Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (25): 6566-6574.doi: 10.12307/2026.463
Previous Articles Next Articles
Effects and mechanisms of glycemic variability on apoptosis in mouse hippocampal neuronal HT-22 cells
Chen Di1, 2, Xu Mengling1, 2, Rao Binchan1, 2, Zhu Liying1,3, Li Xing4, Xu Yongjie2, Pan Wei1, 2
- 1School of Medical Laboratory Science, Guizhou Medical University, Guiyang 550004, Guizhou Province, China; 2Guizhou Provincial Prenatal Diagnosis Center, 3Clinical Laboratory Center, Guizhou Medical University Affiliated Hospital, Guiyang 550004, Guizhou Province, China; 4School of Basic Medical Sciences, Guizhou University of Traditional Chinese Medicine, Guiyang 550025, Guizhou Province, China
-
Received:2025-09-15Revised:2026-02-12Online:2026-09-08Published:2026-04-22 -
Contact:Pan Wei, PhD, Professor, School of Medical Laboratory Science, Guizhou Medical University, Guiyang 550004, Guizhou Province, China; Guizhou Provincial Prenatal Diagnosis Center, Guizhou Medical University Affiliated Hospital, Guiyang 550004, Guizhou Province, China Co-corresponding author: Xu Yongjie, PhD, Guizhou Provincial Prenatal Diagnosis Center, Guizhou Medical University Affiliated Hospital, Guiyang 550004, Guizhou Province, China -
About author:Chen Di, MS candidate, School of Medical Laboratory Science, Guizhou Medical University, Guiyang 550004, Guizhou Province, China; Guizhou Provincial Prenatal Diagnosis Center, Guizhou Medical University Affiliated Hospital, Guiyang 550004, Guizhou Province, China -
Supported by:the Regional Science Foundation Project of the National Natural Science Foundation of China, No. 82260165 (to PW); the National Natural Science Foundation of China (Youth Program), No. 82300920 (to XYJ); the Guizhou Provincial Science and Technology Plan Project, No. ZK[2024] General 199 (to XYJ); the Science and Technology Foundation of Guizhou Provincial Health Commission, No. gzwkj2024-081 (to XYJ); the National Natural Science Foundation Regional Fund Cultivation Program of the Affiliated Hospital of Guizhou Medical University, No. gyfyhsfc-2022-37 (to ZLY); the National Natural Science Foundation of China, No. 82560169 (to ZLY)
CLC Number:
Cite this article
