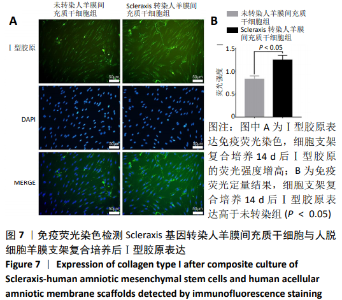
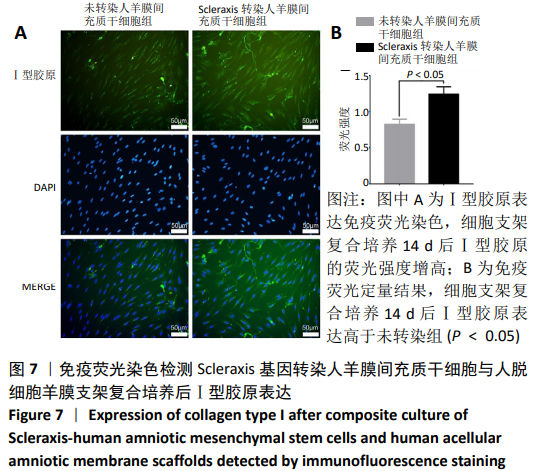
Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (7): 1037-1044.doi: 10.3969/j.issn.2095-4344.2169
Previous Articles Next Articles
Human acellular amniotic membrane scaffold promotes ligament differentiation of human amniotic mesenchymal stem cells modified by Scleraxis in vitro
Zou Gang1, 2, Xu Zhi3, Liu Ziming4, Li Yuwan1, 2, Yang Jibin1, 2, Jin Ying1, 2, Zhang Jun1, 2, Ge Zhen1, 2, Liu Yi1, 2
- 1Department of Orthopedics, Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China; 2Zunyi Medical University-Rochester University Orthopedic Research Center, Zunyi 563000, Guizhou Province, China; 3Second Department of Orthopedics, Hospital of the Jingxian County, Jingxian County 242500, Anhui Province, China; 4Institute of Sports Medicine, Peking University Third Hospital, Beijing 100191, China
-
Received:2020-02-24Revised:2020-02-29Accepted:2020-04-03Online:2021-03-08Published:2020-12-08 -
Contact:Liu Yi, Professor, Master’s supervisor, Department of Orthopedics, Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China; Zunyi Medical University-Rochester University Orthopedic Research Center, Zunyi 563000, Guizhou Province, China -
About author:Zou Gang, Master, Associate chief physician, Department of Orthopedics, Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China; Zunyi Medical University-Rochester University Orthopedic Research Center, Zunyi 563000, Guizhou Province, China Xu Zhi, Master, Second Department of Orthopedics, Hospital of the Jingxian County, Jingxian County 242500, Anhui Province, China -
Supported by:the Joint Fund of Guizhou Provincial Science and Technology Department, No. [2017]7105
CLC Number:
Cite this article
Zou Gang, Xu Zhi, Liu Ziming, Li Yuwan, Yang Jibin, Jin Ying, Zhang Jun, Ge Zhen, Liu Yi. Human acellular amniotic membrane scaffold promotes ligament differentiation of human amniotic mesenchymal stem cells modified by Scleraxis in vitro[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1037-1044.
