Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (34): 8906-8913.doi: 10.12307/2026.842
Previous Articles Next Articles
Interleukin-10 alleviates inflammatory responses after acute tendon injury
Jiang Li1, Peng Guoqiang1, Li Sen2
- 1School of Rehabilitation Medicine, Baicheng Medical College, Baicheng 137000, Jilin Province, China; 2Department of Spine Surgery, Nanjing Drum Tower Hospital Affiliated to Nanjing University Medical School, Nanjing 210000, Jiangsu Province, China
-
Received:2025-08-16Revised:2026-01-07Online:2026-12-08Published:2026-04-13 -
Contact:Li Sen, MD, Chief physician, Department of Spine Surgery, Nanjing Drum Tower Hospital Affiliated to Nanjing University Medical School, Nanjing 210000, Jiangsu Province, China Co-corresponding author: Peng Guoqiang, MS, Associate professor, School of Rehabilitation Medicine, Baicheng Medical College, Baicheng 137000, Jilin Province, China -
About author:Jiang Li, MS, Teaching assistant, School of Rehabilitation Medicine, Baicheng Medical College, Baicheng 137000, Jilin Province, China -
Supported by:Baicheng Municipal Guiding Science and Technology Development Program, No. 2025049 (to JL)
CLC Number:
Cite this article
Jiang Li, Peng Guoqiang, Li Sen. Interleukin-10 alleviates inflammatory responses after acute tendon injury[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 8906-8913.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
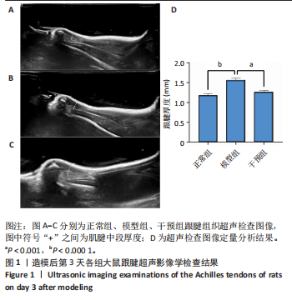
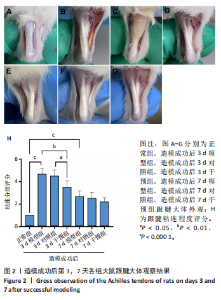
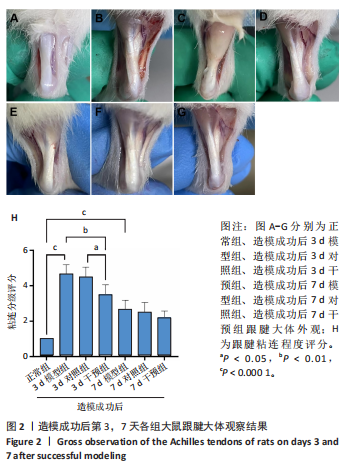
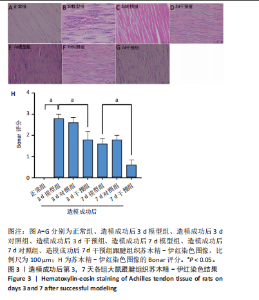
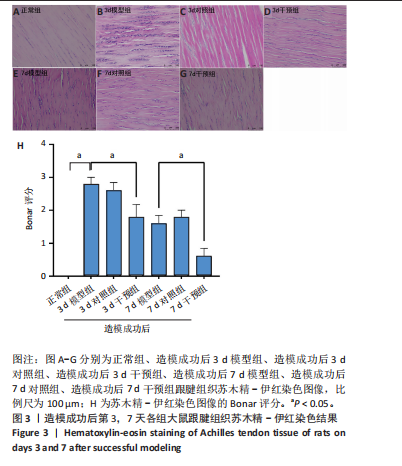
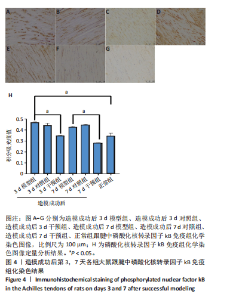
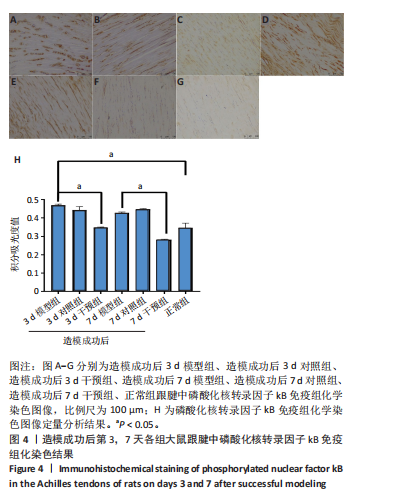
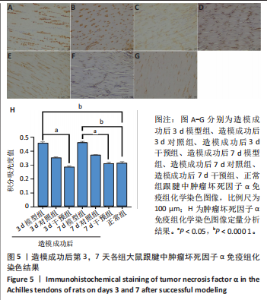
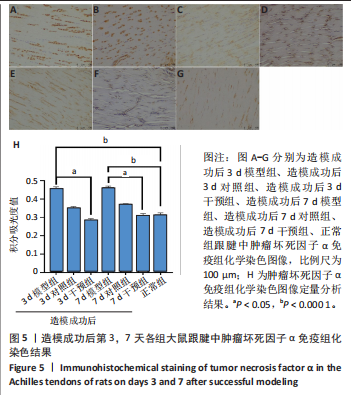
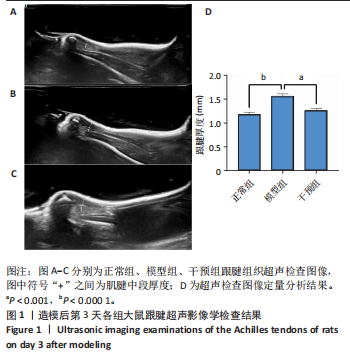
2.1 实验动物数量分析 42只大鼠均完整经历了既定的实验周期,未出现任何死亡情况。所有大鼠在进食、饮水、排便等生理活动方面维持正常状态,并且精神状况良好,均未出现感染等情况。 2.2 超声影像学检查跟腱愈合情况 造模后第3天各组大鼠超声影像学检查结果见图1。正常组跟腱组织边界清晰,组织连续且厚度均匀;模型组跟腱组织边界模糊,跟腱厚度与正常组相比增加(P < 0.000 1);干预组跟腱组织边界清晰度较模型组增强,跟腱厚度较模型组减少(P < 0.001),接近正常组。 2.3 各组大鼠跟腱大体观察结果 各组大鼠跟腱大体观察结果见图2。正常组大鼠跟腱颜色亮白有光泽,肌腱质地柔软且弹性好,无任何粘连。造模成功后第3天,模型组大鼠跟腱肿胀明显,组织表面粗糙并有许多新生血管,颜色偏黄,色泽较暗,质地较硬,跟腱活动时感到一定阻力,将跟腱与皮肤剥离开时存在严重粘连且组织韧性较差;对照组大鼠跟腱大体观察与模型组类似,均存在新生血管、色泽较暗、颜色偏黄的现象,并且跟腱表面存在严重粘连;干预组大鼠跟腱肿胀程度较模型组减轻,新生血管减少,颜色呈淡黄色,跟腱活动时阻力感减轻,相比于模型组在剥离跟腱时较容易,粘连减轻。