Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (18): 2906-2913.doi: 10.12307/2022.702
Previous Articles Next Articles
Visualization analysis of bone defect treatment based on knowledge map
Dong Jiale1, Wei Yuanhao1, Zhang Hongwu2
- 1The First School of Clinical Medicine, 2School of Basic Medical Sciences, Guangdong Provincial Key Laboratory of Construction and Detection in Tissue Engineering, Southern Medical University, Guangzhou 510515, Guangdong Province, China
-
Received:2021-09-23Accepted:2021-10-30Online:2022-06-28Published:2022-01-30 -
Contact:Zhang Hongwu, Associate professor, Master’s supervisor, School of Basic Medical Sciences, Guangdong Provincial Key Laboratory of Construction and Detection in Tissue Engineering, Southern Medical University, Guangzhou 510515, Guangdong Province, China -
About author:Dong Jiale, Successive bachelor-master-doctor program candidate, The First School of Clinical Medicine, Southern Medical University, Guangzhou 510515, Guangdong Province, China -
Supported by:Guangdong Provincial Science and Technology Plan Project, No. 2020A0505100050 (to ZHW); Natural Science Foundation of Guangdong, No. 2020A1515010102 (to ZHW)
CLC Number:
Cite this article
Dong Jiale, Wei Yuanhao, Zhang Hongwu. Visualization analysis of bone defect treatment based on knowledge map[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(18): 2906-2913.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
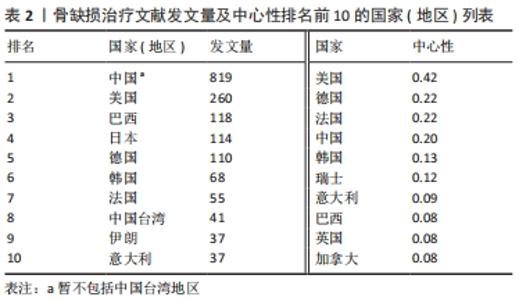
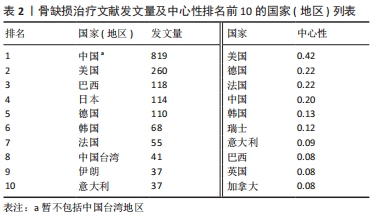
对发文量和中心性进行分析可知:中国(暂不包括中国台湾地区)是图谱中最大的节点,以 819篇的发文量位居首位且为第2位美国的3.15倍,远高于其他各国;美国、巴西、日本在该研究领域发文量居后,为研究领域的主要研究力量,见表2。美国、德国、法国中心性最强,合作最为紧密且集体优势明显,凭借高质量文献在领域内起到关键枢纽的作用。中国起步早、发文多,是骨缺损治疗中的重要研究力量,目前已在组织工程、材料科学、细胞生物学等领域发表了一系列骨缺损治疗的国际性高水平文章,尤其是在骨形态发生蛋白2/地塞米松药物缓释支架、锶代硅酸钙生物活性陶瓷、外泌体/磷酸三钙复合支架等方面提出了独到的见解,取得了一系列富有创新性的成果[19-21],但文章中心性方面仍亟待提高,需力争在增加发文数的同时提高研究质量。"

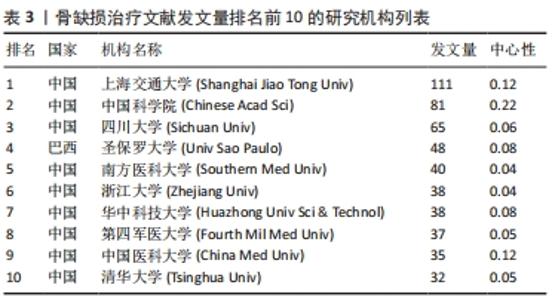
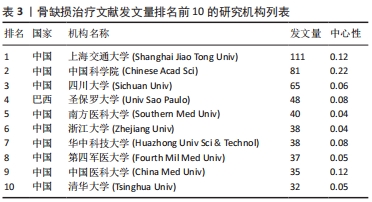
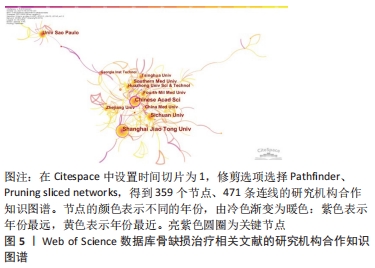
