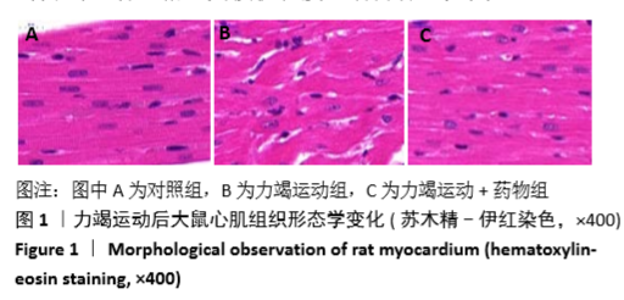
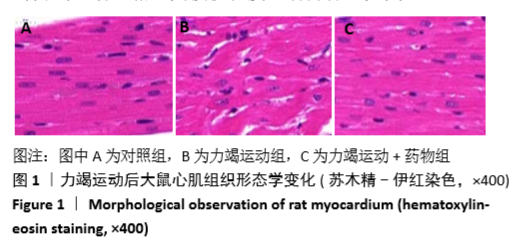
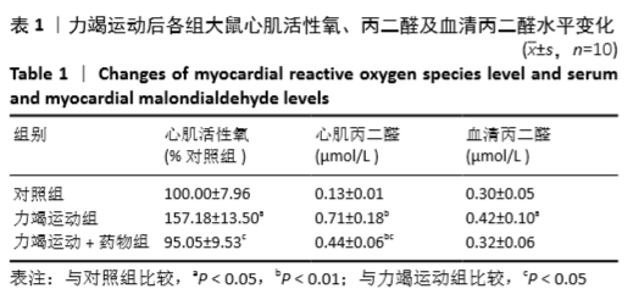
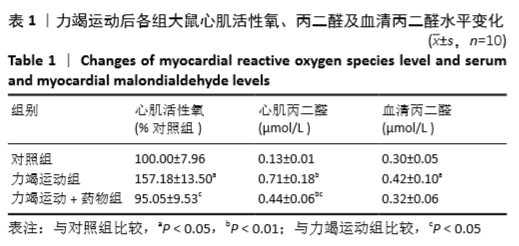
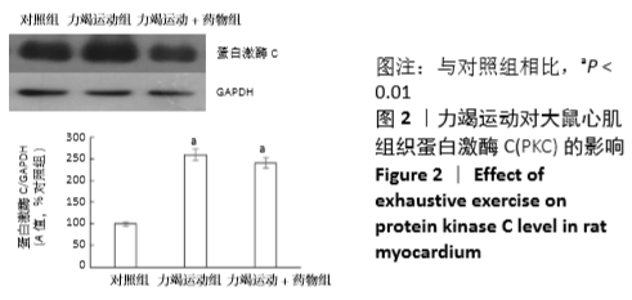
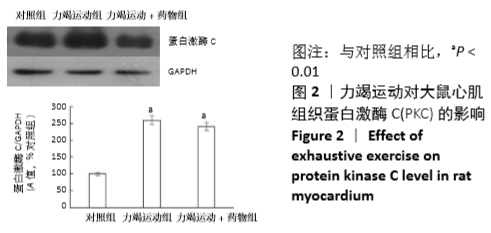
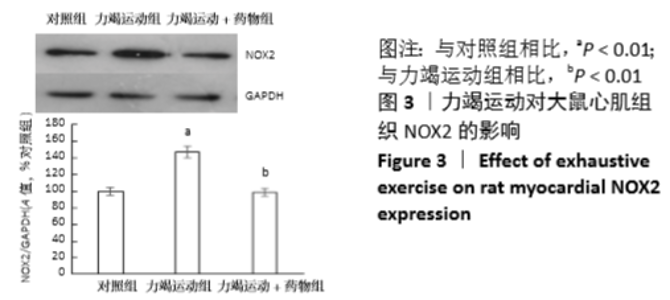
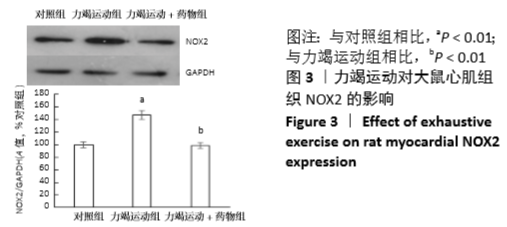
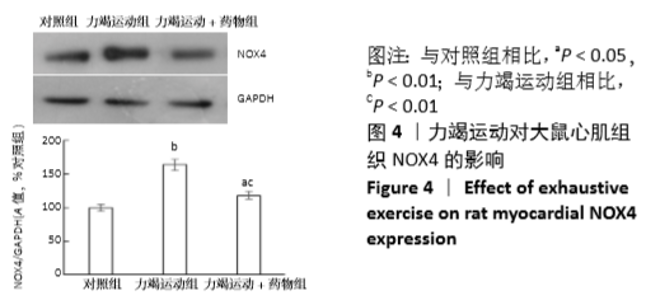
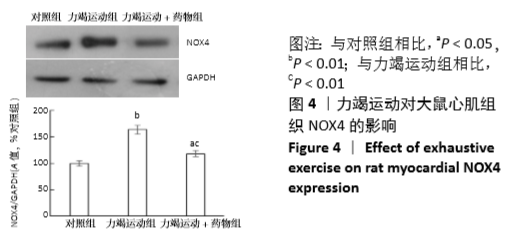
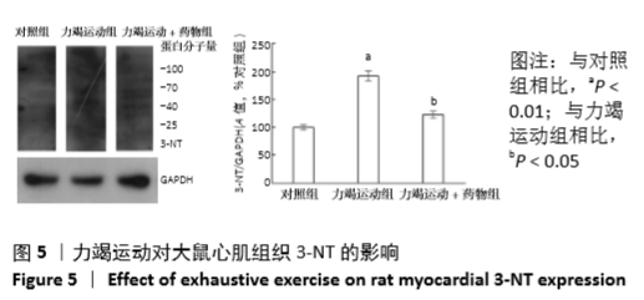
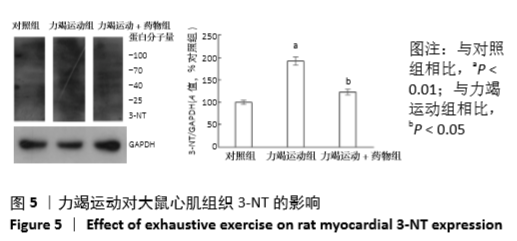
[1] Fenning A, Harrison G, Dwyer D, et al. Cardiac adaptation to endurance exercise in rats. Mol Cell Biochem.2003; 251(1-2):51-59.
[2] 许思毛,上官若男,彭峰林,等. 运动预处理对大负荷跑台运动引起的大鼠心肌损伤干预作用及其机制探讨[J]. 体育科学, 2012, 32(7):45-52.
[3] Wang Y, Xu P, Wang Y, et al. The protection of salidroside of the heart against acute exhaustive injury and molecular mechanism in rat. Oxid Med Cell Longev. 2013;2013(5):507832.
[4] Holmström KM, Finkel T. Cellular mechanisms and physiological consequences of redox-dependent signalling. Nat Rev Mol Cell Biol. 2014;15(6):411-421.
[5] Finkel, T. Signal transduction by reactive oxygen species. J Cell Biol. 2011;194(1):7-15.
[6] Brown I, Griendling KK. Regulation of signal transduction by reactive oxygen species in the cardiovascular system. Circ Res. 2015; 116(3):531-549.
