Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (4): 607-613.doi: 10.3969/j.issn.2095-4344.2376
Previous Articles Next Articles
Characteristics and manufacturing techniques of collagen/inorganic materials for constructing tissue-engineered bone
Liu Liu, Zhou Qingzhu, Gong Zhuo, Liu Boyan, Yang Bin, Zhao Xian
- Department of Plastic Surgery, First Affiliated Hospital of Kunming Medical University, Kunming 650031, Yunnan Province, China
-
Received:2020-03-10Revised:2020-03-17Accepted:2020-04-15Online:2021-02-08Published:2020-11-24 -
Contact:Zhao Xian, MD, Associate professor, Doctoral supervisor, Department of Plastic Surgery, First Affiliated Hospital of Kunming Medical University, Kunming 650031, Yunnan Province, China -
About author:Liu Liu, Master candidate, Department of Plastic Surgery, First Affiliated Hospital of Kunming Medical University, Kunming 650031, Yunnan Province, China -
Supported by:the National Natural Science Foundation of China, No. 81860343; the Funded Project of Basic Research in Yunnan Province, No. 2017FB121; the Funded Project of Yunnan Province Health Science and Technology Reserve Talent Program, No. H-201631
CLC Number:
Cite this article
Liu Liu, Zhou Qingzhu, Gong Zhuo, Liu Boyan, Yang Bin, Zhao Xian. Characteristics and manufacturing techniques of collagen/inorganic materials for constructing tissue-engineered bone[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 607-613.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1 胶原类型的选择 胶原参与形成细胞外基质纤原纤维和微纤原纤维的网络结构,是细胞外基质的主要支架[7]。目前研究应用的胶原较多是从鱼、牛、猪、鼠类等动物的肌腱、尾巴及真皮中获取的,然而随着基因技术的发展,由于避免天然胶原可能导致的致病性和免疫原性缺点,人工基因重组提取的类人胶原蛋白已成功应用于组织工程支架和血管组织[8]。 胶原具有多种特性,比如低免疫原性及良好的通透性、生物相容性和生物降解性,同时胶原还可调节周围组织细胞的形态,增强细胞的迁移、黏附、增殖与分化功能[9]。在无机材料中复合胶原,不仅能改善无机材料的韧性和机械强度,并且胶原的亲水性可促进成骨细胞黏附于移植骨表面[10]。在骨再生过程中,胶原对于生物矿化的作用也是很重要的,一方面,骨骼的矿物质在胶原蛋白基质中形成,胶原调节着矿物质的沉积速率;另一方面,胶原中的带电基团可提供诱导磷灰石成核的位点,有效激发骨修复中生物矿化的开始,且控制矿化过程中2个非常重要的步骤(矿相以无定形前体的形式渗透到原纤维,随后转化为定向的磷灰石晶体),以促进新骨的形成[11]。 Ⅰ型胶原作为最丰富的胶原蛋白以纤原性胶原的形式存在于软组织和骨骼中,构成了骨骼90%以上有机物[7]。大多数胶原/无机材料支架的研究都采用Ⅰ型胶原。SELVARAJU等[12]从猪皮中提取出Ⅰ型胶原,以聚乳酸作为交联剂,与羟基磷灰石构建复合支架。研究发现,复合支架中每种材料的缺点被改善。复合材料处理时,胶原的任何构象都没有改变,且胶原复合后支架的热稳定性和构象稳定性得到提高,更适用于组织工程。LU等[13]采用胶原蛋白与纳米级羟基磷灰石制备多孔纳米羟基磷灰石/Ⅰ型胶原复合支架,研究表明胶原可提高碱性磷酸酶活性,促进细胞在复合支架的黏附,且对细胞无毒。此外,极少数研究会用到其他类型的胶原,例如MAZZONI等[14]采用Ⅱ型胶原复合羟基磷灰石制备复合支架,LIU等[15]采用类人胶原蛋白复合羟基磷灰石制备三维复合支架。以上实验都证明了胶原/无机材料支架具有良好的骨诱导性和再生性。 2.2 无机材料的选择 作为骨移植材料的无机物,必须有着与天然骨质同样的钙、磷成分,能提供无负荷的结构基质支撑新骨生长和愈合,还具备一定的骨传导性、骨诱导性及骨结合性[16]。磷酸钙、生物活性玻璃 [17]、氟磷灰石[18]、凹凸棒石 [19]、天然骨陶瓷等无机材料被应用于组织工程骨支架材料的研究中[20]。磷酸钙具有出色的生物相容性、渗透性、生物吸收性及骨结合性,在组织工程中研究较为成熟。而常用于组织工程的磷酸钙有羟基磷灰石和β-磷酸三钙 [21]。 羟基磷灰石的化学成分与天然硬组织的无机成分相似,与宿主硬组织具有强烈的亲和力。此外,羟基磷灰石还具有一定的刚度和强度,良好的稳定性、生物相容性和成骨诱导性,是无机材料中最出色的候选材料[22]。ZHANG等[23]利用脱氢热成法制备的羟基磷灰石/胶原复合支架可以达到与用自体骨相同的修复效果。近年来,纳米结构的羟基磷灰石比传统羟基磷灰石具有更好的生物相容性、高塑性及力学性能,其化学和结晶性能与骨磷灰石更相似,其所具有超细结构与高表面积更利于细胞在生物材料表面黏附增殖[24]。LIU等[15]制备了胶原/纳米羟基磷灰石复合支架,结果显示与传统的羟基磷灰石相比,纳米纤维的羟基磷灰石因在结构上模拟骨骼结构,其更易降解,同时具有更好的骨诱导性。纳米技术显著提高了羟基磷灰石作为生物材料的优势,并促进其在组织工程骨领域的应用。 β-磷酸三钙具有良好的骨传导性、生物相容性和生物降解性,不仅能提高细胞的黏附能力,还能加速细胞的分化和增殖速度[25]。ARAHIRA等[26]研究大鼠骨髓间充质干细胞培养对β-磷酸三钙/胶原两相复合支架压缩模量变化行为的影响,发现β-磷酸三钙释放的Ca2+和PO43-促使骨髓间充质干细胞向成骨细胞分化,然而单纯β-磷酸三钙支架结构不稳定;复合胶原后,β-磷酸三钙/胶原蛋白复合支架比单纯β-磷酸三钙支架表现出更好的结构稳定性和更多的成骨标记活性。 尽管羟基磷灰石和β-磷酸三钙在化学组成上相似,但它们的生物降解能力却不同,羟基磷灰石稳定性强,降解速度较慢,一旦植入体内,羟基磷灰石仍然可能存在于再生的骨组织中,而β-磷酸三钙则被完全降解[27]。研究发现单一无机材料的机械性能较差,仅限于非承重应用。因此,研究工作集中将这些材料复合制备出双相磷酸钙混合物,达到无机物作为骨支撑材料的首要条件。简单来说,双相磷酸钙混合物就是将不同比例的羟基磷灰石与β-磷酸三钙结合在一起,提高混合物的结构稳定性和生物降解性。SCHWARZ等[28]发现在裂开型缺损中,与天然骨矿物质效果一样,双相磷酸钙具有骨传导性,支持引导性骨再生。同样,KUNERT-KEIL等[29]也发现将羟基磷灰石和β-磷酸三钙按6∶4的比例混合制备出的双相磷酸钙支架,与单纯β-磷酸三钙支架一样具有良好的生物相容性及骨传导性。两者都可完全整合到骨再生组织中,不引起任何炎症或排异反应。