Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (32): 8327-8334.doi: 10.12307/2026.460
Previous Articles Next Articles
Mechanical and fluid dynamic characteristics of S-type triply periodic minimal surface radial functionally graded bone scaffolds
Jia Xianghong, Xu Yan, Zhang Xujing
- School of Mechanical Engineering, Xinjiang University, Urumqi 830017, Xinjiang Uygur Autonomous Region, China
-
Accepted:2026-02-09Online:2026-11-18Published:2026-04-23 -
Contact:Xu Yan, Professor, Doctoral supervisor, School of Mechanical Engineering, Xinjiang University, Urumqi 830017, Xinjiang Uygur Autonomous Region, China -
About author:Jia Xianghong, MS candidate, School of Mechanical Engineering, Xinjiang University, Urumqi 830017, Xinjiang Uygur Autonomous Region, China -
Supported by:Natural Science Foundation Project of Xinjiang Uygur Autonomous Region, No. 2022D01C34 (to XY); National Natural Science Foundation of China, No. 52365053 (to XY)
CLC Number:
Cite this article
Jia Xianghong, Xu Yan, Zhang Xujing. Mechanical and fluid dynamic characteristics of S-type triply periodic minimal surface radial functionally graded bone scaffolds[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8327-8334.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

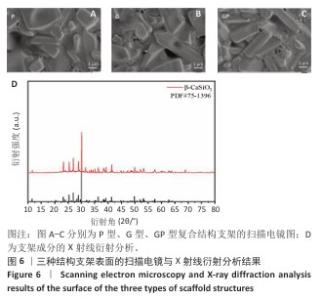
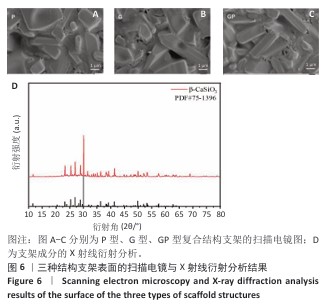
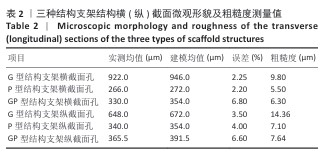
2.1 支架的表征结果 超景深图像(图5)清晰展示了三周期极小曲面结构的微观形貌,整体结构完整,无明显缺陷,数字光处理加工层纹由于液相烧结效应几乎不可见。由于三周期极小曲面结构是参数驱动建模的,因此选择了相对固定的特征结构进行测量,测量发现横截面的加工尺寸略小于设计尺寸,表明加工过程中存在一定误差(表2),误差主要来源于数字光处理加工过程中光的散射和脱脂烧结过程陶瓷件的收缩。粗糙度测量结果显示,由于加工过程的台阶效应,支架纵截面的平均粗糙度要大于横截面。 如图6所示,扫描电镜从微观角度显示了硅灰石陶瓷颗粒边界熔融,陶瓷颗粒之间熔融为一体,形成坚固的晶粒边界,硅灰石陶瓷颗粒生长,长径比增大,坚固的晶粒边界和增大的长径比可以协同提高材料的力学性能。X射线衍射图谱显示了烧结后的支架成分为β-硅酸钙,特征峰(例如2θ=29.8°,34.2°,49.3°)与标准卡片(PDF#75-1396)一致,未检测到杂相,表明烧结过程未引发相变。 "

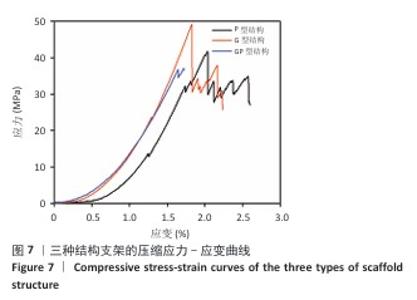
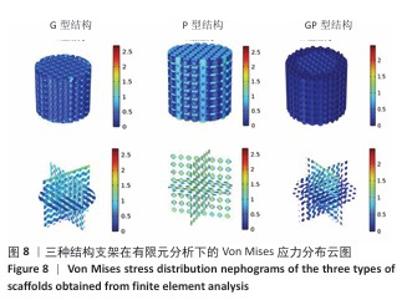
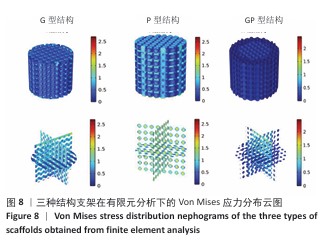
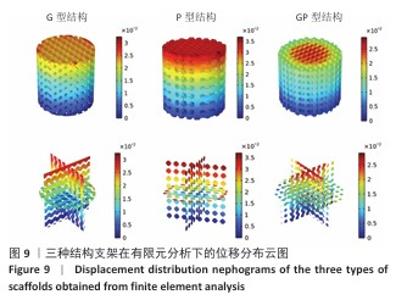
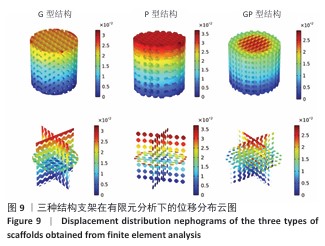
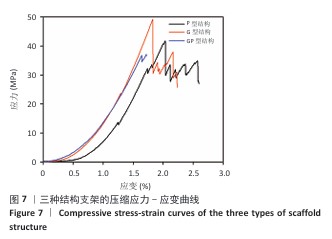
