Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (22): 3571-3577.doi: 10.3969/j.issn.2095-4344.3176
Previous Articles Next Articles
Status and problems of polycaprolactone and its composite materials in bone tissue engineering
Zhang Zhenhua1, 2, Liu Zichen1, 2, Yu Baoqing2
- 1University of Shanghai for Science and Technology, Shanghai 200093, China; 2Department of Orthopedics, Pudong Hospital Affiliated to Fudan University, Shanghai 201399, China
-
Received:2020-05-06Revised:2020-05-12Accepted:2020-07-06Online:2021-08-08Published:2021-01-21 -
Contact:Yu Baoqing, Professor, Chief physician, Department of Orthopedics, Pudong Hospital Affiliated to Fudan University, Shanghai 201399, China -
About author:Zhang Zhenhua, Master candidate, University of Shanghai for Science and Technology, Shanghai 200093, China; Department of Orthopedics, Pudong Hospital Affiliated to Fudan University, Shanghai 201399, China -
Supported by:the National Natural Science Foundation of China, No. 81971753 (to YBQ); the Key Discipline Group Construction Project of Health System in Pudong New Area of Shanghai, No. PWZxq2017-11 (to YBQ); the Shanghai Key Medical Specialty Program, No. ZK2019C01 (to YBQ); the Plateau Discipline Construction Project of Shanghai Pudong New Area, No. PWYgy2018-09 (to YBQ); the Shanghai Medical Leading Talents Program, No. 046 (to YBQ)
CLC Number:
Cite this article
Zhang Zhenhua, Liu Zichen, Yu Baoqing. Status and problems of polycaprolactone and its composite materials in bone tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3571-3577.
share this article
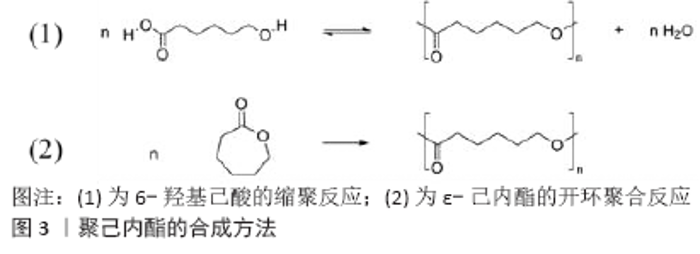
2.1 聚己内酯的合成及其性能 2.1.1 聚己内酯的合成方法 聚己内酯是由分子式为(C6H10O2)n的己酸重复单元组成的,也被称为2-氧戊酮均聚物或6-己内酯聚合物[8]。一般而言聚己内酯的合成有两种方法:6-羟基己酸的缩聚反应或者ε-己内酯的开环聚合反应(图3)[9]。其中,缩聚反应是通过逐步缩合反应开发出来的聚合物合成方法。单体上2个可以相互发生反应的官能团相互反应后使聚合物链得以生长,同时会释放出低分子质量副产物。缩聚反应合成聚己内酯是6-羟基己酸或增长的聚合物链上酸性官能团与另外一个6-羟基己酸的羟基进行的酯化反应,反应副产物是一分子水分子。在聚合过程中,用去除产生水的方法推动反应的进行,最终形成聚合物。通过缩聚反应合成聚己内酯一般使用生物提取的脂肪酶作催化剂或不使用催化剂[10-11]。但开环聚合的方法可以形成分子质量更高且分子质量分布较低的聚合物,所以是公认合成聚己内酯的最优方法[12-14]。开环聚合的催化剂按照机制一般分为阴离子、阳离子、单体活化和配位插入4种,按照种类又可以分为金属类催化剂、有机类催化剂和酶类催化剂等类型,不同方法都会影响聚合物的分子质量及其分布、端基组成和聚合物的结构[8,14]。现如今,金属基催化剂被广泛用于聚己内酯的聚合反应中,其中碱土催化剂因为极低的毒性和高活性优点被广泛用于生物医学应用中,且此种类型催化剂可形成高分子质量、低到中程度分子质量分布的聚己内酯聚合物[8]。对于生物医学应用而言,参与聚己内酯聚合过程的所有分子必须对人类无毒害副作用且不产生具有潜在毒性的副产物,其中钙基和镁基催化剂是最受欢迎的催化剂,而且这两种元素天然存在于生物系统中。近期也有研究表明,将酶用于开环聚合合成聚己内酯是金属基催化剂更安全的替代品[15–17]。用于聚己内酯开环聚合的酶能从不同的原核和真核生物中分离出来,包括假单胞菌属、曲霉属、念珠菌属和伯克霍尔德氏菌等[8]。总的而言,聚己内酯合成催化剂的多样性为聚己内酯的应用提供了多种多样的机会,可以根据对最终产品的性能要求和应用选择现有合适的合成方法。 2.1.2 聚己内酯的性能 聚己内酯是一种可生物降解的聚酯(其他例如聚乙醇酸、聚乳酸),是一种脂肪族半结晶聚合物,熔点范围在59-64 ℃之间,玻璃化转变温度(Tg)约为-60 ℃,所以在室温条件下呈现橡胶态,从而具有高韧性和出色的力学性能(强度、弹性等性能取决于分子质量)[18]。