Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (14): 2209-2214.doi: 10.12307/2024.307
Previous Articles Next Articles
Aerobic exercise inhibits neuroinflammation and alleviates cognitive impairment in Alzheimer’s disease model mice
Deng Longfei1, Zhang Yeting2, Fu Yan1
- 1Southwest University for Nationalities, Chengdu 610225, Sichuan Province, China; 2Civil Aviation Flight University of China, Guanghan 618307, Sichuan Province, China
-
Received:2023-03-14Accepted:2023-04-22Online:2024-05-18Published:2023-07-28 -
Contact:Fu Yan, PhD, Professor, Southwest University for Nationalities, Chengdu 610225, Sichuan Province, China -
About author:Deng Longfei, Master, Lecturer, Southwest University for Nationalities, Chengdu 610225, Sichuan Province, China -
Supported by:Sichuan Provincial Science and Technology Department Project, No. 2020YFH0184 (to ZYT)
CLC Number:
Cite this article
Deng Longfei, Zhang Yeting, Fu Yan. Aerobic exercise inhibits neuroinflammation and alleviates cognitive impairment in Alzheimer’s disease model mice[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(14): 2209-2214.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
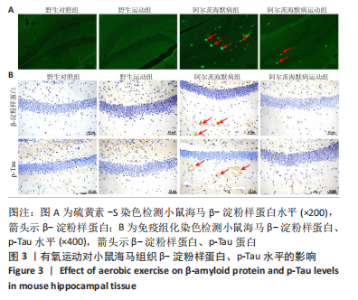
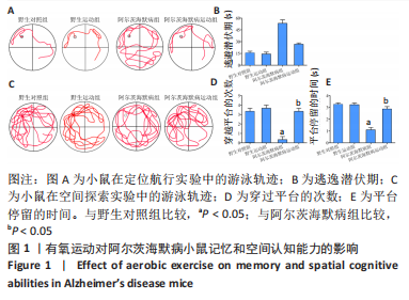
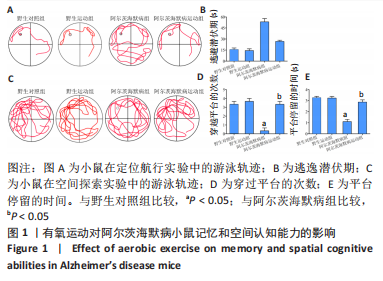
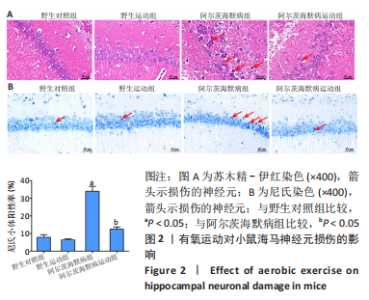
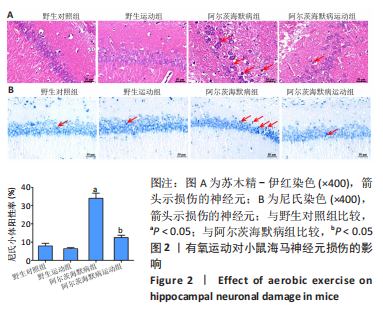
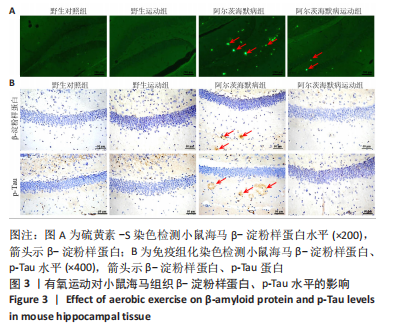
2.3.1 苏木精-伊红染色结果 野生对照组、野生运动组小鼠海马CA1区锥体细胞排列紧密、形态规则,细胞核着色较浅,树突稠密,神经元无明显变性及坏死。与野生对照组比较,阿尔茨海默病组海马锥体细胞排列紊乱,核固缩且染色加深,神经元发生变形,几乎未见完整神经元。与阿尔茨海默病组比较,阿尔茨海默病运动组神经元损伤明显减轻,锥体细胞排列较规则,细胞坏死数量明显减少。 2.3.2 尼氏染色结果 野生对照组、野生运动组海马组织神经元排列紧密、形态规则,无明显的神经元丢失。与野生对照组比较,阿尔茨海默病组神经元排列紊乱,染色质固缩,尼氏体阳性率明显减少(P < 0.05);与阿尔茨海默病组比较,阿尔茨海默病运动组神经元损伤明显缓解,排列较整齐,尼氏体阳性率明显增加(P < 0.05)。 2.4 有氧运动对海马组织β-淀粉样蛋白、p-Tau水平的影响 硫黄素-S染色和免疫组化结果显示,野生对照组、野生运动组小鼠海马CA1区β-淀粉样蛋白、p-Tau基本不表达;与野生对照组比较,阿尔茨海默病组存在明显的β-淀粉样蛋白沉积形成的斑块和p-Tau蛋白形成的神经原纤维缠结;与阿尔茨海默病组比较,阿尔茨海默病运动组β-淀粉样蛋白斑块和神经原纤维缠结明显减少。说明有氧运动能够降低海马组织β-淀粉样蛋白、p-Tau水平,见图3。"
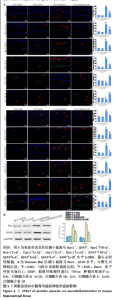
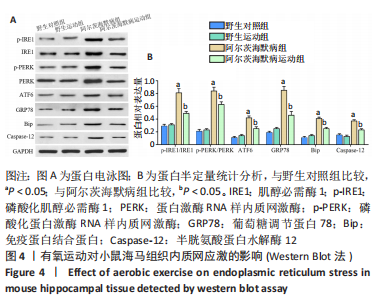
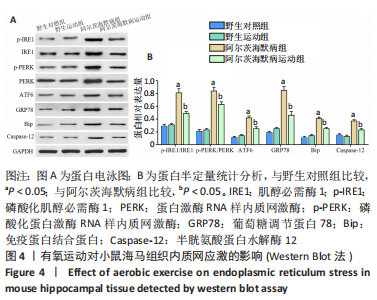
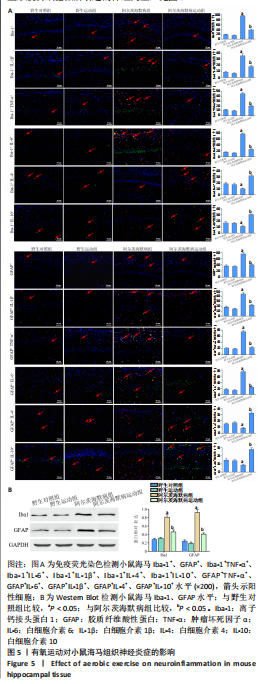