Chen Di, Xu Mengling, Rao Binchan, Zhu Liying, Li Xing, Xu Yongjie, Pan Wei. Effects and mechanisms of glycemic variability on apoptosis in mouse hippocampal neuronal HT-22 cells[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(25): 6566-6574.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
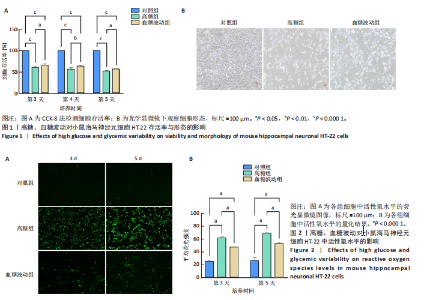
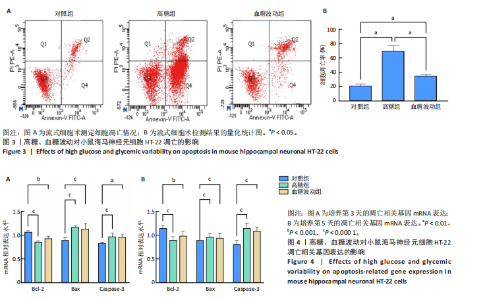
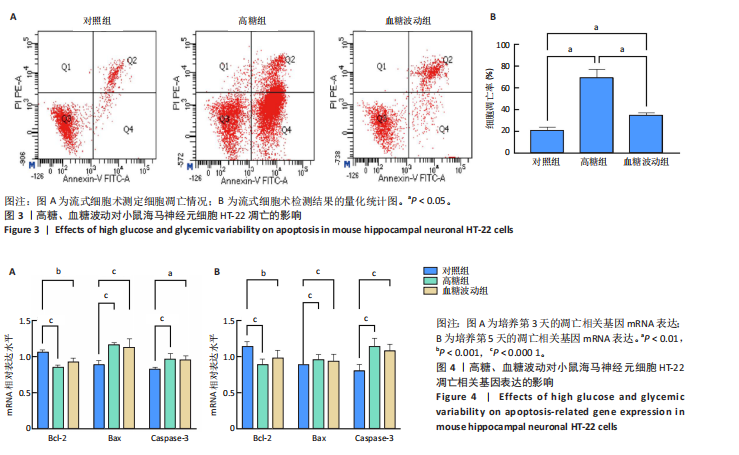
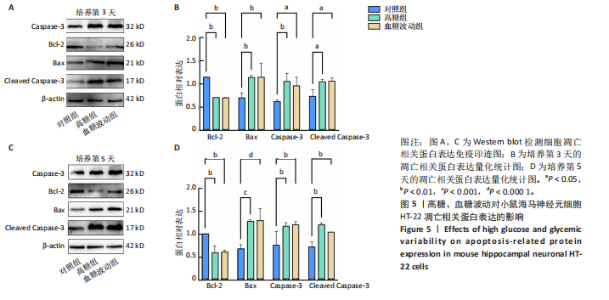
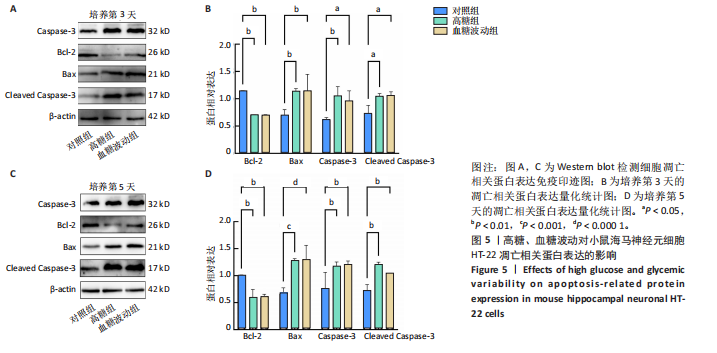
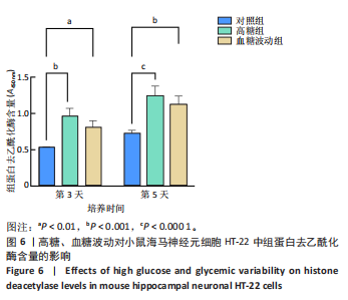
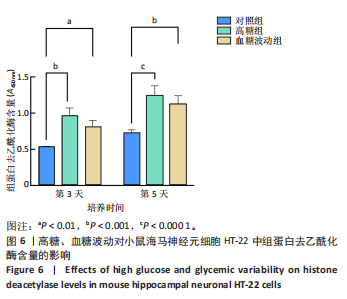
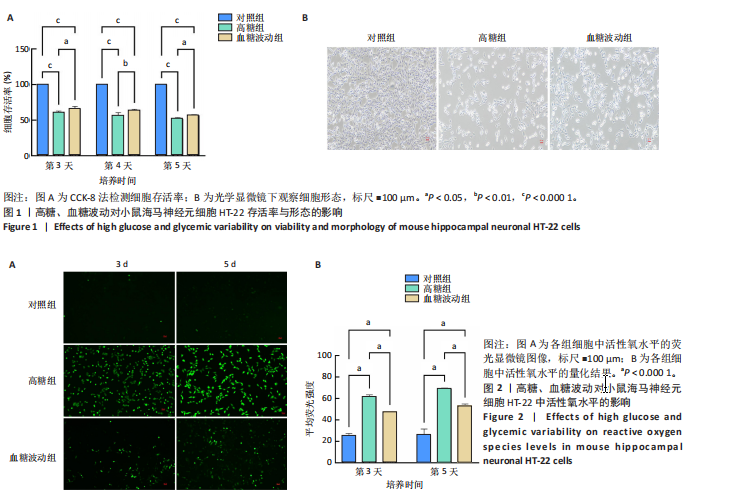
2.1 血糖波动对HT-22细胞存活率及形态影响 CCK-8检测结果显示,培养3,4,5 d后,高糖组、血糖波动组HT-22细胞存活率均低于对照组(P < 0.000 1),血糖波动组HT-22细胞存活率高于高糖组(P < 0.05,P < 0.01),见图1A。 光学显微镜下观察结果显示,对照组细胞生长状态良好,交织成致密网状,突触间相互连接;高糖组、血糖波动组细胞生长受抑制,细胞间突触连接减少,见图1B。 2.2 血糖波动对HT-22细胞中活性氧水平的影响 活性氧是细胞代谢产生的具有高反应性的含氧分子,活性氧过量积累可通过诱导氧化应激、损伤线粒体功能及激活Caspase级联反应,触发或加剧细胞凋亡进程。荧光探针检测结果显示,高糖和血糖波动均显著诱导神经元细胞中的活性氧积累;培养3,5 d后,高糖组、血糖波动组细胞中活性氧水平均高于对照组(P < 0.000 1),高糖组细胞中活性氧水平高于血糖波动组(P < 0.000 1),见图2。 2.3 血糖波动对HT-22细胞凋亡的影响 流式细胞术检测结果显示,高糖与血糖波动均可诱导神经元细胞凋亡;高糖组、血糖波动组细胞凋亡率高于对照组(P < 0.05),高糖组细胞凋亡率高于血糖波动组(P < 0.05),见图3。提示高糖环境和血糖波动可能通过促进细胞凋亡途径对神经元细胞造成损伤。 2.4 血糖波动对HT-22细胞凋亡相关基因表达的影响 RT-qPCR检测结果显示,培养3,5 d后,与对照组比较,高糖组、血糖波动组细胞中Bcl-2 mRNA表达降低(P < 0.001,P < 0.000 1),Bax、Caspase-3 mRNA表达升高(P < 0.01,P < 0.001,P < 0.000 1); 高糖组与血糖波动组之间的Bcl-2、Bax、Caspase-3 mRNA表达比较差异均无显著性意义(P > 0.05),见图4。 2.5 血糖波动对HT-22细胞凋亡相关蛋白表达的影响 Western blot检测结果显示,培养3,5 d后,与对照组比较,高糖组、血糖波动组细胞中Bcl-2蛋白表达降低(P < 0.01),Bax、Caspase-3、Cleaved Caspase-3蛋白表达升高(P < 0.05,P < 0.01,P < 0.001,P < 0.000 1);高糖组与血糖波动组之间的Bcl-2、Bax、Caspase-3、Cleaved Caspase-3蛋白表达比较差异均无显著性意义(P > 0.05),见图5。 2.6 血糖波动对HT-22细胞中组蛋白去乙酰化酶含量的影响 组蛋白去乙酰化酶是一类催化去除组蛋白赖氨酸残基乙酰基团的酶,通过重塑染色质结构抑制基因转录,参与细胞分化、周期调控及肿瘤发生等表观遗传过程。ELISA检测结果显示,培养3,5 d后,高糖组、血糖波动组细胞上清中组蛋白去乙酰化酶含量高于对照组(P < 0.01,P < 0.001,P < 0.000 1),高糖组与血糖波动组细胞上清中组蛋白去乙酰化酶含量比较差异无显著性意义(P > 0.05),见图6。"
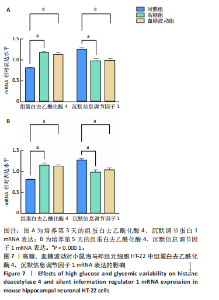
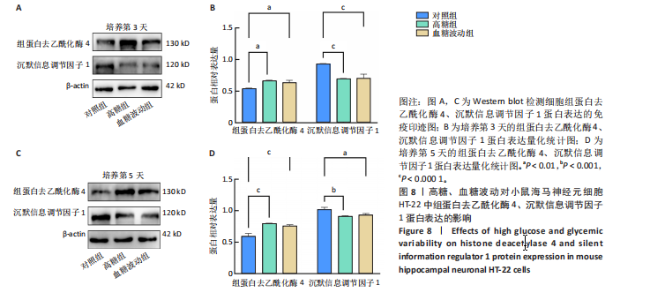
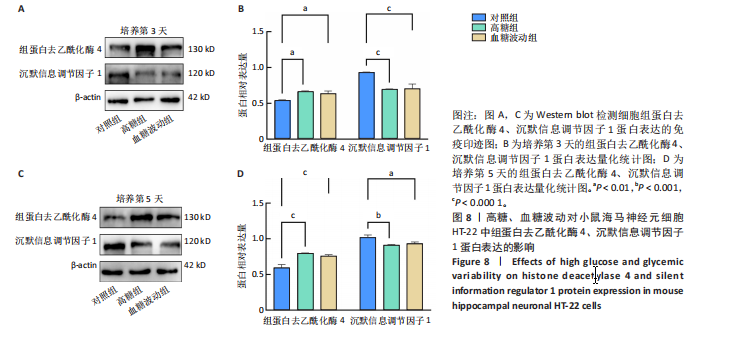
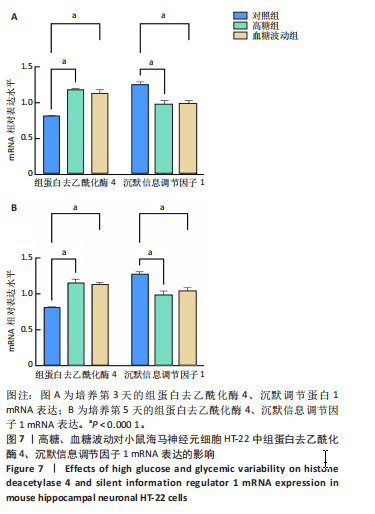
2.7 血糖波动对HT-22细胞中组蛋白去乙酰化酶4、沉默信息调节因子1 mRNA表达的影响 经典的组蛋白去乙酰化酶分为Ⅰ类、Ⅱ类、Ⅲ类和Ⅳ类。根据ELISA结果,此次研究挑选了组蛋白去乙酰化酶4(Ⅱa亚类)、沉默信息调节因子1(Ⅲ类)进行后续实验。 RT-qPCR检测结果显示,培养3,5 d后,与对照组比较,高糖组、血糖波动组细胞中组蛋白去乙酰化酶4 mRNA表达升高(P < 0.000 1),沉默信息调节因子1 mRNA表达降低(P < 0.000 1);高糖组与血糖波动组细胞中组蛋白去乙酰化酶4、沉默信息调节因子1 mRNA表达比较差异无显著性意义(P > 0.05),见图7。 2.8 血糖波动对HT-22细胞中组蛋白去乙酰化酶4、沉默信息调节因子1蛋白表达的影响 Western blot检测结果显示,培养3,5 d后,与对照组比较,高糖组、血糖波动组细胞中组蛋白去乙酰化酶4 蛋白表达升高(P < 0.01,P < 0.000 1),沉默信息调节因子1 蛋白表达降低(P < 0.01,P < 0.001,P < 0.000 1);高糖组与"
