis on the efficacy of bone enhancement methods used for success in dental implants. J Indian Prosthodont Soc. 2017;17(3):219-225. [6] MOUNIR M, MOUNIR S, ABOU-ELFETOUH A, et al. Assessment of vertical ridge augmentation in anterior aesthetic zone using onlay xenografts with titanium mesh versus the inlay bone grafting technique: A randomized clinical trial. Int J Oral Maxillofac Surg. 2017;46(11): 1458-1465. [7] ELNAYEF B, MONJE A, GARGALLO-ALBIOL J, et al. Vertical Ridge Augmentation in the Atrophic Mandible: A Systematic Review and Meta-Analysis. Int J Oral Maxillofac Implants. 2017;32(2):291-312. [8] 邢飞,吴岐佑,者漫,等.无机生物材料在骨修复中的骨免疫调节作用[J].中国修复重建外科杂志,2022,36(4):517-522. [9] 娄静扬,耿欣荣,高慧萌,等.含铜钛合金调控巨噬细胞极化的研究进展[J].口腔疾病防治,2022,30(5):377-380. [10] MA A, YOU Y, CHEN B, et al. Icariin/Aspirin Composite Coating on TiO2 Nanotubes Surface Induce Immunomodulatory Effect of Macrophage and Improve Osteoblast Activity. Coatings. 2020;10(4):427. [11] FANG X, LEI L, JIANG T, et al. Injectable thermosensitive alginate/β-tricalcium phosphate/aspirin hydrogels for bone augmentation. J Biomed Mater Res Part B Appl Biomater. 2018;106(5):1739-1751. [12] ZHANG Y, DING N, ZHANG T, et al. A Tetra-PEG Hydrogel Based Aspirin Sustained Release System Exerts Beneficial Effects on Periodontal Ligament Stem Cells Mediated Bone Regeneration. Front Chem. 2019; 7:682. [13] ZHANG X, MIAO F, NIU L, et al. Berberine carried gelatin/sodium alginate hydrogels with antibacterial and EDTA-induced detachment performances. Int J Biol Macromol. 2021;181:1039-1046. [14] KIM B, BAN E, KIM A. Gelatin-Alginate Coacervates Optimized by DOE to Improve Delivery of bFGF for Wound Healing. Pharmaceutics. 2021;13(12):2112. [15] WANG X, LI S, YU H, et al. The Biocompatibility of Multi-Source Stem Cells and Gelatin-Carboxymethyl Chitosan-Sodium Alginate Hybrid Biomaterials. Tissue Eng Regen Med. 2022;19(3):491-503. [16] AMR M, DYKES I, COUNTS M, et al. 3D printed, mechanically tunable, composite sodium alginate, gelatin and Gum Arabic (SA-GEL-GA) scaffolds. Bioprinting. 2021;22:e00133. [17] CHARRON PN, JACOBS JI, YAO SX, et al. Effects of cryo-processing on the mechanical and biological properties of poly(vinyl alcohol)-gelatin theta-gels. Biointerphases. 2020;15(5): 051004. [18] 徐冉,陈星羽,李志强.羟基磷灰石复合支架负载抗菌性药物:药物与支架的相互作用机制[J].中国组织工程研究,2022,26(27): 4411-4416. [19] CHOCHOLATA P, KULDA V, DVORAKOVA J, et al. Biological Evaluation of Polyvinyl Alcohol Hydrogels Enriched by Hyaluronic Acid and Hydroxyapatite. Int J Mol Sci. 2020;21(16):5719. [20] YANG L, YANG J, QIN X, et al. Ternary composite films with simultaneously enhanced strength and ductility: Effects of sodium alginate-gelatin weight ratio and graphene oxide content. Int J Biol Macromol. 2020;156:494-503. [21] ANAMIZU M, TABATA Y. Design of injectable hydrogels of gelatin and alginate with ferric ions for cell transplantation. Acta Biomater. 2019; 100:184-190. [22] 闫慧敏,杨光,杨波,等.明胶/海藻酸钠/沙蒿胶复合水凝胶的制备及表征[J].工业微生物,2022,52(1):24-33. [23] YUAN L, WU Y, GU QS, et al. Injectable photo crosslinked enhanced double-network hydrogels from modified sodium alginate and gelatin. Int J Biol Macromol. 2017;96:569-577. [24] GONZALEZ JS, ALVAREZ VA. Mechanical properties of polyvinylalcohol/hydroxyapatite cryogel as potential artificial cartilage. J Mech Behav Biomed Mater. 