Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (34): 9017-9023.doi: 10.12307/2026.870
Previous Articles Next Articles
The functions and underlying molecular mechanisms of PIEZO channels in nervous system diseases
Liu Yuxiao1, Huang Sijing1, Geng Longyu1, Gao Beiyao2, Yang Guang3, Ge Ruidong2, Gao Qi1
- 1School of Sports Medicine and Rehabilitation, Beijing Sport University, Beijing 100091, China; 2Department of Rehabilitation Medicine, China-Japan Friendship Hospital, Beijing 100029, China; 3Jingzhong Medical District of the PLA General Hospital, Beijing 100088, China
-
Received:2025-10-29Revised:2026-01-29Online:2026-12-08Published:2026-04-14 -
Contact:Ge Ruidong, PhD, Associate chief therapeutist, Department of Rehabilitation Medicine, China-Japan Friendship Hospital, Beijing 100029, China -
About author:Liu Yuxiao, MS candidate, School of Sports Medicine and Rehabilitation, Beijing Sport University, Beijing 100091, China -
Supported by:National High-Level Hospital Clinical Research Funding, No. 2025-NHLHCRF-PY-41 (to GRD)
CLC Number:
Cite this article
Liu Yuxiao, Huang Sijing, Geng Longyu, Gao Beiyao, Yang Guang, Ge Ruidong, Gao Qi. The functions and underlying molecular mechanisms of PIEZO channels in nervous system diseases[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 9017-9023.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
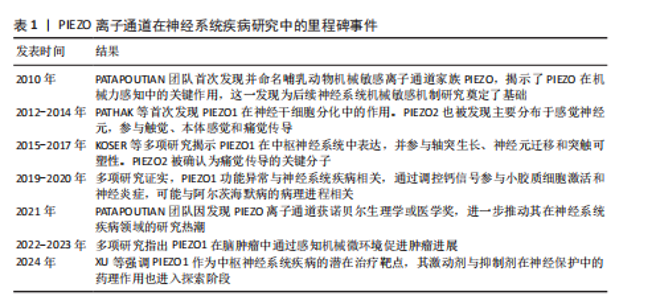
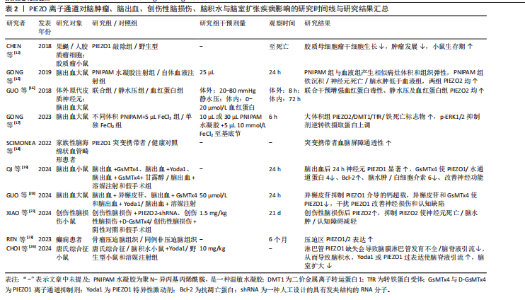
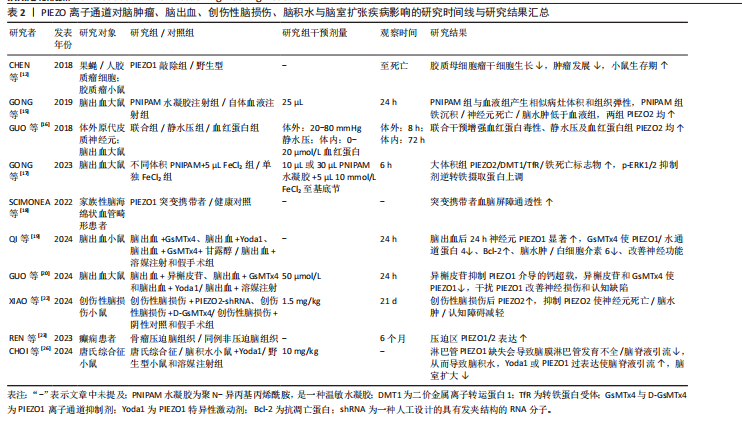
自2010年Patapoutian团队首次发现PIEZO离子通道家族以来,这类机械敏感通道在神经系统疾病中的作用逐渐成为研究热点。PIEZO1和PIEZO2作为机械力感知的核心通道,不仅参与神经发育和痛觉传导等生理过程,其功能异常还与脑出血、阿尔茨海默病、偏头痛等多种神经系统疾病密切相关。现总结PIEZO离子通道在神经系统疾病研究中的里程碑事件,见表1。 2.1 中枢神经系统疾病 2.1.1 脑肿瘤 脑肿瘤是指在大脑或中枢神经系统其他部分形成的异常细胞增生[11]。研究表明,PIEZO1离子通道在胶质瘤的病程中扮演重要角色[12],对PIEZO1进行敲除可以阻止肿瘤细胞有丝分裂,显著减小肿瘤体积,并减少总胶质细胞数量,这表明PIEZO1离子通道不仅参与了肿瘤细胞的增殖过程,而且可能参与肿瘤微环境的硬度重塑过程,间接调控了其他胶质细胞的行为。同时,在人脑胶质母细胞瘤干细胞中,PIEZO1离子通道的表达水平与患者生存期呈负相关。研究进一步对脑肿瘤小鼠模型使用shRNA技术敲低PIEZO1离子通道表达,发现胶质母细胞瘤干细胞的机械敏感性受到抑制,具体表现为细胞对机械刺激做出反应的能力减退、胶质母细胞瘤干细胞系的克隆能力被抑制,并且肿瘤球的形成能力降低,这有效抑制了肿瘤细胞的生长,延长了小鼠的生存时间。PIEZO1离子通道在正常脑组织中表达较低,但在胶质母细胞瘤中高度表达,表达水平与肿瘤恶性程度呈正相关[13],这提示PIEZO1离子通道可能成为治疗恶性胶质瘤的一个潜在靶点。 2.1.2 脑出血 脑出血是指大脑实质内血管破裂导致的出血,有时会扩展到脑室,甚至在极少数情况下到达蛛网膜下腔[14]。PIEZO2离子通道作为机械敏感离子通道,可感知物理压力的变化,从而使钙离子进入细胞内,其含量在脑出血后的病理过程中呈上升趋势[15-16]。在雄性SD大鼠脑出血模型中,随着质量效应(即由于出血导致的物理压力增加)的增强[17],PIEZO2蛋白表达量显著上调,这表明PIEZO2离子通道在神经元中被质量效应激活。同时,铁代谢路径中的关键蛋白质如二价金属转运体1和转铁蛋白受体的表达也相应上调,这将导致更多的铁离子进入细胞内,促进活性氧的生成以及脂质过氧化,最终引发铁死亡,从而间接加剧血红蛋白毒性,这提示PIEZO2蛋白表达增多会促进铁沉积、增强细胞对铁死亡的敏感性,最终加重继发性脑损伤。因此,降低脑出血患者的治疗效应,调控PIEZO2的表达量,可能成为脑出血及继发性脑损伤的一个潜在治疗方向。 PIEZO1离子通道功能异常与脑血管结构完整性密切相关。研究发现,PIEZO1功能获得性突变会导致脑血管内皮细胞持续钙内流,影响细胞间连接蛋白表达,从而增加血脑屏障通透性[18]。