share this article
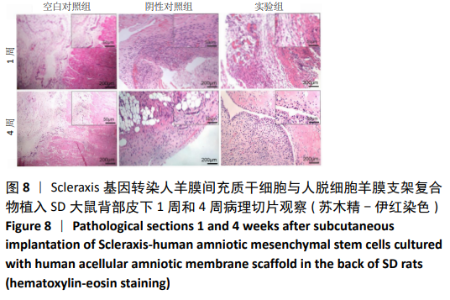
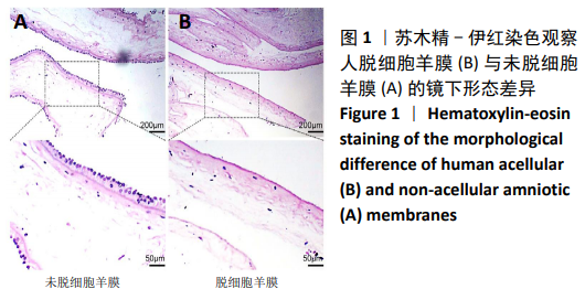
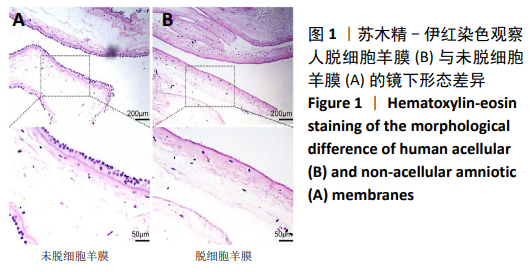
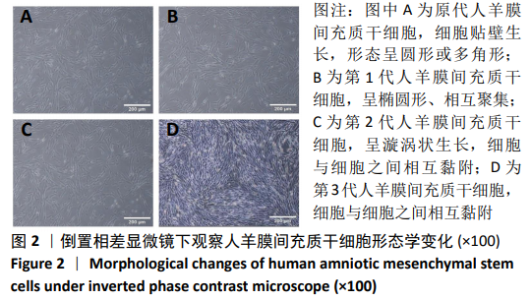
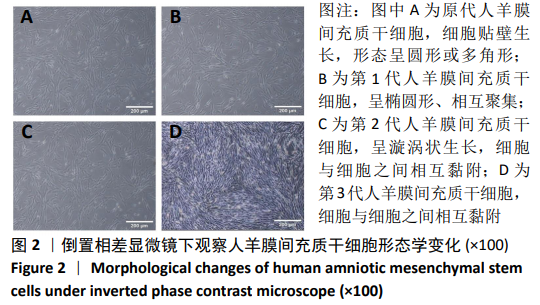
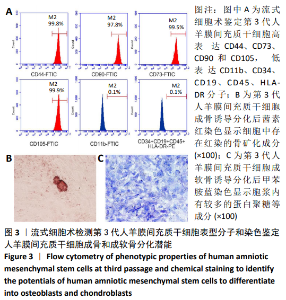
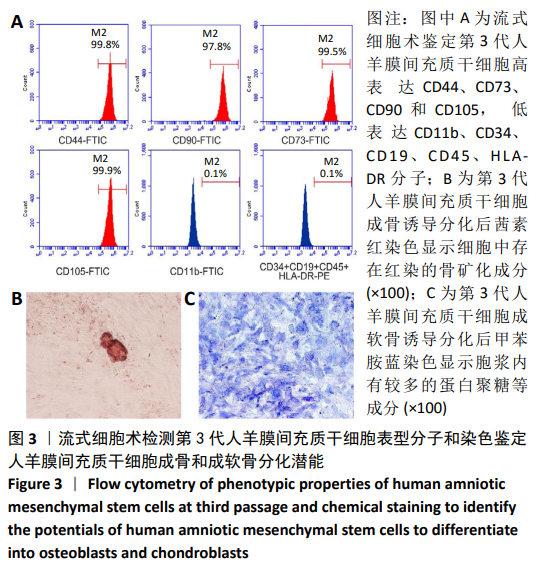
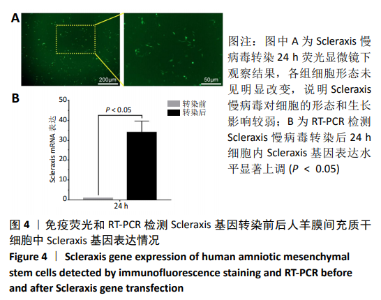
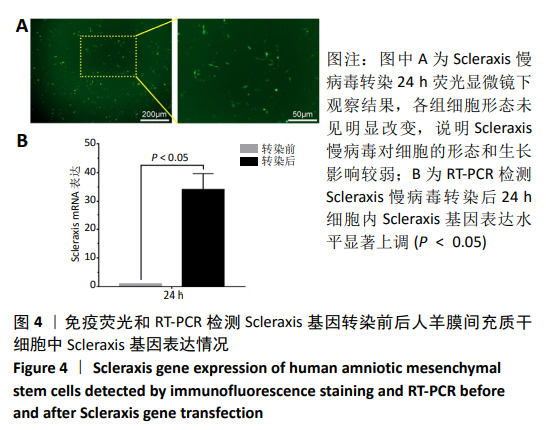
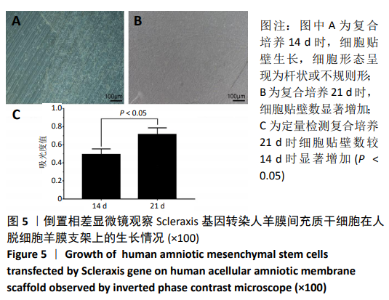
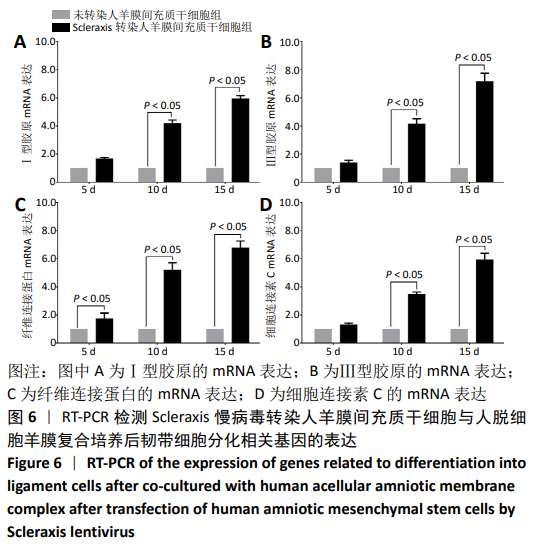
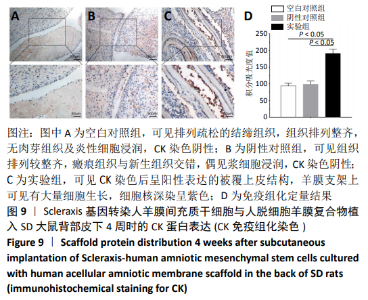
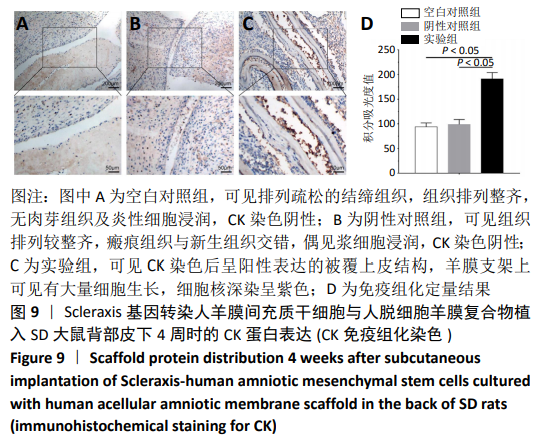
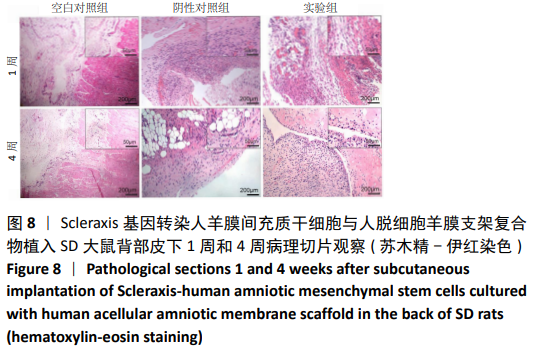
2.8 Scleraxis基因转染人羊膜间充质干细胞与人脱细胞羊膜支架复合物植入大鼠体内的免疫原性 细胞支架复合物移植入SD大鼠背部皮下1周和4周时收集样本行苏木精-伊红染色,4周时收集样本行CK免疫组织化学染色。 苏木精-伊红染色:①空白对照组:1周和4周时筋膜层组织可见排列疏松的脂肪层与脂肪层下方整齐排列的横纹肌,无肉芽组织及炎性细胞浸润。②阴性对照组:1周时皮下组织结构紊乱,层次表现不明显,大量中性粒细胞将组织包绕,基底层偶见浆细胞,局部炎症较为典型;4周时可见术区大量空泡状脂肪细胞及疏松结缔组织,肉芽组织及新生血管减少,偶见中性粒细胞、浆细胞,炎症反应减退。③实验组:1周时相较于阴性对照组炎症反应更为明显,肉芽组织及纤维组织增生,可见术区肌肉萎缩,炎性细胞浸润;4周时炎性细胞明显减少,组织排列趋于整齐,可见淡粉色的被覆上皮组织,高度认为是人脱细胞羊膜结构,其表面可见大量细胞附着生长。见图8。"
| [1] ŚMIGIELSKI R, ZDANOWICZ U, DRWIĘGA M, et al. The anatomy of the anterior cruciate ligament and its relevance to the technique of reconstruction. Bone Joint J. 2016;98-B(8):1020-1026. [2] SOMMERFELDT M, GOODINE T, RAHEEM A, et al. Relationship Between Time to ACL Reconstruction and Presence of Adverse Changes in the Knee at the Time of Reconstruction. Orthop J Sports Med. 2018;6(12):2325967118813917. [3] AGA C, RISBERG MA, FAGERLAND MW, et al. No Difference in the KOOS Quality of Life Subscore Between Anatomic Double-Bundle and Anatomic Single-Bundle Anterior Cruciate Ligament Reconstruction of the Knee: A Prospective Randomized Controlled Trial With 2 Years’ Follow-up. Am J Sports Med. 2018; 46(10):2341-2354. [4] RAN J, HU Y, LE H, et al. Ectopic tissue engineered ligament with silk collagen scaffold for ACL regeneration: A preliminary study. Acta Biomater. 2017;53: 307-317. [5] ZHANG PX, HAN N, KOU YH, et al. Tissue engineering for the repair of peripheral nerve injury. Neural Regen Res. 2019;14(1):51-58. [6] ZHOU H, WANG L, ZHANG C, et al. Feasibility of repairing full-thickness skin defects by iPSC-derived epithelial stem cells seeded on a human acellular amniotic membrane. Stem Cell Res Ther. 2019;10(1):155. [7] XUE SL, LIU K, PAROLINI O, et al. Human acellular amniotic membrane implantation for lower third nasal reconstruction: a promising therapy to promote wound healing. Burns Trauma. 