不过相关成骨基因表达实验显示,双相磷酸钙上调成骨细胞标记基因,而β-磷酸三钙上调破骨细胞标记基因。 此外,富含硅、镁、锌等离子的生物陶瓷、生物玻璃和纳米硅材料会表现出更高的刺激成骨细胞分化和增殖的能力,骨诱导性和传导性也会显著提高,骨缺损修复改善更明显[30-33]。 在移植骨材料选择方面,KAMAKURA等[34]和TANUMA等[35]均认为磷酸八钙/胶原比β-磷酸三钙/胶原、羟基磷灰石/胶原更能促进移植骨再生,不过机械强度较低。而LEI等[36]发现纳米羟基磷灰石/胶原和天然骨陶瓷/胶原2种复合支架均能促进骨相关基因表达,前者上调晚期成骨基因表达,而后者促进早期成骨基因表达,并且天然骨陶瓷/胶原支架比纳米羟基磷灰石/胶原在体内更易降解。因此,在临床中根据用途去选择合适的移植骨材料是十分必要的。 2.3 胶原/无机材料支架的制造技术 在组织工程领域,支架的扩散能力即将营养物充分地送到细胞的能力,对组织工程骨的成功是至关重要的[37]。而支架的几何结构明显影响着支架的扩散能力。 三维多孔支架模拟体内3D几何结构,使移植细胞处于立体培养状态,保留了移植细胞的表型特性及形成矿化基质的能力[38]。支架的孔隙率、孔径和多孔结构同时也影响着细胞的渗透、组织生长、血管浸润、营养供应及支架的机械性能[39]。XU等[40]研究证实三维多孔结构可促进细胞的黏附、分化和增殖,加速骨组织、纤维血管和神经的定植,提高复合支架的生物相容性、骨诱导性及骨传导性。此外,LUO等[41]利用天然的三维纳米纤维细菌纤维素作为模板,通过溶胶凝胶法沉积58S前体玻璃,成功制备出三维多孔纳米纤维58S生物玻璃支架,这种纳米纤维支架具有较高的孔隙率和比表面积,可促进成骨细胞的附着和增殖,实现更好的骨传导性和生物活性。之前也有研究表明,移植细胞与纳米纤维支架的相互作用更好[42]。然而,如何通过改善支架几何机构以达到最佳的骨修复,目前仍然是研究的热点。 传统的制造技术主要有:盐浸、熔融成型法、共聚、冷冻凝胶、冷冻干燥法和热诱导相分离法。采用传统方法混合胶原和无机颗粒灌注来制备复合支架材料易导致支架晶体大小不均匀,限制移植细胞的黏附和生长,且胶原的特性在复合过程中易受到外界的干扰。NEGISHI等[43]发明了一种真空压力浸渍法,通过真空负压作用将胶原均匀复合于支架内部,在保证其特性不变的情况下提高复合支架的生物相容性及生物活性。由于传统制造技术不能精准控制支架内部结构,限制胶原/无机材料支架在骨缺损修复中的应用,因此为了解决传统制造技术存在一些问题,静电纺丝技术和3D打印技术出现。 静电纺丝利用电场产生纳米级纤维,它可以使多种聚合物如胶原蛋白、壳聚糖、聚乳酸等生成纳米纤维结构(从几纳米到数百微米)用于组织再生。YANG等[44]通过静电纺丝法成功制备出羟基磷灰石/胶原蛋白/聚乳酸-羟基乙酸复合纳米纤维,结果证实在纳米级支架上细胞表现出更好的黏附、增殖和分化能力。有研究报道,电纺丝纤维结构似乎更利于各种干细胞的增殖和分化[45]。 3D打印技术通过计算机精准辅助设计能制造出孔径和结构可控的三维支架,且具有个性化修复、操作时间短、浪费少、并发症风险小等特性。LI等[46]对比了3D打印2种不同类型的复合支架-纳米羟基磷灰石和脱蛋白牛骨(图2),结果证实2种复合支架材料在细胞增殖方面无差异,表明3D打印技术能完美复制骨骼结构,适用于组织工程。然而,3D打印技术过程中产生的高温容易使一些热敏性材料变性,故在技术上做出了温度降低的改进。低温3D打印技术是非加热型,保持了复合材料的生物活性,可增强复合支架的成骨能力。INZANA等[47]通过低温3D打印成功制备胶原/磷酸钙复合支架,结果证实该复合支架具有骨传导性和骨结合性。因此,低温3D打印技术在组织工程领域具有可行性,有望突破传统技术制备出性能更佳的人工替代骨。 "

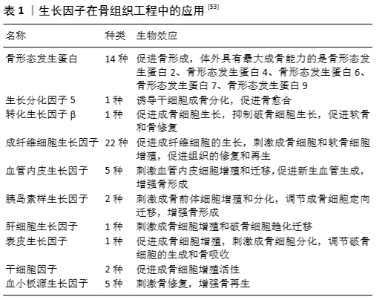
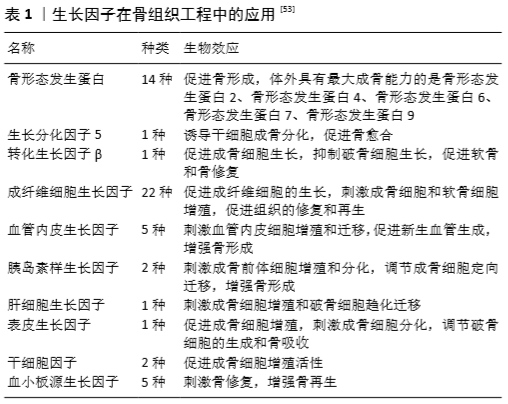
2.4 生物因子在胶原/无机材料支架中的应用 研究表明,胶原/无机材料支架仍然存在着血管化及成骨能力不足的问题,不能达到理想的骨修复效果,因此对其表面修饰生子因子可模仿骨骼环境来增强胶原/无机材料支架的骨愈合能力[48]。目前常见的生长因子有:血小板源性生长因子[49]、血管内皮生长因子[50]、生长分化因子5 [51]、骨形态发生蛋白 [52]、转化生长因子β、成纤维细胞生长因子等(表1)。骨形态发生蛋白是目前已知体外成骨能力最强的生长因子,与生长因子5、转化生长因子β、成纤维细胞生长因子的骨修复效果相似,均能刺激成骨细胞增殖、分化,增加细胞在移植骨材料表面的黏附,促进骨骼愈合。而血小板源性生长因子与血管内皮生长因子是通过募集内皮细胞促进新血管形成和血管生成,为移植骨提供营养和氧气,提高骨组织再生[53]。 除了生长因子外,还有一些生物蛋白及中药成分因子复合到胶原/无机材料支架中同样可提高骨愈合能力。唐宇星等[54]用音猬因子修饰的纳米晶胶原基骨修复大鼠股骨缺损,研究发现音猬因子能够刺激骨细胞增殖、分化,促进移植骨材料的血管化和新骨生成及生长,加快移植骨和受体骨融合,修复骨组织缺损。BRASINIKA等[55]发现L-精氨酸可促进间充质干细胞在羟基磷灰石/胶原复合材料表面的黏附、增殖和分化。此外,淫羊藿苷可促进移植骨支架材料中骨髓间充质干细胞向成骨细胞分化,同时抑制其向破骨细胞分化[19]。槲皮素可激活骨细胞的代谢,促进成骨细胞的增殖、分化[56]。 另外,移植过程中移植骨可能会受到各种细菌及病毒的感染,而骨最主要感染的病原体就是金黄色葡萄球菌。将四环霉素[57-58]、万古霉素等抗生素添加到胶原/无机材料支架中[59],可减少移植过程对移植骨的感染,减少炎症反应和炎症因子的出现,增强成骨活性,促进骨愈合。 通常包裹在支架内部的生物因子会有一个初始的爆发性释放,因此合适的加载方式对支架上生物因子的稳定、持续和可控释放有着至关重要的作用。生物因子加载方法可分为5类:物理吸附/封装、同轴/乳液电纺、微/纳米球封装、多层组装、化学共价固定[60-62],以上方法均适用于胶原/无机材料支架。 2.5 干细胞在胶原/无机材料支架中的应用 干细胞因其丰富来源、低免疫原性及多向分化潜能、能旁分泌各种生物因子及蛋白质,被作为理想的种子细胞应用于组织工程和再生医学中[63]。干细胞分为2类:胚胎干细胞和成体干细胞,它们都具有多能分化潜能,由于胚胎干细胞可能会导致畸胎瘤的产生,并且应用伦理问题未得到解决,故在组织工程骨中主要应用成体干细胞[64]。具有良好成骨潜能的成体干细胞有以下几种:骨髓间充质干细胞[65]、脂肪干细胞[66]、牙髓干细胞[67]、MG-36类成骨细胞[68]、人脐带间充质干细胞[69]。对于骨再生,研究较为成熟的是骨髓间充质干细胞。分离出的骨髓间充质干细胞在体外得到扩增,能保持其分化为多细胞系的能力。MEBARKI等[65]发现在适当的培养条件下,局部移植的骨髓间充质干细胞分泌的胰岛素样生长因子1可以介导小鼠间充质干细胞的趋化,同时刺激宿主间充质干细胞的成骨分化,而骨髓间充质干细胞的成骨分化是被另一种单核细胞趋化蛋白3所诱导的。研究证明局部移植的骨髓间充质干细胞可通过直接或旁分泌协同作用促进骨的整合。此外,经过骨形态发生蛋白[70-71]、骨保护素 [72]、Toll样受体2等基因修饰过的骨髓间充质干细胞[73],其成骨分化能力得到提高,宿主破骨细胞被抑制,促进骨的愈合。 近年来,由于脂肪组织的获取远比骨髓便利,且侵入性小、可获取量大,人们越来越关注脂肪源性干细胞的诱导成骨性和治疗潜力。ZHU等[74]认为脂肪组织是一种丰富、实用、有吸引力的自体细胞替代供体组织来源,与骨髓间充质干细胞相比,脂肪间充质干细胞具有更好的干细胞增殖潜能和多向分化潜能,可以成为骨髓间充质干细胞的替代来源,适合用于同种异体移植和组织工程中。相反,安荣泽等[75]则认为骨髓间充质干细胞的成软骨能力优于脂肪间充质干细胞,骨髓间充质干细胞具有更好的诱导成软骨潜能。就目前来讲,骨髓间充质干细胞在组织工程领域的应用仍然多于脂肪间充质干细胞。 2.6 高分子聚合物在胶原/无机材料支架中的应用 2.6.1 天然高分子材料 众所周知,胶原的热稳定性和机械强度较差,许多外界因素都有可能破坏胶原天然的交联和组装结构。而由多种交联剂稳定的胶原蛋白就具有抗降解性,并且交联形成增加胶原的热稳定性。不过,化学交联剂易产生有毒物质,导致细胞死亡。