但PIEZO1离子通道抑制剂如GsMTx4能够显著恢复受损大脑一侧的血流,减轻脑出血引发的脑水肿和神经功能障碍。研究显示,在发生脑出血后的不同时间点,PIEZO1呈现双向变化,即先短暂上调,随后逐渐下降,最终恢复正常水平[19]。也有研究表明,PIEZO1离子通道的另一种抑制剂异槲皮素通过抑制PIEZO1/NLRP3通路,能够有效减少神经元死亡和炎症反应,改善脑水肿和神经功能[20]。基于此,靶向抑制PIEZO1离子通道已成为减轻继发性脑损伤的新策略,这也为研发基于PIEZO1抑制剂的神经保护药物提供了实验依据。 2.1.3 创伤性脑损伤 创伤性脑损伤是指由外部物理力导致的脑功能紊乱或其他脑病理变化[21]。研究表明,PIEZO2离子通道的表达在大脑组织损伤区域尤其是神经元中显著上调,促进了神经元死亡以及促炎递质如肿瘤坏死因子α和白细胞介素1β的产生[22]。通过使用PIEZO2-shRNA微注射或D-GsMTx4预处理来抑制PIEZO2的功能,能够减轻由创伤性脑损伤引起的包括神经元死亡、脑水肿、脑组织坏死等一系列问题,并改善神经功能和认知功能缺陷。因此,抑制PIEZO2被视为一种减轻创伤性脑损伤的潜在治疗策略。同时,创伤性脑损伤可能引发创伤后癫痫,而机械性压迫可直接激活PIEZO离子通道,参与癫痫发生。例如,REN等[23]报道了1例由硬膜下骨瘤引发的难治性癫痫病例,硬膜下骨瘤压迫脑皮质导致PIEZO1和PIEZO2离子通道上调,引发局部神经元超兴奋性,这一发现将在临床角度将PIEZO离子通道与神经系统功能障碍联系起来,为手术干预机械性病因的癫痫提供理论支持。 2.1.4 脑积水与脑室扩张 脑积水是指大脑室系统内脑脊液的异常积聚,在婴幼儿和老年人群中更为常见[24]。随着脑脊液在脑室中的不断积累,脑室内压力增加,导致脑室体积逐渐增大,从而造成脑室扩张[25]。有研究揭示了PIEZO1离子通道调节大脑液体排泄的重要作用[26],无论是通过遗传手段还是化学手段激活PIEZO1离子通道,都能促进大脑中的液体排泄,这为治疗唐氏综合征类疾病的脑脊液过度聚积问题提供了潜在的治疗思路,同时,PIEZO1离子通道激活可能为那些需要借助侵入性神经外科手术以创建新的液体引流路径的患者提供一种非侵入性的替代疗法。 关于PIEZO离子通道对脑肿瘤、脑出血、创伤性脑损伤、脑积水与脑室扩张疾病影响的研究结果汇总见表2。"
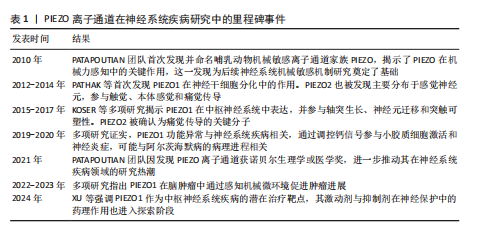
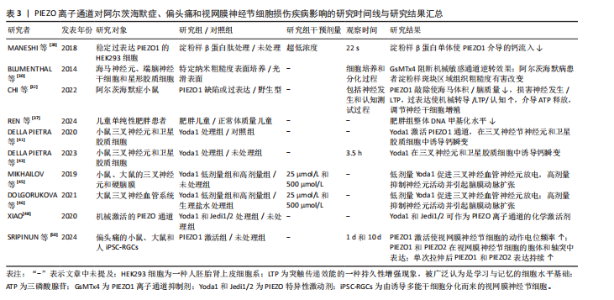
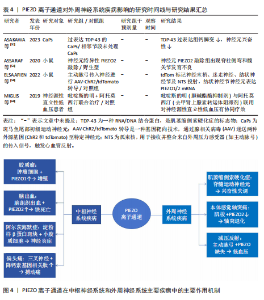
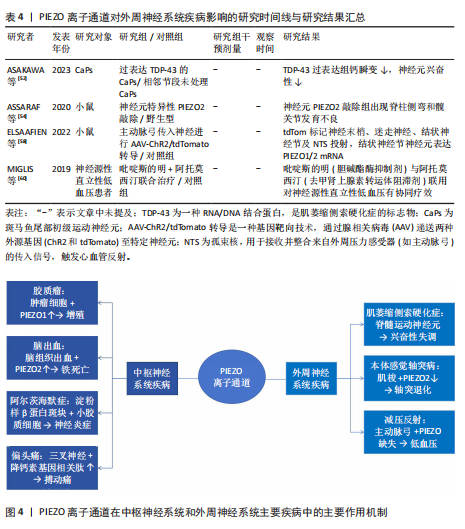
2.1.5 阿尔茨海默病 (1)机制:PIEZO离子通道对于阿尔茨海默病的病程具有多重作用。淀粉样β蛋白是阿尔茨海默病病理特征中的关键分子之一[27]。MANESHI等[28]发现,单体淀粉样β蛋白肽能够在极低浓度下显著抑制PIEZO1离子通道介导的钙离子内流,这种抑制并非简单的物理阻塞,而是通过直接结合PIEZO离子通道的特定区域或干扰与细胞质膜的相互作用,从而改变通道的机械敏感性,影响细胞迁移和细胞骨架动态重组,同时由单体淀粉样β蛋白肽聚集形成的淀粉样β蛋白斑块不仅影响它们所在区域的机械性质,还能够与细胞膜相互作用,从而改变膜结构和力学特性,这种相互作用可能会干扰PIEZO离子通道的功能,从而影响细胞的正常生理功能[29]。因此,在阿尔茨海默病的早期阶段,PIEZO1通道功能障碍可能是导致神经元功能异常的重要因素之一。 阿尔茨海默病发病机制的核心环节是神经炎症,而中枢神经系统的常驻免疫细胞——小胶质细胞在此过程中扮演着关键角色,小胶质细胞的激活和功能状态与疾病进展密切相关。小胶质细胞高度表达PIEZO1离子通道,PIEZO1通道参与小胶质细胞对周围环境机械性质变化的响应,包括迁移、屏障功能和吞噬作用等。对于阿尔茨海默病患者,由于淀粉样β蛋白斑块比周围的脑组织更硬,大脑粗糙度增加[30],小胶质细胞机械敏感性增强可上调反应性星形胶质细胞中PIEZO1蛋白表达[31],进而激活星形胶质细胞的PIEZO1离子通道[32],PIEZO1激活导致持续的钙离子内流,不仅调控细胞的迁移和吞噬功能,更激活促炎因子的成熟和释放,从而放大神经炎症反应。活化的PIEZO1通道作为关键的机械调控枢纽,在神经再生过程中发挥重要作用,这表明PIEZO1离子通道不仅可以检测并响应局部硬度变化[33],还参与胶质细胞功能的调节和炎症反应的控制[34]。 脑毛细血管的功能障碍也是阿尔茨海默病进展的关键因素。脑毛细血管作为大脑中最小的血管,负责感知活跃神经元的代谢需求,并通过调节血流满足这些代谢需求,而阿尔茨海默病患者此功能显著受损,表现为血管僵硬和毛细血管停滞,从而导致脑组织缺氧和代谢废物积累,加剧神经退行性病变[35]。研究表明,PIEZO1离子通道是脑毛细血管内皮细胞的核心机械传感器[36],可通过感知血流动力变化调控血管张力,从而维持脑血流的稳定性。阿尔茨海默病患者PIEZO1离子通道的功能异常,导致脑毛细血管对于机械刺激的响应能力下降,血流调控失调,从而加速疾病的进展。 此外,近年研究发现,PIEZO1离子通道的表观遗传调控与代谢疾病密切相关[37],儿童单纯性肥胖患者PIEZO1基因的异常低甲基化可能通过影响脂质代谢和炎症反应,间接加剧神经退行性病变(如阿尔茨海默病)的进展,这说明PIEZO1不仅是机械转导的关键分子,还可能通过代谢途径参与到神经疾病的病理过程中。 (2)治疗:PIEZO离子通道可以激活小胶质细胞,调节细胞的迁移、屏障功能和吞噬作用,这提示PIEZO离子通道可以作为阿尔茨海默病的潜在治疗靶点[33]。有研究强调了PIEZO离子通道调控大脑多种生理过程及病理状态的重要性[38],特别是在脱髓鞘疾病和阿尔茨海默病等疾病背景下,PIEZO离子通道的活性及调控机制尤为关键。因此,开发针对PIEZO离子通道的治疗策略可能为减缓或逆转阿尔茨海默病病程提供新的方向。 