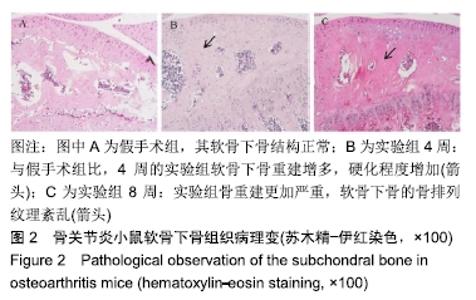
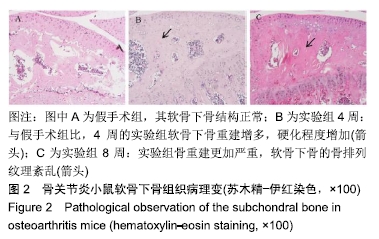
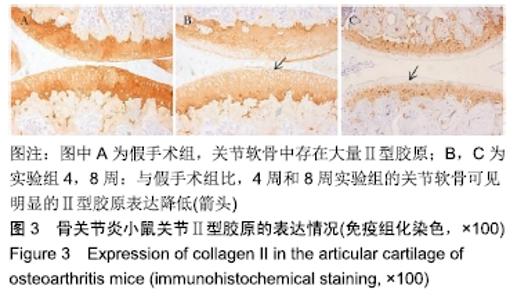
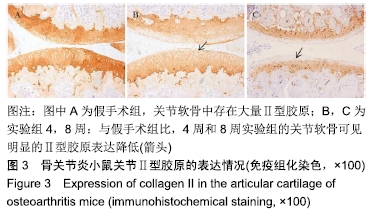
Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (17): 2642-2647.doi: 10.3969/j.issn.2095-4344.2673
Previous Articles Next Articles
Expression and role of retinoblastoma RB1-inducible coiled-coil 1 in the subchondral bone of osteoarthritis
Li Junyan, Fang Hang, Feng Xiaofeng, Cai Daozhang
- The Third Affiliated Hospital of Southern Medical University, Guangzhou 510630, Guangdong Province, China
-
Received:2019-09-18Revised:2019-09-21Accepted:2019-11-04Online:2020-06-18Published:2020-03-28 -
Contact:Cai Daozhang, MD, Chief physician, the Third Affiliated Hospital of Southern Medical University, Guangzhou 510630, Guangdong Province, China -
About author:Li Junyan, Master candidate, the Third Affiliated Hospital of Southern Medical University, Guangzhou 510630, Guangdong Province, China -
Supported by:the National Natural Science Foundation of China, No. 81371990 and 81601945
CLC Number:
Cite this article
Li Junyan, Fang Hang, Feng Xiaofeng, Cai Daozhang. Expression and role of retinoblastoma RB1-inducible coiled-coil 1 in the subchondral bone of osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(17): 2642-2647.
share this article
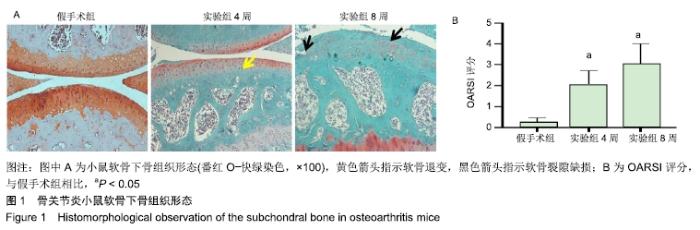
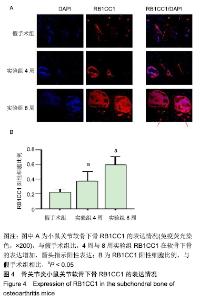
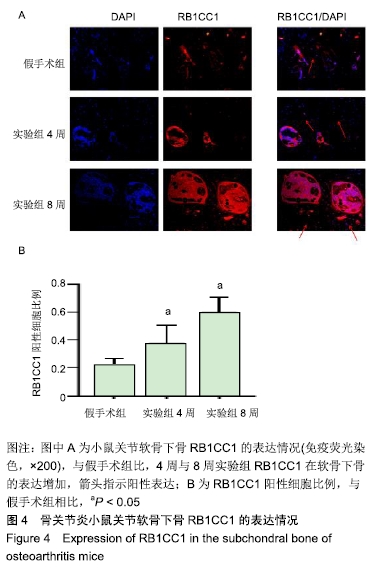
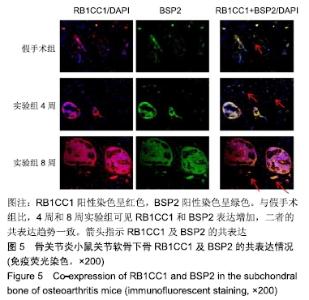
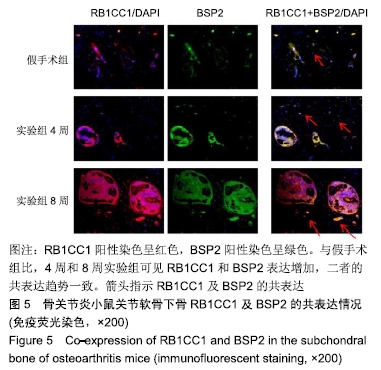
1.4.2 小鼠骨关节炎模型建立 实验组小鼠行右侧膝关节内侧半月板胫骨韧带切除,游离内侧半月板。术前小鼠自由摄取食水,每周更换垫料,造模时间在小鼠10周龄、骨骼肌肉发育成熟后进行。体积分数1%戊巴比妥钠(8 μL/g)进行腹腔麻醉,麻醉起效后将小鼠右膝关节周围的鼠毛剔除,并用棉球蘸取碘伏进行消毒。随后在解剖显微镜下将小鼠右膝关节处皮肤切开,沿髌骨韧带内侧纵行切开肌肉,将其外翻后钝性分离肌肉等组织,暴露胫骨平台与内侧半月板胫骨韧带。将内侧半月板胫骨韧带切断,游离内侧半月板,造成膝关节不稳定以诱导骨关节炎[14],最后生理盐水冲洗,并将外翻的髌骨韧带复位,缝合切口[14-15]。麻醉苏醒后,将小鼠放回原笼中继续饲养,勤换垫料与观察伤口情况,小鼠自由摄取食水、可自由活动。假手术组仅切开关节囊。 1.4.3 组织取材 实验组分别在术后4周与8周进行取材,假手术组在第8周取材,将小鼠断颈处死后取其右侧膝关节,将皮肤肌肉大致剔除,用40 g/L多聚甲醛固定组织标本,放于4 ℃冰箱。第2天用去离子水冲洗后置于摇床上进行脱钙,持续4周左右,脱钙液隔天更换一次。脱钙完成后将标本放入包埋盒并标记分组,用流水冲洗过夜,洗净脱钙液。第2天进行脱水,脱水完成后取出标本,将关节的内侧面朝金属模具底下进行包埋,期间注意保持关节面的平整,包埋完成后将蜡块放入冰箱冻存。 1.4.4 组织切片与脱蜡水化 蜡块切片厚度均为4 μm,切片之后将其置于37 ℃恒温箱干燥过夜。脱蜡水化前,先将组织切片置于60 ℃风箱中烤片1.0-2.0 h,随后在二甲苯(2次)和梯度乙醇(体积分数100%,100%,90%,80%,70%,50%)中按顺序进行脱蜡水化,2次二甲苯和2次无水乙醇每次10 min,其余每次5 min。 1.4.5 番红O快速绿染色与骨关节炎OARSI评分 切片脱蜡水化后铁苏木精染色15 s,水洗后吹干。体积分数1%固绿溶液染色1 min,冰醋酸浸泡数秒后吹干,体积分数0.5%番红O染液染色3 min,吹去多余染料,于显微镜下观察颜色深浅是否合适。随后二甲苯透明,过夜风干,隔天用中性树胶封固,显微镜下观察。切片关节组织的结构改变情况参照国际骨关节炎研究协会(Osteoarthritis Research Society International,OARSI)评分标准,其可反映骨关节炎严重程度,评分越高,骨关节炎软骨损伤越严重。 1.4.6 苏木精-伊红染色 切片脱蜡水化后铁苏木精染色15 s,水洗后吹干切片。伊红染液染色30 s,流水洗去多余染液,二甲苯透明后风干过夜,次日用中性树胶封固定,显微镜观察。 1.4.7 免疫荧光 组织切片脱蜡水化后浸泡于枸橼酸,在60 ℃水浴箱中过夜修复,次日PBS洗3次,每次5 min。