在此情况下,具有良好生物相容性的天然高分子聚合物被作为交联剂应用于组织工程中[12],常用的天然高分子聚合物有壳聚糖、丝素蛋白、透明质酸、明胶等[76]。 壳聚糖作为一种天然高分子材料具有独特的生物相容性、生物降解性、无毒性、抗菌活性、低免疫原性等特性,并且对壳聚糖进行羧甲基化改性可以提高壳聚糖的溶解度和亲水性[77]。KACZMAREK等[78-79]向胶原/无机材料支架中添加多种天然高分子聚合物,如壳聚糖、糖胺聚糖及透明质酸等,发现天然高分子聚合物可降低复合支架的降解速度,减少移植过程中炎症反应的发生,促进细胞增殖,有效提高骨愈合能力。 丝素蛋白是蚕丝主要的组成成分,最丰富的天然聚合物之一,抗拉强度较大,无毒性,无免疫原性,可控制在体内的降解率,在组织工程领域备受关注[80]。丝素蛋白的复合可提高支架的力学性能及细胞对支架的亲和力,改善细胞黏附与生长,促进支架的成骨能力[81-84]。年争好等[83]发现纳米羟基磷灰石、胶原蛋白与丝素蛋白的混合比例为1∶2∶5时,复合支架材料具有适宜的孔径、孔隙率及良好的生物相容性,材料表面利于细胞的黏附、生长,有望用于组织工程骨缺损的修复。 透明质酸、果胶、糖胺多糖等高分子材料模拟天然骨骼中的多糖基质,具有改善胶原复合支架的力学性能、吸水性、降解性及环境稳定性等特性[79,85]。研究发现,透明质酸通过与细胞受体结合来影响细胞功能,导致促炎症细胞因子的表达增加,从而促进间充质和上皮细胞迁移、分化,增强移植骨的血管化和胶原沉积,影响细胞的增殖和肉芽组织基质的发展[86]。 近期,细菌纤维素因为机械强度高、吸水性强及生物相容性好等特性被作为一种可替代增强聚合物的新型生物材料。NOH等[87]将细菌纤维素复合胶原支架,发现细菌纤维素能提高复合支架的结构稳定性,有利于细胞的附着和生长,可能作为一种极具应用前景的新型组织工程支架。此外,磷酸丝氨酸[88]、藻酸盐等其他天然聚合物也常被用于修饰胶原复合支架中[50],调节骨修复过程中的生物矿化反应,促进移植骨材料的血管形成和骨的再生。 2.6.2 人工合成高分子材料 如上所述,其他可降解的人工合成高分子材料,如聚己内酯、聚乳酸、聚乙醇酸、聚乙烯醇等也可应用于骨组织工程中。不过,单独作为支架基底成分的人工合成高分子材料缺乏骨诱导性,不能为成骨细胞提供合适的沉积矿物质环境,因此常将人工合成高分子材料复合到其他支架中。人工合成高分子材料可控制复合支架的结构,促进细胞的黏附与增殖,提高复合支架的生物降解性及成骨能力[89-90]。 聚乳酸具有良好生的物相容性、无细胞毒性、可吸收性等特性,可加速不同材料如羟基磷灰石、生物玻璃和磷酸钙的降解[31-32,91]。WANG等[92]研究纳米羟基磷灰石/胶原/聚乳酸复合支架联合骨髓间充质干细胞治疗兔股骨头缺血性坏死的效果,结果显示聚乳酸加速了支架材料的降解速度,使复合支架具有更好的生物相容性及骨传导性,增强了股骨头缺血性坏死治疗的效果。 聚己内酯是一种线性合成的生物可降解脂肪族聚酯,因其具有良好的热稳定性和最小的炎症反应,较强的机械性能和黏弹力,易受表面修饰,被用于各种组织缺损的修复中[93]。范瑞瑞等[18]研究聚己内酯/Ⅰ型胶原/氟磷灰石复合支架用于牙周组织再生的情况,结果显示聚己内酯使复合支架具有良好的机械性能及黏弹力,提高了复合支架的生物性能。此外,其他合成聚合物如聚乙烯醇[94]、聚乙醇酸等同样可增加复合支架的机械强度[95],允许支架材料表面功能化,增强复合支架的生物活性。 "
| [1] KUMAR G, NARAYAN B. The biology of fracture healing in long bones.Classic Papers Orthop.2014;60-B(2):150-162. [2] COOPER GM, MOONEY MP, GOSAIN AK, et al. Testing the critical size in calvarial bone defects: revisiting the concept of a critical-size defect.Plast Reconstr Surg.2010; 125(6):1685-1692. [3] MYEROFF C, ARCHDEACON M. Autogenous bone graft: donor sites and techniques. J Bone Joint Surg Am.2011;3(23):2227-2236. [4] HUANG J, LIN X, SHI Y, et al. Tissue engineering and regenerative medicine in basic research: a year in review of 2014.Tissue Eng Part B Rev. 2015;21(2): 167-176. [5] 周思佳,姜文学,尤佳.骨缺损修复材料:现状与需求和未来[J].中国组织工程研究, 2018,22(14):2251-2258. [6] TOZZI G, DEMORI A, OLIVEIRA A, et al. Composite Hydrogels For Bone Regeneration. Materials (Basel).2016;9(4):267-291. [7] GELSE K, POSCHL E, AIGNER T. Collagens:tructure, function, and biosynthesis. Adv Drug Deliv Rev.2003;55(12):1531-1546. [8] 郭亚媛.重组人胶原蛋白水凝胶复合成纤维细胞修复皮肤缺损的实验研究[D].西安:西北大学,2019. [9] DONG C, LV Y. Application of collagen scaffold in tissue engineering: recent advances and new perspectives.Polymers (Basel).2016;8(2):42-62. [10] NAKAJIMA M, NAGAO K, TAKEUCHI M, et al. E-SEM CT observation of collagen scaffold cultured osteoblast cells.IEEE(MHS).2016.DOI: 10.1109/MHS. 2016.7824240 [11] Nudelman F, Lausch AJ, Sommerdijk NA, et al. In vitro models of collagen biomineralization. J Structural Biol.2013;183(2):258-269. [12] SELVARAJU S, RAMALINGAM S, RAO JR. Inorganic apatite nanomaterial: modified surface phenomena and its role in developing collagen based polymeric bio-composite (coll-plga/hap) for biological applications. Colloids Surf B Biointerfaces.2018;172:734-742. [13] LU TL, LI YH, WANG YQ, et al. Preparation and characterization of porous nanosized hydroxyapatite/collagen composite as bone scaffold. Adv Mat Res.2013;647:62-66. [14] MAZZONI E, DAGOSTINO A, IAQUINTA MR, et al. Hydroxylapatite-Collagen hybrid scaffold induces human adipose-derived mesenchymal stem cells to osteogenic differentiation in vitro and bone regrowth in patients.Stem Cells Transl Med.2019;9(3):377-388. [15] LIU Y, GU J, FAN D. Fabrication of high-strength and porous hybrid scaffolds based on nano-hydroxyapatite and human-like collagen for bone tissue regeneration. Polymers (Basel).2020;12(1):61-79. [16] DUTTA SR, PASSI D, SINGH P, et al. Ceramic and non-ceramic hydroxyapatite as a bone graft material: a brief review.Ir J Med Sci.2015;184(1):101-106. [17] MAGRIAM P, FERNANDES KR, ASSIS L, et al. Incorporation of collagen and plga in bioactive glass: in vivo biological evaluation.Int J Biol Macromol. 