.2 支架有限元压缩模拟和机械压缩实验结果 静态压缩测试结果表明,3种三周期极小曲面结构支架表现出不同的力学特性(图7)。 G型结构支架的弹性模量为2.90 GPa,压缩强度较高,抗变形能力良好,适合骨组织支撑;P型结构支架的弹性模量为 3.39 GPa,刚度更高,适用于高负荷区域;GP型复合结构支架的弹性模量为3.38 GPa,兼具刚度和弹性恢复能力。 应用有限元分析进一步验证了支架的力学性能,3种结构支架的Von Mises应力分布云图,见图8。G型结构支架的应力分布均匀,最大Mises应力为2.73 MPa,适合高负荷分散;P型结构支架的最大Mises应力为2.68 MPa,致密外层增强了力学稳定性;GP型复合结构支架的最大Mises应力最低(2.47 MPa),显著低于单一结构支架,表明应力分布更均匀,可减少局部应力集中和材料损伤风险。 在100 N轴向载荷条件下,3种支架的位移分布云图,见图9。GP型复合结构支架(外侧P型、内侧G型)的最大位移量为2.75×10-2 mm,位移分布呈现明显的轴向梯度特征,底部区域位移最小(< 0.005 mm),顶部区域位移最大,等值线沿轴向均匀过渡,表明应力传递得到优化,无明显应力集中现象。相比之下,G型结构支架的最大位移量为3.22×10-2 mm,较GP型复合结构支架增大17%;P型结构支架的最大位移量为3.61×10-2 mm,较GP型复合结构支架增大31%。综合分析表明:GP型复合结构支架有效综合了单一结构支架的力学优势,最大位移量最小,该复合设计显著提升了支架的整体刚度和抗变形能力。 尽管GP型复合结构支架的弹性模量(3.38 GPa)与P型结构支架(3.39 GPa)相近,但GP型复合结构支架的最大位移量更小且应力分布更为均匀,表明GP型复合结构支架在保持高刚度的同时,其内部的梯度过渡设计有效优化了载荷传递路径,避免了应力集中,从而赋予了结构更高的效能,使该支架在承受相同载荷时变形更小,即“兼具刚度和弹性恢复能力”。 "
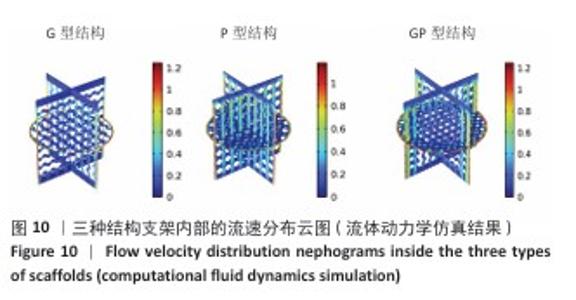
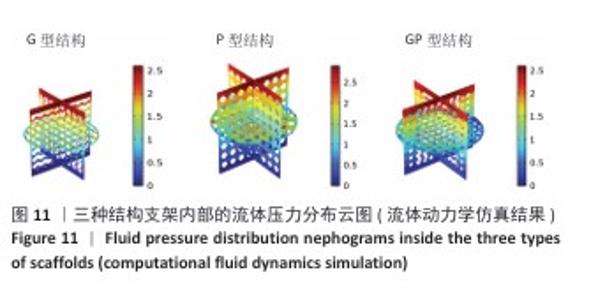
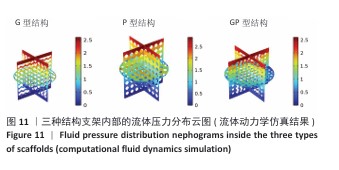
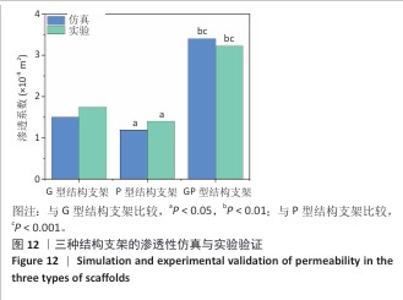
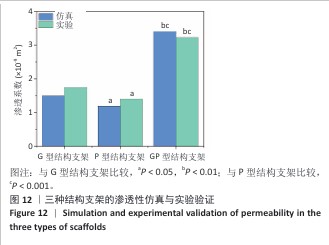
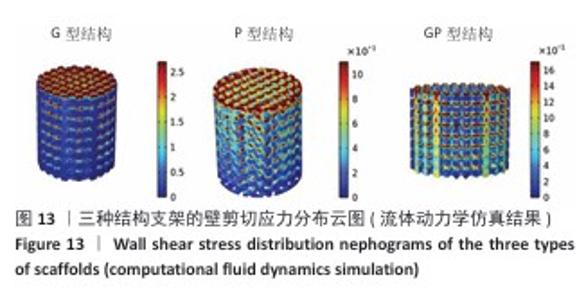
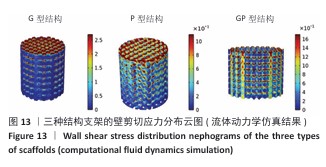
2.3 支架渗透性分析结果 流速分布(图10)显示,G型结构支架凭借三维贯通孔隙网络展现出均匀的流线分布,流道内未观测到明显涡流或滞流区;P型结构支架呈现明显的流速梯度,外缘致密区流速显著降低,而内层通道流速略有提升;GP型复合结构支架的综合表现最优,流线分布结果显示流体在支架内部保持了相对平稳的流动,特别是G型区域流体流速较为均匀,而P型外层流速略有减缓。 压力分布(图11)表明,G型结构支架内部压力场最为均衡,最大压力梯度出现在边缘区域;P型结构支架外缘产生显著压力积聚,形成明显的压力边界层;GP型复合结构支架的压力分布相对最为理想。GP型复合结构支架通过梯度设计保持了较为均匀的压力分布,压力过渡区扩展避免了局部高压区域的形成,形成平缓的液压梯度。 渗透性测试结果(图12)显示,3组结构支架渗透率比较差异有显著性意义(P < 0.01)。G型结构支架的渗透率为1.5×10-9 m2, P型结构支架的渗透率最低(1.2×10-9 m2),而GP型复合结构支架表现出显著的协同增强效应,渗透率最高(3.4×10-9 m2)。GP型复合结构支架的渗透率显著高于G型、P型结构支架(P < 0.01,P < 0.001),G型结构支架的渗透率高于P型结构支架(P < 0.05)。所有支架的渗透率均处于松质骨渗透率的典型参考范围(0.5× 10-9-5.0×10-9 m2)内,表明具备良好的生物传输潜力。 2.4 支架壁剪切应力分布 通过计算流体力学模拟分析了3种三周期极小曲面结构(G型、P型及GP型复合结构)硅灰石支架的壁剪切应力分布特性(图13),结果显示:G型结构支架因高孔隙率(> 50%)和互通孔道设计流体流动平稳,壁剪切应力平均值为0.86 Pa、最大值为1.13 Pa;P型结构支架因本身横纵相交的通道设计,即使和G型结构支架拥有同样的孔隙率,壁剪切应力平均值为1.40 Pa、最大值为2.65 Pa,分别为G型结构支架的1.63和2.35倍,虽然有相同的孔隙率,相比于P型结构支架,G型结构支架的壁剪切应力分布均匀,均处于有效生物力学刺激区间;GP型复合结构综合G型和P型几何特征,虽然外层P型结构致密设计导致流体路径受限,壁剪切应力最大值反而降低(相比P型结构支架),为1.68 Pa,壁剪切应力平均值为1.01 Pa,表明GP型复合结构支架的壁剪切应力分布更 均衡。 "
| [1] CLARKE B. Normal bone anatomy and physiology. Clin J Am Soc Nephrol. 2008;3: S131-S139. [2] 刘凯,艾合买提江·玉素甫.可生物降解金属支架修复长骨严重骨缺损[J].临床骨科杂志,2024,27(1):142-146. [3] LIU K, LIU Y, CAI F, et al. Efficacy comparison of trifocal bone transport using unilateral external fixator for femoral and tibial bone defects caused by infection. BMC Surg. 2022;22(1):141. [4] 朱力鸣,焦昆,刘伟,等.新型同种异体骨修复骨缺损动物实验研究[J].实验动物科学,2023,40(6):73-77. [5] 滕沙格,陈哲,范佳俊,等.同种异体骨植骨临床应用与研究进展[J].