2.4 骨缺损治疗相关文献发文机构分析 可视化图谱可直观展示研究领域内研究力量的分布及各机构间的合作情况,有利于学科发展和成果共享。使用CiteSpace V软件对纳入文献的发文机构情况进行分析(图5):N=359,E=471。发文量排名前5的研究机构分别为上海交通大学(111篇)、中国科学院(81篇)、四川大学(65篇)、圣保罗大学(Univ Sao Paulo,48篇)、南方医科大学(40篇)。中心性排名前5的研究机构分别为中国科学院(0.22)、上海交通大学(0.12)、中国医科大学(0.12)、美国乔治亚理工学院(0.12)、北京大学(0.1)。可见骨缺损治疗文献的发文机构主要为各大医科院校及其附属医院,此外如中国科学院等机构于生物材料学领域也有贡献。"
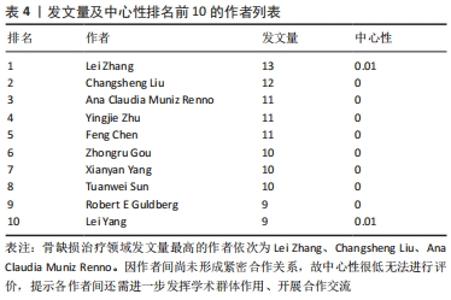
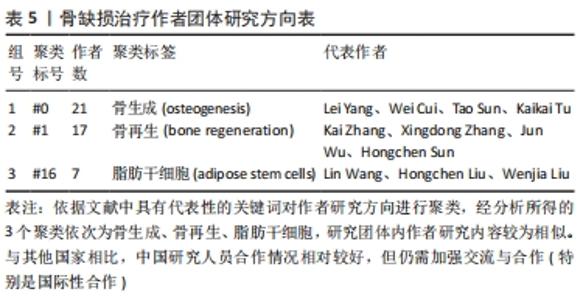
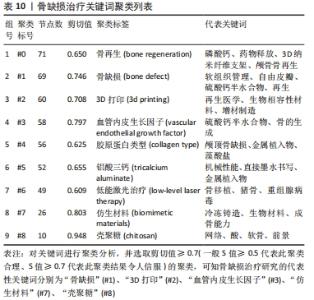
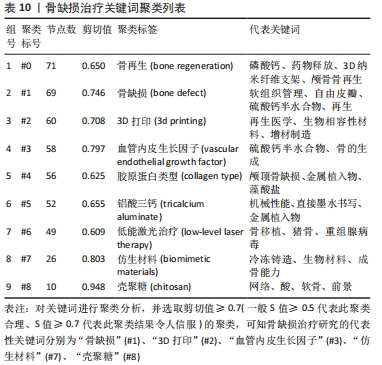
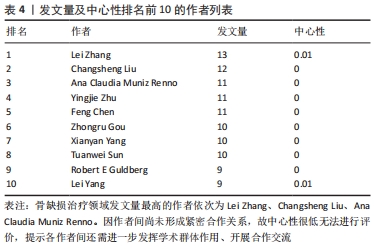
聚类分析利用文献中具有代表性的关键词对作者群体进行划分,从而展示相应小群体的研究方向,提供该学科领域的未来发展趋势。聚类分析发现,作者群体形成了大小不同的3个研究团体,团体内作者间研究内容较为相似,其研究内容可与组内其他成员互为补充,见表4。最大的研究团体由Lei Yang、Wei Cui、Tao Sun、Kaikai Tu等组成,其研究方向主要为探究不同激素和生物材料对小鼠模型骨生成的影响[22-24];第二大研究团体由Kai Zhang、Xingdong Zhang、Jun Wu、Hongchen Sun等组成,其研究方向主要为探究仿生材料在骨再生中的作用[25-27];Lin Wang、Hongchen Liu、Wenjia Liu则主要研究脂肪源性干细胞(ADSCs)骨钙素表达对骨缺损修复的影响[28-29],见表5。"
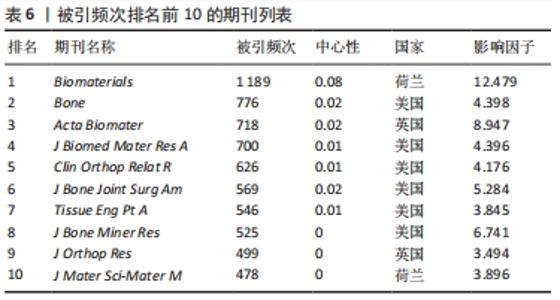
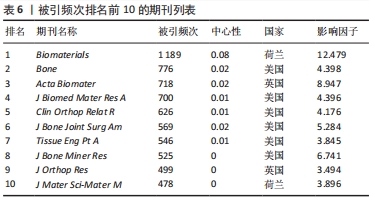
2.6 骨缺损治疗相关文献期刊共被引情况分析 使用CiteSpace V软件对纳入文献的期刊引用情况进行分析(表6),其中被引频次最高的期刊依次为《Biomaterials》(1 189次)、《Bone》(776次)、《Acta Biomater》(718次)、《J Biomed Mater Res A》(700次)、《Clin Orthop Relat R》(626次);中心性最高的期刊依次为《Biomaterials》(0.08)、《Acs Nano》(0.05)、《Brit J Oral Max Surg》(0.05)、《Cancer Res》(0.05)。被引期刊主要集中在骨、生物材料、组织学等,其中《Biomaterials》作为骨缺损治疗领域被引频次和中心性最高的期刊,影响因子较高,在生物材料领域期刊中具有权威地位。"

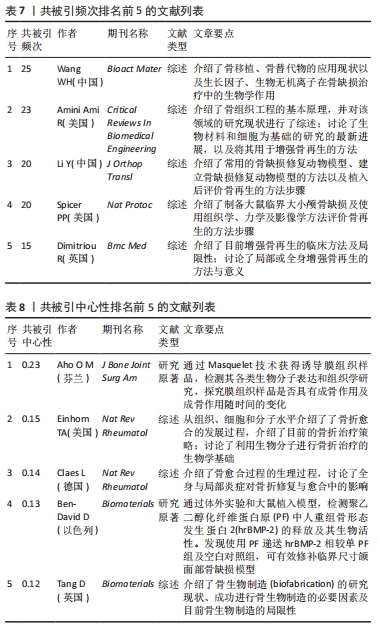
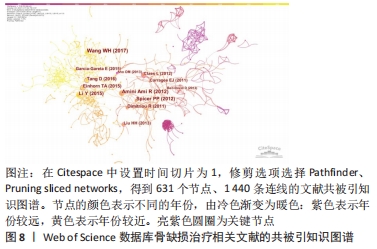
2.7 骨缺损治疗相关文献共被引情况分析 高共被引频次的文献是该领域优秀研究成果的结晶,是该领域的基石,在该领域研究中可为后续的基础及临床研究提供指导。使用CiteSpace V软件对骨缺损治疗领域的文献被引情况进行分析(图8),可归纳出共被引频次及中心性最高的前5名文献(表7、8),对其进行精读可加深对骨缺损治疗领域的了解,指导科研工作者了解国际水平的理论知识与研究方法。由结果可知,近10年来骨缺损领域共被引频次较高的文章对现有的骨移植、骨替代物、骨缺损模型构建及评价、骨再生等方面进行了介绍[30-34]。共被引中心性较高的文章对骨折愈合、骨生物制造进行了介绍[35-37],并探究了两型新型生物材料的成骨及缺损修复效果[38-39]。"

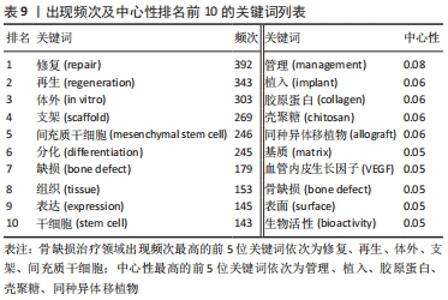
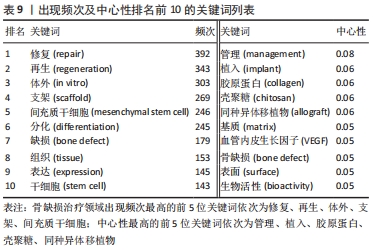
2.8 骨缺损治疗相关文献关键词分析 可视化图谱可为研究者提示该领域内的研究内容。关键词频次的大小可以反映领域内的热门课题,不同时期出现的关键词情况可体现研究领域的发展厘革情况。使用CiteSpace V软件对骨缺损治疗领域的文献关键词进行分析(图9):N=454,E=1 907。频次排名前5的关键词为“修复”“再生”“体外”“支架”“间充质干细胞”;中心性排名前5的关键词为“管理”“植入”“胶原蛋白”“壳聚糖”“同种异体移植物”(表9)。除去与检索策略有关的关键词,骨缺损治疗的研究内容主要有“再生”“体外”“支架”“间充质干细胞”“分化”等,并借助“管理”“植入”“胶原蛋白”等中心词形成一个有机整体。"