2019;134:868-881. [18] 范瑞瑞,李欣聪,刘玉,等.聚己内酯/Ⅰ型胶原/氟磷灰石复合支架的制备及生物相容性研究[J].口腔生物医学,2019,10(1):12-16. [19] 宁钰,秦文,任亚辉,等.载淫羊藿苷/凹凸棒石/Ⅰ型胶原/聚己内酯复合支架修复兔胫骨缺损的实验研究[J].中国修复重建外科杂志, 2019,33(9):1181-1189. [20] HAMMOUCHE S, HAMMOUCHE D, MCNICHOLAS M. Biodegradable bone regeneration synthetic scaffolds: in tissue engineering.Curr Stem Cell Res Ther.2012;7(2):134-142. [21] DOROZHKIN SV, EPPLE M. Biological and medical significance of calcium phosphate. Angew Chem Int Edit.2002;41(17):3130-3146. [22] SINGH A. Hydroxyapatite,a biomaterial: its chemical synthesis, characterization and study of biocompatibility prepared from shell of garden snail, helix aspersa.B Mater Sci.2012;35(6): 1031-1038. [23] ZHANG Z, MA Z, ZHANG Y, et al. Dehydrothermally crosslinked collagen/hydroxyapatite composite for enhanced in vivo bone repair.Colloids Surf B Biointerfaces.2018;163:394-401. [24] SHAKIR M, ZIA I, REHMAN A, et al. Fabrication and characterization of nanoengineered biocompatible n-ha/chitosan-tamarind seed polysaccharide: bio-inspired nanocomposites for bone tissue engineering.Int J Biol Macromol.2018;111:903-916. [25] SILVA LDF, REIS ENRDC, BARBARA TA, et al. Assessment of bone repair in critical-size defect in the calvarium of rats after the implantation of tricalcium phosphate beta (β-tcp).Acta Histochemica.2017;119(6):624-631. [26] ARAHIRA T, TODO M. Variation of mechanical behavior of β-tcp/collagen two phase composite scaffold with mesenchymal stem cell in vitro. J Mech Behav Biomed Mater.2016;8(61):464-474. [27] PINA S, OLIVEIRA JM, REIS RL. Natural-based nanocomposites for bone tissue engineering and regenerative medicine: a review.Adv Mater. 2015; 27(7):1143-1169. [28] SCHWARZ F, HERTEN M, FERRARI D, et al. Guided bone regeneration at dehiscence-type defects using biphasic hydroxyapatite + beta tricalcium phosphate (bone ceramic) or a collagen-coated natural bone mineral (biooss collagen): an immunohistochemical study in dogs.Int J Oral Maxillofac Surg. 2007;36(12):1198-1206. [29] KUNERT-KEIL C, SCHOLZ F, GEDRANGE T, et al. Comparative study of biphasic calcium phosphate with beta-tricalcium phosphate in rat cranial defects--a molecular-biological and histological study.Ann Anat.2015;199:79-84. [30] 张晓敏,宋学文,王维,等.凹凸棒石/Ⅰ型胶原/聚己内酯复合修复兔骨缺损的实验研究[J].中国修复重建外科杂志,2016,30(5):626-633. [31] LAI Y, LI Y, CAO H, et al. Osteogenic magnesium incorporated into plga/tcp porous scaffold by 3d printing for repairing challenging bone defect. Biomaterials.2019;197:209-217. [32] GUTIERREZ-PRIETO SJ, FONSECA LF, SEQUEDA-CASTANEDA LG, et al. Elaboration and biocompatibility of an eggshell-derived hydroxyapatite material modified with si/plga for bone regeneration in dentistry.Int J Dent. 2019;4:1-13. [33] WANG Z, LIU Q, LIU C, et al. Mg2+ in beta-tcp/mg-zn composite enhances the differentiation of human bone marrow stromal cells into osteoblasts through mapk-regulated runx2/osx.J Cell Physiol.2020;235(6):5182-5191. [34] KAMAKURA S, SASAKI K, HOMMA T, et al. The primacy of octacalcium phosphate collagen composites in bone regeneration.J Biomed Mater Res A. 2007;83(3): 725-733. [35] TANUMA Y, MATSUI