现有合成聚己内酯的数均分子质量可在530-630 000之间变化,并可以根据其分子质量进行分级[14]。聚己内酯无毒且与组织相容,因此被广泛用作可吸收缝线、再生治疗和药物输送应用中的支架。室温下,聚己内酯在不同程度上溶于大多数有机溶剂,例如氯仿、二氯甲烷、四氯化碳、苯、甲苯等,但不溶于醇类、石油醚和水[19]。 聚合物支架力学性能决定了它的实用性,支架应该具有对抗植入部位生理应力的强度,并保持和目标组织力学性能的一致性。此外,支架还需要在目标组织恢复正常功能之前起到代替作用[20]。支架的力学性能首先取决于材料的性能,其次和制造成型方法、孔隙率及支架的其他物理参数也有很大关系。RAGAERT等[21]的实验显示其设计的编织状支架刚度参数为3.79 N/mm,远小于此前研究中的最低值(具有相同细丝尺寸的4层聚己内酯支架)11 N/mm。在实际应用中可以通过调节制造方法和支架结构来设计支架的力学性能。 合成聚合物由于其优异的性能在再生医学领域备受欢迎,然而由异物引起的免疫反应是一个需要关注和解决的问题。聚己内酯属于化学惰性、生物相容性和低成本的合成生物可降解聚合物,已被广泛应用于组织工程领域中,但由于其本身性质决定的疏水性不利于细胞的黏附和增殖,为了克服这一问题,进行表面改性、共混等方法都是有效的途径。 降解行为必须是和目标组织相匹配的,随着新组织的生长逐步降解,直到新组织能够支撑自身。降解速度过慢是限制聚己内酯广泛应用的原因之一,由于在其重复单元中存在5个疏水性的-CH2,所以聚己内酯的降解速度极慢,由不同聚酯制成的纳米纤维基体降解速率为:聚乙醇酸>聚乳酸-羟基乙酸共聚物>聚乳酸>聚己内酯[22]。酯基水解是聚己内酯降解的主要机制,在降解过程中酯基的水解会导致链段的随机断裂[23]。一些研究报告称,聚己内酯聚合物支架需要三四年才能完全降解[23]。由于其缓慢的生物降解速率,聚己内酯是缓释药物递送系统的优秀候选材料。除了聚合物的化学结构外,表面改性和支架的宏观结构在降解过程中也起着举足轻重的作用。 2.2 聚己内酯支架的制造方法 总的来说,聚己内酯支架的制备技术取决于所需支架的类型。对于多孔支架,常用有致孔剂浸出、CO2的饱和和释放、3D打印、相分离技术和冷冻干燥等,但在纤维支架的制造中一般使用静电纺丝和熔融静电纺丝等技术。 2.2.1 溶剂浇铸和致孔剂浸出 适当的孔隙率有助于组织的生长和营养物质的扩散,所以孔隙率是设计用于骨组织工程支架的重要因素。这种技术利用在聚合物中嵌入可溶性致孔剂颗粒(例如NaCl和NaHCO3),然后去除致孔剂后会在聚合物基质中形成孔结构。FABBRI等[24]在聚己内酯混合溶液中加入质量比为1∶1的氯化钠/碳酸氢钠作为致孔剂,制作出具有合适孔径和孔隙率的支架,其中孔径和孔隙率可通过适当选择粒度和成孔剂浓度来调整。最近,CUI等[25]将溶剂浇铸和致孔剂浸出法结合起来,可以通过控制氯化钠颗粒的大小和聚己内酯溶液的浓度来调节聚己内酯支架的孔隙率,最终根据不同的处理方法制备成具有高达91%和88%的两种聚己内酯3D支架。 2.2.2 静电纺丝 目前为止,静电纺丝技术是制备高强度可控超细聚合物纤维3D支架最有利的技术。该技术应用广泛,能够应用于各种聚合物溶液及其混合物中,例如聚己内酯和氧化铝、重组蛛丝蛋白和明胶、聚乳酸、聚乳酸-羟基乙酸共聚物、丝素蛋白和聚氨酯等。REN等[26]使用静电纺丝方法制备了聚己内酯/明胶杂化纳米纤维引导性骨再生膜,并证实了其显著的骨再生能力。MISZUK等[27]使用静电纺丝的方法制备了一种新型3D聚己内酯/羟基磷灰石纳米纤维复合支架,为成骨分化和骨形态发生蛋白2诱导的异位骨形成提供了更有利的微环境,成功改善了体外成骨和体内新骨的生成。 静电纺丝时原料状态除了溶液还有熔融形态,此时静电纺丝也称作熔融静电纺丝。熔融静电纺丝的原理保持不变,唯一区别就是所用的聚合物是熔体,聚合物熔体比聚合物溶液更加黏稠。XUE等[28]通过熔融纺丝制备了具有不同负载浓度的聚己内酯/羟基磷灰石复合支架,证明纳米羟基磷灰石的添加有利于改善聚己内酯支架的热稳定性和力学性能;体外矿化实验证明,聚己内酯/羟基磷灰石复合支架的矿物质沉积速度更快,更有利于成骨。REN等[29]使用熔融静电纺丝技术制备了具有互连多孔结构的复聚己内酯-锶代生物活性玻璃(SrBG)复合支架,实验验证该复合支架无细胞毒性,并且可以显著提升MC3T3-E1细胞的碱性磷酸酶活性。 2.2.3 相分离技术 相分离技术主要包括产生少聚合物相和多聚合物相的液体或固液混合物,在降低温度时溶液发生相变,去除发生相变的一相后导致孔的形成[30]。GRANDI等[31]将海藻酸钙作为致孔剂,利用相分离技术制备了具有三维孔结构的聚己内酯支架,实验证明此种方法制备的聚己内酯支架是具有骨组织工程应用所需物理和生物学特性的候选者。 2.2.4 3D打印 3D打印技术现已发展成为一种极具潜力和发展前景的技术,它能够以微米级的尺寸和精度精确制造支架复杂的结构,并且可以在其中掺入分布均匀的细胞[32]。CHO等[33]使用3D打印技术制备了可以将羟基磷灰石暴露在聚己内酯支架表面的复合支架,显著提升了聚己内酯支架的力学性能,并通过碱性侵蚀加速了钙和磷酸根离子的沉积,从而加强了支架的成骨能力。同样的,BITTNER等[34]使用3D打印方法进一步成功制备了可以含有梯度孔隙率、梯度孔径和梯度羟基磷灰石含量的聚己内酯/羟基磷灰石复合支架,这些实验表明可以使用3D打印技术制备出与天然骨软骨单元相似的孔隙率变化及在人小梁骨压缩特性范围内变化的骨组织工程支架。 2.3 聚己内酯及其复合材料在骨组织工程的应用 对于应用于骨组织工程的生物材料而言,足够的力学性能、适合于目标组织的降解性能、一定的骨传导性和骨诱导能力都是必须的[32]。聚己内酯在骨组织工程中的优势在于其具有一定的力学强度、细胞黏附和增殖能力,但其力学性能、降解性能、成骨诱导性仍然有提高改善的需要[35–37]。研究表明,多种生物陶瓷和天然聚合物对聚己内酯进行改性可以分别增强其骨诱导和骨传导能力,以及增强其对骨组织修复的生物相容性[2,38–40]。 常用于骨组织工程中的生物陶瓷主要有羟基磷灰石、β-碳酸钙和双相磷酸钙等。RUMINSKI等[41]使用熔融沉积建模成型方法制备了一种聚己内酯/双相磷酸钙复合支架,用于验证是否有利于促进培养物中脂肪干细胞的成骨分化,从而增强载有脂肪干细胞骨移植物的效用。