Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (25): 6592-6602.doi: 10.12307/2026.261
Previous Articles Next Articles
Gut microbiota and short-chain fatty acids: mechanisms of aerobic exercise regulation in type 2 diabetes
Feng Shuo, Cao Xuan, Guo Xieleiya, Wang Jingfeng, Li Xiaolin
- Graduate School, Harbin Sport University, Harbin 150000, Heilongjiang Province, China
-
Received:2025-09-10Revised:2025-12-31Online:2026-09-08Published:2026-04-22 -
Contact:Li Xiaolin, PhD, Professor, Graduate School, Harbin Sport University, Harbin 150000, Heilongjiang Province, China -
About author:Feng Shuo, MS candidate, Graduate School, Harbin Sport University, Harbin 150000, Heilongjiang Province, China -
Supported by:The Natural Science Foundation Joint Guidance Project of Heilongjiang Province, No. LH2024G003 (to LXL); Harbin Sport University-level Experimental Platform Project, No. LAB2021-06 (to LXL).
CLC Number:
Cite this article
Feng Shuo, Cao Xuan, Guo Xieleiya, Wang Jingfeng, Li Xiaolin. Gut microbiota and short-chain fatty acids: mechanisms of aerobic exercise regulation in type 2 diabetes[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(25): 6592-6602.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
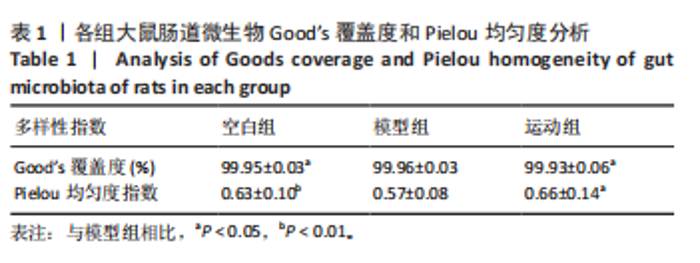
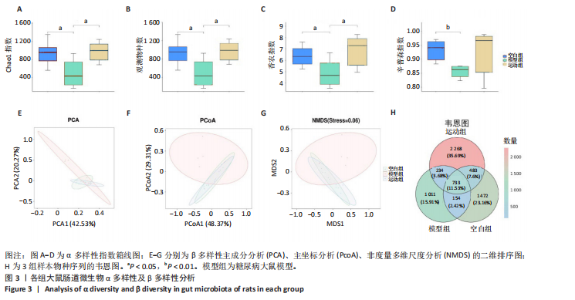
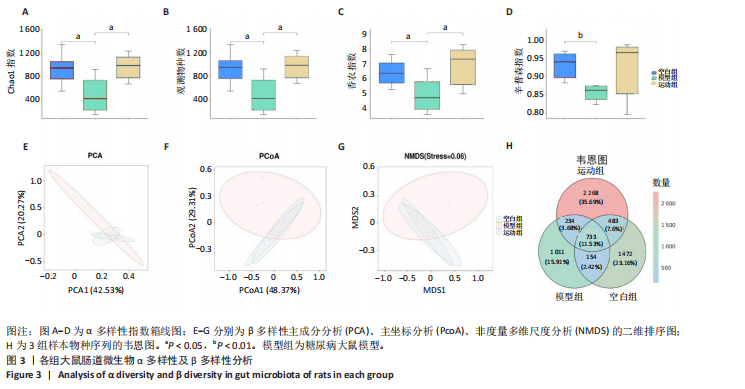
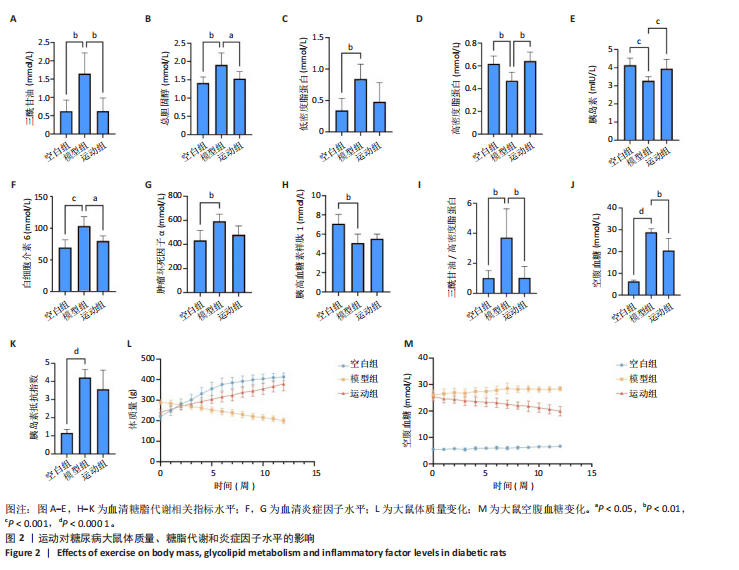
2.1 实验动物数量分析 实验共纳入20只SD雄性大鼠,其中2只大鼠造模失败脱落,余下18只大鼠在干预过程中均无死亡、脱失。 2.2 有氧运动对糖尿病大鼠体质量、血脂、糖脂代谢和炎症因子的影响 如图2所示,12周有氧运动干预逆转了糖尿病大鼠体质量降低。与模型组相比,空腹血糖、三酰甘油、总胆固醇、白细胞介素6水平显著降低(P < 0.05);高密度脂蛋白与胰岛素水平显著升高(P < 0.01);肿瘤坏死因子α、低密度脂蛋白水平与胰岛素抵抗指数虽较模型组有所下降,但未呈现显著性差异(P > 0.05);胰高血糖素样肽1水平有升高趋势但差异不显著(P > 0.05)。 2.3 有氧运动对肠道菌群多样性的影响 2.3.1 α 多样性分析 如表1所示,各组Good’s覆盖度均大于99%,证实测序数据可靠性高,Pielou均匀度指数进一步支持菌群结构均匀性的改善趋势。空白组Chao1指数、观测物种数、香农指数和辛普森指数显著高于模型组(P < 0.05),说明模型组大鼠肠道菌群丰度显著降低,见图3A-D。与模型组相比,运动组肠道菌群Chao1指数、观测物种数、香农指数均显著升高(P < 0.05),见图3A-D。这些结果表明有氧运动能显著恢复糖尿病大鼠肠道菌群的多样性以及均匀性,并在一定程度上提高丰富度。"
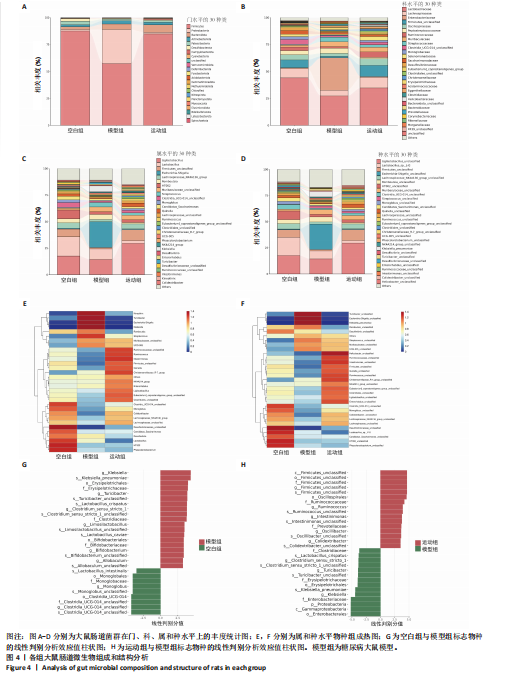
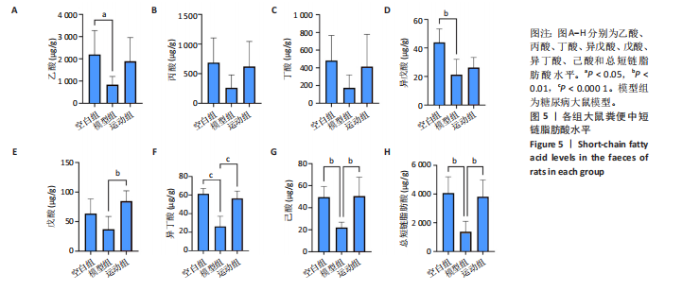
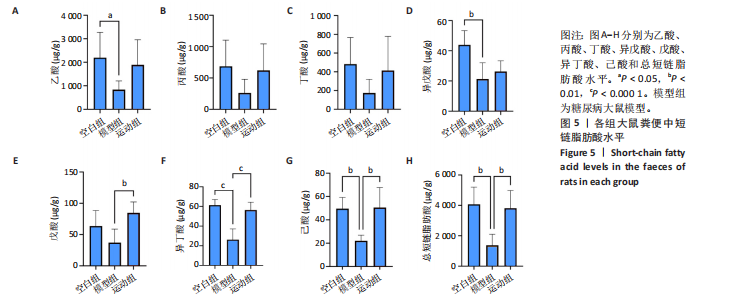
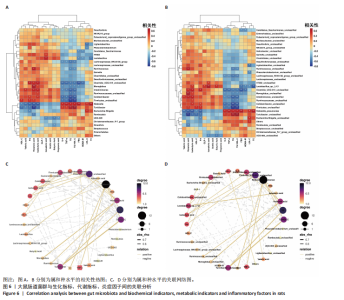
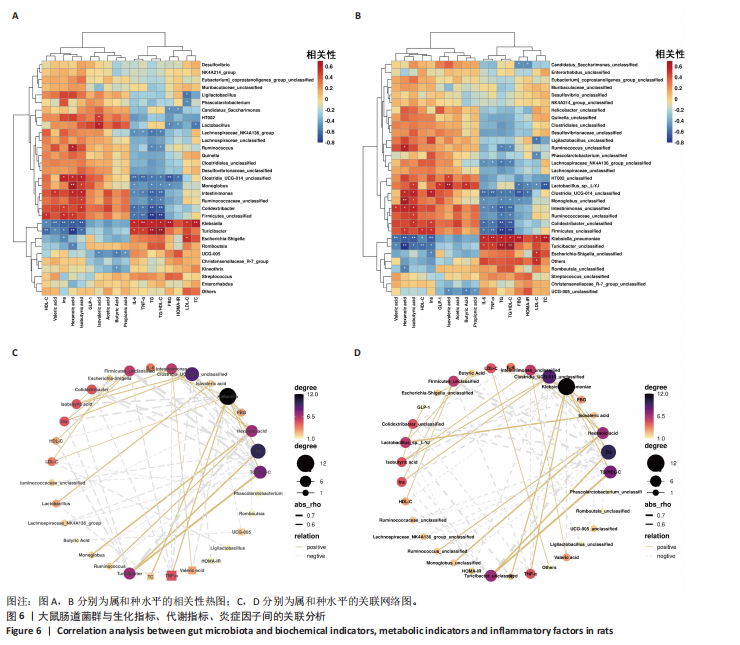