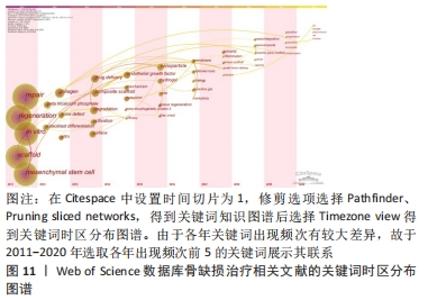
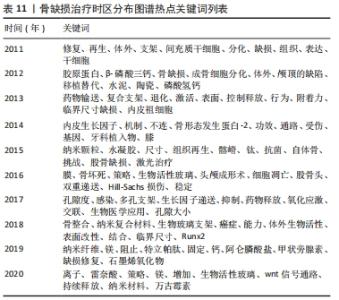
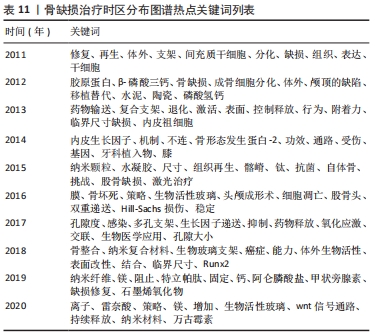
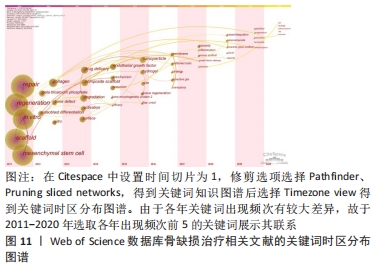
Citespace的时区图谱功能可将关键词知识图谱中涉及的关键词,按发表年份进行可视化展示。其可清晰显示出各时间段本研究领域的发展历程,形象生动地展示研究重点的演进方向,可根据演进趋势对该课题后续研究目标开展评估(图11)。结合出现时间对时间图谱内的热点关键词进行分析(表11),骨缺损治疗领域在2011-2020年前期的研究策略主要是骨缺损的诱导分化、促进再生;从2015年开始,纳米颗粒、水凝胶等材料借助3D打印技术逐渐广泛应用于骨缺损治疗中;近年来,研究者期望在信号通路水平探究骨缺损治疗机制,并期望使用新型复合材料支架联合药物/生长因子控释技术达到更好的治疗效果。此外,对于甲状旁腺素与骨质疏松症的研究也一直是本时间段内的研究热点。"
| [1] SCHLUMBERGER M, MAYR R, KOIDL C, et al. Treatment of tibial nonunion with bone defect using a heterotopic ossification as autologous bone graft: literature overview and case report. Eur J Orthop Surg Traumatol. 2018;28(4):741-746. [2] ZHANG R, LIU J, YU S, et al. Osteoprotegerin (OPG) Promotes Recruitment of Endothelial Progenitor Cells (EPCs) via CXCR4 Signaling Pathway to Improve Bone Defect Repair. Med Sci Monit. 2019;25:5572-5579. [3] HUANG Z, XU J, CHEN J, et al. Photoacoustic stimulation promotes the osteogenic differentiation of bone mesenchymal stem cells to enhance the repair of bone defect. Sci Rep. 2017;7(1):15842. [4] BAUER TW, MUSCHLER GF. Bone graft materials. An overview of the basic science. Clin Orthop Relat Res. 2000;38(371):10-27. [5] BHUMIRATANA S, BERNHARD JC, ALFI DM, et al. Tissue-engineered autologous grafts for facial bone reconstruction. Sci Transl Med. 2016; 8(343):343r-383r. [6] ZWINGENBERGER S, NICH C, VALLADARES RD, et al. Recommendations and considerations for the use of biologics in orthopedic surgery. BioDrugs. 2012;26(4):245-256. [7] 冯晓东,程芳芳,郭军,等.个体化组织工程骨与同种异体骨修复人长骨缺损的临床疗效和安全性对比[J].局解手术学杂志,2020, 29(11):883-889. [8] ZHANG L, YANG G, JOHNSON BN, et al. Three-dimensional (3D) printed scaffold and material selection for bone repair. Acta Biomater. 2019;84:16-33. [9] 徐心怡,于琦,王琪,等.近30年我国健康教育政策的计量分析[J].