造模成功后第7天,模型组大鼠跟腱颜色呈微黄色,表面粗糙程度大幅度减轻,新生血管明显减少,粘连程度减轻,容易剥离;对照组大鼠跟腱内新生血管明显减少,肿胀程度减轻,但与正常组和干预组相比跟腱颜色仍偏黄;干预组大鼠跟腱颜色呈微黄色,肿胀程度大幅度减轻并趋向正常,只存在轻微粘连和活动受限。 2.4 各组大鼠跟腱组织苏木精-伊红染色观察结果 各组大鼠跟腱组织苏木精-伊红染色结果见图3。苏木精-伊红染色显示,正常组大鼠跟腱胶原纤维排列紧密整齐有序,肌腱细胞分布均匀,细胞核呈长梭形,毛细血管很少见,跟腱与腱周组织无粘连。造模成功后第3天,模型组大鼠跟腱胶原纤维排列杂乱,部分纤维卷曲呈波浪状,纤维之间存在间隙,有大量炎症细胞浸润,细胞核呈圆形集中分布,腱周有许多毛细血管生成;对照组大鼠跟腱胶原纤维的排列呈现出与模型组类似的特点;干预组大鼠跟腱纤维排列杂乱程度减轻,炎症细胞浸润减少,长梭形肌腱细胞增多,纤维间的间隙也有所改善。造模成功后第7天,模型组大鼠跟腱胶原纤维排列显著改善,波浪状排列明显减少,肌腱细胞呈圆形向梭形过渡的形态,大部分细胞核排列整齐,分布均匀;对照组大鼠跟腱胶原纤维的卷曲程度减轻、排列更为整齐,圆形肌腱细胞减少;干预组大鼠跟腱胶原纤维排列平行整齐,趋向正常,炎症细胞减少,肌腱细胞多呈长梭形。造模成功后第3,7天,干预组Bonar评分量表评分低于模型组(P < 0.05)。 2.5 免疫组化染色检测各组大鼠跟腱中磷酸化核转录因子κB和肿瘤坏死因子α表达 各组大鼠跟腱中磷酸化核转录因子κB和"
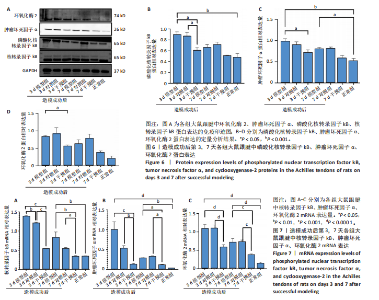
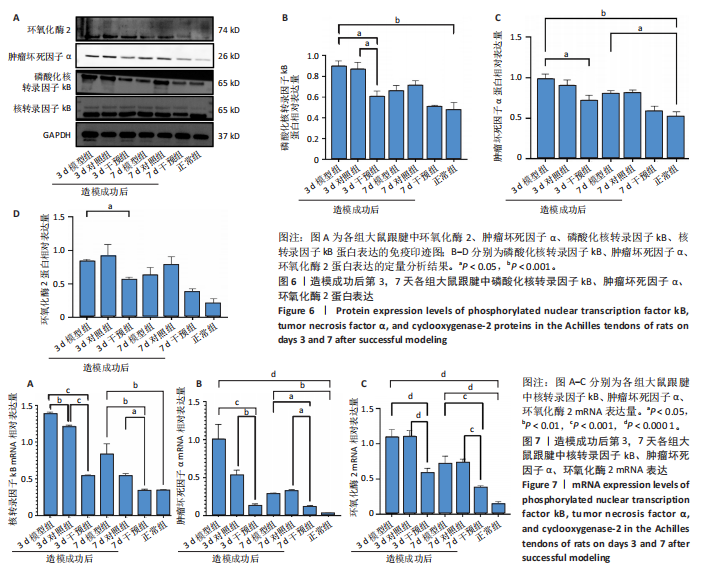
(P < 0.05,P < 0.000 1),干预组造模成功后第3,7天的肿瘤坏死因子α表达低于模型组(P < 0.05)。 2.6 各组大鼠跟腱中相关蛋白表达 Western blot检测各组大鼠跟腱组织中核转录因子κB、磷酸化核转录因子κB、肿瘤坏死因子α、环氧化酶2蛋白表达,见图6。各组大鼠跟腱中核转录因子κB蛋白表达比较差异无显著性意义(P > 0.05),模型组造模成功后第3天的磷酸化核转录因子κB蛋白表达高于正常组、干预组(P < 0.001,P < 0.05);模型组造模材成功后第3,7天的肿瘤坏死因子α蛋白表达高于正常组(P < 0.001,P < 0.05),造模成功后第3天的肿瘤坏死因子α蛋白表达高于干预组(P < 0.05);模型组造模成功后第3天的环氧化酶2蛋白表达高于干预组(P < 0.05)。 2.7 各组大鼠跟腱中相关基因mRNA表达 RT-PCR检测各组大鼠跟腱中核转录因子κB、肿瘤坏死因子α、环氧化酶2 mRNA表达,见图7。模型组造模成功后第3,7天的核转录因子κB mRNA表达高于正常组、干预组(P < 0.001,P < 0.01,P < 0.05),造模成功后第3,7天的肿瘤坏死因子α mRNA表达高于正常组、干预组(P < 0.001,P < 0.01),造模成功后第3,7天的环氧化酶2 mRNA表达高于正常组、干预组(P < 0.000 1,P < 0.001)。"
| [1] LYU K, LIU X, LIU T, et al. miRNAs contributing to the repair of tendon injury. Cell Tissue Res. 2023;393(2):201-215. [2] JIANG L, LU J, CHEN Y, et al. Mesenchymal stem cells: An efficient cell therapy for tendon repair (Review). Int J Mol Med. 2023;52(2):70. [3] ZHANG L, ZHANG Q, CUI L, et al. Kartogenin Combined Platelet-Rich Plasma (PRP) Promoted Tendon-Bone Healing for Anterior Cruciate Ligament (ACL) Reconstruction by Suppressing Inflammatory Response Via targeting AKT/PI3K/NF-κB. Appl Biochem Biotechnol. 2023;195(2):1284-1296. [4] KAMEL SI, ROSAS HG, GORBACHOVA T. Local and Systemic Side Effects of Corticosteroid Injections for Musculoskeletal Indications. AJR Am J Roentgenol. 2024;222(3):e2330458. [5] SHAMROCK AG, DREYER MA, VARACALLO MA. Achilles Tendon Rupture. StatPearls. Treasure Island (FL): StatPearls Publishing, Copyright © 2025, StatPearls Publishing LLC.; 2025. [6] DAMBUZA IM, HE C, CHOI JK, et al. IL-12p35 induces expansion of IL-10 and IL-35-expressing regulatory B cells and ameliorates autoimmune disease. Nat Commun. 