| [1] HOSSAIN MJ, AL-MAMUN M, ISLAM MR. Diabetes mellitus, the fastest growing global public health concern: Early detection should be focused. Health Sci Rep. 2024;7(3):e2004. [2] TAO S, YANG D, ZHANG L, et al. Knowledge domain and emerging trends in diabetic cardiomyopathy: A scientometric review based on CiteSpace analysis. Front Cardiovasc Med. 2022;9:891428. [3] LUO Y, ZHU J, HU Z, et al. Progress in the Pathogenesis of Diabetic Encephalopathy: The Key Role of Neuroinflammation. Diabetes Metab Res Rev. 2024;40(6):e3841. [4] LI Y, ZHANG H, LONG W, et al. Inhibition of NLRP3 and Golph3 ameliorates diabetes-induced neuroinflammation in vitro and in vivo. Aging (Albany NY). 2022;14(21):8745-8762. [5] MA S, BI W, LIU X, et al. Single-Cell Sequencing Analysis of the db/db Mouse Hippocampus Reveals Cell-Type-Specific Insights Into the Pathobiology of Diabetes-Associated Cognitive Dysfunction. Front Endocrinol (Lausanne). 2022;13:891039. [6] CAI J, CAI M, XIA W, et al. Explore the Mechanism of β-Asarone on Improving Cognitive Dysfunction in Rats with Diabetic Encephalopathy. J Alzheimers Dis Rep. 2022;6(1):195-206. [7] XU S, GAO Z, JIANG L, et al. High glucose- or AGE-induced oxidative stress inhibits hippocampal neuronal mitophagy through the Keap1-Nrf2-PHB2 pathway in diabetic encephalopathy. Sci Rep. 2024;14(1):24044. [8] LU M, YE X, DENG X, et al. Inhibition of LCP2 in T cells alleviated apoptosis and oxidative stress via PD-1/PD-L1 in diabetic retinopathy. Int Immunopharmacol. 2025;163:115240. [9] YANG T, FENG Q, SHAO M, et al. The role of metabolic memory in diabetic kidney disease: identification of key genes and therapeutic targets. Front Pharmacol. 2024;15:1379821. [10] DONG H, SUN Y, NIE L, et al. Metabolic memory: mechanisms and diseases. Signal Transduct Target Ther. 2024;9(1):38. [11] TESTA R, BONFIGLI AR, PRATTICHIZZO F, et al. The “Metabolic Memory” Theory and the Early Treatment of Hyperglycemia in Prevention of Diabetic Complications. Nutrients. 