2.1.6 多发性硬化症 多发性硬化症是一种导致中枢神经系统白质脱髓鞘的自身免疫性疾病[39]。研究表明,PIEZO离子通道是多发性硬化症病理过程中的关键角色[40]。轴突中的PIEZO1离子通道激活将负向调控中枢神经系统的髓鞘化过程,而在CD4+ T细胞和调节性T细胞中抑制PIEZO1离子通道表达可减少实验性自身免疫性脑脊髓炎的症状,这提示PIEZO1离子通道可能对CD4+ T细胞和调节性T细胞的功能有抑制作用,并且可能通过影响这些细胞来参与多发性硬化症的病程,这也进一步说明了PIEZO离子通道不仅影响神经元,还在免疫系统中发挥作用,特别是在调节炎症反应方面。 研究表明,激活PIEZO1离子通道可以影响小胶质细胞和巨噬细胞的活动,包括促进炎症反应、影响细胞极化以及改变微环境的刚度[40]。 此外,机械力对于T细胞受体的激活至关重要,PIEZO1离子通道参与T细胞的激活过程,如在形成免疫突触时,机械拉伸可能会激活PIEZO1离子通道,引发钙离子内流,从而诱导calpain(一种钙依赖性的半胱氨酸蛋白酶)活化,促进T细胞受体信号传导。然而,目前PIEZO1离子通道在多发性硬化症相关炎症中的具体作用尚未完全明确,未来的研究可以在此方面进行深入探讨。 2.1.7 偏头痛 (1)机制:PIEZO离子通道在三叉神经痛觉系统中发挥重要作用。脑膜中的三叉神经纤维含有PIEZO1和PIEZO2两种离子通道,当血液以搏动的形式流动时,会周期性地激活这些离子通道,从而产生典型的搏动性疼痛[41-42]。研究表明,PIEZO1离子通道可在小鼠三叉神经痛细胞及脑膜组织中功能性表达[43],并且PIEZO1离子通道激活可以通过FM1-43染料进行标记,证实了PIEZO1离子通道可能直接参与偏头痛特有的搏动性疼痛信号传导过程。此外,PIEZO1离子通道可以通过2种方式被激活[44],一种是直接通过物理机械刺激,另一种则是通过化学激动剂,PIEZO1特异性激动剂Yoda1以及非特异性机械敏感受体激动剂低渗溶液HOS都能够激活三叉神经元中的PIEZO受体。应用Yoda1和HOS后可以显著增加来自硬脑膜三叉神经分支的持续性疼痛信号释放,并且这种激活不仅限于细胞层面的钙离子浓度在短时间内瞬间上升,还增加了疼痛纤维的放电频率;另一方面,Yoda1促进与偏头痛相关的促炎递质降钙素基因相关肽的释放,这表明PIEZO离子通道可能在偏头痛的发生和发展过程中起到关键作用[45]。PIEZO1在偏头痛中呈现剂量依赖性双向调节:低剂量激动剂Yoda1可以激活三叉神经节神经元,增强疼痛信号向三叉颈髓复合体传递;而高剂量Yoda1反而抑制神经元兴奋性,并通过扩张脑膜动脉减轻血管源性疼痛[46],这种双重调节机制增加了PIEZO1在偏头痛病理机制中的复杂性。 (2)治疗:研究表明,在背根神经节细胞中发现的PIEZO1离子通道相较于三叉神经节细胞更为活跃[47],然而在三叉神经元中PIEZO1的mRNA表达水平更高,这意味着在偏头痛条件下,PIEZO1离子通道可能具有潜在上调能力。尽管PIEZO1受体在不同细胞类型中的活性和功能有差异,但它们普遍被认为是偏头痛的关键参与者,可以通过抑制PIEZO离子通道来减轻机械性疼痛[48]。PIEZO1受体不仅参与痛觉信号的传递,而且PIEZO1受体活性受到多种因素的影响,包括机械应力、化学激动剂和抑制剂等。因此,针对PIEZO1离子通道的治疗策略可能为缓解偏头痛提供了一种新的方法。 2.1.8 视网膜神经节细胞损伤 视网膜神经节细胞损伤通常由缺血再灌注引发,视网膜的缺氧和氧化应激将触发细胞死亡[49]。有研究确认了PIEZO1和PIEZO2离子通道存在于视网膜神经节细胞中[50],并且激活离子通道后能够增加视网膜神经节细胞对光的反应频率以及暗背景下的自发信号。此外,短暂的眼内压升高可以显著上调这两种通道的表达,导致视网膜神经节细胞兴奋性增强,能量需求增多,从而产生更多代谢副产品如活性氧,最终导致视网膜神经节细胞功能障碍,这也表明了通过调节PIEZO离子通道的功能可以保护视网膜神经元免受损害。 关于PIEZO离子通道对阿尔茨海默症、偏头痛和视网膜神经节细胞损伤疾病影响的研究结果汇总见表3。 2.2 外周神经系统疾病 2.2.1 肌萎缩侧索硬化症 肌萎缩侧索硬化症是一种复杂的神经退行性疾病,主要特征是控制随意肌活动的运动神经元的逐渐丧失[51]。研究表明,虽然在探讨肌萎缩侧索硬化症机制时主要关注TDP-43蛋白的稳态失调如何扰乱脊髓运动神经元的兴奋性,但也发现了PIEZO离子通道的参与[52]。在过度表达mRFP1-TDP-43z的转基因斑马鱼模型中,压电辅助触觉刺激器可刺激PIEZO离子通道,将机械信号转化为电信号,以调节神经元兴奋性,从而影响肌萎缩侧索硬化症的发展进程。 2.2.2 本体感觉轴突病 有研究揭示了PIEZO2通道在本体感觉轴突病(如延迟性肌肉酸痛和射精后疾病综合征)中的潜在机制[53]。在患有早泄的大鼠模型中,重复剧烈的离心收缩与急性应激反应可能会导致肌肉纺锤体内本体感觉轴突末梢的能量枯竭,导致线粒体功能障碍,进而影响谷氨酸囊泡的释放和本体感觉机械转导PIEZO2离子通道的功能,最终造成一种类似于末端树突退化样的病变。另外,PIEZO2离子通道在肌肉纺锤体和腱器官中感知机械信号,PIEZO2离子通道功能缺失会导致本体感觉输入异常[54-55]。 2.2.3 减压反射异常 PIEZO2是传递机械力以实现触觉感知的主要转换器,Merkel细胞通过PIEZO2调节哺乳动物的触摸感受器功能[56]。同时,PIEZO1离子通道也是触觉感知的转化器,内皮细胞中的阳离子通道PIEZO1通过介导流动引起的ATP释放来控制血压,这意味着PIEZO1不仅参与了神经元对血压的感知,还在维持心血管系统的正常反馈调控中扮演重要角色。例如,在心脏和血管系统中,PIEZO1帮助检测血流的变化并触发相应的生理反应,这对于保持稳定的血压至关重要。因此,PIEZO离子通道可参与到神经元对血压变化的感知以及减压反射过程中,表明它们在神经系统与心血管系统的动态平衡稳定中具有不可或缺的作用[57]。 在高血压的治疗研究中,有研究选择性地标记了主动脉弓处负责监测血压变化的感觉传入神经[58],被标记的迷走神经结节细胞表达PIEZO1和PIEZO2的mRNA,这进一步证明了PIEZO离子通道在心血管调节中的关键地位,尤其是在血压调控方面。同时,PIEZO离子通道参与心血管系统反馈调控,可在血压感知及减压反射方面发挥重要作用[56]。 研究发现,PIEZO1和PIEZO2在压力感受神经元中均有表达[59],且双通道敲除的小鼠表现出显著的压力反射器功能缺陷,血压波动性显著增高,苯肾上腺素诱发的升压反应增强,压力反射敏感性降低,从而导致心脏对血压变化的代偿性心率调节减弱。小鼠静息状态下平均动脉压升高,但当由卧位转为直立位时,由于缺少PIEZO离子通道,压力反射信号传导中断,自主神经系统无法有效代偿血压下降,从而导致直立性低血压[60]。 关于PIEZO离子通道对外周神经系统疾病影响的研究结果汇总见表4。"