实验结果表明与2D培养环境相比,3D培养环境中的成骨细胞分化显著增加;RT-PCR结果显示成骨特异性标志物RUNX-2、胶原蛋白Ⅰ、碱性磷酸酶、骨粘连蛋白和骨钙蛋白的明显上调;此外,免疫荧光染色和Western-Blot结果显示骨钙素的表达显著上调,并且伴随更早期、更强的矿化作用。MOEINI等[42]使用溶剂热法原位制备羟基磷灰石纳米棒增强的聚己内酯支架,根据控制处理温度形成了具有不同长径比羟基磷灰石纳米棒的聚己内酯复合材料。通过对复合支架的表征显示,羟基磷灰石纳米棒的原位合成比异位合成具有更高的弹性模量和强度(约15%),体外矿化实验结果显示了更好的骨再生能力;MTT分析显示,人骨肉瘤细胞系在聚己内酯/羟基磷灰石纳米棒(20%)纳米复合支架上表现出比对照组更高的细胞活力,并且随着孵育时间的增加细胞活力提高,猜测可能是由于合成纳米棒晶体的形态和结晶度导致的。RONCA等[43]使用增材制造方法制备了聚己内酯/羟基磷灰石复合支架,并对其力学性能和生物学性能进行评价,结果显示,添加羟基磷灰石后复合支架的压缩模量从(87.1±7.0) MPa增加到(114.3±9.5) MPa;第7天的共聚焦激光扫描显微图像显示出非常好的细胞附着,细胞遍布支架表面和整个3D支架结构。HASSAN等[44]通过在聚己内酯中添加羟基磷灰石增加了复合纳米纤维的亲水性和润湿性,与纯聚己内酯纳米纤维的接触角相比(127.3±2.4)°,添加羟基磷灰石后的复合纳米纤维接触角能达到(93.7±3.6)°,这显著增加了纳米纤维植入物的表面蛋白质吸收能力;且相对于纯聚己内酯支架2周15.95%的吸水率,聚己内酯/羟基磷灰石复合支架能达到90%的吸水率;体外SBF矿化实验表明,聚己内酯/羟基磷灰石复合纳米纤维在第3天羟基磷灰石开始成核,并在第7天后显示出完全矿化,14 d时纤维的某些部分已被羟基磷灰石层完全覆盖,而纯聚己内酯纤维在第7天之后没有观察到明显的磷灰石层。BAO等[45]使用熔融沉积建模方式构建了具有相互连接的多孔网络聚己内酯/β-碳酸钙3D支架,复合支架显示出对蛋白质(如骨形态发生蛋白)和富含钙微环境更高的亲和力。使用人类胎儿间充质干细胞评估支架的生物学性能,结果显示复合支架显示出更好的生物相容性,最终细胞密度比聚己内酯支架高1.25倍,但是聚己内酯/β-碳酸钙复合支架并没有显示出促进干细胞分化和矿物质沉积的作用。SHIM等[46]将聚己内酯和双相磷酸钙分别改性,然后将双相磷酸钙纳米粒子固定在聚己内酯支架上制备成聚己内酯/双相磷酸钙复合支架,其中聚己内酯支架是使用3D打印技术制备的具有相互连接的多孔支架。体外细胞实验结果显示,聚己内酯/双相磷酸钙复合支架的碱性磷酸酶活性和钙沉积显著增加,极大地促进了成骨细胞活性和MG-63细胞的矿化;体内动物实验显示,聚己内酯/双相磷酸钙复合支架在大鼠胫骨缺损区域新骨的形成显著高于其他对照组。 对于天然聚合物而言,壳聚糖、蚕丝、海藻酸盐和明胶等都是用于聚己内酯改性的可靠选择。KIM等[12]将聚己内酯和各种质量分数的海藻酸盐结合起来制备成具有多层超细支柱3D结构的复合支架,物理表征显示,尽管与纯聚己内酯支架相比复合支架的杨氏模量有所降低,但观察到吸水率和润湿行为得到了显著改善;生物学方面,细胞接种效率和细胞活力得到显著改善,改善了支架的生物相容性;碱性磷酸酶活性检验和矿化分析显示,复合支架可以诱导细胞成骨分化,对成骨有显著积极作用。SHARIFI等[47]使用静电纺丝技术将羧甲基壳聚糖替代了聚己内脂/壳聚糖复合支架中的壳聚糖,物理性能方面改进了聚己内酯/壳聚糖复合支架中不良的超细纤维和分裂纤维,且暴露的羧甲基壳聚糖可以明显改善材料表面亲水性;使用MG63细胞进行实验显示,两种支架都具有良好的细胞相容性,但是聚己内酯/羧甲基壳聚糖复合支架表现出促进细胞增殖的优势,所以聚己内酯/羧甲基壳聚糖电纺支架可以成为骨组织工程应用的优良候选者。GOIMIL等[48]通过超临界CO2发泡法制备了包含聚己内酯基质、丝素蛋白(作为细胞黏附促进剂)和地塞米松(作为成骨诱导剂)的复合支架,首次制备了亚微米尺寸颗粒形式的具有β折叠结构的丝素蛋白气凝胶作为致孔剂,有效防止了支架制备过程中地塞米松的浸出或降解,材料表征显示掺入支架中的这些气凝胶改善了多孔结构并促进了细胞浸润和生物流体的运输;大鼠颅骨缺损模型修复实验显示出复合支架与组织极好的相容性,且没有发炎迹象,并且在缺损的不同位置促进了细胞附着及细胞外基质的发展。WU等[49]使用激光烧结方法制备了聚己内脂/海藻酸盐/聚丙烯酰胺复合支架,物理性能表征显示弹性模量从6.99 MPa(聚己内酯)增加为12.67 MPa(聚己内脂/海藻酸盐/聚丙烯酰胺),断裂伸长率由59%(聚己内酯)增加到112.9%(聚己内脂/海藻酸盐/聚丙烯酰胺);体外细胞实验显示细胞活力5 d内保持94%以上,提高了激光烧结技术在骨组织修复中的潜在应用。AZARUDEEN等[50]使用3D打印方法制备了以聚己内酯为基质的不同浓度的聚己内酯/明胶复合支架,使用间充质干细胞评价其生物学性能,在第21天内支架中明胶含量的增加导致亲水性的增强、更好的孔径分布和相互连接的微孔,增强了细胞的附着、增殖和成骨分化;但随着明胶含量的增加,复合支架的拉伸强度和弹性模量会随之降低,这也是此种支架存在的潜在问题之一。另外,SATTARY等[51]通过静电纺丝技术在聚己内酯/明胶支架中依次添加了羟基磷灰石和维生素D3,实验结果表明,聚己内酯/明胶/羟基磷灰石复合支架具有均匀的纤维直径和最佳的孔隙率水平及适当的孔径,羟基磷灰石的添加可以增强聚己内酯/明胶支架的力学性能;生物学实验表明,细胞在支架上的存活力和黏附性也有所改善;而后又在此支架的基础上添加了维生素D3,使用MG63细胞进行评价显示复合支架的细胞增殖和促成骨能力显著增加。总的来说,通过物理化学表征、形态学特性、高增殖率和良好的细胞黏附性及成骨细胞形态学特征结果证实,掺入羟基磷灰石和维生素 D3的聚己内酯/明胶复合支架是骨组织工程的合适候选者。 近期也有相关创新方法发表,MANAKHOV等[52]使用磁控溅射的方法将生物活性TiCaPCON膜沉积到聚己内酯纳米纤维上,通过调整实验参数达到较高质量的薄膜覆盖,并且避免聚己内酯基材的降解,MC3T3-E1细胞黏附和生长动力学分析表明该支架的生物相容性优良,肌动蛋白纤维的尖端可黏附在基质上,从而使细胞充分铺展;成骨细胞增殖和碱性磷酸酶活性检测表明,与纯聚己内酯支架相比,沉积TiCaPCON膜聚己内酯支架的细胞增殖率和碱性磷酸酶活性分别提高了约4倍和2倍。"
