Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (25): 6575-6583.doi: 10.12307/2026.410
Previous Articles Next Articles
Matrine promotes macrophage polarization to repair myocardial tissue injury in rats
Zhao Yongjian1, Ge Yunxiao2, Yin Yunfei1, Jiang Tingbo1
- 1Department of Cardiology, the First Affiliated Hospital of Soochow University, Suzhou 215006, Jiangsu Province, China; 2Department of Pediatrics, Beijing Friendship Hospital, Capital Medical University, Beijing 100032, China
-
Received:2025-07-06Revised:2025-11-27Online:2026-09-08Published:2026-04-22 -
Contact:Jiang Tingbo, Chief physician, Professor, Department of Cardiology, the First Affiliated Hospital of Soochow University, Suzhou 215006, Jiangsu Province, China -
About author:Zhao Yongjian, MS, Physician, Department of Cardiology, the First Affiliated Hospital of Soochow University, Suzhou 215006, Jiangsu Province, China -
Supported by:the Bo Xi Discipline Program - Cardiology Department, Class II Project of the First Affiliated Hospital of Soochow University (to JTB)
CLC Number:
Cite this article
Zhao Yongjian, Ge Yunxiao, Yin Yunfei, Jiang Tingbo. Matrine promotes macrophage polarization to repair myocardial tissue injury in rats[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(25): 6575-6583.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
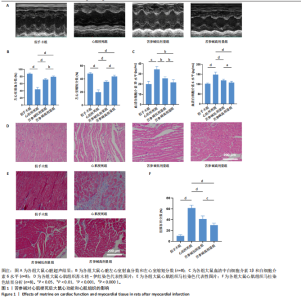
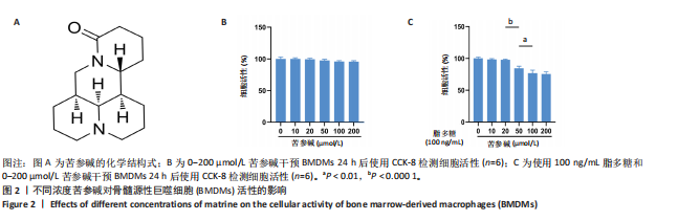
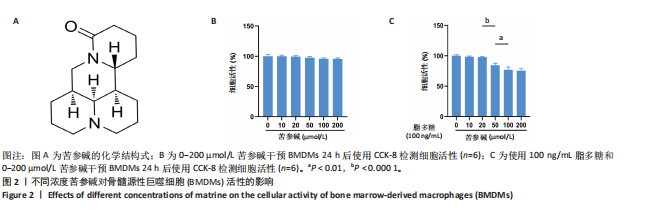
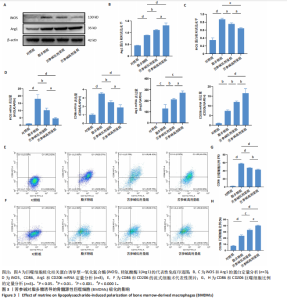
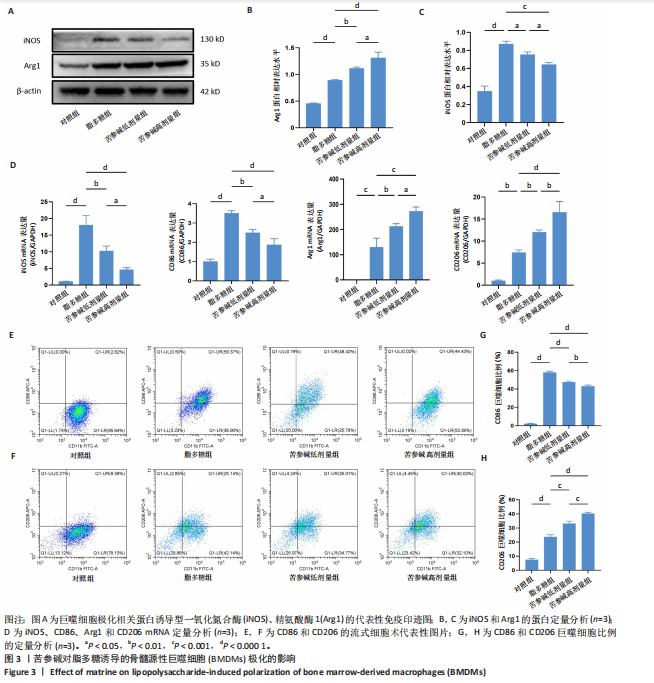
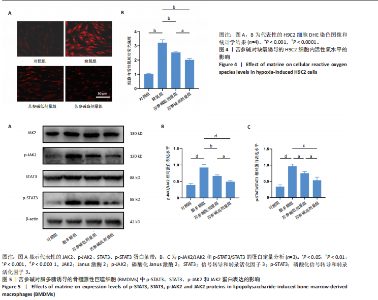
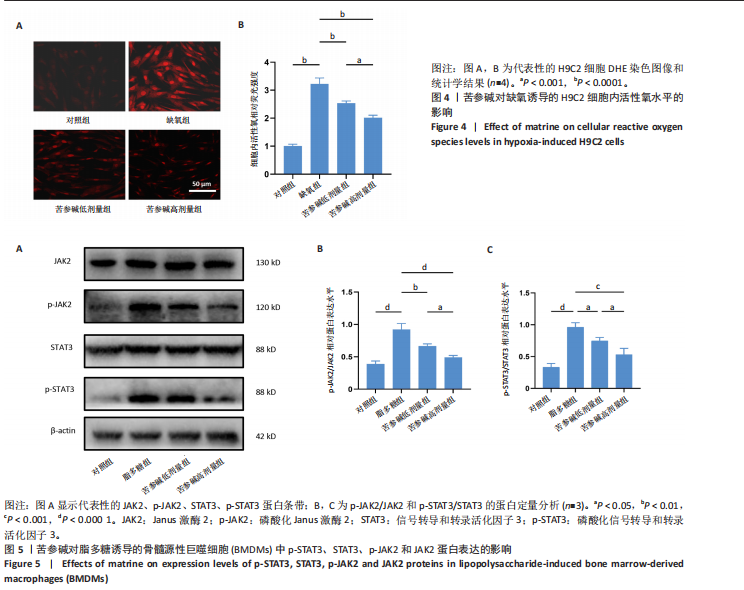
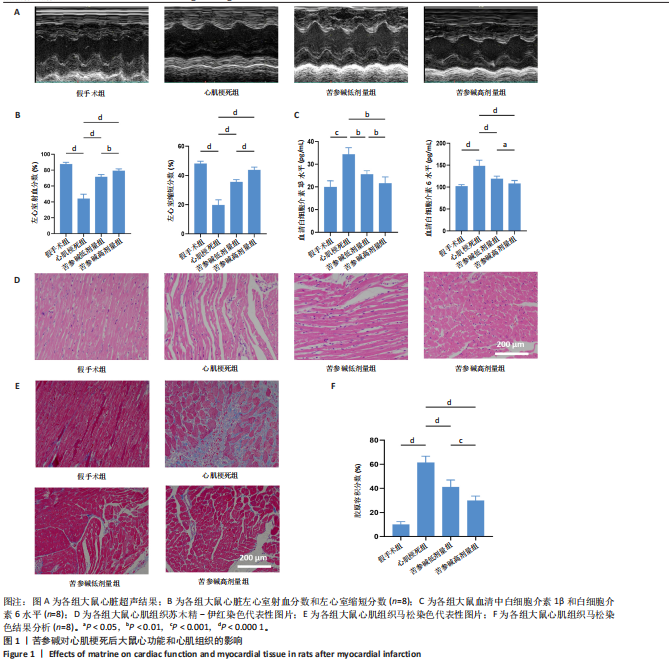
2.1 苦参碱对大鼠心肌梗死后炎症反应和心脏功能的影响 2.1.1 大鼠心脏超声检测结果 术后4 d进行心脏超声检测,结果显示,心肌梗死组大鼠左心室射血分数和左心室缩短分数相较于假手术组显著下降(P < 0.000 1);使用苦参碱治疗后左心室射血分数和左心室缩短分数显著升高(P < 0.000 1),且苦参碱高剂量组治疗效果更为明显(P < 0.01,P < 0.000 1),见图1A,B,这表明苦参碱治疗可以显著改善心肌梗死大鼠的心功能,从而起到保护心肌的作用。 2.1.2 血清ELISA检测结果 心肌梗死组大鼠血清中白细胞介素1β和白细胞介素6水平相较于假手术组明显升高(P < 0.001,P < 0.000 1);而苦参碱组大鼠血清中白细胞介素1β和白细胞介素6水平相较于心肌梗死组则明显下降(P < 0.01,P < 0.000 1),且苦参碱高剂量组降低效果更为显著(P < 0.05,P < 0.01),见图1C,这说明苦参碱可以有效降低大鼠心肌梗死后炎症环境中的炎性细胞因子,从而减弱心肌梗死后的炎症环境。 2.1.3 大鼠心脏苏木精-伊红染色结果 心肌梗死组大鼠心肌组织相较于假手术组心肌纤维排列紊乱,细胞肿胀坏死,心脏间质中的胶原纤维增生、炎症细胞浸润,而苦参碱组大鼠心肌组织相较于心肌梗死组心肌排列相对整齐,细胞形态相对正常,炎症细胞浸润减少,且苦参碱高剂量组心肌排列更为整齐,细胞形态更为正常,炎症细胞浸润显著减少,见图1D,这表明苦参碱可以显著减轻心肌梗死后的心肌损伤,减少心室纤维化,进而保护心肌组织。 