| [1] XU Y, WANG Y, YANG Y, et al. Piezo1: the key regulators in central nervous system diseases. Front Cell Neurosci. 2024;18:1441806. [2] VELASCO-ESTEVEZ M, KOCH N, KLEJBOR I, et al. Mechanoreceptor Piezo1 Is Downregulated in Multiple Sclerosis Brain and Is Involved in the Maturation and Migration of Oligodendrocytes in vitro. Front Cell Neurosci. 2022;16:914985. [3] QIN L, HE T, CHEN S, et al. Roles of mechanosensitive channel Piezo1/2 proteins in skeleton and other tissues. Bone Res. 2021; 9(1):44. [4] LIM J, TAI HH, LIAO WH, et al. ASIC1a is required for neuronal activation via low-intensity ultrasound stimulation in mouse brain. Elife. 2021;10:e61660. [5] XU X, LIU S, LIU H, et al. Piezo Channels: Awesome Mechanosensitive Structures in Cellular Mechanotransduction and Their Role in Bone. Int J Mol Sci. 2021;22(12):6429. [6] SAN SEBASTIAN W, SAMARANCH L, KELLS AP, et al. Gene therapy for misfolding protein diseases of the central nervous system. Neurotherapeutics. 2013;10(3):498-510. [7] ZHAO Q, ZHOU H, LI X, et al. The mechanosensitive Piezo1 channel: a three-bladed propeller-like structure and a lever-like mechanogating mechanism. FEBS J. 2019286(13):2461-2470. [8] LI X, HU J, ZHAO X, et al. Piezo channels in the urinary system. Exp Mol Med. 2022;54(6): 697-710. [9] XIAO B. Mechanisms of mechanotransduction and physiological roles of PIEZO channels. Nat Rev Mol Cell Biol. 2024;25(11):886-903. [10] QIU Z, GUO J, KALA S, et al. The Mechanosensitive Ion Channel Piezo1 Significantly Mediates In Vitro Ultrasonic Stimulation of Neurons. iScience. 2019;21:448-457. [11] PARK JW, LEE K, KIM EE, et al. Brain Tumor Classification by Methylation Profile. J Korean Med Sci. 2023;38(43):e356. [12] CHEN X, WANGGOU S, BODALIA A, et al. A Feedforward Mechanism Mediated by Mechanosensitive Ion Channel PIEZO1 and Tissue Mechanics Promotes Glioma Aggression. Neuron. 