现代预防医学,2020,47(18):3343-3347. [10] 邵一鸣,周志伟,胡振宇,等.基于文献计量学的垂直农场研究现状分析[J].华中农业大学学报,2020,39(06):144-154. [11] 黄娜,刘家玥,黄英杰,等. Web of Science数据库近5年股骨头坏死文献计量学及可视化分析[J].中国组织工程研究,2021,25(17): 2711-2718. [12] 温帅波,韩杰,吴钰坤.来自Web of Science数据库软骨修复15年相关文献的计量学及可视化分析[J]. 中国组织工程研究,2021, 25(17):2657-2663. [13] 刘静,郑丽维,杨晨晨,等.高血压与肠道菌群相关性研究文献计量学分析[J].中国全科医学,2018,21(24):2922-2926. [14] 段明香,徐聪,莫明露,等.基于知识图谱的2型糖尿病发病机制研究的可视化分析[J].重庆医科大学学报,2018,43(06):756-762. [15] 许革新,张云霞,张海英.干细胞治疗阿尔茨海默病的研究现状及发展趋势[J].中国组织工程研究,2019,23(33):5378-5384. [16] 张旭东,董四平,杨威,等.基于CiteSpace的我国临床营养研究可视化分析[J].中国医疗管理科学,2021,11(2):91-96. [17] 陈悦,陈超美,刘则渊,等. CiteSpace知识图谱的方法论功能[J].科学学研究,2015,33(2):242-253. [18] 李杰,陈超美. citespace科技文本挖掘及可视化[M].北京:首都经济贸易大学出版社,2016. [19] LI L, ZHOU G, WANG Y, et al. Controlled dual delivery of BMP-2 and dexamethasone by nanoparticle-embedded electrospun nanofibers for the efficient repair of critical-sized rat calvarial defect. Biomaterials. 2015;37:218-229. [20] LIN K, XIA L, LI H, et al. Enhanced osteoporotic bone regeneration by strontium-substituted calcium silicate bioactive ceramics. Biomaterials. 2013;34(38):10028-10042. [21] ZHANG J, LIU X, LI H, et al. Exosomes/tricalcium phosphate combination scaffolds can enhance bone regeneration by activating the PI3K/Akt signaling pathway. Stem Cell Res Ther. 2016;7(1):136. [22] TAO ZS, ZHOU W S, TU KK, et al. Effect exerted by Teriparatide upon Repair Function of beta-tricalcium phosphate to ovariectomised rat’s femoral metaphysis defect caused by osteoporosis. Injury. 2015; 46(11):2134-2141. [23] CUI W, LIU Q, YANG L, et al. Sustained Delivery of BMP-2-Related Peptide from the True Bone Ceramics/Hollow Mesoporous Silica Nanoparticles Scaffold for Bone Tissue Regeneration. ACS Biomater Sci Eng. 2018;4(1):211-221. [24] TAO ZS, QIANG Z, TU KK, et al. Treatment study of distal femur for parathyroid hormone (1-34) and beta-tricalcium phosphate on bone formation in critical size defects in rats. J Biomater Appl. 2015;30(4): 484-491. [25] YUAN B, WANG L, ZHAO R, et al. A