2017;8(1):719. [7] SHI J, SHI S, XIE W, et al. IL-10 alleviates lipopolysaccharide-induced skin scarring via IL-10R/STAT3 axis regulating TLR4/NF-κB pathway in dermal fibroblasts. J Cell Mol Med. 2021;25(3):1554-1567. [8] KIERAN I, KNOCK A, BUSH J, et al. Interleukin-10 reduces scar formation in both animal and human cutaneous wounds: results of two preclinical and phase II randomized control studies. Wound Repair Regen. 2013;21(3):428-436. [9] NAKAGOME K, DOHI M, OKUNISHI K, et al. In vivo IL-10 gene delivery attenuates bleomycin induced pulmonary fibrosis by inhibiting the production and activation of TGF-beta in the lung. Thorax. 2006;61(10):886-894. [10] SHORT WD, STEEN E, KAUL A, et al. IL-10 promotes endothelial progenitor cell infiltration and wound healing via STAT3. FASEB J. 2022;36(7):e22298. [11] FEARON A, DAHLSTROM JE, TWIN J, et al. The Bonar score revisited: region of evaluation significantly influences the standardized assessment of tendon degeneration. J Sci Med Sport. 2014;17(4):346-350. [12] STEINMANN S, PFEIFER CG, BROCHHAUSEN C, et al. Spectrum of Tendon Pathologies: Triggers, Trails and End-State. Int J Mol Sci. 2020;21(3):844. [13] ATTIA AK, MAHMOUD K, D’HOOGHE P, et al. Outcomes and Complications of Open Versus Minimally Invasive Repair of Acute Achilles Tendon Ruptures: A Systematic Review and Meta-analysis of Randomized Controlled Trials. Am J Sports Med. 2023;51(3):825-836. [14] ZHANG J, BROWN R, HOGAN MV, et al. Mitigating Scar Tissue Formation in Tendon Injuries: Targeting HMGB1, AMPK Activation, and Myofibroblast Migration All at Once. Pharmaceuticals (Basel). 2023;16(12):1739. [15] TRAWEGER A, SCOTT A, KJAER M, et al. Achilles tendinopathy. Nat Rev Dis Primers. 2025;11(1):20. [16] DARRIEUTORT-LAFFITE C, BLANCHARD F, SOSLOWSKY LJ, et al. Biology and physiology of tendon healing. Joint Bone Spine. 2024;91(5):105696. [17] JIANG F, ZHAO H, ZHANG P, et al. Challenges in tendon-bone healing: emphasizing inflammatory modulation mechanisms and treatment. Front Endocrinol (Lausanne). 