[7] Zeng Q, Han Y, Bao Y, et al. 20-HETE increases NADPH oxidase-derived ROS production and stimulates L-type calcium channel via PKC-dependent mechanism in cardiomyocytes. Am J Physiol Heart Circ Physiol . 2010;299(4):H1109-1117.
[8] 成永霞,刘贵波,颜彬,等.PKC/NADPH氧化应激途径对大鼠心脏微血管内皮细胞eNOS脱偶联的影响[J].基础医学与临床,2013, 33(1):82-87.
[9] Koçer G, Sentürk UK, Kuru O, et al. Potential sources of oxidative stress that induce postexercise proteinuria in rats. J Appl Physiol (1985). 2008;104(4):1063-1068.
[10] Bedford TG , Tipton CM , Wilson NC , et al. Maximum oxygen consumption of rats and its changes with various experimental procedures. J Appl Physiol. 1979;47(6):1278-1283.
[11] 陈丽娜, 周刚, 吴乐, 等. 力竭运动对大鼠肾脏的影响[J]. 中国康复理论与实践, 2016, 22(7):789-792.
[12] 王凯. 蛋白激酶C对晚期运动预处理心肌保护效应中缝隙连接蛋白43表达的影响[J]. 中国运动医学杂志, 2015, 34(11): 1079-1084, 1097.
[13] 陈丽娜, 周刚, 吴乐, 等. NADPH氧化酶抑制剂apocynin对力竭运动大鼠运动性蛋白尿的影响[J].中国应用生理学杂志, 2016, 32(2): 116-120.
[14] 李晶晶,徐鹏,平政,等. 运动预适应对力竭大鼠心功能的影响[J]. 解放军医药杂志,2019,31(7): 9-14.
[15] 路富林,余琦,魏明. 运动预处理对大鼠心脏缺血再灌注损伤的保护作用[J]. 郑州大学学报(医学版), 2018,53(3): 323-327.
[16] Muthusamy VR, Kannan S, Sadhaasivam K, et al. Acute exercise stress activates Nrf2/ARE signaling and promotes antioxidant mechanisms in the myocardium. Free Rad Biol Med. 2012;52(2): 366-376.