浙江中西医结合杂志,2020,30(5):433-437. [6] ŠROMOVÁ V, SOBOLA D, KASPAR P. A brief review of bone cell function and importance. Cells. 2023;12(21):2576. [7] 刘畅,周竞宇.基于CT图像的骨骼缺损修复方法研究[J].电脑知识与技术, 2019,15(31):231-233. [8] 王晓康.骨组织工程仿生多孔支架设计及性能分析[D].秦皇岛:燕山大学, 2023. [9] ZHEN C, SHI Y, WANG W, et al. Advancements in gradient bone scaffolds: enhancing bone regeneration in the treatment of various bone disorders. Biofabrication. 2024;16(3):032004. [10] ARMBRECHT G, NGUYEN MINH H, MASSMANN J, et al. Pore Size Distribution and Frequency Dependent Attenuation in Human Cortical Tibia Bone Discriminate Fragility Fractures in Postmenopausal Women With Low Bone Mineral Density. J Bone Miner Res. 2021;5(11):e10536. [11] KUMARI S, KATIYAR S, DARSHNA, et al. Design strategies for composite matrix and multifunctional polymeric scaffolds with enhanced bioactivity for bone tissue engineering. Front Chem. 2022;10:1051678. [12] CHUNG S, KING MW. Design concepts and strategies for tissue engineering scaffolds. Biotechnol Appl Biochem. 2011;58(6):423-438. [13] MIRI Z, HAUGEN HJ, LOCA D, et al. Review on the strategies to improve the mechanical strength of highly porous bone bioceramic scaffolds. J Eur Ceram Soc. 2024;44(1):23-42. [14] BIAN T, ZHAO K, MENG Q, et al. Preparation and properties of calcium phosphate cement/small intestinal submucosa composite scaffold mimicking bone components and Haversian microstructure. Mater Lett. 2018;212:73-77. [15] ZHANG M, LIN R, WANG X, et al. 3D printing of Haversian bone–mimicking scaffolds for multicellular delivery in bone regeneration. Sci Adv. 2020;6(12): eaaz6725. [16] MAYYA A, BANERJEE A, RAJESH R. Haversian microstructure in bovine femoral cortices: An adaptation for improved compressive strength. Mater Sci Eng C Mater Biol Appl. 2016;59:454-463. [17] CHATZIGEORGIOU C, PIOTROWSKI B, MERAGHNI F, et al. Multiscale mechanical analysis for biomimetic implant design based on Triply Periodic Minimal Surfaces (TPMS) lattices: Application to partial replacement of femoral bone. Results Eng. 2025;25:103984. [18] WANG N, MEENASHISUNDARAM GK, KANDILYA D, et al. A biomechanical evaluation on Cubic, Octet, and TPMS gyroid Ti6Al4V lattice structures fabricated by selective laser melting and the effects of their debris on human osteoblast-like cells. Biomater Adv. 2022;137:212829. [19] MIAODA S, YIFAN L, FENGLING L, et al. Bioceramic scaffolds with triply periodic minimal surface architectures guide early-stage bone regeneration. Bioact Mater. 