Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (18): 2921-2926.doi: 10.3969/j.issn.2095-4344.0766
Previous Articles Next Articles
Evaluation methodology of cellular uptake of magnetic nanoparticles
Wang Run-sheng1, 2, Liu Yi-hao3, Mao Ke-ya1
- 1Department of Orthopedics, Chinese PLA General Hospital, Beijing 100853, China; 2Department of Orthopedics, Third Affiliated Hospital of Guangxi University of Chinese Medicine, Liuzhou 545001, Guangxi Zhuang Autonomous Region, China; 3Jiangsu University, Zhenjiang 212013, Jiangsu Province, China
-
Received:2018-01-05Online:2018-06-28Published:2018-06-28 -
Contact:Mao Ke-ya, Chief physician, Department of Orthopedics, Chinese PLA General Hospital, Beijing 100853, China -
About author:Wang Run-sheng, Doctoral candidate, Attending physician, Department of Orthopedics, Chinese PLA General Hospital, Beijing 100853, China; Department of Orthopedics, Third Affiliated Hospital of Guangxi University of Chinese Medicine, Liuzhou 545001, Guangxi Zhuang Autonomous Region, China
CLC Number:
Cite this article
Wang Run-sheng, Liu Yi-hao, Mao Ke-ya. Evaluation methodology of cellular uptake of magnetic nanoparticles[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(18): 2921-2926.
share this article
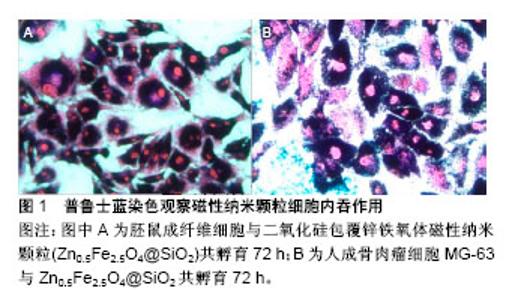
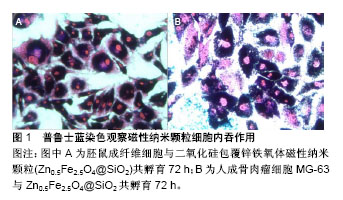
2.1 定性评价方法 2.1.1 普鲁士蓝染色 利用磁性纳米颗粒内含有铁元素,与普鲁士蓝试剂发生反应能生成蓝色沉淀,再通过伊红/核固红复染,增强染色对比,内吞磁性纳米颗粒的细胞被染成蓝色,而未内吞磁性纳米颗粒的细胞未出现蓝色沉淀(图1)。普鲁士蓝染色是研究磁性纳米颗粒细胞内吞最常用的定性研究方法[25-26],因磁性纳米颗粒内吞细胞后多位于细胞质的溶酶体内[27],故光学显微镜下多表现为细胞质染成蓝色,而细胞核未见蓝染[28]。Makridis等[29]采用普鲁士蓝染色观察骨肉瘤SaO-2细胞对MnFe2O4/Fe3O4和CoFe2O4/Fe3O4磁性纳米颗粒细胞内吞情况,共孵育48 h后,SaO-2细胞完全或部分变成蓝色,表明颗粒被肿瘤细胞内吞,当磁性纳米颗粒被肿瘤细胞内吞后再通过交变磁场进行细胞内热疗,提高肿瘤细胞杀伤率。普鲁士蓝染色后,利用酶标仪采用比色法比较吸光度值差异进行定量对比,检测细胞内磁性纳米含量[30],为普鲁士蓝染色定量评估细胞内吞提供新思路。但普鲁士蓝染色也存在缺点,细胞外颗粒洗脱不净可能导致假阳性结果,因此,普鲁士蓝染色评价细胞内吞需要结合荧光显微镜、透射电镜等检查。"
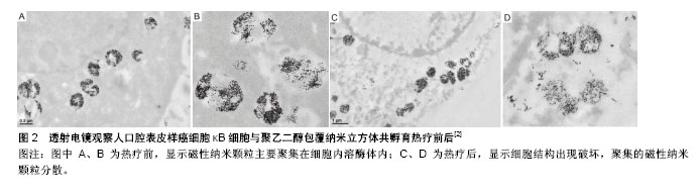
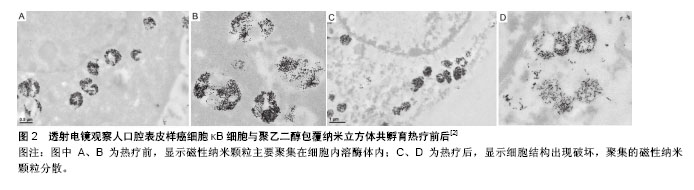
2.1.2 荧光显微镜染色 磁性纳米颗粒经表面修饰后,在其表面插入荧光素,将细胞核、细胞膜或细胞骨架采用不同荧光染料进行染色,通过紫外线激发荧光,当绑定荧光素的磁性纳米颗粒、细胞膜或细胞骨架、细胞核3种荧光重叠时,可观察磁性纳米颗粒在细胞内的位置。采用针对不同细胞器的荧光探针,可观察磁性纳米颗粒内吞细胞后,在亚细胞结构中的位置。Zhang等[27]采用溶酶体荧光探针Lysotracker将溶酶体标记后,可观察绑定荧光素TRITC的磁性纳米颗粒位于溶酶体内。利用溶酶体荧光探针Lysosensor随pH变化的特征,可实时观察动态磁场作用下,由于溶酶体膜破裂导致溶酶体pH变化后绿色荧光的强弱变化情况,判断磁性纳米颗粒在动态磁场中诱导肿瘤细胞凋亡的原因可能与溶酶体膜破裂有关。Shah等[31]将壳核结构磁性纳米颗粒与线粒体靶向促凋亡两性锚定肽连接,再在表面修饰靶向配体iRGD,并绑定荧光素(Alexa-594),通过荧光显微镜观察其对整联蛋白高表达的人胶质瘤细胞系U87具有靶向性,并可增加内吞量,联合热疗作用,可诱导肿瘤细胞通过线粒体途径凋亡。磁性纳米颗粒表面绑定荧光素是荧光显微镜观察磁性纳米颗粒内吞的前提条件,荧光素绑定率可能会影响观察结果。 