biomimetically hierarchical polyetherketoneketone scaffold for osteoporotic bone repair. Sci Adv. 2020;6(50):eabc4704. [26] ZHANG K, ZHOU Y, XIAO C, et al. Application of hydroxyapatite nanoparticles in tumor-associated bone segmental defect. Sci Adv. 2019;5(8):x6946. [27] FU X, ZHOU X, LIU P, et al. The optimized preparation of HA/L-TiO2/D-TiO2 composite coating on porous titanium and its effect on the behavior osteoblasts. Regen Biomater. 2020;7(5):505-514. [28] WANG L, XU X, HUO N, et al. A combination of insulin and ubiquitin A20 promotes osteocalcin expression in adipose-derived stem cells. Biochem Cell Biol. 2013;91(6):513-518. [29] WANG L, WANG H, XU X, et al. Insulin facilitates osteoblast differentiation. Cell Biol Int. 2013;37(11):1157-1161. [30] WANG W, YEUNG K. Bone grafts and biomaterials substitutes for bone defect repair: A review. Bioact Mater. 2017;2(4):224-247. [31] AMINI AR, LAURENCIN CT, NUKAVARAPU SP. Bone tissue engineering: recent advances and challenges. Crit Rev Biomed Eng. 2012;40(5):363-408. [32] LI Y, CHEN SK, LI L, et al. Bone defect animal models for testing efficacy of bone substitute biomaterials. J Orthop Translat. 2015;3(3):95-104. [33] SPICER PP, KRETLOW JD, YOUNG S, et al. Evaluation of bone regeneration using the rat critical size calvarial defect. Nat Protoc. 2012;7(10):1918-1929. [34] DIMITRIOU R, JONES E, MCGONAGLE D, et al. Bone regeneration: current concepts and future directions. BMC Med. 2011;9(1):66. [35] EINHORN TA, GERSTENFELD LC. Fracture healing: mechanisms and interventions. Nat Rev Rheumatol. 2015;11(1):45-54. [36] CLAES L, RECKNAGEL S, IGNATIUS A. Fracture healing under healthy and inflammatory conditions. Nat Rev Rheumatol. 2012;8(3):133-143. [37] TANG D, TARE RS, YANG LY, et al. Biofabrication of bone tissue: approaches, challenges and translation for bone regeneration. Biomaterials. 