| [1] NG KW, HUTMACHER DW, SCHANTZ JT, et al. Evaluation of Ultra-Thin Poly(ε-Caprolactone) Films for Tissue-Engineered Skin. Tissue Eng. 2001;7(4):441-455. [2] ABEDALWAFA M, WANG F, WANG L, et al. Biodegradable poly-epsilon-caprolactone (PCL) for tissue engineering applications: A review. Rev Adv Mater Sci. 2013;34(2):123-140. [3] PUPPI D, CHIELLINI F, PIRAS AM, et al. Polymeric materials for bone and cartilage repair. Prog Polym Sci. 2010;35(4):403-440. [4] PARK YJ, CHA JH, BANG SI, et al. Clinical Application of Three-Dimensionally Printed Biomaterial Polycaprolactone (PCL) in Augmentation Rhinoplasty. J Aesthetic Plast Surg. 2019;43(2):437-446. [5] KANG X, XIE Y, POWELL HM, et al. Adipogenesis of murine embryonic stem cells in a three-dimensional culture system using electrospun polymer scaffolds. Biomaterials. 2007;28(3):450-458. [6] HOLZWARTH JM, MA PX. Biomimetic nanofibrous scaffolds for bone tissue engineering. Biomaterials. 2011;32(36):9622-9629. [7] Wang C, Huang W, Zhou Y, et al. 3D printing of bone tissue engineering scaffolds . Bioact Mater. 2020;5(1):82-91. [8] Labet M, Thielemans W. Synthesis of polycaprolactone: a review. Chem Soc Rev. 2009;38(12):3484-3504. [9] Espinoza SM, Patil HI, San Martin Martinez E, et al. Poly-ε-caprolactone (PCL), a promising polymer for pharmaceutical and biomedical applications: Focus on nanomedicine in cancer. Int J Polym Mater Polym Biomater. 2020;69(2):85-126. [10] STAVILA E, ALBERDA VAN EKENSTEIN GOR, WOORTMAN AJJ, et al. Lipase-Catalyzed Ring-Opening Copolymerization of ε-Caprolactone and β-Lactam. Biomacromolecules. 2014;15(1):234 -241. [11] MAHAPATRO A, KUMAR A, GROSS RA. Mild, Solvent-Free ω-Hydroxy Acid Polycondensations Catalyzed by Candida antarctica Lipase B. Biomacromolecules. 2004;5(1):62-68. [12] KIM YB, KIM GH. PCL/Alginate Composite Scaffolds for Hard Tissue Engineering: Fabrication, Characterization, and Cellular Activities. ACS Comb Sci. 2015;17(2):87-99. [13] CAMA G, MOGOSANU DE, HOUBEN A, et al. 3 - Synthetic biodegradable medical polyesters : Poly-ε-caprolactone. Int J Polym Mater Polym Biomater. 2017;10(3):79-105. [14] MONDAL D, GRIFFITH M, VENKATRAMAN SS. Polycaprolactone-based biomaterials for tissue engineering and drug delivery: Current scenario and challenges. Int J Polym Mater Polym Biomater. 