Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (30): 4856-4861.doi: 10.12307/2023.806
Previous Articles Next Articles
Application of decellularized extracellular matrix composite scaffolds in tissue regeneration
Yang Shun, Zhao Mingyue, Tu Xiling, Gao Li, Yang Kun, Liu Qi
- Department of Periodontology, Stomatological Hospital Affiliated to Zunyi Medical University, Zunyi 563000, Guizhou Province, China
-
Received:2022-10-08Accepted:2022-11-21Online:2023-10-28Published:2023-04-03 -
Contact:Liu Qi, MD, Professor, Department of Periodontology, Stomatological Hospital Affiliated to Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
About author:Yang Shun, Master candidate, Department of Periodontology, Stomatological Hospital Affiliated to Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
Supported by:National Natural Science Foundation of China, No. 82060204 (to GL); Science and Technology Department of Guizhou Province, No. ZK[2021] 437 (to GL)
CLC Number:
Cite this article
Yang Shun, Zhao Mingyue, Tu Xiling, Gao Li, Yang Kun, Liu Qi. Application of decellularized extracellular matrix composite scaffolds in tissue regeneration[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(30): 4856-4861.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1 脱细胞基质支架制备方法 脱细胞是指在保护细胞外基质成分和结构完整性时,去除组织或器官中的细胞及其成分,特别是DNA和RNA[10]。脱细胞因组织来源、厚度、脂质含量、细胞数量和细胞密度等差异,支架制备方法各不相同[11],主要方法有化学(洗涤剂、酸/碱溶液、高渗/低渗溶液、醇、螯合物等)、物理(冻融法、 机械搅拌法、压力法、超临界流体法等)和生物(核酸酶、蛋白酶等),详见表1。其中化学方法可以显著去除细胞成分,但它们不可避免地破坏细胞外基质的超微结构,并且残留的化学试剂毒性较高。相反,物理方法对组织微观结构的破坏性较小、毒性低,但细胞不能完全被去除,破坏了细胞外基质结构。生物方法减少了对其他生物活性成分的损害,清除残留细胞和抗原,但因需要更长的处理时间而残留酶试剂,会引起免疫反应。近年来,为了更好地去除组织细胞及减少对细胞外基质的损伤,常联合两种或两种以上的方法对组织进行脱细胞处理,但同样不可避免对脱细胞基质支架造成损伤[40]。"

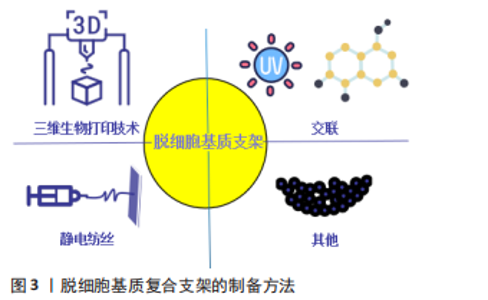
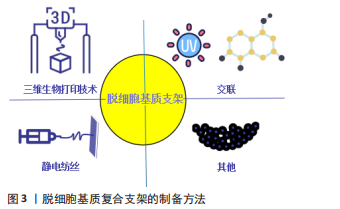
为了更好地发挥脱细胞基质作为天然组织工程支架的优势,同时降低因脱细胞过程而导致的力学性能、结构和生物学性能下降等不利因素,脱细胞基质支架后续的优化处理,如交联、静电纺丝、3D打印等技术显得尤为重要。研究表明,京尼平交联的猪肝脱细胞基质机械性能和促血管生成特性明显提升,并降低了大鼠体内炎症反应[41]。据文献报道,通过静电纺丝技术在脱细胞血管支架外覆以纳米氨基醇聚癸二酸甘油酯,使用肝素修饰血管支架内膜,制成复合型组织工程血管,结果显示脱细胞血管支架经纳米氨基醇聚癸二酸甘油酯包覆后增强了复合型组织工程血管支架的机械性能,支架壁厚度、缝合强度、爆破压力较单纯脱细胞血管支架明显增加,植入大鼠体内 6 周后,复合型组织工程血管通畅性良好,无动脉瘤形成[42],表现为机械性能的提升及良好的组织相容性。研究表明,通过3D生物打印技术将聚己内酯、软骨脱细胞基质与甲基丙烯酸酯化明胶混合配制光敏性生物墨水,制备聚己内酯/甲基丙烯酸酯化明胶/细胞外基质支架,该支架的压缩模量约为正常软骨的3倍,无明显的细胞毒性,利于细胞的增值、迁移,表现出良好的机械性能及生物相容性,为精准化的软骨组织再生提供了一种新的思路[43]。 2.2 脱细胞基质复合支架制备方法及在组织再生中的应用 2.2.1 交联 交联是一种广泛应用于支架性能的改良策略,可以提高脱细胞支架的机械性能,改善其生物力学特性等,因此通常使用交联方法(如化学交联、物理交联和天然交联)来改善脱细胞基质支架的性能[44]。 化学交联:化学交联剂如戊二醛、环氧化合物等,因为植入后细胞毒性相对较高,尤其是支架的严重免疫排斥、血液相容性降低和组织钙化,限制了其应用[45-46]。 物理交联:物理交联方法主要包括光氧化交联、热脱氢及紫外线照射,它可以在不引入外源有毒化学品的情况下达到交联目的,然而由于苛刻的交联条件及较低的交联效率,该技术通常用作辅助方法[47-48]。为了提高物理交联效率,研究发现通过核黄素/紫外线交联脱细胞羊膜,交联后杨氏模量(14 MPa)、抗拉强度(4.9 MPa)与未交联羊膜杨氏模量(2.9 MPa)、抗拉强度(1 MPa)相比提升约5倍,同时显示出优异的抗生物降解性能,利于脂肪来源干细胞的增殖和分化[49]。采用同样方法交联脱细胞人胎盘绒毛膜动脉,提升了交联后脱细胞基质支架的机械强度及光滑度,同时降低了对细胞的毒性作用[50]。 天然交联:同与化学交联剂及物理交联相比,天然交联剂是最理想的交联剂,它能显著提升脱细胞支架的机械性能、抗酶能力,具有更小的细胞毒性。最常用的天然交联剂包括京尼平、单宁酸及原花青素[51]。有文献报道,京尼平交联脱细胞肝组织支架植入大鼠体内21 d,结果显示未交联支架完全降解,京尼平和戊二醛交联的支架保存完好、未见降解,未交联支架组观察到明显的淋巴细胞浸润,京尼平交联组表现出相对轻的淋巴细胞和中性粒细胞浸润,比戊二醛交联组更低[52],表明京尼平交联对提升支架降解性能和免疫原性有更好的作用。