2017;9(5):437. [12] KOWLURU RA, MOHAMMAD G. Epigenetics and Mitochondrial Stability in the Metabolic Memory Phenomenon Associated with Continued Progression of Diabetic Retinopathy. Sci Rep. 2020;10(1):6655. [13] HUANG L, PAN Y, ZHOU K, et al. Correlation Between Glycemic Variability and Diabetic Complications: A Narrative Review. Int J Gen Med. 2023;16:3083-3094. [14] DANSINGER ML, GLEASON JA, MADDALENA J, et al. Lifestyle Modification in Prediabetes and Diabetes: A Large Population Analysis. Nutrients. 2025; 17(8):1333. [15] FONSECA LM, HAWKS ZW, BEERI MS, et al. Cognitive vulnerability to glucose fluctuations: A digital phenotype of neurodegeneration. Alzheimers Dement. 2025;21(2):e70001. [16] LAZAR S, REUREAN-PINTILEI DV, IONITA I, et al. Glycemic Variability and Its Association with Traditional Glycemic Control Biomarkers in Patients with Type 1 Diabetes: A Cross-Sectional, Multicenter Study. J Clin Med. 2025;14(7):2434. [17] WANG H, DENG J, CHEN L, et al. Acute glucose fluctuation induces inflammation and neurons apoptosis in hippocampal tissues of diabetic rats. J Cell Biochem. 2021;122(9):1239-1247. [18] YANG W, SI SC, LUO HY, et al. Cognitive impairment and hippocampal degeneration in aged rat models of type 2 diabetes with induced glycemic fluctuation: A pilot study. Brain Res. 2025;1850:149452. [19] CHEN J, HUANG W, LIANG N. Blood glucose fluctuation and in-hospital mortality among patients with acute myocardial infarction: eICU collaborative research database. PLoS One. 2024;19(4):e0300323. [20] LI W, SONG C, LI X, et al. Procalcitonin contributes to hippocampal neuronal damage and impairment of LTP: implications for cognitive dysfunction in LPS-induced neuroinflammation rat model. Arch Microbiol. 2025;207(6):127. [21] CHU J, LIU CX, SONG R, et al. Ferrostatin-1 protects HT-22 cells from oxidative toxicity. Neural Regen Res. 2020;15(3):528-536. [22] 段云峰,许永劼,杨婷婷,等.高糖诱导HT-22小鼠海马神经元代谢记忆细胞模型的构建及影响[J].天津医药,2024,52(1):44-50. [23] SONTHALIA M, ROY BS, CHANDRAWANSHI D, et al. Histone deacetylase inhibitors as antidiabetic agents: Advances and opportunities. Eur J Pharmacol. 2022;935:175328. [24] SPIRIN P, VEDERNIKOVA V, VOLKAVA T, et al. New and Effective Inhibitor of Class I HDACs, Eimbinostat, Reduces the Growth of Hematologic Cancer Cells and Triggers Apoptosis. Pharmaceutics. 