2016;65(5): 255-265. [15] ZHAO H, NATHANIEL GA, MERENINI PC. Enzymatic ring-opening polymerization (ROP) of lactides and lactone in ionic liquids and organic solvents: digging the controlling factors. RSC Adv. 2017;7(77): 48639-48648. [16] ENGEL J, CORDELLIER A, HUANG L, et al. Enzymatic Ring-Opening Polymerization of Lactones: Traditional Approaches and Alternative Strategies. Chem Cat Chem. 2019;11(20):4983-4997. [17] BOUYAHYA A, BALIEU S, BENIAZZA R, et al. Organotin-bridged ionic liquid as a solvent-free, leaching-resistive catalyst for ring opening polymerization of ε-caprolactone. New J Chem. 2019;43(15): 5872-5878. [18] TOBIAS KM, KIDD CE, MULON PY, et al. Tensile properties of synthetic, absorbable monofilament suture materials before and after incubation in phosphate-buffered saline. Vet Surg. 2020;49(3):550-560. [19] Sinha V R, Bansal K, Kaushik R, et al. Poly-ϵ-caprolactone microspheres and nanospheres: an overview. Int J Appl Pharm. 2004; 278(1):1 -23. [20] PRASADH S, WONG RCW. Unraveling the mechanical strength of biomaterials used as a bone scaffold in oral and maxillofacial defects. Oral Sci Int. 2018;15(2):48 -55. [21] RAGAERT K, DE SOMER F, VAN DE VELDE S, et al. Methods for improved flexural mechanical properties of 3D-plotted PCL-based scaffolds for heart valve tissue engineering. J Mech Eng Sci. 2013;59(11):669-676. [22] MOHAMMADI MS, REZABEIGI E, BERTRAM J, et al. Poly(d,l-Lactic acid) Composite Foams Containing Phosphate Glass Particles Produced via Solid-State Foaming Using CO2 for Bone Tissue Engineering Applications. Polymers. 2020;12(1):12-18. [23] JOHNSTONE B, ALINI M, CUCCHIARINI M, et al. Tissue engineering for articular cartilage repair - the state of the art. Cells Mater. 2013;25: 248-267. [24] FABBRI P, BONDIOLI F, MESSORI M, et al. Porous scaffolds of polycaprolactone reinforced with in situ generated hydroxyapatite for bone tissue engineering. J Mater Sci Mate Med. 2010;21(1):343 -351. [25] CUI Z, LIN L, SI J, et al. Fabrication and characterization of chitosan/OGP coated porous poly(ε-caprolactone) scaffold for bone tissue engineering. J Biomater Sci Polym Ed. 2017;28(9):826-845. [26] REN K, WANG Y, SUN T, et al. Electrospun PCL/gelatin composite nanofiber structures for effective guided bone regeneration membranes. Mater Sci Eng. 2017;78:324-332. [27] MISZUK JM, XU T, YAO Q, et al. Functionalization of PCL-3D electrospun nanofibrous scaffolds for improved BMP2-induced bone formation. Appl Mater Res. 2018;10:194-202. [28] XUE W, CHEN P, WANG F, et al. Melt spinning of nano-hydroxyapatite and polycaprolactone composite fibers for bone scaffold application. J Mater Sci. 2019;54(11):8602-8612. [29] REN J, BLACKWOOD KA, DOUSTGANI A, et al. Melt-electrospun polycaprolactone strontium-substituted bioactive glass scaffolds for bone regeneration. J Biomed Mater Res A. 2014;102(9):3140-3153. [30] ZHONG M, LI J, TANG A, et al. A facile green approach for fabricating bacterial cellulose scaffold with macroporous structure and cell affinity. J Bioact Compat Polym. 