有研究用京尼平、原花青素及戊二醛分别交联脱细胞子宫,支架的机械强度明显提高,通过胶原蛋白含量模拟脱细胞子宫的降解情况,发现未交联脱细胞子宫12 h内被胶原酶完全消化,而交联脱细胞子宫在同一时间点保留了超过80%的质量,表明交联减缓了降解速率,其中戊二醛交联引起明显的细胞死亡和宿主免疫反应,并伴有多种促炎因子的累积,京尼平和原花青素交联后支架显示出良好的生物相容性,低交联度下更能促进细胞增殖[53]。有研究报道,使用5 g/L京尼平溶液交联大鼠脱细胞脊髓支架,极限牵张应力从0.1 MPa提高到0.2 MPa,弹性模量从0.5 MPa提高到1.5 MPa,体外抗酶解能力显示3 d后未交联支架已完全降解,交联组仅部分降解,对细胞无明显毒性[54],表明京尼平交联对提升大鼠脱细胞脊髓支架的机械性能、减缓降解速率更加有利。虽然天然交联剂在提升支架免疫原性及生物相容性方面效果更佳,但如何确定最佳交联剂浓度未有定论,还需要进行深入研究。 2.2.2 静电纺丝技术 静电纺丝纳米纤维制备方法简单、成本低廉、可根据具体要求定制,且能够模拟细胞外基质,促进细胞的增殖和分化,并具有良好的机械性能、生物相容性及抗降解能力等优点[55],现被广泛与脱细胞基质构成复合支架,较好弥补脱细胞基质支架的不足,是目前组织工程研究的热点之一。 GONG等[56]通过静电纺丝技术将聚己内酯与大鼠脱细胞主动脉结合构成复合支架,结果显示复合支架的极限轴向拉力(2.1 MPa)较大鼠脱细胞主动脉(0.6 MPa)明显升高,提示力学性能增加,且表现出更好的血管形态,无血栓及动脉瘤形成,为更好地利用脱细胞支架在血管组织工程中的应用提供了新的思路。GHORBANI等[57]研究了聚己内酯与脱细胞主动脉构成的复合支架修复兔气管软骨缺损的能力,结果表明聚己内酯提供的支撑显著增强了复合支架的机械强度,植入体内4周后无感染及免疫排斥反应发生,提示复合支架具有良好的生物相容性,且再生的组织与天然软骨组织相仿,为气管软骨组织再生提供了一种新的思路。另有研究将聚L-丙交脂-己内脂/明胶与猪冠状动脉脱细胞支架组成双层血管复合支架,最大载荷值(10.32±1.21) N高于单纯动脉脱细胞支架组(1.95±0.79) N,力学性能提升了约5倍,显示出良好的生物相容性,且再生血管的结构、力学性能与天然血管相似,无扩张及动脉瘤形成[58]。该研究表明复合支架可能在血管再生是一种很有前途的策略,但远期的效果还有待进一步观察。MA等[59]制备了一种薄型聚乳酸-乙醇酸/脱细胞化脊髓复合支架,压缩模量(29.8±5.805) kPa较单纯脱细胞化脊髓支架(3.2±0.768) kPa明显增高,机械性能提高了约10倍,聚乳酸-乙醇酸/脱细胞化脊髓支架对肌成纤维细胞的渗透和致密胶原基质的沉积表现出显著增强的韧性,有利于脊髓损伤后内源性神经干细胞的定向迁移、驻留和神经元分化的神经原生态位;然而单纯脱细胞化脊髓支架因结缔组织浸润导致的塌陷显著损害神经原生态位,阐明了复合支架在脊髓损伤中的作用,有望成为脊髓组织工程中的生物材料。有学者将电纺纳米纤维丝素蛋白和人脱细胞羊膜构成三维支架,最大载荷值(1.90±0.18) N高于单纯人脱细胞羊膜(1.30±0.17) N,降解实验显示单纯人脱细胞羊膜的降解速率更快,10 d后已完全降解,而复合支架在14 d后仍有部分支架材料存留,提示改善了支架的降解性及机械性能;同时,复合支架可促进脂肪来源间充质干细胞血管生成因子的表达,如血管内皮生长因子和碱性成纤维细胞生长因子[60],为脂肪或皮肤组织的再生提供一种新的思路和方法。 2.2.3 三维生物打印技术 三维生物打印技术通过医学成像设备获取数据构建组织工程结构,逐层沉积生物墨水(生物活性材料和活细胞的混合物),可以更加精确、个性化模拟自然组织或器官的三维复杂结构,使三维(3D)生物打印成为制造组织或器官仿生结构中最有前景的技术之一[61]。脱细胞基质支架富含天然的细胞外基质成分及多种生长因子等,具有组织或器官特异性的生化和生理特性,可诱导正常组织或器官的再生或重建,有利于3D生物打印的流变和凝胶特性,近年来,被视为理想的生物墨水在三维(3D)生物打印技术中广泛使用。 有研究表明,将猪肝脱细胞基质加载到明胶混合物所制备的新型生物墨水中,在印刷13层的过程中产生了更一致的孔径和形状,其弹性模量(3.5 kPa)比传统脱细胞基质生物墨水(0.38 kPa)增加了9.17倍,机械性能显著提高,这很好地揭示了新型生物墨水在3D打印层堆叠方面表现出的优异性能,为复杂的肝脏组织再生提供了一种新的思路[62]。另外,YU等[63]利用数字光处理制备可光交联的组织特异性猪源心脏脱细胞基质生物墨水,发现能够对特异性组织复杂微结构的再生和机械性能进行良好控制,可能是因为生物墨水的微观结构对细胞排列和功能的引导作用,这项研究表明,三维生物打印的脱细胞基质生物墨水对精准化心脏组织工程是一项很有前途的策略。JANG等[64]首次使用维生素B2诱导的紫外线交联和热凝胶固化猪源心脏脱细胞基质生物墨水,弹性模量由0.18 kPa提高到15.74 kPa,提升约87倍,且具有与天然心脏组织相当的弹性模量(10-15 kPa),通过三维生物打印可以构建多达10层的稳定结构,利于心脏祖细胞存活,且心肌特异性基因MEF2C、GATA4、NKX2.5显著上调,增加了心肌分化。该研究结果表明,通过三维层层打印的猪源心脏脱细胞基质生物墨水更能模拟心脏祖细胞生存的微环境,促进心脏祖细胞的分化,为更加精准化的心脏组织再生提供了思路。VISSCHER等[65]将猪耳郭软骨脱细胞基质使用甲基丙烯酸化制备成光交联水凝胶,并与软骨细胞一起构成可三维生物打印的生物墨水用于耳郭软骨重建,结果表明,生物墨水打印出解剖型耳郭表现出良好的力学性能和结构完整性,有利于软骨细胞的存活、分化,且产生软骨细胞外基质成分,如胶原和糖胺聚糖。有理由相信,基于此方法的三维生物打印技术,可能为未来个性化耳软骨重建的提供一种新的治疗方法。研究报道,将猪骨骼肌和血管脱细胞基质的生物墨水行同轴喷嘴印刷来模拟血管化肌肉的层次结构,显示出更好的血管化肌肉再生[66],结果表明,肌肉和血管脱细胞基质生物油墨在空间上定位在印刷结构内,可以为肌肉和血管区域的细胞分化提供有利的微环境,这为精准化的功能性肌肉组织再生提供了一种潜在可行性。ZHANG等[67]将丝素蛋白和关节软骨脱细胞基质制备成丝素蛋白-脱细胞基质生物墨水,与骨髓间充质干细胞混合用于3D生物打印,结果表明具有更加精准的外形、与软组织相当的压缩模量,利于骨髓间充质干细胞的成活及分化,软骨特异性标志基因SOX-9、Ⅱ型胶原、蛋白聚糖、Ⅰ型胶原在14,28 d显著上调,为更加精准化的软骨组织再生提供了一种新的策略。有研究表明,将猪肝脱细胞基质与明胶、聚乙二醇混合后,进一步用酪氨酸酶交联制备新型生物墨水,相较于非交联猪肝脱细胞基质生物墨水,新型生物墨水储存模量增加了32倍,通过三维打印出了更加精准、稳定的结构,并保持较高细胞活力(85%-93%),肝特征性标志基因甲胎蛋白、白蛋白、KRT19、MK167显著上调[68],这为更好发挥脱细胞基质在精准化肝脏组织再生中的优势,提供了一种潜在可行性。 2.2.4 其他 纳米颗粒:具有独特的物理化学特性和多功能性,常用来修饰脱细胞基质支架,提高机械强度、结构稳定性及抗降解等性能[69]。有研究表明,将银纳米颗粒修饰脱细胞大鼠食管支架后,抗降解、压痕及钙化能力增强,降低了支架的免疫原性,且促进血管生成及提高了支架的生物相容性[70]。