[17] Frankiewicz-Jóźko A, Faff J, Sieradzan-Gabelska B. Changes in concentrations of tissue free radical marker and serum creatine kinase during the post-exercise period in rats. Eur J Appl Physiol Occup Physiol. 1996;74(5):470-474.
[18] Huang CC, Tsai SC, Lin WT. Potential ergogenic effects of l-arginine against oxidative and inflammatory stress induced by acute exercise in aging rats. Exp Gerontol. 2008;43(6):571-577.
[19] 谢永磊, 徐宛玲, 黄亚男, 等. 大豆肽通过抑制氧化应激和NF-κB信号通路保护力竭运动大鼠心脏功能[J]. 中国老年学杂志, 2019, 39(5):1158-1161.
[20] Qiu Y, Ping P, Tang XL, et al. Direct evidence that protein kinase C plays an essential role in the development of late preconditioning against myocardial stunning in conscious rabbits and that epsilon is the isoform involved. J Clin Invest. 1998; 101(10):2182.
[21] Zhao J, Renner O, Wightman L, et al. The Expression of Constitutively Active Isotypes of Protein Kinase C to Investigate Preconditioning. J Biol Chem. 1998;273(36):23072-23079.
[22] Bowman JC, Steinberg SF, Jiang T, et al. Expression of protein kinase C β in the heart causes hypertrophy in adult mice and sudden death in neonates. J Clin Invest. 1997;100(9):2189-2195.
[23] Braz JC, Bueno OF, Molkentin WJD. PKCα Regulates the Hypertrophic Growth of Cardiomyocytes through Extracellular Signal-Regulated Kinase1/2 (ERK1/2). J Cell Biol. 2002;156(5):905-919.
[24] Pinton P, Rimessi A, Marchi S, et al. Protein Kinase C β and Prolyl Isomerase 1 Regulate Mitochondrial Effects of the Life-Span Determinant p66Shc. Science. 2007;315(5812):659-663.
[25] Voris JP, Sitailo LA, Rahn HR, et al. Functional alterations in protein kinase C beta II expression in melanoma. Pigment Cell Melanoma Res. 2009; 23(2):216-224.
[26] Drummond GR, Selemidis S, Griendling KK, et al. Combating oxidative stress in vascular disease: NADPH oxidases as therapeutic targets. Nat Rev Drug Discov. 2011;10(6):453-471.
[27] Giordano FJ. Oxygen, oxidative stress, hypoxia, and heart failure. J Clin Invest. 2005; 115(3):500-508.
[28] Lassegue B, San MA, Griendling KK. Biochemistry,physiol-ogy,and pathophysiology of NADPH oxidases in the cardi-ovascular system. Circ Res. 2012;110(10):1364-1390.
[29] Bey EA, Xu B, Bhattacharjee A, et al. Protein kinase C delta is required for p47phox phosphorylation and translocation in activated human monocytes. J Immunol. 2004;173(9):5730-5738.
[30] Petry A, Djordjevic T, Weitnauer M, et al. NOX2 and NOX4 mediate proliferative response in endothelial cells. Antioxid Redox Signal. 2006; 8(9-10):1473-1484.
[31] Kim YM, Kim SJ, Tatsunami R, et al. ROS-induced ROS Release Orchestrated by Nox4, Nox2 and Mitochondria in VEGF Signaling and Angiogenesis. Am J Physiol Cell Physiol . 2017;312(6):C749-C764.
[32] Raad H, Paclet M H, Boussetta T, et al. Regulation of the phagocyte NADPH oxidase activity: phosphorylation of gp91phox/NOX2 by protein kinase C enhances its diaphorase activity and binding to Rac2, p67phox, and p47phox. FASEB J. 2009;23(4):1011-1022.
[33] 丁勇,王建月,许思毛.PKCδ/NOX途径介导一次性力竭运动大鼠心肌过氧化损伤的研究[J]. 北京体育大学学报, 2016, 39(3):73-80, 105.
[34] Xu H, Goettsch C, Xia N, et al. Differential roles of PKCα and PKCɛ in controlling the gene expression of Nox4 in human endothelial cells. Free Radic Biol Med. 2008;44(8):1656-1667.
[35] Gill EK, Edgar KS, Wilson AJ, et al. 7 NOX4 NADPH oxidase is a key regulator of endothelial cell function in experimental diabetes. Br Heart J. 2018;104(Suppl 4):A7.
[36] 余薇, 闵清, 郭霜. NADPH氧化酶在高糖诱导的H9C2心肌细胞损伤中的作用[J]. 中国药理学通报, 2015,31(10):1379-1382.
[37] 黄礼兵,邹蓉,朱明慧,等.NADPH氧化酶抑制剂Apocynin预防脂多糖心肌纤维化的研究[J].实用药物与临床,2017,20(10): 1119-1122.
[38] Lokuta AJ. Increased Nitration of Sarcoplasmic Reticulum Ca2+-ATPase in Human Heart Failure. Circulation. 2005;111(8):988-995. |