2.1.4 大鼠心脏马松染色结果 与假手术组相比,心肌梗死组大鼠心肌组织有大量胶原纤维浸润(P < 0.000 1),而苦参碱组相较于心肌梗死组胶原纤维则浸润显著减少(P < 0.000 1),且苦参碱高剂量组效果更为显著(P < 0.001),见图1E,F,这说明苦参碱可以显著减少心肌梗死后胶原纤维的浸润,改善心肌梗死后的心肌纤维化,从而保护心脏。 综上所述,体内实验证实了苦参碱能够有效减轻心肌梗死后的炎症反应,改善心脏功能,保护心肌组织,并且苦参碱对心肌组织的保护作用存在一定的剂量依赖性。 2.2 苦参碱对骨髓源性巨噬细胞活性的影响 2.2.1 不同浓度苦参碱对骨髓源性巨噬细胞活性的影响 苦参碱是一种喹唑啉类化合物,化学结构式见图2A。为评估苦参碱对骨髓源性巨噬细胞的药物安全性,采用CCK-8法检测细胞活性,以确定苦参碱在细胞培养中的安全浓度范围,见图2B,结果发现各个浓度(0,10,20,50,100,200 μmol/L)苦参碱对骨髓源性巨噬细胞的活性没有显著影响。 2.2.2 不同浓度苦参碱在脂多糖刺激下对骨髓源性巨噬细胞活性的影响 在不同浓度(0,10,20,50,100,200 μmol/L)苦参碱处理的培养基中加入100 ng/mL脂多糖干预24 h后,采用CCK-8检测不同浓度苦参碱对骨髓源性巨噬细胞活性的影响,见图2C,与20 μmol/L苦参碱相比,50 μmol/L苦参碱显著降低了细胞活性(P < 0.000 1)。因此,综合上述实验结果,在保证骨髓源性巨噬细胞足够活性的条件下,最终确定使用10 μmol/L苦参碱作为低剂量治疗组,使用20 μmol/L苦参碱作为高剂量治疗组,共同干预24 h进行后续实验。 2.3 苦参碱能够促进巨噬细胞由M1型向M2型极化 2.3.1 Western blot 检测结果 与对照组相比,脂多糖组诱导型一氧化氮合酶和精氨酸酶1蛋白表达增加(P < 0.000 1);与脂多糖组相比,苦参碱组诱导型一氧化氮合酶蛋白表达减少(P < 0.05,P < 0.001),而精氨酸酶1蛋白表达持续增加(P < 0.01,P < 0.000 1),且苦参碱高剂量组效果更加明显(P < 0.05),见图3A-C,这表明苦参碱能有效抑制脂多糖引发的巨噬细胞M1型极化,促进巨噬细胞向M2型极化。 2.3.2 RT-qPCR检测结果 与对照组相比,脂多糖组诱导型一氧化氮合酶、CD86、精氨酸酶1和CD206 mRNA表达增加(P < 0.01,P < 0.001,P < 0.000 1);与脂多糖组相比,苦参碱组诱导型一氧化氮合酶和CD86 mRNA表达减少(P < 0.01,P < 0.000 1),精氨酸酶1和CD206 mRNA表达持续增加(P < 0.01,P < 0.001,P < 0.000 1),在苦参碱高剂量组更加明显(P < 0.05,P < 0.01),见图3D。这进一步验证了苦参碱可以促进巨噬细胞由M1型向M2型极化,发挥抗炎作用。 2.3.3 流式细胞术检测结果 CD11b、CD86和CD206分别是巨噬细胞、M1型巨噬细胞和M2型巨噬细胞的特异性表面标志物。通过流式细胞术分析4组细胞表面标志物CD86和CD206的表达,与对照组相比,脂多糖组M1型和M2型巨噬细胞比例显著升高。与脂多糖组相比,苦参碱组M1型巨噬细胞比例明显下降而M2型巨噬细胞比例明显增加,并且苦参碱高剂量组效果更加明显,见图3E-H。这表明苦参碱可以有效抑制脂多糖对M1型巨噬细胞的诱导极化,减少M1型巨噬细胞比例,促进M2型极化,减弱炎症反应。 综上所述,研究表明苦参碱能够有效抑制脂多糖诱导的巨噬细胞M1型极化,促进M2型极化,发挥抗炎保护作用,并且苦参碱的治疗效果存在剂量依赖性。 2.4 苦参碱对H9C2细胞内活性氧水平的影响 与对照组相比,缺氧组细胞中活性氧水平明显升高(P < 0.000 1);与缺氧组相比,苦参碱组H9C2细胞内活性氧水平明显降低(P < 0.000 1),且苦参碱高剂量组降低程度更为明显(P < 0.001),见图4A,B。这表明苦参碱可以减轻H9C2细胞因缺氧导致的氧化应激损伤,从而起到保护心肌细胞的作用,并且治疗效果存在剂量依赖性。 2.5 苦参碱抑制脂多糖诱导骨髓源性巨噬细胞中p-JAK2、p-STAT3蛋白表达 与对照组相比,脂多糖组p-JAK2、p-STAT3蛋白表达水平明显升高(P < 0.000 1);与脂多糖组相比,苦参碱组p-JAK2、p-STAT3蛋白表达水平明显降低(P < 0.05,P < 0.01,P < 0.001,P < 0.000 1),且苦参碱高剂量组降低程度更为明显(P < 0.05),见图5A-C。这表明苦参碱可以抑制骨髓源性巨噬细胞中JAK2和STAT3的磷酸化水平,从而减弱炎症反应,起到保护作用。"
| [1] JENSEN RV, HJORTBAK MV, BØTKER HE. Ischemic Heart Disease: An Update. Semin Nucl Med. 2020;50(3):195-207. [2] ANDREADOU I, CABRERA-FUENTES HA, DEVAUX Y, et al. Immune cells as targets for cardioprotection: new players and novel therapeutic opportunities. Cardiovasc Res. 2019;115(7):1117-1130. [3] HERNANDEZ AF, UDELL JA, JONES WS, et al. Effect of Empagliflozin on Heart Failure Outcomes After Acute Myocardial Infarction: Insights From the EMPACT-MI Trial. Circulation. 