K, KAWAI T, et al. Comparison of bone regeneration between octacalcium phosphate/collagen composite and beta-tricalcium phosphate in canine calvarial defect.Oral Surg Oral Med Oral Pathol Oral Radiol.2013;115(1):9-17. [36] LEI X, GAO J, XING F, et al. Comparative evaluation of the physicochemical properties of nano-hydroxyapatite/collagen and natural bone ceramic/collagen scaffolds and their osteogenesis-promoting effect on mc3t3-e1 cells.Regen Biomater.2019;6(6):361-371. [37] KARANDE T, ONG J, AGRAWAL C. Diffusion in musculoskeletal tissue engineering scaffolds: design issues related to porosity, permeability, architecture, and nutrient mixing. Ann Biomed Eng.2004;32(12):1728-1743. [38] WEI J, HAN J, ZHAO Y, et al. The importance of three-dimensional scaffold structure on stemness maintenance of mouse embryonic stem cells. Biomaterials.2014;35(27):7724-7733. [39] LOH QL, CHOONG C. Three-Dimensional scaffolds for tissue engineering applications: role of porosity and pore size.Tissue Eng Part B Rev. 2013; 19(6):485-502. [40] XU Q, LU H, ZHANG J, et al. Tissue engineering scaffold material of porous nanohydroxyapatite/polyamide 66.Int J Nanomedicine.2010;5:331-335. [41] LUO H, ZHANG Y, LI G, et al. Sacrificial template method for the synthesis of three-dimensional nanofibrous 58s bioglass scaffold and its in vitro bioactivity and cell responses.J Biomater Appl.2017;32(2):265-275. [42] HSU FY, HUNG YS, LIOU HM, et al. Electrospun hyaluronate–collagen nanofibrous matrix and the effects of varying the concentration of hyaluronate on the characteristics of foreskin fibroblast cells.Acta Biomater. 2010;6(6):2140-2147. [43] NEGISHI J, FUNAMOTO S, HASHIMOTO Y, et al. Pla-collagen composite scaffold fabrication by vacuum pressure impregnation.Tissue Eng Part C Methods. 2019;25(12):742-747. [44] YANG X, LI Y, HE W, et al. Hydroxyapatite/Collagen coating on plga electrospun fibers for osteogenic differentiation of bone marrow mesenchymal stem cells.J Biomed Mater Res A.2018;106(11):2863-2870. [45] LI Z, ZHOU Y, YAO H, et al. Greener synthesis of electrospun collagen/hydroxyapatite composite fibers with an excellent microstructure for bone tissue engineering.Int J Nanomedicine.2015;10:3203-3215. [46] LI Q, LEI X, WANG X, et al. Hydroxyapatite/Collagen three-dimensional printed scaffolds and their osteogenic effects on human bone marrow-derived mesenchymal stem cells.Tissue Eng Part A.2019;25(17-18):1261-1271. [47] INZANA JA, OLVERA D, FULLER SM, et al. 3D printing of composite calcium phosphate and collagen scaffolds for bone regeneration.Biomaterials. 2014; 35(13):4026-4034. [48] KORIA P. Delivery of growth factors for tissue regeneration and wound healing.Biodrugs.2012;26(3):163-175. [49] PHIPPS MC, XU Y, BELLIS SL. Delivery of platelet-derived growth factor as a chemotactic factor for mesenchymal stem cells by bone-mimetic electrospun scaffolds.Plos One.2012;7(7): E40831. [50] QUINLAN E, LOPEZ-NORIEGA A, THOMPSON EM, et al. Controlled release of vascular endothelial growth factor from spray-dried alginate microparticles in collagen-hydroxyapatite scaffolds for promoting vascularization and bone repair.J Tissue Eng Regen Med.2017;11(4): 1097-1109. [51] 梁峰,贾长青,谭树森,等.GDF-5体外转染BMSCs移植修复兔退变椎间盘的实验研究[J].解剖科学进展,2011,17(4):368-371. [52] CAI Y, TONG S, ZHANG R, et al. In vitro evaluation of a bone morphogenetic protein2 nanometer hydroxyapatite collagen scaffold for bone regeneration. Mol Med Rep.2018;17(4): 5830-5836. [53] YUN YR, JANG JH, JEON E, et al. Administration Of Growth Factors For Bone Regeneration.Regen Med.2012;7(3): 369-385. [54] 唐宇星,赵庆,杨中萌,等.