2018;6:34. [8] 杨继彬,朱喜忠,熊华章,等.人羊膜间充质干细胞与人脱细胞羊膜支架的生物相容性[J].中国组织工程研究杂志,2018,22(5):742-747. [9] LIU Z, LI Y, REN Y, et al. Enhancement of in vitro proliferation and bioactivity of human anterior cruciate ligament fibroblasts using an in situ tissue isolation method and basic fibroblast growth factor culture conditions: A pilot analysis. Medicine (Baltimore). 2019;98(22):e15907. [10] LARSON BL, YU SN, PARK H, et al. Chondrogenic, hypertrophic, and osteochondral differentiation of human mesenchymal stem cells on three-dimensionally woven scaffolds. J Tissue Eng Regen Med. 2019;13(8):1453-1465. [11] HASSAN G, KASEM I, SOUKKARIEH C, et al. A Simple Method to Isolate and Expand Human Umbilical Cord Derived Mesenchymal Stem Cells: Using Explant Method and Umbilical Cord Blood Serum. Int J Stem Cells. 2017;10(2):184-192. [12] HE A, LIU L, LUO X, et al. Repair of osteochondral defects with in vitro engineered cartilage based on autologous bone marrow stromal cells in a swine model. Sci Rep. 2017;7:40489. [13] LIU R, ZHANG X, FAN Z, et al. Human amniotic mesenchymal stem cells improve the follicular microenvironment to recover ovarian function in premature ovarian failure mice. Stem Cell Res Ther. 2019;10(1):299. [14] ABBASI-KANGEVARI M, GHAMARI SH, SAFAEINEJAD F, et al. Potential Therapeutic Features of Human Amniotic Mesenchymal Stem Cells in Multiple Sclerosis: Immunomodulation, Inflammation Suppression, Angiogenesis Promotion, Oxidative Stress Inhibition, Neurogenesis Induction, MMPs Regulation, and Remyelination Stimulation. Front Immunol. 2019;10:238. [15] YUAN Z, BIAN Y, MA X, et al. LncRNA H19 Knockdown in Human Amniotic Mesenchymal Stem Cells Suppresses Angiogenesis by Associating with EZH2 and Activating Vasohibin-1. Stem Cells Dev. 2019;28(12):781-790. [16] GOHI BFCA, LIU XY, ZENG HY, et al. Enhanced efficiency in isolation and expansion of hAMSCs via dual enzyme digestion and micro-carrier. Cell Biosci. 2020;10:2. [17] BIAN Y, DU Y, WANG R, et al. A comparative study of HAMSCs/HBMSCs transwell and mixed coculture systems. IUBMB Life. 2019;71(7):1048-1055. [18] ZHANG C, DU Y, YUAN H, et al. HAMSCs/HBMSCs coculture system ameliorates osteogenesis and angiogenesis against glucolipotoxicity. Biochimie. 