2024;15:1485876. [18] DAKIN SG, DUDHIA J, SMITH RK. Resolving an inflammatory concept: the importance of inflammation and resolution in tendinopathy. Vet Immunol Immunopathol. 2014;158(3-4):121-127. [19] CHEN R, AI L, ZHANG J, et al. Dendritic Cell-Derived Exosomes Promote Tendon Healing and Regulate Macrophage Polarization in Preventing Tendinopathy. Int J Nanomedicine. 2024;19:11701-11718. [20] THOMOPOULOS S, HATTERSLEY G, ROSEN V, et al. The localized expression of extracellular matrix components in healing tendon insertion sites: an in situ hybridization study. J Orthop Res. 2002;20(3):454-463. [21] NAJAFI Z, RAHMANIAN-DEVIN P, BARADARAN RAHIMI V, et al. Challenges and opportunities of medicines for treating tendon inflammation and fibrosis: A comprehensive and mechanistic review. Fundam Clin Pharmacol. 2024;38(5): 802-841. [22] CHEN J, SHENG R, MO Q, et al. Controlled TPCA-1 delivery engineers a pro-tenogenic niche to initiate tendon regeneration by targeting IKKβ/NF-κB signaling. Bioact Mater. 2025;44:319-338. [23] LANE RA, MIGOTSKY N, HAVLIOGLU N, et al. The effects of NF-κB suppression on the early healing response following intrasynovial tendon repair in a canine model. J Orthop Res. 2023;41(10):2295-2304. [24] LI H, LI Y, LUO S, et al. The roles and mechanisms of the NF-κB signaling pathway in tendon disorders. Front Vet Sci. 2024;11:1382239. [25] ZHANG P, HU J, LIU X, et al. Antiadhesion Biomaterials in Tendon Repair: Application Status and Future Prospect. Tissue Eng Part B Rev. 2025;31(1):20-30. [26] SHARMA P , MAFFULLI N. Biology of tendon injury: healing, modeling and remodeling. J Musculoskelet Neuronal Interact. 2006;6(2):181-190. [27] HAST MW, ZUSKOV A, SOSLOWSKY LJ. The role of animal models in tendon research. Bone Joint Res. 2014;3(6):193-202. [28] LAKE SP, ANSORGE HL, SOSLOWSKY LJ. Animal models of tendinopathy. Disabil Rehabil. 2008;30(20-22):1530-1541. [29] VIDAL L, LOPEZ-GARZON M, VENEGAS V, et al. A Novel Tendon Injury Model, Induced by Collagenase Administration Combined with a Thermo-Responsive Hydrogel in Rats, Reproduces the Pathogenesis of Human Degenerative Tendinopathy. Int J Mol Sci. 2024;25(3):1868. [30] DUNICAN AL, LEUENROTH SJ, GRUTKOSKI P, et al. TNFalpha-induced suppression of PMN apoptosis is mediated through interleukin-8 production. Shock. 