2016;83:363-382. [38] AHO O M, LEHENKARI P, RISTINIEMI J, et al. The mechanism of action of induced membranes in bone repair. J Bone Joint Surg Am. 2013;95(7): 597-604. [39] BEN-DAVID D, SROUJI S, SHAPIRA-SCHWEITZER K, et al. Low dose BMP-2 treatment for bone repair using a PEGylated fibrinogen hydrogel matrix. Biomaterials. 2013;34(12):2902-2910. [40] 叶鹰.文献计量法和内容分析法的理论基础及软件工具比较[J]. 评价与管理,2005,3(3):24-26. [41] TOMAS CA, PETER I. Informetric analyses on the world wide web: methodological approaches to ‘webometrics’. J Documentation. 1997; 53(4):404-426. |
| [1] | Xue Yadong, Zhou Xinshe, Pei Lijia, Meng Fanyu, Li Jian, Wang Jinzi . Reconstruction of Paprosky III type acetabular defect by autogenous iliac bone block combined with titanium plate: providing a strong initial fixation for the prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1424-1428. |
| [2] | Li Zhiyi, He Pengcheng, Bian Tianyue, Xiao Yuxia, Gao Lu, Liu Huasheng. Bibliometric and visualized analysis of ferroptosis mechanism research [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1202-1209. |
| [3] | Zhao Jing, Liu Xiaobo, Zhang Yue, Zhang Jiaming, Zhong Dongling, Li Juan, Jin Rongjiang. Visualization analysis of neuromuscular electrical stimulation therapy based on CiteSpace: therapeutic effects, hot spots, and developmental trends [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1234-1241. |
| [4] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [5] | Tan Guozhong, Tu Xinran, Guo Liyang, Zhong Jialin, Zhang Yang, Jiang Qianzhou. Biosafety evaluation of three-dimensional printed gelatin/sodium alginate/58S bioactive glass scaffolds for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 521-527. |
| [6] | Liu Ming, Wang Kai. Theaflavin-3-gallate modified nano-hydroxyapatite/polycaprolactone composite porous scaffold in bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3480-3486. |
| [7] | Tang Zhenzhou, Gu Yong, Chen Liang. Preparation of modified dextran composite hydrogel with osteogenetic effect and in vitro experiment [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3521-3527. |