2014;34:47-56. [25] XU M, QIN M, ZHANG X, et al. Porous PVA/SA/HA hydrogels fabricated by dual-crosslinking method for bone tissue engineering. J Biomater Sci Polym Ed. 2020;31(6):816-831. [26] KOUSER R, VASHIST A, ZAFARYAB M, et al. Biocompatible and mechanically robust nanocomposite hydrogels for potential applications in tissue engineering. Mater Sci Eng C. 2018;84:168-179. [27] GARNICA-PALAFOX IM, SANCHEZ-AREVALO FM, VELASQUILLO C, et al. Mechanical and structural response of a hybrid hydrogel based on chitosan and poly(vinyl alcohol) cross-linked with epichlorohydrin for potential use in tissue engineering. J Biomater Sci Polym Ed. 2014; 25(1):32-50. [28] 张杰,张元晶,丁玉琴,等.生物医用高分子材料研究热点[J].高分子材料科学与工程,2021,37(9):182-190. [29] YOSHIKAWA T, NAKAJIMA H, UEMURA T, et al. In vitro bone formation induced by immunosuppressive agent tacrolimus hydrate (FK506). TISSUE Eng. 2005;11(3-4):609-617. [30] PEREZ RA, MESTRES G. Role of pore size and morphology in musculo-skeletal tissue regeneration. Mater Sci Eng C. 2016;61:922-939. [31] Vo TTN, Morgan S, McCormick C, et al. Modelling drug release from polymer-free coronary stents with microporous surfaces. Int J Pharm. 2018;544(2):392-401. [32] MAGNUSSEN RA, GUILAK F, VAIL TP. Cartilage degeneration in post-collapse cases of osteonecrosis of the human femoral head: Altered mechanical properties in tension, compression, and shear. J Orthop Res. 2005;23(3):576-583. [33] BOBBERT F, LIETAERT K, EFTEKHARI AA, et al. Additively manufactured metallic porous biomaterials based on minimal surfaces: A unique combination of topological, mechanical, and mass transport properties. Acta Biomater. 2017;53:572-584. [34] 高泽,石志良,李锋,等.材料和孔隙率对可降解支架内骨形成的影响[J].医用生物力学,2021,36(4):582-588. [35] MA P, WU W, WEI Y, et al. Biomimetic gelatin/chitosan/polyvinyl alcohol/nano-hydroxyapatite scaffolds for bone tissue engineering. Mater Des. 2021;207:109865. [36] 任泽宇,姜宏,石永芳.3D打印PLLA/HA复合组织工程骨支架及性能表征[J].燕山大学学报,2022,46(2):116-122. [37] 杨怡天,王璐,姚蔚,等.生物支架与巨噬细胞在骨再生中的相互影响及应用[J].中国组织工程研究,2023,27(7):1071-1079. [38] REEVES AR, SPILLER KL, FREYTES DO, et al. Controlled release of cytokines using silk-biomaterials for macrophage polarization. Biomaterials. 2015;73:272-283. [39] LECH M, ANDERS H. Macrophages and fibrosis: How resident and infiltrating mononuclear phagocytes orchestrate all phases of tissue injury and repair. Biochim Biophys Acta Mol Basis Dis. 2013;1832(7): 989-997. [40] LIU Y, FANG S, LI X, et al. Aspirin inhibits LPS-induced macrophage activation via the NF-κB pathway. Sci Rep. 2017;7(1):11549. [41] ZHANG Y, DOU X, ZHANG L, et al. Facile fabrication of a biocompatible composite gel with sustained release of aspirin for bone regeneration. Bioact Mater. 2022;11:130-139. [42] LI Y, LI Q, LI H, et al. An effective dual‐factor modified 3D‐printed PCL scaffold for bone defect repair. J Biomed Mater Res B Appl Biomater. 2020;108(5):2167-2179. [43] 陈甜甜,王聪,刘婉嫕,等.pH响应性纤维素纳米纤丝/海藻酸钠基水凝胶的制备及其释药性研究[J]. 中国造纸学报,2022,37(1): 16-21. [44] ZHANG J, WANG Q, WANG A. In situ generation of sodium alginate/hydroxyapatite nanocomposite beads as drug-controlled release matrices. Acta Biomater. 2010;6(2):445-454. [45] BAJPAI AK, SAINI R. Preparation and characterization of biocompatible spongy cryogels of poly(vinyl alcohol)-gelatin and study of water sorption behaviour. Polym Int. 2005;54(9):1233-1242. [46] BÖLGEN N, PLIEVA F, GALAEV IY, et al. Cryogelation for preparation of novel biodegradable tissue-engineering scaffolds. J Biomater Sci Polym Ed. 2007;18(9):1165-1179. [47] ZOU Z, WANG L, ZHOU Z, et al. Simultaneous incorporation of PTH(1–34) and nano-hydroxyapatite into Chitosan/Alginate Hydrogels for efficient bone regeneration. Bioact Mater. 2021;6(6):1839-1851. |