2023;25:374-386. [20] LV J, JIN W, LIU W, et al. Selective laser melting fabrication of porous Ti6Al4V scaffolds with triply periodic minimal surface architectures: structural features, cytocompatibility, and osteogenesis. Front Bioeng Biotechnol. 2022;10:899531. [21] CASTRO APG, RUBEN RB, GONÇALVES SB, et al. Numerical and experimental evaluation of TPMS Gyroid scaffolds for bone tissue engineering. Comput Methods Biomech Biomed Engin. 2019;22(6):567-573. [22] DU X, RONAYNE S, LEE SS, et al. 3D-printed PEEK/silicon nitride scaffolds with a triply periodic minimal surface structure for spinal fusion implants. ACS Applied Bio Mater. 2023;6(8):3319-3329. [23] SUN QD, SUN JS, GUO K, et al. Compressive mechanical properties and energy absorption characteristics of SLM fabricated Ti6Al4V triply periodic minimal surface cellular structures. Mech Mater. 2022;166:104241. [24] ATAOLLAH S N, MARYAM T, ARSALAN M, et al. Mechanical Characterisation and Numerical Modelling of TPMS-Based Gyroid and Diamond Ti6Al4V Scaffolds for Bone Implants: An Integrated Approach for Translational Consideration. Bioengineering. 2022;9(10):504. [25] CAO Y, LAI S, WU W, et al. Design and mechanical evaluation of additively-manufactured graded TPMS lattices with biodegradable polymer composites. J Mater Res Technol. 2023;23:2868-2880. [26] ZENG C, WANG W. Modeling method for variable and isotropic permeability design of porous material based on TPMS lattices. Tribol Int. 2022;176:107913. [27] PIRES THV, DUNLOP JWC, CASTRO APG, et al. Wall shear stress analysis and optimization in tissue engineering TPMS scaffolds. Materials. 2022;15(20):7375. [28] ZHOU J, GUI Y, XU Q, et al. Investigation of permeability and biocompatibility of TPMS structures printed by laser powder bed fusion using Ti64-5Cu alloy for orthopedic implants. Mater Lett. 2024;355:135552. [29] 孙亚迪,马剑雄,王岩,等.三周期极小曲面骨支架微观结构对支架性能的影响研究进展[J].中国修复重建外科杂志,2023,37(10):1314-1318. [30] KARAMAN D, ASL HG. The effects of sheet and network solid structures of similar TPMS scaffold architectures on permeability, wall shear stress, and velocity: A CFD analysis. Med Eng Phys. 2023;118:104024. [31] STRÖMBERG N. Optimal grading of TPMS-based lattice structures with transversely isotropic elastic bulk properties. Eng Optim. 