2.1.3 共聚焦显微镜 磁性纳米颗粒、细胞膜、细胞核荧光标记后,通过激光激发荧光探针发出不同颜色荧光,观察磁性纳米颗粒在细胞内的位置,所得图像较荧光显微镜更为清晰,而且可通过断层扫描、三维重建观察细胞及亚细胞结构。共聚焦显微镜是研究磁性纳米颗粒内吞、颗粒细胞内示踪、磁性纳米颗粒热疗、载药等治疗后细胞特征形态变化的重要观察手段[32]。Sanz等[7]将磁性纳米颗粒采用Alexa 488荧光探针标记,细胞核采用DAPI荧光探针标记,通过共聚焦显微镜观察磁性纳米颗粒内吞入细胞后,主要位于细胞质中,细胞核中无磁性纳米颗粒,磁热疗后细胞核出现形态改变,且磁热疗较水浴热疗细胞核形态变化(核固缩)更明显。Connord等[33]设计微型动态磁场,将微型磁场置于细胞培养皿中,通过共聚焦显微镜动态观察内吞荧光标记磁性纳米颗粒的肿瘤细胞死亡,可能与溶酶体膜的通透性改变、ROS释放有关,而且观察到内吞入肿瘤细胞的磁性纳米颗粒,在微型动态磁场的变化下由散乱分布而数分钟内变为线性排列现象。 2.1.4 透射电镜 透射电镜是观察磁性纳米颗粒与细胞关系最直接的评价方法[34],可观察到磁性纳米颗粒在细胞内的分布及与细胞器的关系,并可观察治疗前后磁性纳米颗粒在细胞内的聚集、分散情况。Di Corato等[35]通过透射电镜观察5种常用磁性纳米颗粒在4-37 ℃下与细胞的关系,发现4 ℃时磁性纳米颗粒主要位于细胞膜上,而37 ℃时磁性纳米颗粒被肿瘤细胞内吞进入细胞内,在溶酶体内聚集,从而验证细胞内吞磁性纳米颗粒是主动内吞,需消耗能量。Guardia等[2]通过透射电镜可观察到与细胞共孵育后,磁性纳米颗粒主要位于细胞溶酶体内,且在溶酶体内聚集,进行磁热疗后,磁性纳米颗粒随着溶酶体膜的破裂而逐渐分散(图2)。Zhang 等[27]将磁性纳米颗粒荧光标记并连接溶酶体膜相关蛋白1抗体,内吞入细胞后与溶酶体膜特异性紧密结合,在低频动态磁场作用下使磁性纳米颗粒发生旋转,通过机械力撕裂溶酶体膜导致溶酶体内容物释放,用透射电镜观察到在动态磁场施加前后,细胞内的磁性纳米颗粒由聚集状态变成分散状态。"

2.1.5 光热显微镜 光热显微镜又称光热单粒子显微镜,可观察非荧光标记的纳米颗粒,主要依赖于检测粒子光热吸收特性的不同检测粒子,其信号强弱与激光束强度和纳米颗粒光热吸收率呈正比例[36],是将宏观的光热光谱扩展到纳米级领域,具有高灵敏度和选择性,这项技术目前主要用于观察金属纳米颗粒,如金、银、量子点、单畴碳纳米管。Bogart等[37]采用光热显微镜观察右旋糖酐包埋的磁性纳米颗粒细胞内吞过程,并与荧光显微镜、普鲁士蓝染色、透视电镜观察结果进行对比,结果提示光热显微镜观察的结果与以上传统方法观察结果一致,不但可观察固定的细胞、冰冻切片,而且可用于观察活细胞。除定性观察外,又根据光热信号的差异进行定量分析,与电感耦合等离子体质谱分析仪和普鲁士蓝染色比色法两种定量分析结果一致。光热显微镜具有以下优势:对细胞无损伤,可进行活细胞观察;具有高敏感性和分辨率,可精确观察低剂量磁性纳米颗粒在细胞内的位置。 2.1.6 原子力显微镜 原子力显微镜主要用于包括绝缘体在内的固体材料表面结构分析,通过检测样品表面和微型力敏感元件之间的极微弱原子间相互作用力来研究物质的表面结构及性质,可纳米级分辨率获得表面形貌结构信息及表面粗糙度信息,也用于分析细胞表面特征及生物大分子。Silva等[38]通过原子力显微镜动态观察聚乙二醇2000修饰磁性纳米颗粒内吞入NIH-3T3细胞的过程,将磁性纳米颗粒与NIH-3T3共孵育30 min后,可实时观察到细胞膜表面由于磁性纳米颗粒内吞逐渐形成的内吞坑和凹陷(图3),为磁性纳米颗粒细胞内吞提供实时、直观的观察方法。 2.2 定量评价方法 2.2.1 电感耦合等离子体 包括电感耦合等离子体质谱分析仪和电感耦合等离子体原子发射光谱仪,前者是检测离子质谱,后者是检测光学光谱,通过电感耦合等离子体检测,可明确内吞磁性纳米颗粒内金属元素的含量,联合细胞计数可分析单个细胞平均磁性纳米颗粒内吞量,进行定量评价,电感耦合等离子体是目前检测磁性纳米颗粒内吞最常用、最为准确的定量方法。Datta等[22]采用电感耦合等离子体质谱分析仪研究人乳腺癌细胞MDA-MB 231对壳聚糖包覆MnFe2O4磁性纳米颗粒的内吞作用,分别检测24,48,72 h内吞量,发现内吞量呈时间依耐性。Kyoung等[39]采用电感耦合等离子体联合透射电镜、共聚焦显微镜研究脉冲磁场、永磁场、无磁场作用下,磁性纳米颗粒内吞量的变化,发现脉冲磁场可减少磁性纳米颗粒在细胞膜表面聚集,较永磁场和无磁场更利于磁性纳米颗粒的内吞。 2.2.2 磁化强度测定 先测定单位质量磁性纳米颗粒的磁化强度,当磁性纳米颗粒被细胞内吞后,将细胞外磁性纳米颗粒冲洗后,再将内吞磁性纳米颗粒的细胞消化、离心,通过磁强计测定标本中磁性纳米颗粒的磁化强度,计算磁性纳米颗粒的质量,结合细胞计数进行定量分析。Zysler等[40]采用磁化强度测定方法可对组织内的磁性纳米颗粒进行定量分析,其测量精度可达1 μg。Silva等[38]采用磁化强度测量方法比较3种细胞系对聚乙二醇350和聚乙二醇2000包覆超顺磁性纳米颗粒的内吞量差异进行定量分析。但该方法对磁强计的测量精度要求高,如细胞数量少、内吞的磁性纳米颗粒总质量少会导致误差较大。 2.2.3 普鲁士蓝染色比色法 根据细胞内吞磁性纳米颗粒量不同,细胞内含有铁元素质量不同,利用普鲁士蓝试剂反应后细胞悬液色阶差异,采用分光光度计或酶标仪进行检测,可间接得到细胞内吞磁性纳米颗粒的含量变化趋势。Yallapu等[30]研究磁性纳米颗粒与血清蛋白共孵育后理化及生物特性的变化,普鲁士蓝染色后采用酶标仪观测发现,人前列腺癌C4-2B和胰腺癌细胞Panc-1对与血清共孵育的磁性纳米颗粒内吞量,随血清浓度增高内吞量增大,从而验证了磁性纳米颗粒与血清混合后会影响细胞对磁性纳米颗粒的内吞。 "