2018;152: 121-133. [19] CHEN W, XIAO S, WEI Z, et al. Schwann Cell-Like Cells Derived from Human Amniotic Mesenchymal Stem Cells Promote Peripheral Nerve Regeneration through a MicroRNA-214/c-Jun Pathway. Stem Cells Int. 2019;2019:2490761. [20] LI Y, LIU Z, TANG Y, et al. Schnurri-3 regulates BMP9-induced osteogenic differentiation and angiogenesis of human amniotic mesenchymal stem cells through Runx2 and VEGF. Cell Death Dis. 2020;11(1):72. [21] BIAN Y, MA X, WANG R, et al. Human amnion-derived mesenchymal stem cells promote osteogenesis of human bone marrow mesenchymal stem cells against glucolipotoxicity. FEBS Open Bio. 2018;9(1):74-81. [22] TOPOLUK N, HAWKINS R, TOKISH J, et al. Amniotic Mesenchymal Stromal Cells Exhibit Preferential Osteogenic and Chondrogenic Differentiation and Enhanced Matrix Production Compared With Adipose Mesenchymal Stromal Cells. Am J Sports Med. 2017;45(11):2637-2646. [23] CRANE JL, XIAN L, CAO X. Role of TGF-β Signaling in Coupling Bone Remodeling. Methods Mol Biol. 2016;1344:287-300. [24] 邹刚,李豫皖,金瑛,等.TGF-β1联合VEGF对人羊膜间充质干细胞向韧带成纤维细胞体外分化作用的研究[J].中国修复重建外科杂志,2017,31(5): 582-593. [25] KAGUE E, HUGHES SM, LAWRENCE EA, et al. Scleraxis genes are required for normal musculoskeletal development and for rib growth and mineralization in zebrafish. FASEB J. 2019;33(8):9116-9130. [26] HSIEH CF, YAN Z, SCHUMANN RG, et al. In Vitro Comparison of 2D-Cell Culture and 3D-Cell Sheets of Scleraxis-Programmed Bone Marrow Derived Mesenchymal Stem Cells to Primary Tendon Stem/Progenitor Cells for Tendon Repair. Int J Mol Sci. 2018;19(8). pii: E2272. [27] SAKABE T, SAKAI K, MAEDA T, et al. Transcription factor scleraxis vitally contributes to progenitor lineage direction in wound healing of adult tendon in mice. J Biol Chem. 2018;293(16):5766-5780. [28] ALBERTON P, POPOV C, PRÄGERT M, et al. Conversion of human bone marrow-derived mesenchymal stem cells into tendon progenitor cells by ectopic expression of scleraxis. Stem Cells Dev. 2012;21(6):846-858. [29] NICHOLS AEC, WERRE SR, DAHLGREN LA. Transient Scleraxis Overexpression Combined with Cyclic Strain Enhances Ligament Cell Differentiation. Tissue Eng Part A. 2018;24(19-20):1444-1455. [30] 朱喜忠,刘子铭,吴术红.Scleraxis慢病毒基因感染人羊膜间充质干细胞向肌腱细胞的定向分化[J].