2.3.2 β 多样性分析 如图3E-G所示,基于加权UniFrac距离的β分析:空白组与模型组样本分布距离较远(R=0.40,P < 0.005),表明糖尿病显著改变肠道菌群结构;运动组样本分布趋近空白组,且与模型组分离明显(R=0.24,P < 0.05),表明有氧运动逆转了糖尿病状态下的菌群紊乱,使菌群向正常状态靠拢。 2.3.3 菌群组成特异性分析 韦恩图结果显示:3组共有733个物种,其中运动组特有物种数量最多(2 268个),显著高于空白组(1 011个)与模型组(1 472个),此外运动组与空白组共有物种数量较模型组略有提升,这表明运动干预不仅增加菌群共享物种,还显著提升特定菌种比例,进一步印证运动改善菌群丰富度的作用,见图3H。 2.4 有氧运动对肠道菌群组成和结构的影响 根据肠道微生物多层级分类分析,在门水平上,厚壁菌门(Firmicutes)为各组核心优势菌群,模型组厚壁菌门丰度较空白组降低,同时变形菌门(Proteobacteria)丰度升高,见图4A。此外,在科水平上,模型组脱硫弧菌科(Desulfovibrionaceae)和氨基酸球菌科(Acidaminococcaceae)丰度降低,而肠杆菌科(Enterobacteriaceae)和丹毒丝菌科(Erysipelotrichaceae)丰度升高,见图4B。这一变化在属水平进一步体现为乳杆菌属(Lactobacillus)、罗姆布茨菌属(Romboutsia)等有益菌属减少,以及埃希氏菌-志贺氏菌属(Escherichia-Shigella)、克雷伯氏菌属(Klebsiella)等潜在致病菌属富集。运动干预后,螺杆菌科(Helicobacteraceae)和普雷沃菌科(Prevotellaceae)丰度上调,对应属水平联合乳杆菌属(Ligilactobacillus)和肠单胞菌属(Intestinimonas)丰度恢复,见图4C,E,提示运动可能通过增强短链脂肪酸产生菌的代谢功能缓解菌群失调。在种水平上,模型组中未分类的脱硫弧菌科(Desulfovibrionaceae_unclassified)和未分类的单球菌属(Monoglobus_unclassified)丰度降低,见图4D,F,与科、属水平中脱硫弧菌科及单球菌属的减少趋势一致,表明该类群可能在糖尿病病程中受到系统性抑制。同时,肺炎克雷伯氏菌(Klebsiella_pneumoniae)及未分类的埃希氏菌-志贺氏菌(Escherichia-Shigella_unclassified)在模型组显著富集,与其所属的变形菌门(Proteobacteria)及肠杆菌科(Enterobacteriaceae)在门、科水平的升高形成连锁效应,进一步验证了变形菌门相关致病菌在糖尿病模型中的潜在负面影响。运动干预后,未分类的瘤胃球菌属(Ruminococcus_unclassified)和肠单胞菌属(Intestinimonas_unclassified)丰度上调,与其所属的瘤胃球菌科(Ruminococcaceae)及厚壁菌门(Firmicutes)的恢复趋势相符,提示运动可能通过促进纤维降解菌及产丁酸菌的定植改善肠道微环境。 根据线性判别分析效应值分析结果显示,模型组的标志性菌群以克雷伯氏菌属(Klebsiella)、肺炎克雷伯氏菌(Klebsiella-pneumoniae)及苏黎世杆菌属(Turicibacter)为主,而运动干预后,瘤胃球菌属(Ruminococcus)、肠单胞菌属(Intestinimonas)及普雷沃氏菌科(Prevotellaceae)成为优势菌群,见图4G,H。 2.5 运动对大鼠粪便中短链脂肪酸的影响 有氧运动调控肠道菌群结构,显著提升糖尿病大鼠肠道短链脂肪酸合成能力。模型组样本中乙酸、异戊酸、异丁酸、己酸及总短链脂肪酸水平较空白组显著降低(P < 0.05);运动干预后,戊酸、异丁酸、己酸及总短链脂肪酸水平显著回升(P < 0.05),见图5,表明中等强度有氧运动可部分恢复糖尿病大鼠肠道短链脂肪酸合成功能。 2.6 关联网络分析 相关性热图横坐标代表代谢和生化指标,纵坐标代表肠道菌群(属、种),红色表示正相关,蓝色表示负相关;颜色的深浅直观展示菌群与代谢和生化指标的相关性,同时进行相关性显著检验,见图6A,B。在微生物共现网络图中,不同的颜色、形状和线条粗细代表相关性的强弱以及协同或竞争作用,见图6C,D。图中节点的大小与其他微生物属的连接数(即degree)相关,节点越大,表示该微生物属与其他属的相关性越强。通过Spearman相关系数(r值)进行度量。相关性系数越高,表示相关性越强。通过观察节点的大小和连线数量,可以识别出网络中的核心菌群。克雷伯氏菌属(Klebsiella)、Clostridia_UCG_014_unclassified和肺炎克雷伯氏菌种(Klebsiella_pneumoniae)等节点具有较多的连线,表明"
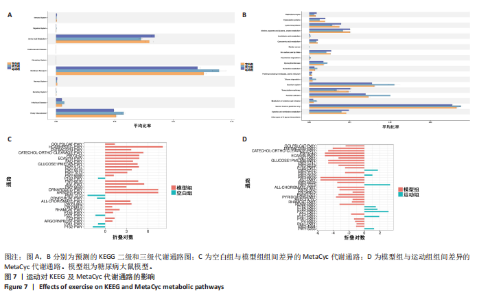
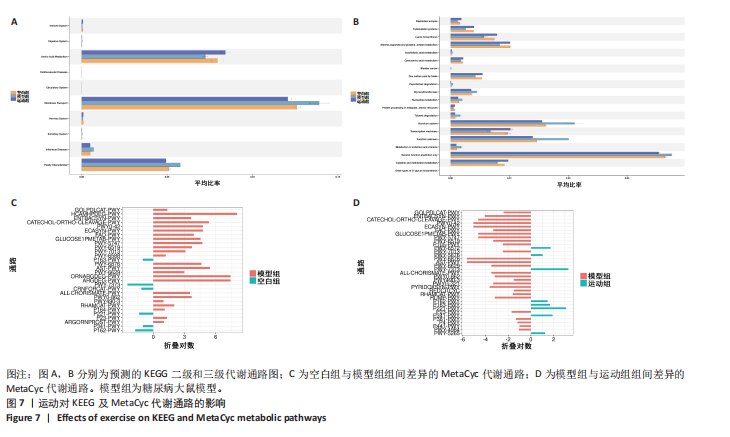
它们在群落中具有较高的中心度。研究结果显示,厚壁菌门(Firmicutes)中科利德克斯特菌属(Colidextribacter)、肠单胞菌属(Intestinimonas)、单球菌属(Monoglobus)及拉克诺螺旋菌科NK4A136群(Lachnospiraceae_NK4A136_group)等有益菌属的丰度在运动组显著升高,且与空腹血糖、胰岛素抵抗指数、三酰甘油/高密度脂蛋白、总胆固醇、炎症因子等代谢指标呈负相关,而与己酸、戊酸、异丁酸等短链脂肪酸水平呈正相关,提示上述菌属可能通过促进短链脂肪酸的生物合成,增强肠道屏障功能并抑制系统性炎症,从而改善糖脂代谢稳态。相反,变形菌门(Proteobacteria)中的条件致病菌属克雷伯氏菌属(Klebsiella)、苏黎世杆菌属(Turicibacter)及埃希氏菌-志贺氏菌(Escherichia-Shigella)在模型组显著富集,其丰度与代谢指标呈正相关,与短链脂肪酸水平呈负相关,见图6A,C,提示上述菌属可能通过破坏短链脂肪酸产生加剧胰岛素抵抗与炎症反应。此外,乳杆菌属(Lactobacillus)丰度与空腹血糖、胰岛素抵抗指数的负相关性进一步支持了益生菌在代谢调控中的关键作用。 图6B,D结果显示,未分类的厚壁菌门(Firmicutes_unclassified)、未分类的科利德克斯特菌属(Colidextribacter_unclassified)及Lactobacillus_sp._L-YJ等菌种与白细胞介素6、三酰甘油/高密度脂蛋白等指标呈强负相关,且 Lactobacillus_sp._L-YJ 与丁酸、异丁酸及异戊酸呈正相关,进一步验证了属水平分析结果的可靠性。