用山羊血清在室温下封闭1 h后加入RB1CC1(1∶100)与BSP2(1∶100)混合—抗工作液,在4 ℃条件下过夜孵育14-16 h,次日PBS洗3次,每次5 min。避光滴加兔抗羊(594)(1∶500)与鼠抗羊(488)(1∶500)混合荧光二抗,孵育1 h后PBS洗3次,每次5 min,注意避光。最后用DAPI染核封固。在荧光显微镜下,观察切片组织中RB1CC1及BSP2的表达情况。 1.4.8 免疫组化 脱蜡水化与枸橼酸修复步骤同上。加体积分数3%过氧化物酶抑制剂,室温孵育10 min,PBS洗3次,每次5 min。用山羊血清在室温条件下封闭1 h。加入Ⅱ型胶原(1∶100)—抗液在4 ℃条件下过夜孵育14-16 h,次日PBS洗3次,每次5 min。室温下滴加兔抗羊(1∶200)二抗,孵育1 h后PBS洗3次,每次5 min。DAB避光显色,苏木素染核,脱水透明后中性树胶封固,显微镜下观察拍照。 1.5 主要观察指标 ①切片组织学改变与骨关节炎OARSI 评分;②组织中Ⅱ型胶原、RB1CC1及BSP2的表达水平。 1.6 统计学分析 用EXCEL2013(美国微软公司)整理汇总资料,Photoshop CS6软件(美国Adobe Systems公司)处理图片,Graph Pad Prism8软件做图分析(美国Graph Pad公司),SPSS 23.0软件(美国IBM公司)进行统计分析。进行单因素方差分析和多重比较检测评估假手术组与4周及8周实验组的差异,P < 0.05为差异有显著性意义。 "
| [1] CROSS M, SMITH E, HOY D, et al. The global burden of hip and knee osteoarthritis: estimates from the global burden of disease 2010 study. Ann Rheum Dis. 2014;73(7):1323-1330. [2] HUNTER DJ, SCHOFIELD D, CALLANDER E. The individual and socioeconomic impact of osteoarthritis. Nat Rev Rheumatol. 2014;10(7): 437-441. [3] LAMBOVA SN, MÜLLER-LADNER U. Osteoarthritis - Current Insights in Pathogenesis, Diagnosis and Treatment. Curr Rheumatol Rev. 2018; 14(2): 91-97. [4] NELSON AE, ALLEN KD, GOLIGHTLY YM, et al. A systematic review of recommendations and guidelines for the management of osteoarthritis: The chronic osteoarthritis management initiative of the U.S. bone and joint initiative. Semin Arthritis Rheum. 2014;43(6):701-712. [5] MURPHY L, HELMICK CG. The impact of osteoarthritis in the United States: a population-health perspective: A population-based review of the fourth most common cause of hospitalization in U.S. adults. Orthop Nurs. 2012;31(2):85-91. [6] MURAOKA T, HAGINO H, OKANO T, et al. Role of subchondral bone in osteoarthritis development: a comparative study of two strains of guinea pigs with and without spontaneously occurring osteoarthritis. Arthritis Rheum. 2007;56(10):3366-3374. [7] DORE DA. The role of subchondral bone in osteoarthritis. Hobart, Australia: University of Tasmania. 2011. [8] MARUOTTI N, CORRADO A, CANTATORE FP. Osteoblast role in osteoarthritis pathogenesis. J Cell Physiol. 2017;232(11):2957-2963. [9] OCHI Y, CHANO T, IKEBUCHI K, et al. RB1CC1 activates the p16 promoter through the interaction with hSNF5. Oncol Rep. 2011;26(4):805-812. [10] LI L, WANG G, HU JS, et al. RB1CC1-enhanced autophagy facilitates PSCs activation and pancreatic fibrogenesis in chronic pancreatitis. Cell Death Dis. 2018;9(10):952. [11] LI S, QIANG Q, SHAN H, et al. MiR-20a and miR-20b negatively regulate autophagy by targeting RB1CC1/FIP200 in breast cancer cells. Life Sci. 2016;147:143-152. [12] CARAMÉS B, HASEGAWA A, TANIGUCHI N, et al. Autophagy activation by rapamycin reduces severity of experimental osteoarthritis. Ann Rheum Dis. 2012;71(4):575-581. [13] ALMONTE-BECERRIL M, NAVARRO-GARCIA F, GONZALEZ-ROBLES A, et al. Cell death of chondrocytes is a combination between apoptosis and autophagy during the pathogenesis of Osteoarthritis within an experimental model. Apoptosis. 