2025;17(4):416. [25] MCCLARTY BM, RODRIGUEZ G, DONG H. Class 1 histone deacetylases differentially modulate memory and synaptic genes in a spatial and temporal manner in aged and APP/PS1 mice. Brain Res. 2024;1837:148951. [26] CAO W, JI Z, ZHU S, et al. Bioinformatic identification and experiment validation reveal 6 hub genes, promising diagnostic and therapeutic targets for Alzheimer’s disease. BMC Med Genomics. 2024;17(1):6. [27] 许永劼,许雯,陈钢,等.2种高糖诱导海马神经元模型应用及优势比较[J].中国比较医学杂志,2021,31(8):1-8. [28] LI Y, WANG G, ZHANG Y, et al. Biological effects of exosomes derived from 2D and 3D culture adipose stem cells on JEC Cell proliferation and migration. Front Bioeng Biotechnol. 2025;13:1541150. [29] TELFORD WG, KOMORIYA A, PACKARD BZ, et al. Multiparametric analysis of apoptosis by flow cytometry. Methods Mol Biol. 2011;699:203-227. [30] LI M, WANG Y, KE Q, et al. The inhibitory and anti-inflammatory effects of TMP269 on peste des petits ruminants virus replication. Virulence. 2025;16(1):2495838. [31] ALPEROVICH NY, VASILYEVA OB, SCHAFFTER SW. Prevention of ribozyme catalysis through cDNA synthesis enables accurate RT-qPCR measurements of context-dependent ribozyme activity. RNA. 2025;31(5):633-645. [32] LIN X, WANG S, LIU X, et al. Enhanced sensitivity in PCSK9 detection using binding-induced DNA walker-triggered Argonaute protein-based DNA circuit. Mikrochim Acta. 2025;192(8):509. [33] LIU D, ZHENG Z, CHEN Z, et al. Ferroptosis in Müller cells under hyperglycemia: mechanisms and therapeutic implications for diabetic retinopathy-associated optic neuroinflammation. Int Ophthalmol. 2025; 45(1):302. [34] HONGMEI H, MAOJUN Y, TING LI, et al. Neferine inhibits the progression of diabetic nephropathy by modulating the miR-17-5p/nuclear factor E2-related factor 2 axis. J Tradit Chin Med. 2024;44(1):44-53. [35] DEN BRABER N, VOLLENBROEK-HUTTEN MMR, TEUNISSEN SEM, et al. The Contribution of Postprandial Glucose Levels to Hyperglycemia in Type 2 Diabetes Calculated from Continuous Glucose Monitoring Data: Real World Evidence from the DIALECT-2 Cohort. Nutrients. 2024;16(20):3557. [36] KO J, JANG S, KWON W, et al. Protective Effect of GIP against Monosodium Glutamate-Induced Ferroptosis in Mouse Hippocampal HT-22 Cells through the MAPK Signaling Pathway. Antioxidants (Basel). 