2021;53(11):1871-1883. [32] YANNI Z, NA S, MENGRAN Z, et al. The contribution of pore size and porosity of 3D printed porous titanium scaffolds to osteogenesis. Mater Sci Eng C Mater Biol Appl. 2022;133:112651. [33] CHEN Z, YAN X, YIN S, et al. Influence of the pore size and porosity of selective laser melted Ti6Al4V ELI porous scaffold on cell proliferation, osteogenesis and bone ingrowth. Mater Sci Eng C. 2020;106:110289. [34] NAUMAN EA, FONG K, KEAVENY TM. Dependence of intertrabecular permeability on flow direction and anatomic site. Ann Biomed Eng. 1999;27(4):517-524. [35] LI L, WANG P, LIANG H, et al. Design of a Haversian system-like gradient porous scaffold based on triply periodic minimal surfaces for promoting bone regeneration. J Adv Res. 2023;54:89-104. [36] GÜNTHER F, WAGNER M, PILZ S, et al. Design procedure for triply periodic minimal surface based biomimetic scaffolds. J Mech Behav Biomed Mater. 2022; 126:104871. |
| [1] | Hu Liqun, Xiao Dongqin, Ma Chenxi, Li Zhuohan, Yan Jiyuan, Li Zhong, He Kui, Duan Ke. Performance of calcium sulfate-magnesium oxide composites as anti-infective bone graft materials [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8309-8318. |
| [2] | Yang Yanjun, Zhu Lin, Gu Yongchun, Yan Zhanjun. Mechanism by which magnesium implant-activated integrin α10β1 promotes osteogenic differentiation of periosteal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8319-8326. |
| [3] | Yang Ping, Qi Xiaoyang, Lei Zhijie, Chen Yixin, Qiu Xusheng. Novel collagen membrane in repairing skull bone defects in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8364-8371. |
| [4] | Yu Cenqi, Liu Yang, Yu Jianfeng, Kang Kang, Deng Yaoge, Xia Xiaowei, Zhang Yijian, Zhu Xuesong. Biomimetic black phosphorus nanosystem regulates synovial macrophage polarization for osteoarthritis treatment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8378-8390. |
| [5] | Zhu Jisheng, Teng Jianxiang, Zou Zihao, Pan Jiazhao, Zhou Tianqi, Shu Xiaolin, He Cheng, Yuan Daizhu, Tian Xiaobin . Application of photocrosslinkable nanofiber scaffold loaded with decellularized cartilage matrix in cartilage tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8402-8412. |