2010;15(5):631-638. [14] FANG H, BEIER F. Mouse models of osteoarthritis: modelling risk factors and assessing outcomes. Nat Rev Rheumatol. 2014;10(7):413-421. [15] GLASSON SS, ASKEW R, SHEPPARD B, et al. Characterization of and osteoarthritis susceptibility in ADAMTS-4-knockout mice. Arthritis Rheum. 2004;50(8):2547-2558. [16] VON DER MARK K, KIRSCH T, NERLICH A, et al. Type X collagen synthesis in human osteoarthritic cartilage. Indication of chondrocyte hypertrophy. Arthritis Rheum. 1992;35(7):806-811. [17] KOBAYASHI M, SQUIRES GR, MOUSA A, et al. Role of interleukin-1 and tumor necrosis factor alpha in matrix degradation of human osteoarthritic cartilage. Arthritis Rheum. 2005;52(1):128-135. [18] CHOI MC, MARUYAMA T, CHUN CH, et al. Alleviation of Murine Osteoarthritis by Cartilage-Specific Deletion of IκBζ. Arthritis Rheumatol. 2018;70(9):1440-1449. [19] BURR DB. Increased biological activity of subchondral mineralized tissues underlies the progressive deterioration of articular cartilage in osteoarthritis. J Rheumatol. 2005;32(6):1156-1159. [20] LI G, YIN J, GAO J, et al. Subchondral bone in osteoarthritis: insight into risk factors and microstructural changes. Arthritis Res Ther. 2013;15(6): 223. [21] FUNCK-BRENTANO T, COHEN-SOLAL M. Subchondral bone and osteoarthritis. Curr Opin Rheumatol. 2015;27(4):420-426. [22] RADIN EL, PAUL IL, ROSE RM. Role of mechanical factors in pathogenesis of primary osteoarthritis. Lancet. 1972;1(7749):519-522. [23] CHAN PMB, WEN C, YANG WC, et al. Is subchondral bone cyst formation in non-load-bearing region of osteoarthritic knee a vascular problem?. Med Hypotheses. 2017;109:80-83. [24] LIN C, SHAO Y, ZENG C, et al. Blocking PI3K/AKT signaling inhibits bone sclerosis in subchondral bone and attenuates post-traumatic osteoarthritis [published correction appears in J Cell Physiol. 2019 Jun;234(6):9873]. J Cell Physiol. 2018;233(8):6135-6147. [25] FANG H, HUANG L, WELCH I, et al. Early Changes of Articular Cartilage and Subchondral Bone in The DMM Mouse Model of Osteoarthritis. Sci Rep. 2018;8(1):2855. [26] XUE J, XUE J, ZHANG J, et al. miR-130b-3p/301b-3p negatively regulated Rb1cc1 expression on myogenic differentiation of chicken primary myoblasts. Biotechnol Lett. 2017;39(11):1611-1619. [27] CHEN J, YAN L, WANG H, et al. ZBTB38, a novel regulator of autophagy initiation targeted by RB1CC1/FIP200 in spinal cord injury. Gene. 2018; 678:8-16. [28] CORONA VELAZQUEZ AF, JACKSON WT. So Many Roads: the