2.6 有氧运动对海马组织神经炎症的影响 免疫荧光染色和Western Blot结果显示,与野生对照组比较,阿尔茨海默病组小鼠海马CA1区Iba-1+、Iba-1+TNF-α+、Iba-1+IL-6+、Iba-1+IL-1β+、GFAP+、GFAP+TNF-α+、GFAP+IL-6+、GFAP+IL-1β+阳性细胞数、Iba-1、GFAP蛋白水平明显增加,Iba-1+IL-4+、Iba-1+IL-10+、GFAP+IL-4+、GFAP+IL-10+阳性细胞数明显减少(P < 0.05);与阿尔茨海默病组比较,阿尔茨海默病运动组Iba-1+、Iba-1+TNF-α+、Iba-1+IL-6+、Iba-1+IL-1β+、GFAP+、GFAP+TNF-α+、GFAP+IL-6+、GFAP+IL-1β+阳性细胞数、Iba-1、GFAP蛋白水平明显减少,Iba-1+IL-4+、Iba-1+IL-10+、GFAP+IL-4+、GFAP+IL-10+阳性细胞数明显增加(P < 0.05)。说明有氧运动抑制海马组织小胶质细胞/星形胶质细胞激活引起的神经炎症,见图5。"
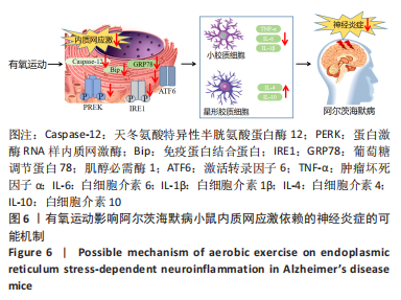
2.7 有氧运动影响阿尔茨海默病小鼠内质网应激依赖的神经炎症的可能机制 此次研究观察有氧运动对APP/PS1双转基因小鼠海马组织内质网应激、神经炎症和小鼠认知的影响,发现有氧运动能减轻海马神经元损伤;减少β-淀粉样蛋白沉积形成的斑块和p-Tau蛋白形成的神经原纤维缠结;通过降低未折叠蛋白反应的3条途径(IRE1、PERK、ATF6)及内质网应激激活的相关蛋白GRP78、Bip、Caspase-12表达抑制内质网应激;通过抑制小胶质细胞(Iba-1)和星形胶质细胞(GFAP)的激活及促炎因子TNF-α、IL-6、IL-1β水平,升高抗炎因子IL-4、IL-10水平减轻内质网应激依赖的神经炎症,见图6。"

| [1] OSSENKOPPELE R, VAN DER KANT R, HANSSON O. Tau biomarkers in Alzheimer’s disease: towards implementation in clinical practice and trials. Lancet Neurol. 2022;21(8):726-734. [2] VILLAIN N, PLANCHE V, LEVY R. High-clearance anti-amyloid immunotherapies in Alzheimer’s disease. Part 1: Meta-analysis and review of efficacy and safety data, and medico-economical aspects. Rev Neurol (Paris). 2022;178(10):1011-1030. [3] PÉREZ MJ, IVANYUK D, PANAGIOTAKOPOULOU V, et al. Loss of function of the mitochondrial peptidase PITRM1 induces proteotoxic stress and Alzheimer’s disease-like pathology in human cerebral organoids. Mol Psychiatry. 2021;26(10):5733-5750. [4] WEBERS A, HENEKA MT, GLEESON PA. The role of innate immune responses and neuroinflammation in amyloid accumulation and progression of Alzheimer’s disease. Immunol Cell Biol. 2020;98(1):28-41. [5] 任娜, 苏中昊, 凌子成, 等. 阿尔茨海默病中内质网应激与胶质细胞介导的神经炎症之间的联系[J]. 中国细胞生物学学报,2021,43(10):2045-2053. [6] 万航娟. 欧前胡素对阿尔茨海默病模型小鼠SIRT1/eIF2α通路的影响[D]. 赣州:赣南医学院,2022. [7] 李敏. 硫化氢对术后认知功能障碍的拮抗作用:涉及到抑制海马内质网应激及神经炎症[D]. 衡阳:南华大学,2017. [8] YU F, VOCK DM, ZHANG L, et al. Cognitive Effects of Aerobic Exercise in Alzheimer’s Disease: A Pilot Randomized Controlled Trial. J Alzheimers Dis. 2021;80(1):233-244. [9] 杜佳, 付燕, 范佳, 等. 有氧运动干预Aβ_(1-42)诱导阿尔茨海默病大鼠海马突触结构与突触蛋白的改变[J]. 