2022;11(2):189. [37] SUN Y, LU Y, SAREDY J, et al. ROS systems are a new integrated network for sensing homeostasis and alarming stresses in organelle metabolic processes. Redox Biol. 2020;37:101696. [38] GONZÁLEZ P, LOZANO P, ROS G, et al. Hyperglycemia and Oxidative Stress: An Integral, Updated and Critical Overview of Their Metabolic Interconnections. Int J Mol Sci. 2023;24(11):9352. [39] KATO A, NIHEI W, YAKO H, et al. Imeglimin improves hyperglycemia and hypoglycemia-induced cell death and mitochondrial dysfunction in immortalized adult mouse Schwann IMS32 cells. J Diabetes Investig. 2025. doi: 10.1111/jdi.70092. [40] DU W, SHI G, SHAN CM, et al. Mechanisms of chromatin-based epigenetic inheritance. Sci China Life Sci. 2022;65(11):2162-2190. [41] MAKKAR R, BEHL T, ARORA S. Role of HDAC inhibitors in diabetes mellitus. Curr Res Transl Med. 2020;68(2):45-50. [42] JIN T, WANG Z, FAN F, et al. HDAC1 Promotes Mitochondrial Pathway Apoptosis and Inhibits the Endoplasmic Reticulum Stress Response in High Glucose-Treated Schwann Cells via Decreased U4 Spliceosomal RNA. Neurochem Res. 2024;49(10):2699-2724. [43] COLUSSI C, ACETO G, RIPOLI C, et al. Cytoplasmic HDAC4 recovers synaptic function in the 3×Tg mouse model of Alzheimer’s disease. Neuropathol Appl Neurobiol. 2023;49(1):e12861. [44] KUMAR KK, ABURAWI EH, LJUBISAVLJEVIC M, et al. Exploring histone deacetylases in type 2 diabetes mellitus: pathophysiological insights and therapeutic avenues. Clin Epigenetics. 2024;16(1):78. [45] LI M, SONG J, NIU X, MO F, et al. Baicalein ameliorates cognitive decline induced by chronic cerebral hypoperfusion through the SIRT1-mediated Notch1 pathway to improve angiogenesis and suppress neuroinflammation. Front Aging Neurosci. 2025;17:1521353. [46] ZHAO Y, ZHANG AP, BAO BY, et al. Sirt1 protects lupus nephritis by inhibiting the NLRP3 signaling pathway in human glomerular mesangial cells. Open Life Sci. 2025;20(1):20221038. [47] LI Y, YISHAJIANG S, CHEN Y, et al. TRPC5-mediated NLRP3 inflammasome activation contributes to myocardial cell pyroptosis in chronic intermittent hypoxia rats. Acta Cardiol. 2024;79(7):796-804. [48] 许永劼,陈钢,林海容,等.慢病毒干扰HDAC4对HT-22小鼠海马神经元凋亡的影响及稳转株建立[J].中国老年学杂志,2024,44(16):3919-3923. [49] 王安迪.