2000;14(3):284-288; discussion 288-289. [31] GARBACKI N, TITS M, ANGENOT L, et al. Inhibitory effects of proanthocyanidins from Ribes nigrum leaves on carrageenin acute inflammatory reactions induced in rats. BMC Pharmacol. 2004;4:25. [32] JIANG L, LIU T, LYU K, et al. Inflammation-related signaling pathways in tendinopathy. Open Life Sci. 2023;18(1):20220729. [33] DE SOUZA S, ROSARIO CLAUDIO J, SIM J, et al. Interleukin-10 signaling in somatosensory neurons controls CCL2 release and inflammatory response. Brain Behav Immun. 2024;116:193-202. [34] WANG S, YAO Z, CHEN L, et al. Preclinical assessment of IL-1β primed human umbilical cord mesenchymal stem cells for tendon functional repair through TGF-β/IL-10 signaling. Heliyon. 2023;9(11):e21411. |
| [1] | Cao Yong, Teng Hongliang, Tai Pengfei, Li Junda, Zhu Tengqi, Li Zhaojin. Interactions between cytokines and satellite cells in muscle regeneration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1808-1817. |
| [2] | Cai Ziming, Yu Qinghe, Ma Pengfei, Zhang Xin, Zhou Longqian, Zhang Chongyang, Lin Wenping. Heme oxygenase-1 alleviates lipopolysaccharide-induced inflammatory response in nucleus pulposus mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1624-1631. |
| [3] | He Jiale, Huang Xi, Dong Hongfei, Chen Lang, Zhong Fangyu, Li Xianhui. Acellular dermal matrix combined with adipose-derived stem cell exosomes promotes burn wound healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1699-1710. |
| [4] | Xia Linfeng, Wang Lu, Long Qianfa, Tang Rongwu, Luo Haodong, Tang Yi, Zhong Jun, Liu Yang. Human umbilical cord mesenchymal stem cell-derived exosomes alleviate blood-brain barrier damage in mice with septic encephalopathy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1711-1719. |
| [5] | Cui Lianxu, Li Haomin, Xu Junrong, Tan Baodong, Lu Dahong, Peng Siwei, Wang Jinhui. Effect of umbilical cord mesenchymal stem cell conditioned medium on tissue repair after traumatic craniocerebral injury in miniature pigs [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1730-1735. |
| [6] | Hou Chaowen, Li Zhaojin, Kong Jianda, Zhang Shuli. Main physiological changes in skeletal muscle aging and the multimechanism regulatory role of exercise [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1464-1475. |