2024;149(21):1627-1638. [4] CARBERRY J, MARQUIS-GRAVEL G, O’MEARA E, et al. Where Are We With Treatment and Prevention of Heart Failure in Patients Post-Myocardial Infarction? JACC Heart Fail. 2024;12(7):1157-1165. [5] NONG J, GLASSMAN PM, MUZYKANTOV VR. Targeting vascular inflammation through emerging methods and drug carriers. Adv Drug Deliv Rev. 2022;184: 114180. [6] ONG SB, HERNÁNDEZ-RESÉNDIZ S, CRESPO-AVILAN GE, et al. Inflammation following acute myocardial infarction: Multiple players, dynamic roles, and novel therapeutic opportunities. Pharmacol Ther. 2018;186:73-87. [7] YANG XM, CUI L, WHITE J, et al. Mitochondrially targeted Endonuclease III has a powerful anti-infarct effect in an in vivo rat model of myocardial ischemia/reperfusion. Basic Res Cardiol. 2015;110(2):3. [8] KIM Y, NURAKHAYEV S, NURKESH A, et al. Macrophage Polarization in Cardiac Tissue Repair Following Myocardial Infarction. Int J Mol Sci. 2021;22(5):2715. [9] YANG Y, WU A, DENG AN, et al. Macrophages after myocardial infarction: Mechanisms for repairing and potential as therapeutic approaches. Int Immunopharmacol. 2024;143(Pt 3):113562. [10] SUN X, LI Y, DENG Q, et al. Macrophage Polarization, Metabolic Reprogramming, and Inflammatory Effects in Ischemic Heart Disease. Front Immunol. 2022;13: 934040. [11] LIU S, CHEN J, SHI J, et al. M1-like macrophage-derived exosomes suppress angiogenesis and exacerbate cardiac dysfunction in a myocardial infarction microenvironment. Basic Res Cardiol. 2020;115(2):22. [12] WANG Y, PENG M, YANG X, et al. Total alkaloids in Fritillaria cirrhosa D. Don alleviate OVA-induced allergic asthma by inhibiting M2 macrophage polarization. J Ethnopharmacol. 2025;337(Pt 3):118935. [13] DICK SA, MACKLIN JA, NEJAT S, et al. Self-renewing resident cardiac macrophages limit adverse remodeling following myocardial infarction. Nat Immunol. 2019; 20(1):29-39. [14] RURIK JG, AGHAJANIAN H, EPSTEIN JA. Immune Cells and Immunotherapy for Cardiac Injury and Repair. Circ Res. 2021;128(11):1766-1779. [15] BAO H, WANG X, ZHOU H, et al. PCSK9 regulates myofibroblast transformation through the JAK2/STAT3 pathway to regulate fibrosis after myocardial infarction. Biochem Pharmacol. 2024;220:115996. [16] WANG H, SUN N, SUN P, et al. Matrine regulates autophagy in ileal epithelial cells in a porcine circovirus type 2-infected murine model. Front Microbiol. 