妊娠期糖尿病中LPS通过HDAC1影响SIRT1相关胰岛素通路的机制研究[D].重庆:重庆医科大学,2022. [50] HUANG X, SHI Y, CHEN H, et al. Isoliquiritigenin prevents hyperglycemia-induced renal injuries by inhibiting inflammation and oxidative stress via SIRT1-dependent mechanism. Cell Death Dis. 2020;11(12):1040. [51] HUANG Z, LI Y, XIE Y, et al. Jiawei Erzhiwan Ameliorates Androgenetic Alopecia by Regulating the SIRT1/JNK/p38 MAPK Pathway. Drug Des Devel Ther. 2025;19:2393-2409. [52] XU Y, LI H, CHEN G, et al. Radix polygoni multiflori protects against hippocampal neuronal apoptosis in diabetic encephalopathy by inhibiting the HDAC4/JNK pathway. Biomed Pharmacother. 2022;153:113427. [53] WU LD, LIU Y, LI F, et al. Glucose fluctuation promotes cardiomyocyte apoptosis by triggering endoplasmic reticulum (ER) stress signaling pathway in vivo and in vitro. Bioengineered. 2022;13(5):13739-13751. |
| [1] | Liu Anting, Lu Jiangtao, Zhang Wenjie, He Ling, Tang Zongsheng, Chen Xiaoling. Regulation of AMP-activated protein kinase by platelet lysate inhibits cadmium-induced neuronal apoptosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1800-1807. |
| [2] | Yuan Xiaoshuang, Yang Xu, Yang Bo, Chen Xiaoxu, Tian Ting, Wang Feiqing, Li Yanju, Liu Yang, Yang Wenxiu. Effect of conditioned medium of diffuse large B-cell lymphoma cells on proliferation and apoptosis of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1632-1640. |
| [3] | He Jiale, Huang Xi, Dong Hongfei, Chen Lang, Zhong Fangyu, Li Xianhui. Acellular dermal matrix combined with adipose-derived stem cell exosomes promotes burn wound healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1699-1710. |
| [4] | Jia Jinwen, Airefate·Ainiwaer, Zhang Juan. Effects of EP300 on autophagy and apoptosis related to allergic rhinitis in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1439-1449. |
| [5] | Liu Yu, Lei Senlin, Zhou Jintao, Liu Hui, Li Xianhui. Mechanisms by which aerobic and resistance exercises improve obesity-related cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1171-1183. |
| [6] | Wang Zhengye, Liu Wanlin, Zhao Zhenqun. Advance in the mechanisms underlying miRNAs in steroid-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1207-1214. |