PESARAKLOU等[71]用氧化铈纳米颗粒修饰人皮肤脱细胞基质支架,不仅对细胞没有毒性,还提高了细胞存活率,并且自由基消除能力、胶原蛋白含量及抗拉伸强度也明显改善。 甲氧基聚乙二醇:是一种有效、无毒、无免疫原性的细胞表面免疫伪装材料,可与细胞表面的氨基酸残基共价结合,通过甲氧基聚乙二醇诱导的电荷和空间伪装减少或防止宿主免疫排斥反应[72-73]。LIU等[74]采用甲氧基聚乙二醇修饰脱细胞脂肪基质,显示出更低的免疫原性及更好的脂肪组织再生,其机制可能与甲氧基聚乙二醇修饰后Treg细胞计数增加,进而通过分泌白细胞介素10、白细胞介素13和转化生长因子β1提高了M2/M1巨噬细胞比率,但还需进一步深入研究。 生长因子:能够通过与特定细胞表面受体的相互作用,促进细胞分化、介导缺损组织的修复,在组织工程学中发挥重要作用。SU等[75]将表皮生长因子负载到透明质酸-脱细胞腹膜支架中,观察到表皮生长因子的持续释放,皮肤创伤愈合率提高,促进了皮肤附属物的再生,表皮和真皮更厚。同样,Qiu等[76]将胶质源性神经营养因子负载到犬脱细胞神经基质支架中,植入到比格犬的坐骨神经缺损处,结果发现缺损的神经组织再生和生理功能恢复有明显改善,表明将生长因子负载到脱细胞基质支架中,对于组织再生有更积极的正向作用。 综上所述,脱细胞基质复合支架制备方法主要有交联、静电纺丝技术及三维生物打印技术及其他方法(如纳米颗粒、甲氧基聚乙二醇等),见图3。不同复合支架的性能较单纯的脱细胞基质支架有一定改善,通过交联(提升2-5倍)[49,54]、静电纺丝(提升1.46-10倍)[56,58-60]、三维生物打印的生物墨水(提升9.17-87倍)均能提升支架的机械性能[62,64,68],同时减缓支架的降解速率,对更好引导组织再生有积极的正向作用;其中,脱细胞生物墨水通过三维生物打印技术能打印出更加稳定、个性化复合支架,为更好利用脱细胞基质支架在组织再生中的优势提供了一种新的策略。"
| [1] CHOUDHURY D, YEE M, SHENG ZLJ, et al. Decellularization systems and devices: State-of-the-art. Acta Biomater. 2020;115:51-59. [2] PARVEEN S, SINGH SP, PANICKER MM, et al. Amniotic membrane as novel scaffold for human iPSC-derived cardiomyogenesis. In Vitro Cell Dev Biol Anim. 2019;55(4):272-284. [3] TAYLOR DA, SAMPAIO LC, FERDOUS Z, et al. Decellularized matrices in regenerative medicine. Acta Biomater. 2018;74:74-89. [4] LIU F, ZHOU H, DU W, et al. Hair follicle stem cells combined with human allogeneic acellular amniotic membrane for repair of full thickness skin defects in nude mice. J Tissue Eng Regen Med. 2020;14(5):723-735. [5] OHATA K, OTT HC. Human-scale lung regeneration based on decellularized matrix scaffolds as a biologic platform. Surg Today. 2020;50(7):633-643. [6] AKBAY E, ONUR MA. Investigation of survival and migration potential of differentiated cardiomyocytes transplanted with decellularized heart scaffold. J Biomed Mater Res A. 2019;107(3):561-570. [7] SASSI L, AJAYI O, CAMPINOTI S, et al. A Perfusion Bioreactor for Longitudinal Monitoring of Bioengineered Liver Constructs. Nanomaterials (Basel). 2021;11(2): 275. [8] WANG F, LIU X, YU Y, et al. Decellularized kidney scaffold alters the healing response in chronic renal failure. J Biomed Mater Res A. 2021;109(11):2101-2110. [9] DA LC, HUANG YZ, XIE HQ, et al. Membranous Extracellular Matrix-Based Scaffolds for Skin Wound Healing. Pharmaceutics. 2021;13(11):1796. [10] DENNER J, TÖNJES RR. Infection barriers to successful xenotransplantation focusing on porcine endogenous retroviruses. Clin Microbiol Rev. 2012;25(2):318-343. [11] ZHANG Q, HU Y, LONG X, et al. Preparation and Application of Decellularized ECM-Based Biological Scaffolds for Articular Cartilage Repair: A Review. Front Bioeng Biotechnol. 2022;10:908082. [12] ALSHAIKH AB, PADMA AM, DEHLIN M. Decellularization of the mouse ovary: comparison of different scaffold generation protocols for future ovarian bioengineering. J Ovarian Res. 2019;12(1):58. [13] SONG YH, MAYNES MA, HLAVAC N, et al. Development of novel apoptosis-assisted lung tissue decellularization methods. Biomater Sci. 2021;9(9):3485-3498. [14] WILLEMSE J, VERSTEGEN MMA, VERMEULEN A, et al. Fast, robust and effective decellularization of whole