2018;100(4):799-815.e7. [13] FU W, HOU X, DING L, et al. Piezo1-related physiological and pathological processes in glioblastoma. Front Cell Dev Biol. 2025;13: 1536320. [14] QURESHI AI, TUHRIM S, BRODERICK JP, et al. Spontaneous intracerebral hemorrhage. N Engl J Med. 2001;344(19):1450-1460. [15] GONG Y, GONG Y, HOU Z, et al. Establishment of an Experimental Intracerebral Haemorrhage Model for Mass Effect Research using a Thermo-sensitive Hydrogel. Sci Rep. 2019;9(1):13838. [16] GUO T, REN P, LI X, et al. Neural Injuries Induced by Hydrostatic Pressure Associated With Mass Effect after Intracerebral Hemorrhage. Sci Rep. 2018;8(1):9195. [17] GONG Y, DENG J, WU Y, et al. Role of mass effect on neuronal iron deposition after intracerebral hemorrhage. Exp Neurol. 2023;368:114475. [18] SCIMONE C, DONATO L, ALIBRANDI S, et al. Evidences of PIEZO1 involvement in cerebral cavernous malformation pathogenesis. Microvasc Res. 2022;141:104342. [19] QI M, LIU R, ZHANG F, et al. Roles of mechanosensitive ion channel PIEZO1 in the pathogenesis of brain injury after experimental intracerebral hemorrhage. Neuropharmacology. 2024;251:109896. [20] GUO T, CHEN G, YANG L, et al. Piezo1 inhibitor isoquercitrin rescues neural impairment mediated by NLRP3 after intracerebral hemorrhage. Exp Neurol. 2024;379:114852. [21] KHELLAF A, KHAN DZ, HELMY A. Recent advances in traumatic brain injury. J Neurol. 2019;266(11): 2878-2889. [22] XIAO Y, ZHANG Y, YUAN W, et al. Piezo2 Contributes to Traumatic Brain Injury by Activating the RhoA/ROCK1 Pathways. Mol Neurobiol. 2024; 61(10):7419-7430. [23] REN Y, LIU Y, WU H, et al. Subdural osteoma in an adolescent patient with epilepsy: an unusual case report and literature review. Childs Nerv Syst. 2023;39(11):3281-3288. [24] HOCHSTETLER A, RASKIN J, BLAZER-YOST BL. Hydrocephalus: historical analysis and considerations for treatment. Eur J Med Res. 2022;27(1):168. [25] EDWARDS RJ, DOMBROWSKI SM, LUCIANO MG, et al. Chronic hydrocephalus in adults. Brain Pathol. 2004;14(3):325-336. [26] CHOI D, PARK E, CHOI J, et al. Piezo1 regulates meningeal lymphatic vessel drainage and alleviates excessive CSF accumulation. Nat Neurosci. 2024;27(5):913-926. [27] MCKHANN G, DRACHMAN D, FOLSTEIN M, et al. Clinical diagnosis of Alzheimer’s disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology. 1984;34(7):939-944. [28] MANESHI MM, ZIEGLER L, SACHS F, et al. Enantiomeric Aβ peptides inhibit the fluid shear stress response of PIEZO1. Sci Rep. 2018;8(1): 14267. [29] PARK M, HOANG GM, NGUYEN T, et al. Effects of transcranial ultrasound stimulation pulsed at 40 Hz on Aβ plaques and brain rhythms in 5×FAD mice. Transl Neurodegener. 