2019;34(6):442-452. [31] GRANDI C, DI LIDDO R, PAGANIN P, et al. Porous alginate/poly(ε-caprolactone) scaffolds: Preparation, characterization and in vitro biological activity. Int J Mol Med. 2011;27(3):455-467. [32] PARK JH, JUNG SY, LEE CK, et al. A 3D-printed polycaprolactone/β-tricalcium phosphate mandibular prosthesis: A pilot animal study. Laryngoscope. 2020;130(2):358-366. [33] CHO YS, CHOI S, LEE SH, et al. Assessments of polycaprolactone/hydroxyapatite composite scaffold with enhanced biomimetic mineralization by exposure to hydroxyapatite via a 3D-printing system and alkaline erosion. Eur Polym J. 2019;113:340-348. [34] BITTNER SM, SMITH BT, DIAZ-GOMEZ L, et al. Fabrication and mechanical characterization of 3D printed vertical uniform and gradient scaffolds for bone and osteochondral tissue engineering. Acta Biomater. 2019;90:37-48. [35] PARK JS, KIM JM, LEE SJ, et al. Surface hydrolysis of fibrous poly(ε-caprolactone) scaffolds for enhanced osteoblast adhesion and proliferation. Macromol Res. 2007;15(5):424-429. [36] KARUPPUSWAMY P, REDDY VENUGOPAL J, NAVANEETHAN B, et al. Polycaprolactone nanofibers for the controlled release of tetracycline hydrochloride. Mater Lett. 2015;141:180-186. [37] DIAS AM, DA SILVA FG, DE FIGUEIREDO MONTEIRO AP, et al. Polycaprola ctone nanofibers loaded oxytetracycline hydrochloride and zinc oxide for treatment of periodontal disease. J Mater Sci Eng. 2019; 103:109798. [38] MOTAMEDIAN SR. Smart scaffolds in bone tissue engineering: A systematic review of literature. J Stem Cells. 2015;7(3):657-667. [39] ROI A, ARDELEAN LC, ROI CI, et al. Oral Bone Tissue Engineering: Advanced Biomaterials for Cell Adhesion, Proliferation and Differentiation. Materials. 2019;12(14):128-137. [40] GHOSAL K, MANAKHOV A, ZAJÍČKOVÁ L, et al. Structural and Surface Compatibility Study of Modified Electrospun Poly(ε-caprolactone) (PCL) Composites for Skin Tissue Engineering. AAPS Pharm Sci Tech. 2017; 18(1):72-81. [41] RUMINSKI S, OSTROWSKA B, JAROSZEWICZ J, et al. Three-dimensional printed polycaprolactone-based scaffolds provide an advantageous environment for osteogenic differentiation of human adipose-derived stem cells. J Tissue Eng Regener Med. 2018;12(1):473-485. [42] MOEINI S, MOHAMMADI MR, SIMCHI A. In-situ solvothermal processing of polycaprolactone/hydroxyapatite nanocomposites with enhanced mechanical and biological performance for bone tissue engineering. Bioact Mater. 