| [6] | Li Xuanze, Fang Hanhong, Xu Zhe. Preparation and cytocompatibility of sodium alginate-hydroxyapatite-graphene oxide hydrogels [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8420-8426. |
| [7] | Liu Bangding, Tang Yongliang, Li Ni, Ren Bo. Quercetin-loaded hydrogel materials for treatment of infected bone defects [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8427-8435. |
| [8] | Zhang Zhanyue, Zhao Lijun, Zhang Chunyang, Zhang Zhongqi, Fu Kang, Zhang Zhihong. Application and development of polyetheretherketone material in skull defect repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8569-8576. |
| [9] | Zhao Zhanghong, Jin Dongsheng, Ruan Shiqiang, Huang Wenliang, Wan Yu, Tian Renyuan, Deng Jiang. In vitro osteogenic and anti-inflammatory properties of icariin sustained-release microsphere three-dimensional scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6710-6718. |
| [10] | Liu Weiwei, Gou Yuanbin, Cui Xiaoxue, Li Xin, Liu Dawei, Shi Mengrou, Chen Bin, Li Zhifeng. Comparison of different 3D-printed allogeneic bone and artificial polymer composite porous scaffold materials for repairing cranial bone defects in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6744-6751. |
| [11] | Wu Ningyuan, Wei Zhiyi, Feng Hao, Gao Ming. Frontiers and hot topics of nanobiomedicine in delaying the progression of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6907-6915. |
| [12] | Ye Gaoqi, Gong Yukang, Chen Dejin, Gao Wenshan. Selenium effect on human bone health and its application in bone materials [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6916-6922. |
| [13] | Liu Xuemiao, Zhang Yuchang, Zhang Weiguo, Tian Kang, Wang Xing. Three-dimensional bioprinting and tendon repair: application advances and future directions [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6923-6929. |
| [14] | Wang Liang, Zhang Xin, He Wei, Wang Jian. Clinical application and prospects of MXene-based materials for the repair of bone defects [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5282-5294. |
| [15] | Tang Hao, Zhong Qian, Wu Honghan, Wu Hengpeng, Wu Xingkai, Wa Qingde. 3D-printed biodegradable polyester-based scaffolds in bone regeneration therapy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5304-5311. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||