2021;10(1):48. [30] BLUMENTHAL NR, HERMANSON O, HEIMRICH B, et al. Stochastic nanoroughness modulates neuron-astrocyte interactions and function via mechanosensing cation channels. Proc Natl Acad Sci U S A. 2014;111(45):16124-16129. [31] SATOH K, HATA M, TAKAHARA S, et al. A novel membrane protein, encoded by the gene covering KIAA0233, is transcriptionally induced in senile plaque-associated astrocytes. Brain Res. 2006;1108(1):19-27. [32] CHI S, CUI Y, WANG H, et al. Astrocytic Piezo1-mediated mechanotransduction determines adult neurogenesis and cognitive functions. Neuron. 2022;110(18):2984-2999.e8. [33] IVKOVIC S, MAJOR T, MITIC M, et al. Fatty acids as biomodulators of Piezo1 mediated glial mechanosensitivity in Alzheimer’s disease. Life Sci. 2022;297:120470. [34] GENG J, SHI Y, ZHANG J, et al. TLR4 signalling via Piezo1 engages and enhances the macrophage mediated host response during bacterial infection. Nat Commun. 2021;12(1):3519. [35] SCHELTENS P, DE STROOPER B, KIVIPELTO M, et al. Alzheimer’s disease. Lancet. 2021;397(10284): 1577-1590. [36] LIM XR, ABD-ALHASEEB MM, IPPOLITO M, et al. Endothelial Piezo1 channel mediates mechano-feedback control of brain blood flow. Nat Commun. 2024;15(1):8686. [37] REN Y, HUANG P, HUANG X, et al. Alterations of DNA methylation profile in peripheral blood of children with simple obesity. Health Inf Sci Syst. 2024;12(1):26. [38] ZHENG Q, LIU H, YU W, et al. Mechanical properties of the brain: Focus on the essential role of Piezo1-mediated mechanotransduction in the CNS. Brain Behav. 2023;13(9):e3136. [39] OH J, VIDAL-JORDANA A, MONTALBAN X. Multiple sclerosis: clinical aspects. Curr Opin Neurol. 2018; 31(6):752-759. [40] YANG K, HE X, WU Z, et al. The emerging roles of piezo1 channels in animal models of multiple sclerosis. Front Immunol. 2022;13:976522. [41] DELLA PIETRA A, MIKHAILOV N, GINIATULLIN R. The Emerging Role of Mechanosensitive Piezo Channels in Migraine Pain. Int J Mol Sci. 2020; 21(3):696. [42] BURISH MJ, HAN C, MAWATARI K, et al. The first-line cluster headache medication verapamil alters the circadian period and elicits sex-specific sleep changes in mice. Chronobiol Int. 2021;38(6):839-850. [43] DELLA PIETRA A, MIKHAILOV N, GINIATULLIN R. FM1-43 Dye Memorizes Piezo1 Activation in the Trigeminal Nociceptive System Implicated in Migraine Pain. Int J Mol Sci. 2023;24(2):1688. [44] GINIATULLIN R. Ion Channels of Nociception. Int J Mol Sci. 2020;21(10):3553. [45] MIKHAILOV N, LESKINEN J, FAGERLUND I, et al. Mechanosensitive meningeal nociception via Piezo channels: Implications for pulsatile pain in migraine? Neuropharmacology. 2019;149:113-123. [46] DOLGORUKOVA A, ISAEVA JE, VERBITSKAYA E, et al. Differential effects of the Piezo1 agonist Yoda1 in the trigeminovascular system: An electrophysiological and intravital microscopy study in rats. Exp Neurol. 