2017;2(3):146-155. [43] RONCA D, LANGELLA F, CHIERCHIA M, et al. Bone Tissue Engineering: 3D PCL-based Nanocomposite Scaffolds with Tailored Properties. Proc CIRP. 2016;49:51-54. [44] HASSAN MI, SULTANA N, HAMDAN S. Bioactivity assessment of poly(ε -caprolactone)/hydroxyapatite electrospun fibers for bone tissue engineering application. J Nanomat. 2014;3(5):15-27 [45] BAO C, CHONG MSK, QIN L, et al. Effects of tricalcium phosphate in polycaprolactone scaffold for mesenchymal stem cell-based bone tissue engineering. Mat Tech. 2019;3(5):1-7. [46] SHIM KS, KIM SE, YUN YP, et al. Surface immobilization of biphasic calcium phosphate nanoparticles on 3D printed poly(caprolactone) scaffolds enhances osteogenesis and bone tissue regeneration. J Ind Eng Chem. 2017;55:101-109. [47] SHARIFI F, ATYABI SM, NOROUZIAN D, et al. Polycaprolactone/carboxymethyl chitosan nanofibrous scaffolds for bone tissue engineering application. Int J Biol Macromol. 2018;115:243-248. [48] GOIMIL L, SANTOS-ROSALES V, DELGADO A, et al. scCO2-foamed silk fibroin aerogel/poly(ε-caprolactone) scaffolds containing dexamethasone for bone regeneration. J CO2 Util. 2019;31:51-64. [49] WU J, YANG R, ZHENG J, et al. Fabrication and improvement of PCL/alginate/PAAm scaffold via selective laser sintering for tissue engineering. Micro Nano Lett. 2019;14(8):852-855. [50] AZARUDEEN RS, HASSAN MN, YASSIN MA, et al. 3D printable Polycaprolactone-gelatin blends characterized for in vitro osteogenic potency. React Funct Polym. 2020;146:104445. [51] SATTARY M, KHORASANI M T, RAFIENIA M, et al. Incorporation of nanohydroxyapatite and vitamin D3 into electrospun PCL/Gelatin scaffolds: The influence on the physical and chemical properties and cell behavior for bone tissue engineering. Polym Adv Technol. 2018; 29(1):451-462. [52] MANAKHOV A, PERMYAKOVA ES, ERSHOV S, et al. Bioactive TiCaPCON-coated PCL nanofibers as a promising material for bone tissue engineering. Appl Surf Sci. 2019;479:796-802. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Xu Feng, Kang Hui, Wei Tanjun, Xi Jintao. Biomechanical analysis of different fixation methods of pedicle screws for thoracolumbar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1313-1317. |
| [3] | Jiang Yong, Luo Yi, Ding Yongli, Zhou Yong, Min Li, Tang Fan, Zhang Wenli, Duan Hong, Tu Chongqi. Von Mises stress on the influence of pelvic stability by precise sacral resection and clinical validation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1318-1323. |
| [4] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [5] | Zhang Yu, Tian Shaoqi, Zeng Guobo, Hu Chuan. Risk factors for myocardial infarction following primary total joint arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1340-1345. |