音猬因子修饰纳米晶胶原基骨复合骨髓间充质干细胞修复股骨缺损[J].中国组织工程研究,2017,21(14):2180-2185. [55] BRASINIKA D, TSIGKOU O, TSETSEKOU A, et al. Bioinspired synthesis of hydroxyapatite nanocrystals in the presence of collagen and l-arginine: candidates for bone regeneration.J Biomed Mater Res B Appl Biomater.2016;104(3):458-469. [56] SONG JE, TIAN J, KOOK YJ, et al. A Bmscs-laden quercetin/duck’s feet collagen/hydroxyapatite sponge for enhanced bone regeneration.J Biomed Mater Res A.2020;108(3):784-794. [57] MARTIN V, RIBEIRO IA, ALVES MM, et al. Engineering a multifunctional 3d-printed pla-collagen-minocycline-nanohydroxyapatite scaffold with combined antimicrobial and osteogenic effects for bone regeneration. Mater Sci Eng C Mater Biol Appl.2019;101:15-26. [58] SEMYARI H, SALEHI M, TALEGHANI F, et al. Fabrication and characterization of collagen-hydroxyapatite-based composite scaffolds containing doxycycline via freeze-casting method for bone tissue engineering.J Biomater Appl. 2018;33(4):501-513. [59] SUCHÝ T, ŠUPOVÁ M, KLAPKOVÁ E, et al. The sustainable release of vancomycin and its degradation products from nanostructured collagen/hydroxyapatite composite layers. J Pharm Sci.2016;105(3):1288-1294. [60] HAJIMIRI M, SHAHVERDI S, KAMALINIA G, et al. Growth factor conjugation: strategies and applications.J Biomed Mater Res A.2015;103(2):819-838. [61] KORIA, PIYUSH. Delivery of growth factors for tissue regeneration and wound healing.Biodrugs.2012;26(3):163-175. [62] LV Y, CHEN G. Immobilization and application of electrospun nanofiber scaffold-based growth factor in bone tissue engineering.Curr Pharm Des. 2015;21(15):1967-1978. [63] SEONG JM, KIM BC, PARK JH, et al. Stem cells in bone tissue engineering. Biomed Mater.2010;5(6):1-16. [64] HOWARD D, BUTTERY LD, SHAKESHEFF KM, et al. Tissue engineering: strategies, stem cells and scaffolds.J Anat.2008;213(1): 66-72. [65] MEBARKI M, COQUELIN L, LAYROLLE P, et al. Enhanced human bone marrow mesenchymal stromal cell adhesion on scaffolds promotes cell survival and bone formation.Acta Biomater.2017;59:94-107. [66] CALABRESE G, GIUFFRIDA R, FABBI C, et al. Collagen-hydroxyapatite scaffolds induce human adipose derived stem cells osteogenic differentiation in vitro.Plos One.2016;11(3): E0151181. [67] TRIVEDI S, SRIVASTAVA K, SALUJA TS, et al. Hydroxyapatite-collagen augments osteogenic differentiation of dental pulp stem cells.Odontology. 2020;108(2):251-259. [68] CECEN B, KOZACI D, YUKSEL M, et al. Biocompatibility of mg-63 cells on collagen, poly-l-lactic acid, hydroxyapatite scaffolds with different parameters.J Appl Biomater Funct Mater.2015; 13(1):10-16. [69] YE B, LUO X, LI Z, et al. Rapid biomimetic mineralization of collagen fibrils and combining with human umbilical cord mesenchymal stem cells for bone defects healing.Mater Sci Eng C Mater Biol Appl.2016;68(1):43-51. [70] 王翔宇,刘强,卢向东,等.转基因干细胞复合纳米羟基磷灰石胶原/聚乳酸修复骨缺损的实验研究[J].中国药物与临床,2012,12(7):857-860. [71] YAN X, ZHOU Z, GUO L, et al. BMP7-overexpressing bone marrow-derived mesenchymal stem cells (bmscs) are more effective than wild-type bmscs in healing fractures.Exp Ther Med.2018;16(2):1381-1388. [72] LIU X, BAO C, XU HHK, et al. Osteoprotegerin gene-modified bmscs with hydroxyapatite scaffold for treating critical-sized mandibular defects in ovariectomized osteoporotic rats.Acta Biomaterialia.2016;42(9):378-388. [73] ZHOU Q, GU X, DONG J, et al. The use of tlr2 modified bmscs for enhanced bone regeneration in the inflammatory micro-environment.Artif Cells Nanomed Biotechnol.2019;47(1):3329-3337. [74] ZHU Y, LIU T, SONG K, et al. Adipose-derived stem cell: a better stem cell than bmsc.Cell Biochem Funct.2008;26(6):664-675. [75] 安荣泽,赵俊延,王兆杰.