| [1] Hervault A,Thanh NT.Magnetic nanoparticle-based therapeutic agents for thermo-chemotherapy treatment of cancer.Nanoscale. 2014;6(20): 11553-11573.[2] Guardia P,Di Corato R,Lartigue L,et al.Water-Soluble Iron Oxide Nanocubes with High Values of Specific Absorption Rate for Cancer Cell Hyperthermia Treatment.ACS Nano.2012;6(4):3080-3091.[3] Fang J,Nakamura H,Maeda H.The EPR effect: unique features of tumor blood vessels for drug delivery, factors involved, and limitations and augmentation of the effect.Adv Drug Deliv Rev. 2011;63(3): 136-151.[4] Ding W,Guo L.Immobilized transferrin Fe3O4@SiO2 nanoparticle with high doxorubicin loading for dual-targeted tumor drug delivery.Int J Nanomedicine.2013;8:4631-4639.[5] Kaur P,Aliru ML,Chadha AS,et al.Hyperthermia using nanoparticles-promises and pitfalls.Int J Hyperther. 2016;32:76-88.[6] Dennis CL,Ivkov R.Physics of heat generation using magnetic nanoparticles for hyperthermia.Int J Hyperther. 2013;29:715-729.[7] Sanz B,Calatayud MP,Torres TE,et al.Magnetic hyperthermia enhances cell toxicity with respect to exogenous heating. Biomaterials. 2017;114:62-70.[8] Kaur P,Aliru ML,Chadha AS,et al.Hyperthermia using nanoparticles- promises and pitfalls.Int J Hyperther. 2016;32:76-88.[9] Dennis CL,Ivkov R.Physics of heat generation using magnetic nanoparticles for hyperthermia.Int J Hyperther 2013;29:715-729.[10] Zhou Z,Sun Y,Shen J,etal.Iron/iron oxide core/shell nanoparticles for magnetic targeting MRI and near-infrared photothermal therapy. Biomaterials.2014;35(26):7470-7478.[11] Tong S,Hou SJ,Zheng ZL,et al.Coating Optimization of Superparamagnetic Iron Oxide Nanoparticles for High T-2 Relaxivity. Nano Lett.2010;10:4607-4613.[12] Lee SY,Yang CY,Peng CL,et al.A theranostic micelleplex co-delivering SN-38 and VEGF siRNA for colorectal cancer therapy.Biomaterials. 2016;86:92-105. [13] Baldi G,Ravagli C,Mazzantini F,et al.In vivo anticancer evaluation of the hyperthermic efficacy of anti-human epidermal growth factor receptor-targeted PEG-based nanocarrier containing magnetic nanoparticles.Int J Nanomedicine.2014;9:3037-3056.[14] Lee N,Kim H,Choi SH,et al.Magnetosome-like ferrimagnetic iron oxide nanocubes for highly sensitive MRI of single cells and transplanted pancreatic islets.Proc Natl Acad Sci U S A. 2011;108(7):2662-2667.