human livers using mild detergents and pressure controlled perfusion. Mater Sci Eng C Mater Biol Appl. 2020;108:110200. [15] FARAG A, HASHIMI SM, VAQUETTE C, et al. Assessment of static and perfusion methods for decellularization of PCL membrane-supported periodontal ligament cell sheet constructs. Arch Oral Biol. 2018;88:67-76. [16] WOLLMANN L, SUSS P, MENDONÇA J, et al. Characterization of Decellularized Human Pericardium for Tissue Engineering and Regenerative Medicine Applications. Arq Bras Cardiol. 2019;113(1):11-17. [17] SYED O, WALTERS NJ, DAY RM, et al. Evaluation of decellularization protocols for production of tubular small intestine submucosa scaffolds for use in oesophageal tissue engineering. Acta Biomater. 2014;10(12):5043-5054. [18] AEBERHARD PA, GROGNUZ A, PENEVEYRE C, et al. Efficient decellularization of equine tendon with preserved biomechanical properties and cytocompatibility for human tendon surgery indications. Artif Organs. 2020;44(4):E161-E171. [19] ROTH SP, BREHM W, GROß C, et al. Transforming Growth Factor Beta 3-Loaded Decellularized Equine Tendon Matrix for Orthopedic Tissue Engineering. Int J Mol Sci. 2019;20(21):5474. [20] ROSMARK O, ÅHRMAN E, MÜLLER C, et al. Quantifying extracellular matrix turnover in human lung scaffold cultures. Sci Rep. 2018;8(1):5409. [21] PORZIONATO A, STOCCO E, BARBON S, et al. Tissue-Engineered Grafts from Human Decellularized Extracellular Matrices: A Systematic Review and Future Perspectives. Int J Mol Sci. 2018;19(12):4117. [22] CHENG J, WANG C, GU Y. Combination of freeze-thaw with detergents: A promising approach to the decellularization of porcine carotid arteries. Biomed Mater Eng. 2019;30(2):191-205. [23] RABBANI M, ZAKIAN N, ALIMORADI N. Contribution of Physical Methods in Decellularization of Animal Tissues. J Med Signals Sens. 2021;11(1):1-11. [24] FERDOWSI KHOSROSHAHI A, SOLEIMANI RAD J, KHEIRJOU R, et al. Adipose tissue-derived stem cells upon decellularized ovine small intestine submucosa for tissue regeneration: An optimization and comparison method. J Cell Physiol. 2020;235(2):1556-1567. [25] FERNÁNDEZ-PÉREZ J, AHEARNE M. The impact of decellularization methods on extracellular matrix derived hydrogels. Sci Rep. 2019;9(1):14933. [26] SIMSA R, VILA XM, SALZER E, et al. Effect of fluid dynamics on decellularization efficacy and mechanical properties of blood vessels. PLoS One. 2019;14(8):e0220743. [27] GUIMARAES AB, CORREIA AT, ALVES BP, et al. Evaluation of a Physical-Chemical Protocol for Porcine Tracheal Decellularization. Transplant Proc. 2019;51(5):1611-1613. [28] YUSOF F, SHA’BAN M, AZHIM A. Development of decellularized meniscus using closed sonication treatment system: potential scaffolds for orthopedics tissue engineering applications. Int J Nanomedicine. 2019;14:5491-5502. [29] GULER S, ASLAN B, HOSSEINIAN P, et al. Supercritical Carbon Dioxide-Assisted Decellularization of Aorta and Cornea. Tissue Eng Part C Methods. 