脂肪干细胞与骨髓间充质干细胞成软骨能力的比较[J].中国组织工程研究,2013,17(32):5793-5798. [76] VILLA MM, WANG L, HUANG J, et al. Improving the permeability of lyophilized collagen-hydroxyapatite scaffolds for cell-based bone regeneration with a gelatin porogen.J Biomed Mater Res B Appl Biomater. 2016;104(8):1580-1590. [77] ZAHRA, SHARIATINIA. Carboxymethyl chitosan: properties and biomedical applications.Int J Biol Macromol.2018;120(Pt B):1406-1419. [78] KACZMAREK B, SIONKOWSKA A, GOLYNSKA M, et al. In vivo study on scaffolds based on chitosan, collagen, and hyaluronic acid with hydroxyapatite. Int J Biol Macromol.2018;118(Pt A): 938-944. [79] KACZMAREK B, SIONKOWSKA A, OSYCZKA AM. Physicochemical properties of scaffolds based on mixtures of chitosan,collagen and glycosaminoglycans with nano-hydroxyapatite addition.Int J Biol Macromol.2018;118(Pt B): 1880-1883. [80] NGUYEN TP, NGUYEN QV, NGUYEN VH, et al. Silk fibroin-based biomaterials for biomedical applications: a review.Polymers.2019;11(12):1933-1958. [81] LAI GJ, SHALUMON KT, CHEN SH, et al. Composite chitosan/silk fibroin nanofibers for modulation of osteogenic differentiation and proliferation of human mesenchymal stem cells.Carbohydr Polym.2014;11(13):288-297. [82] AGRAWAL P, PRAMANIK K. Enhanced chondrogenic differentiation of human mesenchymal stem cells in silk fibroin/chitosan/glycosaminoglycan scaffolds under dynamic culture condition.Differentiation.2019;110:36-48 [83] 年争好,李晖,李瑞欣,等.纳米羟基磷灰石/胶原蛋白/丝素蛋白复合骨组织工程支架材料的生物相容性[J].中国组织工程研究, 2015, 19(8):1149-1154. [84] 孙凯,李瑞欣,范猛,等.打印丝素蛋白/胶原蛋白支架的制备及性能[J].中国组织工程研究,2017,21(2):280-285. [85] WENPO F, GAOFENG L, SHUYING F, et al. Preparation and characterization of collagen-hydroxyapatite/pectin composite.Int J Biol Macromol. 2015;74: 218-223. [86] HOUACINE C, YOUSAF SS, KHAN I, et al. Potential of natural biomaterials in nano-scale drug delivery.Curr Pharm Des.2018;24(43):5188-5206. [87] NOH YK, DOS SANTOS DA COSTA A, PARK YS, et al. Fabrication of bacterial cellulose-collagen composite scaffolds and their osteogenic effect on human mesenchymal stem cells.Carbohydr Polym.2019;219:210-218. [88] 杨春蓉,王迎军,陈晓峰.纳米羟基磷灰石/胶原/磷酸丝氨酸仿生复合骨组织工程支架材料的制备及表征[J].科学通报,2013,58(3):267-271. [89] AKINO N, TACHIKAWA N, MIYAHARA T, et al. Vertical ridge augmentation using a porous composite of uncalcined hydroxyapatite and poly-dl-lactide enriched with types 1 and 3 collagen.Int J Implant Dent.2019;5(1):16-23. [90] YU X, TANG X, GOHIL SV, et al. Biomaterials for bone regenerative engineering. Adv Healthc Mater.2015;4(9):1268-1285. [91] HUANG J, XIA X, ZOU Q, et al. The long-term behaviors and differences in bone reconstruction of three polymer-based scaffolds with different degradability.J Mater Chem B.2019;7(48): 7690-7703. [92] WANG L, XU L, PENG C, et al. The effect of bone marrow mesenchymal stem cell and nano-hydroxyapatite/collagen i/poly-l-lactic acid scaffold implantation on the treatment of avascular necrosis of the femoral head in rabbits.Exp Ther Med.2019;18(3):2021-2028. [93] SIDDIQUI N, ASAWA S, BIRRU B, et al. Pcl-Based composite scaffold matrices for tissue engineering applications.Mol Biotechnol.2018;60(7):506-532. [94] KIM H, YANG GH, KIM G. Three-dimensional gelatin/pva scaffold with nanofibrillated collagen surface for applications in hard-tissue regeneration.Int J Biol Macromol.2019;135:21-28. [95] TOOSI S, NADERI-MESHKIN H, KALALINIA F, et al. Bone defect healing is induced by collagen sponge/polyglycolic acid.J Mater Sci Mater Med. 2019; 30(3):33. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Hu Kai, Qiao Xiaohong, Zhang Yonghong, Wang Dong, Qin Sihe. Treatment of displaced intra-articular calcaneal fractures with cannulated screws and plates: a meta-analysis of 15 randomized controlled trials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1465-1470. |