[15] Nandwana V,Ryoo SR,Kanthala S,et al.Engineered Theranostic Magnetic Nanostructures: Role of Composition and Surface Coating on Magnetic Resonance Imaging Contrast and Thermal Activation.ACS Appl Mater Interfaces. 2016;8(11):6953-6961.[16] Knecht LD,Ali N,Wei Y,et al.Nanoparticle-mediated remote control of enzymatic activity.ACS Nano. 2012;6(10):9079-9086.[17] Guo M,Que C,Wang C,et al.Multifunctional superparamagnetic nanocarriers with folate-mediated and pH-responsive targeting properties for anticancer drug delivery.Biomaterials. 2011;32(1): 185-194.[18] Li F,Lu J,Kong X,et al.Dynamic Nanoparticle Assemblies for Biomedical Applications.Adv Mater. 2017;29(14). doi:10.1002/adma.201605897.[19] Zhang ZQ,Song SC.Multiple hyperthermia-mediated release of TRAIL/SPION nanocomplex from thermosensitive polymeric hydrogels for combination cancer therapy. Biomaterials.2017;132:16-27.[20] Yoo D,Jeong H,Noh SH,et al.Magnetically triggered dual functional nanoparticles for resistance-free apoptotic hyperthermia.Angew Chem Int Ed Engl. 2013;52(49):13047-13051.[21] Yang HW,Liu HL,Li ML,et al.Magnetic gold-nanorod/ PNIPAAmMA nanoparticles for dual magnetic resonance and photoacoustic imaging and targeted photothermal therapy. Biomaterials. 2013;34(22): 5651-5660.[22] Datta NR,Krishnan S,Speiser DE,et al.Magnetic nanoparticle-induced hyperthermia with appropriate payloads: Paul Ehrlich's "magic (nano)bullet" for cancer theranostics? Cancer Treat Rev. 2016;50: 217-227.[23] Tarangelo A,Dixon SJ.Nanomedicine: An iron age for cancer therapy. Nat Nanotechnol.2016;11(11):921-922.[24] Goya GF,Asín L,Ibarra MR.Cell death induced by AC magnetic fields and magnetic nanoparticles:Current state and perspectives.Int J Hyperthermia.2013;29(8):810-818.[25] Xie J,Zhang Y,Yan C,et al.High-performance PEGylated Mn-Zn ferrite nanocrystals as a passive-targeted agent for magnetically induced cancer theranostics.Biomaterials. 2014;35(33):9126-9136. [26] Yallapu MM,Othman SF,Curtis ET,et al.Curcumin-loaded magnetic nanoparticles for breast cancer therapeutics and imaging applications.Int J Nanomedicine.2012;7:1761-1779.[27] Zhang E,Kircher MF,Koch M,et al.Dynamic magnetic fields remote-control apoptosis via nanoparticle rotation.ACS Nano. 2014;8(4):3192-3201.[28] Oh Y,Lee N,Kang HW,et al.In vitro study on apoptotic cell death by effective magnetic hyperthermia with chitosan-coated MnFe(2)O(4).Nanotechnology. 2016;27(11):115101-115122.