Multifaceted Regulation of Autophagy Induction. Mol Cell Biol. 2018;38(21): e00303-18. [29] NISHIMURA I, CHANO T, KITA H, et al. RB1CC1 protein suppresses type II collagen synthesis in chondrocytes and causes dwarfism. J Biol Chem. 2011;286(51):43925-43932. [30] BOULEFTOUR W, GRANITO RN, VANDEN-BOSSCHE A, et al. Bone Shaft Revascularization After Marrow Ablation Is Dramatically Accelerated in BSP-/- Mice, Along With Faster Hematopoietic Recolonization. J Cell Physiol. 2017;232(9):2528-2537. [31] BOULEFTOUR W, JUIGNET L, VERDIÈRE L, et al. Deletion of OPN in BSP knockout mice does not correct bone hypomineralization but results in high bone turnover. Bone. 2019;120:411-422. [32] LI Z, WANG L, WEI J, et al. Bone-strengthening pill (BSP) promotes bone cell and chondrocyte repair, and the clinical and experimental study of BSP in the treatment of osteonecrosis of the femoral head. Oncotarget. 2017;8(57):97079-97089. [33] BOLAMPERTI S, VILLA I, SPINELLO A, et al. Evidence for Altered Canonical Wnt Signaling in the Trabecular Bone of Elderly Postmenopausal Women with Fragility Femoral Fracture. Biomed Res Int. 2016;2016:8169614. [34] SHAH M, GBURCIK V, REILLY P, et al. Local origins impart conserved bone type-related differences in human osteoblast behaviour. Eur Cell Mater. 2015;29:155-176. [35] PESESSE L, SANCHEZ C, WALSH DA, et al. Bone sialoprotein as a potential key factor implicated in the pathophysiology of osteoarthritis. Osteoarthritis Cartilage. 2014;22(4):547-556. [36] CUI Z, CRANE J, XIE H, et al. Halofuginone attenuates osteoarthritis by inhibition of TGF-β activity and H-type vessel formation in subchondral bone. Ann Rheum Dis. 2016;75(9):1714-1721. [37] IKEBUCHI K, CHANO T, OCHI Y, et al. RB1CC1 activates the promoter and expression of RB1 in human cancer. Int J Cancer. 2009;125(4):861-867. [38] SURANENI MV, MOORE JR, ZHANG D, et al. Tumor-suppressive functions of 15-Lipoxygenase-2 and RB1CC1 in prostate cancer. Cell Cycle. 2014;13(11):1798-1810. [39] LI X, WAN X, CHEN H, et al. Identification of miR-133b and RB1CC1 as independent predictors for biochemical recurrence and potential therapeutic targets for prostate cancer. Clin Cancer Res. 2014;20(9): 2312-2325. [40] LI J, FU Z, JIANG H, et al. IGF2-derived miR-483-3p contributes to macrosomia through regulating trophoblast proliferation by targeting RB1CC1. Mol Hum Reprod. 2018;24(9):444-452. [41] CORONA VELAZQUEZ A, CORONA AK, KLEIN KA, et al. Poliovirus induces autophagic signaling independent of the ULK1 complex. Autophagy. 2018;14(7):1201-1213. [42] GRUNWALD DS, OTTO NM, PARK JM, et al. GABARAPs and LC3s have opposite roles in regulating ULK1 for autophagy induction. Autophagy. 