值得注意的是,肺炎克雷伯氏菌(Klebsiella_pneumoniae)与代谢指标的正相关性及其对短链脂肪酸的抑制作用,证明特定病原菌种可能通过竞争性抑制短链脂肪酸生成直接参与2型糖尿病病理进程。 2.7 预测的代谢通路结果 2.7.1 代谢通路统计结果 采用PICRUSt2进行代谢通路注释,进一步揭示了糖尿病模型组及运动干预组肠道菌群能量代谢与短链脂肪酸合成通路的动态变化。基于KEGG数据库的代谢通路分析结果显示,运动干预后大鼠肠道菌群的代谢功能在多个二级和三级代谢通路中呈现明显变化,预测功能主要涉及代谢类别,其中氨基酸代谢(Amino acid metabolism)、辅助因子和维生素代谢(Metabolism of cofactors and vitamins)比率较高,见图7A。更深层次的三级代谢通路表明与氨基酸代谢中的丙氨酸、天冬氨酸和谷氨酸代谢(Alanine,aspartate and glutamate metabolism),半胱氨酸和蛋氨酸代谢(Cysteine and methionine metabolism)相关联,见图7B。 2.7.2 代谢通路差异分析结果 获得代谢通路的丰度数据后,使用宏基因组测序分析法找出MetaCyc组间具有显著差异代谢通路,见图7C,D。与空白组相比,模型组中能量代谢相关通路如 HCAMHPDGE-PWY、ECASYN-PWY、PWY0-42、GLUCOSE1PMETAB- PWY、GOLPDLCAT-PWY上调,这些通路与能量代谢、碳水化合物代谢、脂质分解和葡萄糖代谢相关,表明糖尿病状态下肠道菌群通过增强碳水化合物及脂质分解代谢以适应宿主的能量需求失衡,但这一过程可能伴随促炎因子释放及胰岛素抵抗的恶化。模型组中甘氨酸代谢通路PWY-7373及短链脂肪酸合成通路CRNFORCAT-PWY和P163-PWY下调,与属水平观察到的短链脂肪酸产生菌如科利德克斯特菌属(Colidextribacter)、肠单胞菌属(Intestinimonas)丰度降低一致,表明菌群功能紊乱通过抑制短链脂肪酸生成进一步破坏肠道代谢稳态。与模型组相比,运动组上"
| [1] ZHANG H, WEI J, XUE R, et al. Berberine lowers blood glucose in type 2 diabetes mellitus patients through increasing insulin receptor expression. Metabolism. 2010;59(2):285-292. [2] ALMUGADAM BS, YANG P, TANG L. Analysis of jejunum microbiota of HFD/STZ diabetic rats. Biomed Pharmacother. 2021;138:111094. [3] LI G, FENG H, MAO XL, et al. The effects of probiotics supplementation on glycaemic control among adults with type 2 diabetes mellitus: a systematic review and meta-analysis of randomised clinical trials. J Transl Med. 2023; 21(1):442. [4] ZHANG Y, WANG J, GE W, et al. Camel milk peptides alleviate hyperglycemia by regulating gut microbiota and metabolites in type 2 diabetic mice. Food Res Int. 2023;173(Pt 1):113278. [5] LI D, LI Y, YANG S, et al. Diet-gut microbiota-epigenetics in metabolic diseases: From mechanisms to therapeutics. Biomed Pharmacother. 2022; 153:113290. [6] CINTADO E, MUELA P, MARTÍN-RODRÍGUEZ L, et al. Gut microbiota regulates exercise-induced hormetic modulation of cognitive function. EBioMedicine. 2025;119:105876. [7] MUGUERZA-RODRÍGUEZ L, MIER A, PONCE-GONZÁLEZ JG, et al. Systematic Review on the Importance of Gut Microbiota in the Regulation of Type 2 Diabetes Through Physical Activity and Exercise. Curr Issues Mol Biol. 2025;47(7):505. [8] SU W, TAO M, MA L, et al. Dose-response relationships of resistance training in Type 2 diabetes mellitus: a meta-analysis of randomized controlled trials. Front Endocrinol (Lausanne). 2023;14:1224161. [9] HASHIMOTO Y, TAKAHASHI F, OKAMURA T, et al. Diet, exercise, and pharmacotherapy for sarcopenia in people with diabetes. Metabolism. 