[29] Makridis A,Tziomaki M,Topouridou K,et al.A novel strategy combining magnetic particle hyperthermia pulses with enhanced performance binary ferrite carriers for effective in vitro manipulation of primary human osteogenic sarcoma cells.Int J Hyperthermia. 2016;32(7): 778-785. [30] Yallapu MM,Chauhan N,Othman SF,et al.Implications of protein corona on physico-chemical and biological properties of magnetic nanoparticles.Biomaterials.2015;46:1-12.[31] Shah BP,Pasquale N,De G,et al.Core-shell nanoparticle-based peptide therapeutics and combined hyperthermia for enhanced cancer cell apoptosis.ACS Nano.2014;8(9):9379-9387.[32] Creixell M,Bohórquez AC,Torres-Lugo M,et al.EGFR-targeted magnetic nanoparticle heaters kill cancer cells without a perceptible temperature rise.ACS Nano.2011;5(9):7124-7129.[33] Connord V,Clerc P,Hallali N,et al.Real-Time Analysis of Magnetic Hyperthermia Experiments on Living Cells under a Confocal Microscope.Small.2015;11(20):2437-2445.[34] Ding X,Liu Y,Li J,et al.Hydrazone-bearing PMMA-functionalized magnetic nanocubes as pH-responsive drug carriers for remotely targeted cancer therapy in vitro and in vivo.ACS Appl Mater Interfaces. 2014;6(10):7395-7407.[35] Di Corato R,Espinosa A,Lartigue L,et al.Magnetic hyperthermia efficiency in the cellular environment for different nanoparticle designs.Biomaterials. 2014;35(24):6400-6411. [36] Berciaud S,Lasne D,Blab GA,et al.Photothermal Heterodyne Imaging of Individual Metallic Nanoparticles: Theory versus Experiment.Phys Rev B.2006;73:045424.[37] Bogart LK,Taylor A,Cesbron Y,et al.Photothermal microscopy of the core of dextran-coated iron oxide nanoparticles during cell uptake.ACS Nano.2012;6(7):5961-5971.[38] Silva AH,Lima E Jr,Mansilla MV,et al.Superparamagnetic iron-oxide nanoparticles mPEG350- and mPEG2000-coated: cell uptake and biocompatibility evaluation.Nanomedicine. 2016;12(4):909-919.[39] Kyoung Ah Min,Meong Cheol Shin,Faquan Yu,et al.Pulsed Magnetic Field Improves the Transport of Iron Oxide Nanoparticles through Cell Barriers.Acs Nano. 2013;7(3):2161-2171.[40] Zysler RD,Lima E Jr,Vasquez Mansilla M,et al.A New Quantitative Method to Determine the Uptake of SPIONs in Animal Tissue and Its Application to Determine the Quantity of Nanoparticles in the Liver and Lung of Balb-c Mice Exposed to the SPIONs.J Biomed Nanotechnol. 2013;9(1):142-145.[41] Hildebrandt B, Peter Wustp, Thoralf Kerner T, et al. The cellular and molecular basis of hyperthermia.Eur J Cancer Care (Engl). 2002;43: 33-56.