2019. doi:10.1080/15548627.2019.1632620. [43] MORSELLI E, SHEN S, RUCKENSTUHL C, et al. p53 inhibits autophagy by interacting with the human ortholog of yeast Atg17, RB1CC1/FIP200. Cell Cycle. 2011;10(16):2763-2769. [44] GUO M, MU Y, YU D, et al. Comparison of the expression of TGF-β1, E-cadherin, N-cadherin, TP53, RB1CC1 and HIF-1α in oral squamous cell carcinoma and lymph node metastases of humans and mice. Oncol Lett. 2018;15(2):1639-1645. [45] YAO J, JIA L, KHAN N, et al. Deletion of autophagy inducer RB1CC1 results in degeneration of the retinal pigment epithelium. Autophagy. 2015;11(6):939-953. [46] ZHANG Y, VASHEGHANI F, LI YH, et al. Cartilage-specific deletion of mTOR upregulates autophagy and protects mice from osteoarthritis. Ann Rheum Dis. 2015;74(7):1432-1440. [47] CARAMÉS B, TANIGUCHI N, OTSUKI S, et al. Autophagy is a protective mechanism in normal cartilage, and its aging-related loss is linked with cell death and osteoarthritis. Arthritis Rheum. 2010;62(3): 791-801. [48] ZHU X, YANG S, LIN W, et al. Roles of Cell Cyle Regulators Cyclin D1, CDK4, and p53 in Knee Osteoarthritis. Genet Test Mol Biomarkers. 2016;20(9):529-534. [49] ZHONG G, LONG H, MA S, et al. miRNA-335-5p relieves chondrocyte inflammation by activating autophagy in osteoarthritis. Life Sci. 2019; 226:164-172. [50] CHENG NT, MENG H, MA LF, et al. Role of autophagy in the progression of osteoarthritis: The autophagy inhibitor, 3-methyladenine, aggravates the severity of experimental osteoarthritis. Int J Mol Med. 2017;39(5): 1224-1232. [51] XUE JF, SHI ZM, ZOU J, et al. Inhibition of PI3K/AKT/mTOR signaling pathway promotes autophagy of articular chondrocytes and attenuates inflammatory response in rats with osteoarthritis. Biomed Pharmacother. 2017;89:1252-1261. |
| [1] | Peng Zhihao, Feng Zongquan, Zou Yonggen, Niu Guoqing, Wu Feng. Relationship of lower limb force line and the progression of lateral compartment arthritis after unicompartmental knee arthroplasty with mobile bearing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1368-1374. |
| [2] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Comparison of the short-term efficacy of extracorporeal shock wave therapy for middle-aged and elderly knee osteoarthritis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1471-1476. |
| [3] | Chen Jiming, Wu Xiaojing, Liu Tianfeng, Chen Haicong, Huang Chengshuo. Effects of silymarin on liver injury and bone metabolism induced by carbon tetrachloride in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1224-1228. |
| [4] | Liu Xiangxiang, Huang Yunmei, Chen Wenlie, Lin Ruhui, Lu Xiaodong, Li Zuanfang, Xu Yaye, Huang Meiya, Li Xihai. Ultrastructural changes of the white zone cells of the meniscus in a rat model of early osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1237-1242. |