| [8] | Li Duchenhui, Tian Ai, Tang Zhenglong. Angiogenesis induced by bone bioscaffold materials [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3602-3608. |
| [9] | Tian Yuhang, Liu Yadong, Cui Yutao, Liu He, Li Shaorong, Wang Gan, Fan Yi, Peng Chuangang, Wu Dankai. Application of chitosan biomaterial scaffold in the treatment of infectious bone defects [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(21): 3415-3420. |
| [10] | Zhang Chunyu, Hu Baoyang, Feng Yao, Zhang Wanfen, Sun Leiye, Yang Yan. Performance optimization of a new type of calcium silicate (based) scaffold matched with bone tissue regeneration in the bone defect area [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(21): 3421-3428. |
| [11] | Wang Lu, Li Limei, Li Qing, Yang Hongcai, Lan Xiaoqian, Hu Yingrui. Application of polyurethane materials in the repair of osteoporotic bone defects [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(21): 3429-3434. |
| [12] | Wang Jinsi, Wang Shengfa, Wu Zhuguo, He Xiaoling, Wang Xinyu, Luo Xiaoyu, Zhao Yi, Zhang Jingying. Design and biological activity of beta-tricalcium phosphate biomimetic bone scaffold based on triply periodic minimal surfaces [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(21): 3291-3297. |
| [13] | Li Mingxin, Li Jun, Wang Wenchao, Song Ping, Lei Haoyuan, Gui Xingyu, Zhang Chengyun, Zhou Changchun, Liu Lei. Cell-carrying porous methacrylate anhydride gelatin three-dimensional scaffolds and their effects on cell behavior [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(16): 2532-2539. |
| [14] | Wei Zhangao, Xu Linghan, Wu Zichen, Tang Hao, Chen Jialong. Application of inorganic nonmetallic artificial bone materials in vivo [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(16): 2584-2589. |
| [15] | Peng Huizhen, Cai Mingxiang, Liu Xiangning. Angiogenesis regulation in bone repair: new ideas and new methods [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(15): 2400-2405. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||