2023;144:155585. [10] ALGHADIR AH, GABR SA, IQBAL A. The effects of supervised aerobic training on dyslipidaemia among diabetic older patients. BMC Endocr Disord. 2024;24(1):212. [11] 张润红,刘尚武,高建军.基于肠道菌群和血清炎症因子及肠黏膜屏障功能探讨八段锦训练在改善T2DM中的作用 [J].中国微生态学杂志, 2024,36(11):1303-1309. [12] 蔡祎晴,刘新,李光宗.太极拳应用于2型糖尿病的研究进展[J].中华中医药杂志,2023,38(1):298-300. [13] BALDUCCI S, SACCHETTI M, HAXHI J, et al. Physical exercise as therapy for type 2 diabetes mellitus. Diabetes Metab Res Rev. 2014;30 Suppl 1: 13-23. [14] NIYAZI A, YASREBI SMA, YAZDANIAN M, et al. High-Intensity Interval Versus Moderate-Intensity Continuous Exercise Training on Glycemic Control, β Cell Function, and Aerobic Fitness in Women with Type 2 Diabetes. Biol Res Nurs. 2024;26(3):449-459. [15] GHAFFAR T, UBALDI F, VOLPINI V, et al. The Role of Gut Microbiota in Different Types of Physical Activity and Their Intensity: Systematic Review and Meta-Analysis. Sports (Basel). 2024;12(8):221. [16] LIN S, HU Y, DING S, et al. Effects of different pre-conditioning exercise on leptin synthesis and its downstream signalling pathway in T2DM rats. Iran J Basic Med Sci. 2025;28(1):31-37. [17] HAYS KE, PFAFFINGER JM, RYZNAR R. The interplay between gut microbiota, short-chain fatty acids, and implications for host health and disease. Gut Microbes. 2024;16(1):2393270. [18] COLLINS SL, STINE JG, BISANZ JE, et al. Bile acids and the gut microbiota: metabolic interactions and impacts on disease. Nat Rev Microbiol. 2023; 21(4):236-247. [19] SAMPATH KUMAR A, MAIYA AG, SHASTRY BA, et al. Exercise and insulin resistance in type 2 diabetes mellitus: A systematic review and meta-analysis. Ann Phys Rehabil Med. 2019;62(2):98-103. [20] BRINKMANN C. Road map for personalized exercise medicine in T2DM. Trends Endocrinol Metab. 2023;34(12):789-798. [21] WEGIERSKA AE, CHARITOS IA, TOPI S, et al. The Connection Between Physical Exercise and Gut Microbiota: Implications for Competitive Sports Athletes. Sports Med. 2022;52(10):2355-2369. [22] MARTTINEN M, ALA-JAAKKOLA R, LAITILA A, et al. Gut Microbiota, Probiotics and Physical Performance in Athletes and Physically Active Individuals. Nutrients. 2020;12(10):2936. [23] HAN Y, QUAN H, JI W, et al. Moderate-intensity continuous training and high-intensity interval training alleviate glycolipid metabolism through modulation of gut microbiota and their metabolite SCFAs in diabetic rats. Biochem Biophys Res Commun. 2024;735:150831. [24] YANG Y, WANG Y, WANG Y, et al. PCSK9 inhibitor effectively alleviated cognitive dysfunction in a type 2 diabetes mellitus rat model. PeerJ. 