2017;23(9): 540-547. [30] CHOU PR, LIN YN, WU SH, et al. Supercritical Carbon Dioxide-decellularized Porcine Acellular Dermal Matrix combined with Autologous Adipose-derived Stem Cells: Its Role in Accelerated Diabetic Wound Healing. Int J Med Sci. 2020; 17(3):354-367. [31] GIL-RAMÍREZ A, ROSMARK O, SPÉGEL P, et al. Pressurized carbon dioxide as a potential tool for decellularization of pulmonary arteries for transplant purposes. Sci Rep. 2020;10(1):4031. [32] WATANABE N, MIZUNO M, MATSUDA J, et al. Comparison of High-Hydrostatic-Pressure Decellularized Versus Freeze-Thawed Porcine Menisci. J Orthop Res. 2019;37(11):2466-2475. [33] GILPIN A, YANG Y. Decellularization Strategies for Regenerative Medicine: From Processing Techniques to Applications. Biomed Res Int. 2017;2017:9831534. [34] POULIOT RA, YOUNG BM, LINK PA, et al. Porcine Lung-Derived Extracellular Matrix Hydrogel Properties Are Dependent on Pepsin Digestion Time. Tissue Eng Part C Methods. 2020;26(6):332-346. [35] LI J, CAI Z, CHENG J, et al. Characterization of a heparinized decellularized scaffold and its effects on mechanical and structural properties. J Biomater Sci Polym Ed. 2020;31(8):999-1023. [36] RAHMAN S, GRIFFIN M, NAIK A, et al. Optimising the decellularization of human elastic cartilage with trypsin for future use in ear reconstruction. Sci Rep. 2018; 8(1):3097. [37] MCCRARY MW, VAUGHN NE, HLAVAC N, et al. Novel Sodium Deoxycholate-Based Chemical Decellularization Method for Peripheral Nerve. Tissue Eng Part C Methods. 2020;26(1):23-36. [38] DONG M, ZHAO L, WANG F, et al. Rapid porcine corneal decellularization through the use of sodium N-lauroyl glutamate and supernuclease. J Tissue Eng. 2019;10:2041731419875876. [39] PHAN NV, WRIGHT T, RAHMAN MM, et al. In Vitro Biocompatibility of Decellularized Cultured Plant Cell-Derived Matrices. ACS Biomater Sci Eng. 2020;6(2):822-832. [40] HUSSEY GS, DZIKI JL, BADYLAK SF. Extracellular matrix-based materials for regenerative medicine. Nat Rev Mater. 2018;3(7):159-173. [41] GAO M, WANG Y, HE Y, et al. Comparative evaluation of decellularized porcine liver matrices crosslinked with different chemical and natural crosslinking agents. Xenotransplantation. 2019;26(1):e12470. [42] 刘俊,张晓膺. 纳米APS外膜肝素化内膜小口径组织工程血管的实验研究[J].上海交通大学学报(医学版),2017,37(3):337-343. [43] 杨振,李浩,付力伟,等.3D生物打印负载转化生长因子β3的软骨复合支架[J].中国组织工程研究,2021,25(34):5445-5452. [44] ZHANG X, CHEN X, HONG H, et al. Decellularized extracellular matrix scaffolds: Recent trends and emerging strategies in tissue engineering. Bioact Mater. 2021; 10:15-31. [45] ZHANG XZ, JIANG YL, HU JG, et al. Procyanidins-crosslinked small intestine submucosa: A bladder patch promotes smooth muscle regeneration and bladder function restoration in a rabbit model. Bioact Mater. 2020;6(6):1827-1838. [46] ARRIZABALAGA JH, NOLLERT MU. Human Amniotic Membrane: A Versatile Scaffold for Tissue Engineering. ACS Biomater Sci Eng. 2018;4(7):2226-2236. [47] DAVIDENKO N, BAX DV, SCHUSTER CF, et al. Optimisation of UV irradiation as a binding site conserving method for crosslinking collagen-based scaffolds. J Mater Sci Mater Med. 2016;27(1):14. [48] YAO Q, ZHENG YW, LAN QH, et al. Recent development and biomedical applications of decellularized extracellular matrix biomaterials. Mater Sci Eng C Mater Biol Appl. 2019;104:109942. [49] ARRIZABALAGA JH, NOLLERT MU. Riboflavin-UVA crosslinking of amniotic membranes and its influence on the culture of adipose-derived stem cells. J Mech Behav Biomed Mater. 