中国组织工程研究,2023,27(33):5256-5262. [10] XIA J, LI BX, YIN LY, et al. Treadmill exercise decreases β-amyloid burden in APP/PS1 transgenic mice involving regulation of the unfolded protein response. Neurosci Lett. 2019;703:125-131. [11] 韩梦杰. 有氧运动通过BDNF-CREB信号通路对帕金森病小鼠的神经保护作用及机制研究[D]. 开封:河南大学,2021. [12] APÁTIGA-PÉREZ R, SOTO-ROJAS LO, CAMPA-CÓRDOBA BB, et al. Neurovascular dysfunction and vascular amyloid accumulation as early events in Alzheimer’s disease. Metab Brain Dis. 2022;37(1):39-50. [13] JOHANSSON ME, CAMERON IGM, VAN DER KOLK NM, et al. Aerobic Exercise Alters Brain Function and Structure in Parkinson’s Disease: A Randomized Controlled Trial. Ann Neurol. 2022;91(2):203-216. [14] 张业廷, 付燕, 李雪, 等. 有氧运动对阿尔茨海默症小鼠学习记忆及海马神经形态结构的影响[J]. 中国组织工程研究,2023,27(20):3188-3194. [15] 夏杰, 赵娜, 王璟,等. 有氧运动对APP/PS1小鼠海马内质网应激-细胞自噬偶联的影响[J]. 体育科学,2022,42(8):55-65. [16] NODA Y, KUZUYA A, TANIGAWA K, et al. Fibronectin type III domain-containing protein 5 interacts with APP and decreases amyloid β production in Alzheimer’s disease. Mol Brain. 2018;11(1):61. [17] LIU Y, CHU JMT, YAN T, et al. Short-term resistance exercise inhibits neuroinflammation and attenuates neuropathological changes in 3xTg Alzheimer’s disease mice. J Neuroinflammation. 2020;17(1):4. [18] ISHIWATA-KIMATA Y, HATA T, KIMATA Y. Self-association status-dependent inactivation of the endoplasmic reticulum stress sensor Ire1 by C-terminal tagging with artificial peptides. Biosci Biotechnol Biochem. 2022;86(6):739-746. [19] MANDULA JK, CHANG S, MOHAMED E, et al. Ablation of the endoplasmic reticulum stress kinase PERK induces paraptosis and type I interferon to promote anti-tumor T cell responses. Cancer Cell. 2022;40(10):1145-1160.e9. [20] SALVAGNO C, MANDULA JK, RODRIGUEZ PC, et al. Decoding endoplasmic reticulum stress signals in cancer cells and antitumor immunity. Trends Cancer. 2022;8(11):930-943. [21] KOU JJ, SHI JZ, HE YY, et al. Luteolin alleviates cognitive impairment in Alzheimer’s disease mouse model via inhibiting endoplasmic reticulum stress-dependent neuroinflammation. Acta Pharmacol Sin. 2022;43(4):840-849. [22] LI XQ, QIN YP, YE S, et al. Protective effect of Huangpu Tongqiao capsule against