2024; 12:e17676. [25] PENGAM M, GOANVEC C, MOISAN C, et al. Moderate intensity continuous versus high intensity interval training: Metabolic responses of slow and fast skeletal muscles in rat. PLoS One. 2023; 18(10):e0292225. [26] HØYDAL MA, WISLØFF U, KEMI OJ, et al. Running speed and maximal oxygen uptake in rats and mice: practical implications for exercise training. Eur J Cardiovasc Prev Rehabil. 2007;14(6):753-760. [27] XU Z, CHEN Y, YU D, et al. Optimal exercise intensity and volume to impact rats with Traditional Chinese Medicine phlegm-dampness constitution. Sports Med Health Sci. 2020;3(1):28-33. [28] YAO Y, CHEN H, YAN L, et al. Berberine alleviates type 2 diabetic symptoms by altering gut microbiota and reducing aromatic amino acids. Biomed Pharmacother. 2020;131:110669. [29] ZHU L, SHA L, LI K, et al. Dietary flaxseed oil rich in omega-3 suppresses severity of type 2 diabetes mellitus via anti-inflammation and modulating gut microbiota in rats. Lipids Health Dis. 2020;19(1):20. [30] MURUGESAN R, KUMAR J, LEELA KV, et al. The role of gut microbiota and bacterial translocation in the pathogenesis and management of type 2 diabetes mellitus: Mechanisms, impacts, and dietary therapeutic strategies. Physiol Behav. 2025;293:114838. [31] LIU L, ZHANG J, CHENG Y, et al. Gut microbiota: A new target for T2DM prevention and treatment. Front Endocrinol (Lausanne). 2022;13:958218. [32] 刘昭志,彭莉.运动、肠道菌群和2型糖尿病关系的研究进展[J].微生物学报,2025,65(1):1-16. [33] YANG L, LIN H, LIN W, et al. Exercise Ameliorates Insulin Resistance of Type 2 Diabetes through Motivating Short-Chain Fatty Acid-Mediated Skeletal Muscle Cell Autophagy. Biology (Basel). 2020;9(8):203. [34] YAZDANI F, SHAHIDI F, KARIMI P. The effect of 8 weeks of high-intensity interval training and moderate-intensity continuous training on cardiac angiogenesis factor in diabetic male rats. J Physiol Biochem. 2020;76(2): 291-299. [35] RAHIM HA, DAMIRCHI A, BABAEI P. Comparison of HIIT and MICT and further detraining on metabolic syndrome and asprosin signaling pathway in metabolic syndrome model of rats. Sci Rep. 2024;14(1):11313. [36] TORQUATI L, GAJANAND T, COX ER, et al. Effects of exercise intensity on gut microbiome composition and function in people with type 2 diabetes. Eur J Sport Sci. 2023;23(4):530-541. [37] RIBEIRO FM, RIBEIRO CFA, G ACM, et al. Limited Effects of Low-to-Moderate Aerobic Exercise on the Gut Microbiota of Mice Subjected to a High-Fat Diet. Nutrients. 