| [6] | Wei Wei, Li Jian, Huang Linhai, Lan Mindong, Lu Xianwei, Huang Shaodong. Factors affecting fall fear in the first movement of elderly patients after total knee or hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1351-1355. |
| [7] | Wang Jinjun, Deng Zengfa, Liu Kang, He Zhiyong, Yu Xinping, Liang Jianji, Li Chen, Guo Zhouyang. Hemostatic effect and safety of intravenous drip of tranexamic acid combined with topical application of cocktail containing tranexamic acid in total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1356-1361. |
| [8] | Xiao Guoqing, Liu Xuanze, Yan Yuhao, Zhong Xihong. Influencing factors of knee flexion limitation after total knee arthroplasty with posterior stabilized prostheses [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1362-1367. |
| [9] | Huang Zexiao, Yang Mei, Lin Shiwei, He Heyu. Correlation between the level of serum n-3 polyunsaturated fatty acids and quadriceps weakness in the early stage after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1375-1380. |
| [10] | Zhang Chong, Liu Zhiang, Yao Shuaihui, Gao Junsheng, Jiang Yan, Zhang Lu. Safety and effectiveness of topical application of tranexamic acid to reduce drainage of elderly femoral neck fractures after total hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1381-1386. |
| [11] | Wang Haiying, Lü Bing, Li Hui, Wang Shunyi. Posterior lumbar interbody fusion for degenerative lumbar spondylolisthesis: prediction of functional prognosis of patients based on spinopelvic parameters [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1393-1397. |
| [12] | Lü Zhen, Bai Jinzhu. A prospective study on the application of staged lumbar motion chain rehabilitation based on McKenzie’s technique after lumbar percutaneous transforaminal endoscopic discectomy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1398-1403. |
| [13] | Chen Xinmin, Li Wenbiao, Xiong Kaikai, Xiong Xiaoyan, Zheng Liqin, Li Musheng, Zheng Yongze, Lin Ziling. Type A3.3 femoral intertrochanteric fracture with augmented proximal femoral nail anti-rotation in the elderly: finite element analysis of the optimal amount of bone cement [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1404-1409. |
| [14] | Du Xiupeng, Yang Zhaohui. Effect of degree of initial deformity of impacted femoral neck fractures under 65 years of age on femoral neck shortening [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1410-1416. |
| [15] | Zhang Shangpu, Ju Xiaodong, Song Hengyi, Dong Zhi, Wang Chen, Sun Guodong. Arthroscopic suture bridge technique with suture anchor in the treatment of acromioclavicular dislocation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1417-1422. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||