Alzheimer’s disease through inhibiting the apoptosis pathway mediated by endoplasmic reticulum stress in vitro and in vivo. Saudi Pharm J. 2022;30(11):1561-1571. [23] HONG JY, HONG SG, LEE J, et al. Exercise training ameliorates cerebrovascular dysfunction in a murine model of Alzheimer’s disease: role of the P2Y2 receptor and endoplasmic reticulum stress. Am J Physiol Heart Circ Physiol. 2020;318(6):H1559-H1569. [24] 刘涛. 运动对内质网应激诱导阿尔茨海默病模型大鼠海马细胞凋亡的影响[J]. 体育科学,2012,32(4):72-76. [25] HU J, CHEN Q, ZHU HR, et al. Microglial Piezo1 senses Aβ fibril stiffness to restrict Alzheimer’s disease. Neuron. 2023;111(1):15-29.e8. [26] LEE HG, WHEELER MA, QUINTANA FJ. Function and therapeutic value of astrocytes in neurological diseases. Nat Rev Drug Discov. 2022;21(5):339-358. [27] PAN RY, HE L, ZHANG J, et al. Positive feedback regulation of microglial glucose metabolism by histone H4 lysine 12 lactylation in Alzheimer’s disease. Cell Metab. 2022; 34(4):634-648.e6. [28] WANG CY, CHEN SS, GUO HY, et al. Forsythoside A Mitigates Alzheimer’s-like Pathology by Inhibiting Ferroptosis-mediated Neuroinflammation via Nrf2/GPX4 Axis Activation. Int J Biol Sci. 2022;18(5):2075-2090. [29] XIE LS, LIU Y, ZHANG N, et al. Electroacupuncture Improves M2 Microglia Polarization and Glia Anti-inflammation of Hippocampus in Alzheimer’s Disease. Front Neurosci. 2021;15:689629. |
| [1] | Zhang Yuxin, Yu Cong, Zhang Cui, Ding Jianjun, Chen Yan. Differences in postural control ability between older adults with mild cognitive impairment and those with normal cognition under different single-task and dual-task conditions [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(8): 1643-1649. |
| [2] | Xie Liugang, Cui Shuke, Guo Nannan, Li Aoyu, Zhang Jingrui. Research hotspots and frontiers of stem cells for Alzheimer’s disease [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1475-1485. |
| [3] | Qian Kun, Li Ziqing, Sun Shui . Endoplasmic reticulum stress in the occurrence and development of common degenerative bone diseases [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1285-1295. |
| [4] |
Li Tian, Ren Yuhua, Gao Yanping, Su Qiang.