| [7] | You Huijuan, Wu Shuzhen, Rong Rong, Chen Liyuan, Zhao Yuqing, Wang Qinglu, Ou Xiaowei, Yang Fengying. Macrophage autophagy in lung diseases: two-sided effects [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1516-1526. |
| [8] | Yin Yongcheng, Zhao Xiangrui, Yang Zhijie, Li Zheng, Li Fang, Ning Bin. Effect and mechanism of peroxiredoxin 1 in microglial inflammation after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1106-1113. |
| [9] | Zhang Di, Zhao Jun, Ma Guangyue, Sun Hui, Jiang Rong. Mechanism of depression-like behavior in chronic social defeat stress mice based on high-throughput sequencing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1139-1146. |
| [10] | Li Haojing, Wang Xin, Song Chenglin, Zhang Shengnan, Chen Yunxin. Therapeutic efficacy of extracorporeal shock wave therapy in the upper trapezius muscle area combined with exercise control training in patients with chronic non-specific neck pain [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1162-1170. |
| [11] | Liu Yu, Lei Senlin, Zhou Jintao, Liu Hui, Li Xianhui. Mechanisms by which aerobic and resistance exercises improve obesity-related cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1171-1183. |
| [12] | Yu Huifen, Mo Licun, Cheng Leping. The position and role of 5-hydroxytryptamine in the repair of tissue injury [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1196-1206. |
| [13] | Wen Xiaolong, Weng Xiquan, Feng Yao, Cao Wenyan, Liu Yuqian, Wang Haitao. Effects of inflammation on serum hepcidin and iron metabolism related parameters in patients with type 2 diabetes mellitus: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1294-1301. |
| [14] | Chen Yixian, Chen Chen, Lu Liheng, Tang Jinpeng, Yu Xiaowei. Triptolide in the treatment of osteoarthritis: network pharmacology analysis and animal model validation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 805-815. |
| [15] | Ma Hong, Ding Xueling, Wang Qi, Lyu Hui, Asya Albusm, Cheng Xinyi, Ma Xiang. Expression and significance of tumor necrosis factor alpha, nuclear factor kappaB and ionized calcium binding adaptor molecule-1 in the hippocampus of mice with aortic dissection [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 858-863. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||