| [3] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Comparison of the short-term efficacy of extracorporeal shock wave therapy for middle-aged and elderly knee osteoarthritis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1471-1476. |
| [4] | Xu Feng, Kang Hui, Wei Tanjun, Xi Jintao. Biomechanical analysis of different fixation methods of pedicle screws for thoracolumbar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1313-1317. |
| [5] | Jiang Yong, Luo Yi, Ding Yongli, Zhou Yong, Min Li, Tang Fan, Zhang Wenli, Duan Hong, Tu Chongqi. Von Mises stress on the influence of pelvic stability by precise sacral resection and clinical validation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1318-1323. |
| [6] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [7] | Zhang Yu, Tian Shaoqi, Zeng Guobo, Hu Chuan. Risk factors for myocardial infarction following primary total joint arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1340-1345. |
| [8] | Wei Wei, Li Jian, Huang Linhai, Lan Mindong, Lu Xianwei, Huang Shaodong. Factors affecting fall fear in the first movement of elderly patients after total knee or hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1351-1355. |
| [9] | Wang Jinjun, Deng Zengfa, Liu Kang, He Zhiyong, Yu Xinping, Liang Jianji, Li Chen, Guo Zhouyang. Hemostatic effect and safety of intravenous drip of tranexamic acid combined with topical application of cocktail containing tranexamic acid in total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1356-1361. |
| [10] | Xiao Guoqing, Liu Xuanze, Yan Yuhao, Zhong Xihong. Influencing factors of knee flexion limitation after total knee arthroplasty with posterior stabilized prostheses [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1362-1367. |
| [11] | Huang Zexiao, Yang Mei, Lin Shiwei, He Heyu. Correlation between the level of serum n-3 polyunsaturated fatty acids and quadriceps weakness in the early stage after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1375-1380. |
| [12] | Zhang Chong, Liu Zhiang, Yao Shuaihui, Gao Junsheng, Jiang Yan, Zhang Lu. Safety and effectiveness of topical application of tranexamic acid to reduce drainage of elderly femoral neck fractures after total hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1381-1386. |
| [13] | Wang Haiying, Lü Bing, Li Hui, Wang Shunyi. Posterior lumbar interbody fusion for degenerative lumbar spondylolisthesis: prediction of functional prognosis of patients based on spinopelvic parameters [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1393-1397. |
| [14] | Lü Zhen, Bai Jinzhu. A prospective study on the application of staged lumbar motion chain rehabilitation based on McKenzie’s technique after lumbar percutaneous transforaminal endoscopic discectomy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1398-1403. |
| [15] | Chen Xinmin, Li Wenbiao, Xiong Kaikai, Xiong Xiaoyan, Zheng Liqin, Li Musheng, Zheng Yongze, Lin Ziling. Type A3.3 femoral intertrochanteric fracture with augmented proximal femoral nail anti-rotation in the elderly: finite element analysis of the optimal amount of bone cement [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1404-1409. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||