Mechanism of agomelatine alleviating anxiety- and depression-like behaviors in APP/PS1 transgenic mice #br#
#br#
[J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1176-1182.
|
| [5] | Zhao Xiaoxuan, Liu Shuaiyi, Li Qi, Xing Zheng, Li Qingwen, Chu Xiaolei. Different exercise modalities promote functional recovery after peripheral nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1248-1256. |
| [6] | Wang Yuru, Li Siyuan, Xu Ye, Zhang Yumeng, Liu Yang, Hao Huiqin. Effects of wogonin on joint inflammation in collagen-induced arthritis rats via the endoplasmic reticulum stress pathway [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(5): 1026-1035. |
| [7] | Xu Zhenhua, Li Yanjie, Qin Hewei, Liu Haoyuan, Zhu Bochao, Wang Yupu. Traditional Chinese medicine monomer in treatment of neuroinflammation after spinal cord injury: effects of nuclear transcription factor kappa B signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(3): 590-598. |
| [8] | Hu Shujuan, Cheng Ping, Zhang Xiao, Ding Yiting, Liu Xuan, Pu Rui, Wang Xianwang. Effects of different exercise interventions on carboxylesterase 1 and inflammatory factors in skeletal muscle of type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(2): 269-278. |
| [9] | Zhu Bochao, Li Yanjie, Qin Hewei, Zhao Nannan, Liu Haoyuan, Xu Zhenhua, Wang Yupu. Jiawei Chunze Decoction treats urinary retention after spinal cord injury in rats based on the regulation of endoplasmic reticulum stress apoptosis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(2): 371-378. |
| [10] | Wang Ji, Zhang Min, Li Wenbo, Yang Zhongya, Zhang Long. Effect of aerobic exercise on glycolipid metabolism, skeletal muscle inflammation and autophagy in type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1200-1205. |
| [11] | Li Longyang, Zhang Songjiang, Zhao Xianmin, Zhou Chunguang, Gao Jianfeng. Electroacupuncture intervention on the proliferation and differentiation of hippocampal neurons and oligodendrocytes in Alzheimer’s disease model mice [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1029-1035. |
| [12] | Xie Yanli, Wei Siang, Zhang Guodong. Effects of treadmill exercise on metabolism and chronic neuroinflammation in type 1 diabetes mice of different sexes [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(35): 5577-5583. |
| [13] | Lu Donglei, Feng Zhanpeng, Cao Liquan, Tang Yi, Tan Sijie, Yu Zhongtao. Exercise intervention methods for senile sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(35): 5723-5731. |
| [14] | Xu Zheng, Zhao Xiaoqin, Chen Xiaodan, Wang Jiapu, Bao Fenmiao, Yu Liang, Li Junping, Wei Yan. Aerobic exercise upregulates the thioredoxin system and inhibits cardiomyocyte apoptosis in aging rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(34): 5508-5515. |
| [15] | Wen Huaneng, Lin Run, Wang Yixiao, Wang Bingshui, Liu Lu, Liu Chuanyao, Cai Canxin, Cui Shaoyang, Xu Mingzhu. Effects of electroacupuncture with “Zhi San Zhen” on Notch signaling pathway and synaptic plasticity in 5xFAD mice [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(32): 5148-5153. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||