中国组织工程研究,2017,21(33):5382-5387. [31] NOVAKOVA SS, MAHALINGAM VD, FLORIDA SE, et al. Tissue-engineered tendon constructs for rotator cuff repair in sheep. J Orthop Res. 2018;36(1):289-299. [32] CHEN C, LIU F, TANG Y, et al. Book-Shaped Acellular Fibrocartilage Scaffold with Cell-loading Capability and Chondrogenic Inducibility for Tissue-Engineered Fibrocartilage and Bone-Tendon Healing. ACS Appl Mater Interfaces. 2019;11(3):2891-2907. [33] SANAEI P, CUMMINGS LJ, WATERS SL, et al. Curvature- and fluid-stress-driven tissue growth in a tissue-engineering scaffold pore. Biomech Model Mechanobiol. 2019;18(3):589-605. [34] ZHANG Z, ZENG L, YANG J, et al. Amniotic membrane-derived stem cells help repair osteochondral defect in a weight-bearing area in rabbits. Exp Ther Med. 2017;14(1):187-192. [35] CHEHELCHERAGHI F, EIMANI H, HOMAYOONSADRAIE S, et al. Effects of Acellular Amniotic Membrane Matrix and Bone Marrow-Derived Mesenchymal Stem Cells in Improving Random Skin Flap Survival in Rats. Iran Red Crescent Med J. 2016;18(6):e25588. [36] ZHANG L, ZOU D, LI S, et al. An Ultra-thin Amniotic Membrane as Carrier in Corneal Epithelium Tissue-Engineering. Sci Rep. 2016;6:21021. [37] TANG K, WU J, XIONG Z, et al. Human acellular amniotic membrane: A potential osteoinductive biomaterial for bone regeneration. J Biomater Appl. 2018;32(6):754-764. [38] JORGE LF, FRANCISCO JC, BERGONSE N, et al. Tracheal repair with acellular human amniotic membrane in a rabbit model. J Tissue Eng Regen Med. 2018; 12(3):e1525-e1530. [39] ZHENG Y, JI S, WU H, et al. Topical administration of cryopreserved living micronized amnion accelerates wound healing in diabetic mice by modulating local microenvironment. Biomaterials. 2017;113:56-67. [40] DESPEYROUX A, DURET C, GONDEAU C, et al. Mesenchymal stem cells seeded on a human amniotic membrane improve liver regeneration and mouse survival after extended hepatectomy. J Tissue Eng Regen Med. 2018;12(4):1062-1073. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Min Youjiang, Yao Haihua, Sun Jie, Zhou Xuan, Yu Hang, Sun Qianpu, Hong Ensi. Effect of “three-tong acupuncture” on brain function of patients with spinal cord injury based on magnetic resonance technology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-8. |