2021;339:113634. [47] DELLA PIETRA A, GÓMEZ DABÓ L, MIKULENKA P, et al. Mechanosensitive receptors in migraine: a systematic review. J Headache Pain. 2024;25(1):6. [48] XIAO B. Levering Mechanically Activated Piezo Channels for Potential Pharmacological Intervention. Annu Rev Pharmacol Toxicol. 2020;60:195-218. [49] FENG Y, HU C, CUI K, et al. GSK840 Alleviates Retinal Neuronal Injury by Inhibiting RIPK3/MLKL-Mediated RGC Necroptosis After Ischemia/Reperfusion. Invest Ophthalmol Vis Sci. 2023; 64(14):42. [50] SRIPINUN P, SEE LP, NIKONOV S, et al. Piezo1 and Piezo2 channels in retinal ganglion cells and the impact of Piezo1 stimulation on light-dependent neural activity. bioRxiv [Preprint]. 2024:2024.06.25.599602. [51] HARDIMAN O, AL-CHALABI A, CHIO A, et al. Amyotrophic lateral sclerosis. Nat Rev Dis Primers. 2017;3:17071. [52] ASAKAWA K, HANDA H, KAWAKAMI K. Dysregulated TDP-43 proteostasis perturbs excitability of spinal motor neurons during brainstem-mediated fictive locomotion in zebrafish. Dev Growth Differ. 2023;65(8):446-452. [53] SONKODI B, KOPA Z, NYIRÁDY P. Post Orgasmic Illness Syndrome (POIS) and Delayed Onset Muscle Soreness (DOMS): Do They Have Anything in Common? Cells. 2021;10(8):1867. [54] ASSARAF E, BLECHER R, HEINEMANN-YERUSHALMI L, et al. Piezo2 expressed in proprioceptive neurons is essential for skeletal integrity. Nat Commun. 2020;11(1):3168. [55] KRÖGER S. Proprioception 2.0: novel functions for muscle spindles. Curr Opin Neurol. 2018; 31(5):592-598. [56] YANG H, TENORIO LOPES L, BARIONI NO, et al. The molecular makeup of peripheral and central baroreceptors: stretching a role for Transient Receptor Potential (TRP), Epithelial Sodium Channel (ENaC), Acid Sensing Ion Channel (ASIC), and Piezo channels. Cardiovasc Res. 2022; 118(15):3052-3070. [57] VALENZA G, MATIĆ Z, CATRAMBONE V. The brain-heart axis: integrative cooperation of neural, mechanical and biochemical pathways. Nat Rev Cardiol. 2025;22(8):537-550. [58] ELSAAFIEN K, HARDEN SW, JOHNSON DN, et al. A Novel Organ-Specific Approach to Selectively Target Sensory Afferents Innervating the Aortic Arch. Front Physiol. 2022;13:841078. [59] ZENG WZ, MARSHALL KL, MIN S, et al. PIEZOs mediate neuronal sensing of blood pressure and the baroreceptor reflex. Science. 2018;362(6413):464-467. [60] MIGLIS MG, MUPPIDI S. Ion channels PIEZOs identified as the long-sought baroreceptor mechanosensors for blood pressure control, and other updates on autonomic research. Clin Auton Res. 2019;29(1):9-11. |