2020;106:103729. [50] SCHNEIDER KH, ROHRINGER S, KAPELLER B, et al. Riboflavin-mediated photooxidation to improve the characteristics of decellularized human arterial small diameter vascular grafts. Acta Biomater. 2020;116:246-258. [51] MA B, WANG X, WU C, et al. Crosslinking strategies for preparation of extracellular matrix-derived cardiovascular scaffolds. Regen Biomater. 2014;1(1):81-89. [52] WANG Y, BAO J, WU X, et al. Genipin crosslinking reduced the immunogenicity of xenogeneic decellularized porcine whole-liver matrices through regulation of immune cell proliferation and polarization. Sci Rep. 2016;6:24779. [53] YAO Q, ZHENG YW, LIN HL, et al. Exploiting crosslinked decellularized matrix to achieve uterus regeneration and construction. Artif Cells Nanomed Biotechnol. 2020;48(1):218-229. [54] 蒋涛,任先军,阴洪,等.京尼平交联大鼠脱细胞脊髓支架的制备及其生物力学特性研究[J]. 中华创伤杂志,2014,30(2):180-184. [55] LIU W, BI W, SUN Y, et al. Biomimetic organic-inorganic hybrid hydrogel electrospinning periosteum for accelerating bone regeneration. Mater Sci Eng C Mater Biol Appl. 2020;110:110670. [56] GONG W, LEI D, LI S, et al. Hybrid small-diameter vascular grafts: Anti-expansion effect of electrospun poly ε-caprolactone on heparin-coated decellularized matrices. Biomaterials. 2016;76:359-370. [57] GHORBANI F, MORADI L, SHADMEHR MB, et al. In-vivo characterization of a 3D hybrid scaffold based on PCL/decellularized aorta for tracheal tissue engineering. Mater Sci Eng C Mater Biol Appl. 2017;81:74-83. [58] 冉小琳.利用猪冠状动脉脱细胞基质和PLCL/明胶纺丝构建双层小口径人工血管的研究[D].重庆:重庆大学,2018. [59] MA YH, SHI HJ, WEI QS, et al. Developing a mechanically matched decellularized spinal cord scaffold for the in situ matrix-based neural repair of spinal cord injury. Biomaterials. 2021;279:121192. [60] GHOLIPOURMALEKABADI M, SAMADIKUCHAKSARAEI A, SEIFALIAN AM, et al. Silk fibroin/amniotic membrane 3D bi-layered artificial skin. Biomed Mater. 2018; 13(3):035003. [61] PARK W, GAO G, CHO DW. Tissue-Specific Decellularized Extracellular Matrix Bioinks for Musculoskeletal Tissue Regeneration and Modeling Using 3D Bioprinting Technology. Int J Mol Sci. 2021;22(15):7837. [62] KIM MK, JEONG W, LEE SM, et al. Decellularized extracellular matrix-based bio-ink with enhanced 3D printability and mechanical properties. Biofabrication. 2020;12(2):025003. [63] YU C, MA X, ZHU W, et al.Scanningless and continuous 3D bioprinting of human tissues with decellularized extracellular matrix. Biomaterials. 2019;194:1-13. [64] JANG J, KIM TG, KIM BS, et al. Tailoring mechanical properties of decellularized extracellular matrix bioink by vitamin B2-induced photo-crosslinking. Acta Biomater. 2016;33:88-95. [65] VISSCHER DO, LEE H, VAN ZUIJLEN PPM, et al. A photo-crosslinkable cartilage-derived extracellular matrix bioink for auricular cartilage tissue engineering. Acta Biomater. 