2019;11(1):149. [38] ZHANG C, FANG B, ZHANG N, et al. The Effect of Bifidobacterium animalis subsp. lactis MN-Gup on Glucose Metabolism, Gut Microbiota, and Their Metabolites in Type 2 Diabetic Mice. Nutrients. 2024;16(11):1691. [39] XIAO S, LIU C, CHEN M, et al. Scutellariae radix and coptidis rhizoma ameliorate glycolipid metabolism of type 2 diabetic rats by modulating gut microbiota and its metabolites. Appl Microbiol Biotechnol. 2020;104(1): 303-317. [40] SU C, MAO Z, QI P, et al. Hypoglycemic and intestinal microbiota-regulating effects of melanoidins in diabetic mice. J Sci Food Agric. 2025;105(4): 2294-2308. [41] O’SULLIVAN JF, MORNINGSTAR JE, YANG Q, et al. Dimethylguanidino valeric acid is a marker of liver fat and predicts diabetes. J Clin Invest. 2017;127(12):4394-4402. [42] BAOTHMAN OA, ZAMZAMI MA, TAHER I, et al. The role of Gut Microbiota in the development of obesity and Diabetes. Lipids Health Dis. 2016;15:108. [43] MA X, LIU J, JIANG L, et al. Dynamic changes in the gut microbiota play a critical role in age-associated cognitive dysfunction via SCFAs and LPS synthesis metabolic pathways during brain aging. Int J Biol Macromol. 2025;304(Pt 2):140945. [44] HE QL, WANG HC, MA YK, et al. Changes in the Microbiota and their Roles in Patients with Type 2 Diabetes Mellitus. Curr Microbiol. 2023;80(4):132. [45] LI L, WANG X, ZHOU Y, et al. Physalis alkekengi L. Calyx Extract Alleviates Glycolipid Metabolic Disturbance and Inflammation by Modulating Gut Microbiota, Fecal Metabolites, and Glycolipid Metabolism Gene Expression in Obese Mice. Nutrients. 2023;15(11):2507. [46] YI ZY, CHEN L, WANG Y, et al. The potential mechanism of Liu-Wei-Di-Huang Pills in treatment of type 2 diabetic mellitus: from gut microbiota to short-chain fatty acids metabolism. Acta Diabetol. 2022;59(10):1295-1308. [47] RIAL SA, RAVAUT G, MALARET TB, et al. Hexanoic, Octanoic and Decanoic Acids Promote Basal and Insulin-Induced Phosphorylation of the Akt-mTOR Axis and a Balanced Lipid Metabolism in the HepG2 Hepatoma Cell Line. Molecules. 2018;23(9):2315. [48] YU C, LIU S, CHEN L, et al. Effect of exercise and butyrate supplementation on microbiota composition and lipid metabolism. J Endocrinol. 2019; 243(2):125-135. [49] ZHANG WH, JIN ZY, YANG ZH, et al. Fecal Microbiota Transplantation Ameliorates Active Ulcerative Colitis by Downregulating Pro-inflammatory Cytokines in Mucosa and Serum. Front Microbiol. 2022;13:818111. [50] DING H, XU Y, CHENG Y, et al. Gut microbiome profile of Chinese hypertension patients with and without type 2 diabetes mellitus. BMC Microbiol. 