| [1] | Zhang Yirui, Gu Ye, Qian Zhengtao, Wu Zerui, Xie Heng, Tang Yihan, Gu Yingchu, Fang Tao, Wang Qiufei, Peng Yuqin, Geng Dechun, Xu Yaozeng. Molecular mechanisms and therapeutic targets of mechanical stress regulating osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 9048-9055. |
| [2] | Kan Weiyi, Wang Linrong, Cheng Leping. The interaction and balance between cellular senescence and tissue repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6297-6305. |
| [3] | Huang Sijing, Cui Rui, Geng Longyu, Gao Beiyao, Ge Ruidong, Jiang Shan. Application and molecular mechanism of extracorporeal shock wave for anti-fibrosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(17): 4417-4429. |
| [4] | Gao Xilin, Wu Si Zhang Chao Zhu Liguo, Fu Bifeng, Wang Ping. Mechanotransduction proteins in intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(3): 579-589. |
| [5] | Long Chenyan, Cheng Biao, Tian Ju. Cellular and molecular mechanisms of platelet-rich plasma in promoting wound healing [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(13): 2793-2801. |
| [6] | Xiong Wanqi, Li Zhenhao, Cui Yan, Liu Jiahe, Li Chenzhi, Wu Mingjian, Li Yancheng, Yang Fan, Liu Baoyi. Effects of biomechanics on biological characteristics of osteoblasts [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(21): 3407-3412. |
| [7] | Min Ziyang, Munire·Aili, Zheng Yunhao, Zeng Xingzhi, Bian Nanyan, Deng Shuangshan, Xie Jing. Mechanism and characteristics of mechanical microenvironment of extracellular matrix and intercellular interaction [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 4034-4045. |
| [8] | An Dong, Liu Yang, Yang Tongjiang. Application of extracorporeal shock wave therapy in burn wound repair and post-burn scar treatment [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(20): 3265-3272. |
| [9] | Chen Jun-liang, He Yun, Xiao Jin-gang. Effect of continuous versus intermittent force on the proliferation and differentiation of bone marrow stromal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(13): 1969-1973. |
| [10] | Zhang Bao-hua, Qiu Fu-cheng, Dong Ci1, Han Rui, Zhang Yong-zhi, Liu Hui-miao,Xie Bing-chuan, Zhang Li-na, Wang Wen-ting, Wang Yan-yong, Zhang Zhen-qing, Gu Ping,Yan Bao-yong. Neural stem cell transplantation for central nervous system diseases via the cerebrospinal fluid [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 974-978. |
| [11] | Yan Peng, Zheng Wei-dong, Zhang Ji-kai, Tan Yun-bing, Li Gao-feng, Li Guang-chun, Song Cheng, Yang Xiao-yu . Histological changes and safety of long-term acral nerve root stimulation electrode placement in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2013, 17(37): 6587-6593. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||