2024;15:1455049. [17] WANG X, WU FP, HUANG YR, et al. Matrine suppresses NLRP3 inflammasome activation via regulating PTPN2/JNK/SREBP2 pathway in sepsis. Phytomedicine. 2023;109:154574. [18] XIAO Y, YU Y, HU L, et al. Matrine Alleviates Sepsis-Induced Myocardial Injury by Inhibiting Ferroptosis and Apoptosis. Inflammation. 2023;46(5):1684-1696. [19] ZHANG Y, CUI L, GUAN G, et al. Matrine suppresses cardiac fibrosis by inhibiting the TGF‑β/Smad pathway in experimental diabetic cardiomyopathy. Mol Med Rep. 2018;17(1):1775-1781. [20] HU C, ZHANG X, WEI W, et al. Matrine attenuates oxidative stress and cardiomyocyte apoptosis in doxorubicin-induced cardiotoxicity via maintaining AMPKα/UCP2 pathway. Acta Pharm Sin B. 2019;9(4):690-701. [21] YAP J, IREI J, LOZANO-GERONA J, et al. Macrophages in cardiac remodelling after myocardial infarction. Nat Rev Cardiol. 2023;20(6):373-385. [22] YE X, LIN ZJ, HONG GH, et al. Pyroptosis inhibitors MCC950 and VX-765 mitigate myocardial injury by alleviating oxidative stress, inflammation, and apoptosis in acute myocardial hypoxia. Exp Cell Res. 2024;438(2):114061. [23] CARLSON S, HELTERLINE D, ASBE L, et al. Cardiac macrophages adopt profibrotic/M2 phenotype in infarcted hearts: Role of urokinase plasminogen activator. J Mol Cell Cardiol. 2017;108:42-49. [24] THORP EB. Cardiac macrophages and emerging roles for their metabolism after myocardial infarction. J Clin Invest. 2023;133(18):e171953. [25] LI L, CAO J, LI S, et al. M2 Macrophage-Derived sEV Regulate Pro-Inflammatory CCR2+ Macrophage Subpopulations to Favor Post-AMI Cardiac Repair. Adv Sci (Weinh). 2023;10(14):e2202964. [26] KANURI B, SREEJIT G, BISWAS P, et al. Macrophage heterogeneity in myocardial infarction: Evolution and implications for diverse therapeutic approaches. iScience. 2024;27(7):110274. [27] LI Z, WANG S, QIN Y, et al. Gabapentin attenuates cardiac remodeling after myocardial infarction by inhibiting M1 macrophage polarization through the peroxisome proliferator-activated receptor-γ pathway. Eur J Pharmacol. 2024;967:176398. [28] DING P, LIU J, MENG Y, et al. MFG-E8 facilitates heart repair through M1/M2 polarization after myocardial infarction by inhibiting CaMKII. Int Immunopharmacol. 2024;126:111216. [29] 常金霞,刘羽飞,牛少辉,等.巨噬细胞极化在组织修复过程中的可视化分析[J].