2021;121:193-203. [66] CHOI YJ, JUN YJ, KIM DY, et al. A 3D cell printed muscle construct with tissue-derived bioink for the treatment of volumetric muscle loss. Biomaterials. 2019; 206:160-169. [67] ZHANG X, LIU Y, LUO C, et al. Crosslinker-free silk/decellularized extracellular matrix porous bioink for 3D bioprinting-based cartilage tissue engineering. Mater Sci Eng C Mater Biol Appl. 2021;118:111388. [68] KHATI V, RAMACHANDRAIAH H, PATI F, et al. 3D Bioprinting of Multi-Material Decellularized Liver Matrix Hydrogel at Physiological Temperatures. Biosensors (Basel). 2022;12(7):521. [69] SALEH TM, AHMED EA, YU L, et al. Incorporation of nanoparticles into transplantable decellularized matrices: Applications and challenges. Int J Artif Organs. 2018;41(8):421-430. [70] SALEH T, AHMED E, YU L, et al. Characterization of silver nanoparticle-modified decellularized rat esophagus for esophageal tissue engineering: Structural properties and biocompatibility. J Biosci Bioeng. 2019;128(5):613-621. [71] PESARAKLOU A, MAHDAVI-SHAHRI N, HASSANZADEH H, et al.Use of cerium oxide nanoparticles: a good candidate to improve skin tissue engineering. Biomed Mater. 2019;14(3):035008. [72] KYLUIK-PRICE DL, SCOTT MD. Effects of methoxypoly (Ethylene glycol) mediated immunocamouflage on leukocyte surface marker detection, cell conjugation, activation and alloproliferation. Biomaterials. 2016;74:167-177. [73] LE Y, SCOTT MD. Immunocamouflage: the biophysical basis of immunoprotection by grafted methoxypoly(ethylene glycol) (mPEG). Acta Biomater. 2010;6(7):2631-2641. [74] LIU K, HE Y, YAO Y, et al. Methoxy polyethylene glycol modification promotes adipogenesis by inducing the production of regulatory T cells in xenogeneic acellular adipose matrix. Mater Today Bio. 2021;12:100161. [75] SU Z, MA H, WU Z, et al. Enhancement of skin wound healing with decellularized scaffolds loaded with hyaluronic acid and epidermal growth factor. Mater Sci Eng C Mater Biol Appl. 2014;44:440-448. [76] QIU S, RAO Z, HE F, et al. Decellularized nerve matrix hydrogel and glial-derived neurotrophic factor modifications assisted nerve repair with decellularized nerve matrix scaffolds. J Tissue Eng Regen Med. 2020;14(7):931-943. [77] VELNAR T, BAILEY T, SMRKOLJ V. The wound healing process: an overview of the cellular and molecular mechanisms. J Int Med Res. 2009;37(5):1528-1542. [78] SUTHERLAND AJ, CONVERSE GL, HOPKINS RA, et al. The bioactivity of cartilage extracellular matrix in articular cartilage regeneration. Adv Healthc Mater. 2015;4(1):29-39. [79] NICA C, LIN Z, SCULEAN A, et al.Adsorption and Release of Growth Factors from Four Different Porcine-Derived Collagen Matrices. Materials (Basel). 2020;13(11):2635. |
| [1] | Lai Pengyu, Liang Ran, Shen Shan. Tissue engineering technology for repairing temporomandibular joint: problems and challenges [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(在线): 1-9. |
| [2] | Cai Yaohao, Lang Lyu, Li Hong. Assessing the bone mass of the residual alveolar ridge in the first molar for implant placement by cone-beam computed tomography [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(8): 1572-1577. |