[42] Gordon RT,Hines JR,Gordon D.Intracellular hyperthermia. A biophysical approach to cancer treatment via intracellular temperature and biophysical alterations.Med Hypotheses. 1979;5(1):83-102.[43] Min KA,Shin MC,Yu F,et al.Pulsed Magnetic Field Improves the Transport of Iron Oxide Nanoparticles through Cell Barriers.ACS Nano. 2013;7(3):2161-2171.[44] Luetke A,Meyers PA,Lewis I,et al.Osteosarcoma treatment - where do we stand? A state of the art review.Cancer Treat Rev. 2014;40(4): 523-532.[45] Mou X,Ali Z,Li S,et al.Applications of Magnetic Nanoparticles in Targeted Drug Delivery System.J Nanosci Nanotechnol. 2015;15(1): 54-62.[46] Grossman JH,McNeil SE.Nanotechnology in Cancer Medicine.Physics Today.2012;65(8):38-42.[47] Yoo D,Jeong H,Noh SH,et al.Magnetically triggered dual functional nanoparticles for resistance-free apoptotic hyperthermia.Angew Chem Int Ed Engl. 2013;52(49):13047-13051. |
| [1] | Chen Ziyang, Pu Rui, Deng Shuang, Yuan Lingyan. Regulatory effect of exosomes on exercise-mediated insulin resistance diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 4089-4094. |
| [2] | Chen Yang, Huang Denggao, Gao Yuanhui, Wang Shunlan, Cao Hui, Zheng Linlin, He Haowei, Luo Siqin, Xiao Jingchuan, Zhang Yingai, Zhang Shufang. Low-intensity pulsed ultrasound promotes the proliferation and adhesion of human adipose-derived mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3949-3955. |
| [3] | Yang Junhui, Luo Jinli, Yuan Xiaoping. Effects of human growth hormone on proliferation and osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3956-3961. |
| [4] | Sun Jianwei, Yang Xinming, Zhang Ying. Effect of montelukast combined with bone marrow mesenchymal stem cell transplantation on spinal cord injury in rat models [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3962-3969. |
| [5] | Gao Shan, Huang Dongjing, Hong Haiman, Jia Jingqiao, Meng Fei. Comparison on the curative effect of human placenta-derived mesenchymal stem cells and induced islet-like cells in gestational diabetes mellitus rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3981-3987. |
| [6] | Hao Xiaona, Zhang Yingjie, Li Yuyun, Xu Tao. Bone marrow mesenchymal stem cells overexpressing prolyl oligopeptidase on the repair of liver fibrosis in rat models [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3988-3993. |
| [7] | Liu Jianyou, Jia Zhongwei, Niu Jiawei, Cao Xinjie, Zhang Dong, Wei Jie. A new method for measuring the anteversion angle of the femoral neck by constructing the three-dimensional digital model of the femur [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3779-3783. |