中国组织工程研究,2025,29(7):1486-1496. [30] JUNG M, DODSWORTH M, THUM T. Inflammatory cells and their non-coding RNAs as targets for treating myocardial infarction. Basic Res Cardiol. 2018;114(1):4. [31] MA S, HE H, REN X, et al. Luteolin ameliorates periodontitis by modulating mitochondrial dynamics and macrophage polarization via the JAK2/STAT3 pathway. Int Immunopharmacol. 2025;144:113612. [32] ZHAO Y, TAN M, YIN Y, et al. Comprehensive macro and micro views on immune cells in ischemic heart disease. Cell Prolif. 2024;57(12):e13725. [33] CAI S, ZHAO M, ZHOU B, et al. Mitochondrial dysfunction in macrophages promotes inflammation and suppresses repair after myocardial infarction. J Clin Invest. 2023;133(4):e159498. [34] ZUO W, SUN R, JI Z, et al. Macrophage-driven cardiac inflammation and healing: insights from homeostasis and myocardial infarction. Cell Mol Biol Lett. 2023;28(1):81. [35] MAO N, YU Y, CUI J, et al. Effect of Matrine on growth performance, gut health, and gut microbiota in chickens infected with avian pathogenic Escherichia coli. Poult Sci. 2025;104(1):104520. [36] MAO N, YU Y, LU X, et al. Preventive effects of matrine on LPS-induced inflammation in RAW 264.7 cells and intestinal damage in mice through the TLR4/NF-κB/MAPK pathway. Int Immunopharmacol. 2024;143(Pt 2):113432. [37] ZHU C, ZHANG M, GONG S, et al. Identification of Matrine as a Kirsten rats Arcomaviral oncogene homolog inhibitor alleviating chemotherapy-induced neuropathic pain. Phytomedicine. 2024;132:155841. [38] MO RL, LI Z, ZHANG P, et al. Matrine inhibits invasion and migration of gallbladder cancer via regulating the PI3K/AKT signaling pathway. Naunyn Schmiedebergs Arch Pharmacol. 2024;397(10):8129-8143. [39] QIAO WT, YAO X, LU WH, et al. Matrine exhibits antiviral activities against PEDV by directly targeting Spike protein of the virus and inducing apoptosis via the MAPK signaling pathway. Int J Biol Macromol. 2024;270(Pt 2):132408. [40] DE BOCK CE, DEMEYER S, DEGRYSE S, et al. HOXA9 Cooperates with Activated JAK/STAT Signaling to Drive Leukemia Development. Cancer Discov. 2018;8(5):616-631. [41] MCINNES IB, SCHETT G. Pathogenetic insights from the treatment of rheumatoid arthritis. Lancet. 2017;389(10086):2328-2337. [42] LI Y, ZHANG X, CUI L, et al. Salvianolic acids enhance cerebral angiogenesis and neurological recovery by activating JAK2/STAT3 signaling pathway after ischemic stroke in mice. J Neurochem. 