| [7] | Yang Xiao, Bai Yuehui, Zhao Tiantian, Wang Donghao, Zhao Chen, Yuan Shuo. Cartilage degeneration in temporomandibular joint osteoarthritis: mechanisms and regenerative challenges [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 926-935. |
| [8] | Bao Zhuoma, Hou Ziming, Jiang Lu, Li Weiyi, Zhang Zongxing, Liu Daozhong, Yuan Lin. Effect and mechanism by which Pterocarya hupehensis skan total flavonoids regulates the proliferation, migration and apoptosis of fibroblast-like synoviocytes [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 816-823. |
| [9] | Ma Runqiu, Yang Huixia, Li Xuer, Bai Zhigang, Li Guizhong, Hao Yinju, Ma Shengchao, Jiang Yideng. Mechanism of glucocorticoid-induced mitochondrial dysfunction in osteoblasts in steroid-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 8845-8851. |
| [10] | Mu Bingtao, Guo Minfang, Hu Fenqi, Liu Qiyuan, Jia Hui, Xu Mingyuan, Chen Jiayuan, Zhang Huiyu, Meng Tao, Yu Jiezhong. Mitochondrial kinetic mechanism by which triptolide alleviates hydrogen peroxide-induced apoptosis in SH-SY5Y cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 8986-8993. |
| [11] | Wang Zhengye, Liu Wanlin, Zhao Zhenqun. Mechanism by which vascular endothelial growth factor A targets regulation of angiogenesis in the treatment of steroid-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 671-679. |
| [12] | Zhang Zhilong, Wang Haiying, Ma Fenghua, Hou Yanjie. Regulating mitochondrial dynamics balance in nucleus pulposus cells inhibits cell apoptosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7520-7528. |
| [13] | Zhang Jingyi, Ma Fang, Liu Honglin, Wang Jianjun, Xia Tongtong, Yang Jiaqi, Wang Yajing, Shen Jiangyong, Jiang Yideng. Regulatory role of ADAMTS8 in proliferation and apoptosis of hypertrophic scar fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7260-7266. |
| [14] | Wang Qing, Yang Zhichao, Liu Jian, Liang Yajie, Tang Yibin, Guo Yu, Song Guobin, Ma Cungen. Grape seed proanthocyanidin oligomers alleviate demyelination in cuprizone-fed mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(25): 6463-6471. |
| [15] | Wang Zhengye, Liu Wanlin, Zhao Zhenqun. Mechanisms of miRNAs involved in cartilage development: new strategies and targets [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6289-6296. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||