| [8] | Meng Lingjie, Qian Hui, Sheng Xiaolei, Lu Jianfeng, Huang Jianping, Qi Liangang, Liu Zongbao. Application of three-dimensional printing technology combined with bone cement in minimally invasive treatment of the collapsed Sanders III type of calcaneal fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3784-3789. |
| [9] | Qian Xuankun, Huang Hefei, Wu Chengcong, Liu Keting, Ou Hua, Zhang Jinpeng, Ren Jing, Wan Jianshan. Computer-assisted navigation combined with minimally invasive transforaminal lumbar interbody fusion for lumbar spondylolisthesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3790-3795. |
| [10] | Hu Jing, Xiang Yang, Ye Chuan, Han Ziji. Three-dimensional printing assisted screw placement and freehand pedicle screw fixation in the treatment of thoracolumbar fractures: 1-year follow-up [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3804-3809. |
| [11] | Shu Qihang, Liao Yijia, Xue Jingbo, Yan Yiguo, Wang Cheng. Three-dimensional finite element analysis of a new three-dimensional printed porous fusion cage for cervical vertebra [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3810-3815. |
| [12] | Wang Yihan, Li Yang, Zhang Ling, Zhang Rui, Xu Ruida, Han Xiaofeng, Cheng Guangqi, Wang Weil. Application of three-dimensional visualization technology for digital orthopedics in the reduction and fixation of intertrochanteric fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3816-3820. |
| [13] | Sun Maji, Wang Qiuan, Zhang Xingchen, Guo Chong, Yuan Feng, Guo Kaijin. Development and biomechanical analysis of a new anterior cervical pedicle screw fixation system [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3821-3825. |
| [14] | Lin Wang, Wang Yingying, Guo Weizhong, Yuan Cuihua, Xu Shenggui, Zhang Shenshen, Lin Chengshou. Adopting expanded lateral approach to enhance the mechanical stability and knee function for treating posterolateral column fracture of tibial plateau [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3826-3827. |
| [15] | Zhu Yun, Chen Yu, Qiu Hao, Liu Dun, Jin Guorong, Chen Shimou, Weng Zheng. Finite element analysis for treatment of osteoporotic femoral fracture with far cortical locking screw [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3832-3837. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||