2017;143(1):87-99. [43] RAO T, TONG H, LI J, et al. Exploring the role and mechanism of hyperoside against cardiomyocyte injury in mice with myocardial infarction based on JAK2/STAT3 signaling pathway. Phytomedicine. 2024;128:155319. [44] ZHANG Y, WANG D, ZHAO Z, et al. Nephronectin promotes cardiac repair post myocardial infarction via activating EGFR/JAK2/STAT3 pathway. Int J Med Sci. 2022;19(5):878-892. [45] YU L, ZHANG Y, CHEN Q, et al. Formononetin protects against inflammation associated with cerebral ischemia-reperfusion injury in rats by targeting the JAK2/STAT3 signaling pathway. Biomed Pharmacother. 2022;149:112836. [46] ZHANG Y, WANG S, DAI X, et al. Simiao San alleviates hyperuricemia and kidney inflammation by inhibiting NLRP3 inflammasome and JAK2/STAT3 signaling in hyperuricemia mice. J Ethnopharmacol. 2023;312:116530. |
| [1] | Yu Cenqi, Liu Yang, Yu Jiangfeng, Kang Kang, Deng Yaoge, Xia Xiaowei, Zhang Yijian, Zhu Xuesong. Biomimetic Black Phosphorus Nanosystem Regulates Synovial Macrophage Polarization for Osteoarthritis Treatment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(在线): 1-13. |
| [2] | Liu Yang, Liu Donghui , Xu Lei, Zhan Xu, Sun Haobo, Kang Kai. Role and trend of stimuli-responsive injectable hydrogels in precise myocardial infarction therapy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2072-2080. |
| [3] | Song Puzhen, Ma Hebin, Chen Hongguang, Zhang Yadong. Effect of bone marrow mesenchymal stem cell-derived exosomes combined with transforming growth factor beta 1 on macrophages [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1616-1623. |
| [4] | Chen Yulin, He Yingying, Hu Kai, Chen Zhifan, Nie Sha Meng Yanhui, Li Runzhen, Zhang Xiaoduo , Li Yuxi, Tang Yaoping. Effect and mechanism of exosome-like vesicles derived from Trichosanthes kirilowii Maxim. in preventing and treating atherosclerosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1768-1781. |
| [5] | Peng Zhiwei, Chen Lei, Tong Lei. Luteolin promotes wound healing in diabetic mice: roles and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1398-1406. |
| [6] | You Huijuan, Wu Shuzhen, Rong Rong, Chen Liyuan, Zhao Yuqing, Wang Qinglu, Ou Xiaowei, Yang Fengying. Macrophage autophagy in lung diseases: two-sided effects [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1516-1526. |