2023;23(1):254. [51] LI D, FENG G, LI Y, et al. Benefits of Huang Lian mediated by gut microbiota on HFD/STZ-induced type 2 diabetes mellitus in mice. Front Endocrinol (Lausanne). 2023;14:1120221. [52] TAPPAUF N, LAMERS Y, SHAM HP, et al. Multiomics profiling and parenteral nutrition weaning in pediatric patients with intestinal failure: A longitudinal cohort study. JPEN J Parenter Enteral Nutr. 2025;49(4):451-459. [53] ZHANG L, CHU J, HAO W, et al. Gut Microbiota and Type 2 Diabetes Mellitus: Association, Mechanism, and Translational Applications. Mediators Inflamm. 2021;2021:5110276. [54] QUIROGA R, NISTAL E, ESTÉBANEZ B, et al. Exercise training modulates the gut microbiota profile and impairs inflammatory signaling pathways in obese children. Exp Mol Med. 2020;52(7):1048-1061. [55] KERN T, BLOND MB, HANSEN TH, et al. Structured exercise alters the gut microbiota in humans with overweight and obesity-A randomized controlled trial. Int J Obes (Lond). 2020;44(1):125-135. [56] WANG Y, CHEN J, NI Y, et al. Exercise-changed gut mycobiome as a potential contributor to metabolic benefits in diabetes prevention: an integrative multi-omics study. Gut Microbes. 2024;16(1):2416928. |
| [1] | You Huijuan, Wu Shuzhen, Rong Rong, Chen Liyuan, Zhao Yuqing, Wang Qinglu, Ou Xiaowei, Yang Fengying. Macrophage autophagy in lung diseases: two-sided effects [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1516-1526. |
| [2] | Liu Yu, Lei Senlin, Zhou Jintao, Liu Hui, Li Xianhui. Mechanisms by which aerobic and resistance exercises improve obesity-related cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1171-1183. |
| [3] | Wen Fan, Xiang Yang, Zhu Huan, Tuo Yanfang, Li Feng. Exercise improves microvascular function in patients with type 2 diabetes [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1225-1235. |
| [4] | Chen Qiang, Wu Wenjuan, Jiang Shuhua, Huang Da. Physical exercise improves physical function in burn patients: a systematic review and meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1269-1281. |
| [5] | Wen Xiaolong, Weng Xiquan, Feng Yao, Cao Wenyan, Liu Yuqian, Wang Haitao. Effects of inflammation on serum hepcidin and iron metabolism related parameters in patients with type 2 diabetes mellitus: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1294-1301. |