| [3] | Yu Jingbang, Wu Yayun. Regulatory effect of non-coding RNA in pulmonary fibrosis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(8): 1659-1666. |
| [4] | Wang Qiuyue, Jin Pan, Pu Rui . Exercise intervention and the role of pyroptosis in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(8): 1667-1675. |
| [5] | Yuan Weibo, Liu Chan, Yu Limei. Potential application of liver organoids in liver disease models and transplantation therapy [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(8): 1684-1692. |
| [6] | Cao Yue, Ye Xinjian, Li Biyao, Zhang Yining, Feng Jianying. Effect of extracellular vesicles for diagnosis and therapy of oral squamous cell carcinoma [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1523-1530. |
| [7] | Sun Yuting, Wu Jiayuan, Zhang Jian. Physical factors and action mechanisms affecting osteogenic/odontogenic differentiation of dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1531-1540. |
| [8] | Peng Hongcheng, Peng Guoxuan, Lei Anyi, Lin Yuan, Sun Hong, Ning Xu, Shang Xianwen, Deng Jin, Huang Mingzhi . Role and mechanism of platelet-derived growth factor BB in repair of growth plate injury [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1497-1503. |
| [9] | Li Shuai, Liu Hua, Shang Yonghui, Liu Yicong, Zhao Qihang, Liu Wen. Stress distribution on the maxilla when wearing the Twin-block appliance for Class II malocclusion [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(5): 881-887. |
| [10] | Ding Zhili, Huang Jie, Jiang Qiang, Li Tusheng, Liu Jiang, Ding Yu. Constructing rabbit intervertebral disc degeneration models by different methods under X-ray guidance: a comparative study [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(5): 995-1002. |
| [11] | Liu Haoyang, Xie Qiang, Shen Mengran, Ren Yansong, Ma Jinhui, Wang Bailiang, Yue Debo, Wang Weiguo . Application, research hotspots, and shortcomings of degradable zinc-based alloys in bone defect repair and reconstruction [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 839-845. |
| [12] | Zhang Yu, Xu Ruian, Fang Lei, Li Longfei, Liu Shuyan, Ding Lingxue, Wang Yuexi, Guo Ziyan, Tian Feng, Xue Jiajia. Gradient artificial bone repair scaffold regulates skeletal system tissue repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 846-855. |
| [13] | Xiao Fang, Huang Lei, Wang Lin. Magnetic nanomaterials and magnetic field effects accelerate bone injury repair [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 827-838. |
| [14] | Yu Shuangqi, Ding Fan, Wan Song, Chen Wei, Zhang Xuejun, Chen Dong, Li Qiang, Lin Zuoli. Effects of polylactic acid-glycolic acid copolymer/lysine-grafted graphene oxide nanoparticle composite scaffolds on osteogenic differentiation of MC3T3 cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 707-712. |
| [15] |
Zhang Bo, Zhang Zhen, Jiang Dong.
Tannic acid modified interpenetrating network hydrogel promotes tissue remodeling of ruptured Achilles tendon after surgery#br#
#br#
[J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 721-729.
|
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||