| [7] | Cao Wenqi, Feng Xiuzhi, Zhao Yi, Wang Zhimin, Chen Yiran, Yang Xiao, Ren Yanling. Effect of macrophage polarization on osteogenesis-angiogenesis coupling in type 2 diabetic osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 917-925. |
| [8] | Luan Chuankai, Zhu Lei. Role and mechanism of exercise-regulated miRNAs in cardiac remodeling after acute myocardial infarction [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 9024-9031. |
| [9] | Wang Yanfei, Jin Lianhai, Li Qingya, Fu Yuanfei, Tan Huangsheng, Deng Pengwei, Gao Kun. Synovial fluid exosome-mediated crosstalk between synoviocytes and chondrocytes in development and progression of knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 9032-9040. |
| [10] | Zhao Canbin, Zeng Ping, Shi Weiqi, Liu Jinfu, Ding Qiang, Guo Liang, Wang Weiwei, Tao Hongcheng, Guo Yafeng, Qin Ying. Mechanism by which luteolin regulates macrophage polarization in the treatment of knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 8868-8877. |
| [11] | Yu Cenqi, Liu Yang, Yu Jianfeng, Kang Kang, Deng Yaoge, Xia Xiaowei, Zhang Yijian, Zhu Xuesong. Biomimetic black phosphorus nanosystem regulates synovial macrophage polarization for osteoarthritis treatment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8378-8390. |
| [12] | Zhang Xiaoxu, Tian Zhenli, Xie Tingting. Roles of pregnane X receptor in sodium arsenite-induced oxidative stress and inflammatory injury in human normal hepatocytes [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6259-6266. |
| [13] | Jiang Chao, Che Yanjun. Biological mechanisms and future research trends of cartilaginous endplate degeneration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 5915-5924. |
| [14] | Jiang Qiang, Ding Yu, Ding Zhili, Han Jiaheng. Mechanisms by which mitochondria-endoplasmic reticulum interaction stress mediates activation of inflammatory vesicles in nerve roots of lumbar intervertebral disc herniation rabbits modulated by acupotomy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6110-6121. |
| [15] | Xie Peisen, Guan Zhenpeng, Wei Xianjie, Zhang Keshi, Kang Qingyuan, Xiao Wentao, Guo Xiaoshuai. Cerium dioxide nanoparticles regulate expression of inflammatory factors in M1 macrophages and affect fibroblast co-culture system [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(2): 375-383. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||