| [5] | Liu Cong, Liu Su. Molecular mechanism of miR-17-5p regulation of hypoxia inducible factor-1α mediated adipocyte differentiation and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1069-1074. |
| [6] | Cao Xuhan, Bai Zixing, Sun Chengyi, Yang Yanjun, Sun Weidong. Mechanism of “Ruxiang-Moyao” herbal pair in the treatment of knee osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 746-753. |
| [7] | Li Yonghua, Feng Qiang, Tan Renting, Huang Shifu, Qiu Jinlong, Yin Heng. Molecular mechanism of Eucommia ulmoides active ingredients treating synovitis of knee osteoarthritis: an analysis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 765-771. |
| [8] | Song Shan, Hu Fangyuan, Qiao Jun, Wang Jia, Zhang Shengxiao, Li Xiaofeng. An insight into biomarkers of osteoarthritis synovium based on bioinformatics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 785-790. |
| [9] | Deng Zhenhan, Huang Yong, Xiao Lulu, Chen Yulin, Zhu Weimin, Lu Wei, Wang Daping. Role and application of bone morphogenetic proteins in articular cartilage regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 798-806. |
| [10] | Zheng Xiaolong, He Xiaoming, Gong Shuidi, Pang Fengxiang, Yang Fan, He Wei, Liu Shaojun, Wei Qiushi. Bone turnover characteristics in patients with alcohol-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 657-661. |
| [11] | Liu Xin, Yan Feihua, Hong Kunhao. Delaying cartilage degeneration by regulating the expression of aquaporins in rats with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 668-673. |
| [12] | Ma Zetao, Zeng Hui, Wang Deli, Weng Jian, Feng Song. MicroRNA-138-5p regulates chondrocyte proliferation and autophagy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 674-678. |
| [13] | Chen Junyi, Wang Ning, Peng Chengfei, Zhu Lunjing, Duan Jiangtao, Wang Ye, Bei Chaoyong. Decalcified bone matrix and lentivirus-mediated silencing of P75 neurotrophin receptor transfected bone marrow mesenchymal stem cells to construct tissue-engineered bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 510-515. |
| [14] | Ma Zhijie, Li Jingyu, Cao Fang, Liu Rong, Zhao Dewei. Influencing factors and biological property of novel biomedical materials: porous silicon carbide coated with bioactive tantalum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 558-563. |
| [15] | Shi Xiaoxiu, Mao Shilong, Liu Yang, Ma Xingshuang, Luo Yanfeng. Comparison of tantalum and titanium (alloy) as orthopedic materials: physical and chemical indexes, antibacterial and osteogenic ability [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 593-599. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||