| [6] | Yan Chengbo, Luo Qiuchi, Fan Jiabing, Gu Yeting, Deng Qian, Zhang Junmei. Effect of type 2 diabetes mellitus on orthodontic tooth movement and bone microstructure parameters on the tension side in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 824-831. |
| [7] | Gu Fucheng, Yang Meixin, Wu Weixin, Cai Weijun, Qin Yangyi, Sun Mingyi, Sun Jian, Geng Qiudong, Li Nan. Effects of Guilu Erxian Glue on gut microbiota in rats with knee osteoarthritis: machine learning and 16S rDNA analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1058-1072. |
| [8] | Wen Fan, Zhu Huan, Hu Qinghua, Tuo Yanfang, Zhou Shufeng, Hu Jiangping, Wang Kangfeng. Eight-week aerobic exercise and aerobic exercise combined with full-body vibration training improve the microcirculation function of obese college students [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 8994-9001. |
| [9] | Wang Jingfeng, Feng Shuo, Cao Xuan, Li Xiaolin. Lycium barbarum polysaccharide-mediated intestinal flora remodeling improves glycolipid abnormalities in type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7592-7602. |
| [10] |
Zhao Yanan, Cao Liquan, Tan Sijie.
A new perspective on exercise for the prevention and treatment of type 2 diabetes mellitus: pyroptosis#br#
#br#
[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7673-7679.
|
| [11] | Fan Zhiliang, Chai Yihui, Chen Guanglei, Li Qian, Gu Chunsong, Chen Yunzhi, Li Wen, Wu Damei, Pu Xiang. Pathogenesis and potential therapeutic targets of idiopathic pulmonary fibrosis: analysis of data from a large-scale genome-wide association study [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7465-7474. |
| [12] | Liu Meng, Hou Shilun. Effects of blood flow restriction training and aerobic exercise on energy expenditure in young men [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(25): 6603-6609. |
| [13] | Wang Wei, Chen Jun, Jia Shaohui, Xue Xinxuan, Dong Kunwei. Mechanisms by which exercise regulates gut microbiota in the prevention and treatment of non-alcoholic fatty liver disease [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6328-6336. |
| [14] | Zhang Zheng, Zhang Yibo, Xu Bin, Yan Shichao, Guo Hui. Sarcopenia and non-alcoholic fatty liver disease: analysis of the gut microbiota [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6000-6009. |
| [15] | Li Yiguang, Guo Haonan, Ding Xiaotao, Yuan Mengyao, Jiang Lijin, Fan Xinfeng, Feng Yan. Visual analysis of research hotspots in the field of gut microbiota in the elderly at home and abroad [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6071-6080. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||