| [1] | Lai Pengyu, Liang Ran, Shen Shan. Tissue engineering technology for repairing temporomandibular joint: problems and challenges [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(在线): 1-9. |
| [2] | Cao Yue, Ye Xinjian, Li Biyao, Zhang Yining, Feng Jianying. Effect of extracellular vesicles for diagnosis and therapy of oral squamous cell carcinoma [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1523-1530. |
| [3] | Sun Yuting, Wu Jiayuan, Zhang Jian. Physical factors and action mechanisms affecting osteogenic/odontogenic differentiation of dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1531-1540. |
| [4] | Li Jialin, Zhang Yaodong, Lou Yanru, Yu Yang, Yang Rui. Molecular mechanisms underlying role of mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1512-1522. |
| [5] | Ding Zhili, Huang Jie, Jiang Qiang, Li Tusheng, Liu Jiang, Ding Yu. Constructing rabbit intervertebral disc degeneration models by different methods under X-ray guidance: a comparative study [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(5): 995-1002. |
| [6] | Wu Guangtao, Qin Gang, He Kaiyi, Fan Yidong, Li Weicai, Zhu Baogang, Cao Ying . Causal relationship between immune cells and knee osteoarthritis: a two-sample bi-directional Mendelian randomization analysis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(5): 1081-1090. |
| [7] | Liu Yani, Yang Jinghuan, Lu Huihui, Yi Yufang, Li Zhixiang, Ou Yangfu, Wu Jingli, Wei Bing . Screening of biomarkers for fibromyalgia syndrome and analysis of immune infiltration [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(5): 1091-1100. |
| [8] | Li Shuai, Liu Hua, Shang Yonghui, Liu Yicong, Zhao Qihang, Liu Wen. Stress distribution on the maxilla when wearing the Twin-block appliance for Class II malocclusion [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(5): 881-887. |
| [9] | Yu Shuangqi, Ding Fan, Wan Song, Chen Wei, Zhang Xuejun, Chen Dong, Li Qiang, Lin Zuoli. Effects of polylactic acid-glycolic acid copolymer/lysine-grafted graphene oxide nanoparticle composite scaffolds on osteogenic differentiation of MC3T3 cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 707-712. |
| [10] | Dang Xiaowen, Huang Hailiang, Huang Lei, Wang Yajie . Research frontiers and hotspots of carbon nanomaterials in biomedical field over the past 10 years [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 752-760. |
| [11] | Wang Sifan, He Huiyu, Yang Quan, Han Xiangzhen. miRNA-378a overexpression of macrophage cell line composite collagen sponge: anti-inflammation and tissue repair promotion [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 789-799. |
| [12] | Xiao Fang, Huang Lei, Wang Lin. Magnetic nanomaterials and magnetic field effects accelerate bone injury repair [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 827-838. |
| [13] | Li Mingzhe, Ye Xiangling, Wang Bing, Yu Xiang. Preparation and osteogenic properties of liquid crystal display light-cured polylactic acid scaffold loaded with nano-tantalum [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 670-677. |
| [14] | Wu Ao, Yu Peng, Teng Jiawen, Kong Peng, Bian Sishan. Analysis of oxidative stress-related genes and immune infiltration in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(2): 302-311. |
| [15] | Tian Jiayu, Li Duohua, Zhang Feng, Feng Hu, Sun Wei. Construction and performance evaluation of polycaprolactone/nanodiamond-phospholipid composite materials [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(16): 3380-3387. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||