| [4] | Zhang Chong, Liu Zhiang, Yao Shuaihui, Gao Junsheng, Jiang Yan, Zhang Lu. Safety and effectiveness of topical application of tranexamic acid to reduce drainage of elderly femoral neck fractures after total hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1381-1386. |
| [5] | Wang Haiying, Lü Bing, Li Hui, Wang Shunyi. Posterior lumbar interbody fusion for degenerative lumbar spondylolisthesis: prediction of functional prognosis of patients based on spinopelvic parameters [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1393-1397. |
| [6] | Zhang Shangpu, Ju Xiaodong, Song Hengyi, Dong Zhi, Wang Chen, Sun Guodong. Arthroscopic suture bridge technique with suture anchor in the treatment of acromioclavicular dislocation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1417-1422. |
| [7] | Chen Jinping, Li Kui, Chen Qian, Guo Haoran, Zhang Yingbo, Wei Peng. Meta-analysis of the efficacy and safety of tranexamic acid in open spinal surgery [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1458-1464. |
| [8] | Shu Wenbo, Chen Mengchi, Li Hua, Huang Liqian, Huang Binbin, Zhang Wenhai, Wu Yachen, Wang Zefeng, Li Qiaoli, Liu Peng. Correlation between body fat distribution and characteristics of daily physical activity in college students [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1277-1283. |
| [9] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [10] | Wu Xun, Meng Juanhong, Zhang Jianyun, Wang Liang. Concentrated growth factors in the repair of a full-thickness condylar cartilage defect in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1166-1171. |
| [11] | Shen Jinbo, Zhang Lin. Micro-injury of the Achilles tendon caused by acute exhaustive exercise in rats: ultrastructural changes and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1190-1195. |
| [12] | Zeng Zhen, Hu Jingwei, Li Xuan, Tang Linmei, Huang Zhiqiang, Li Mingxing. Quantitative analysis of renal blood flow perfusion using contrast-enhanced ultrasound in rats with hemorrhagic shock during resuscitation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1201-1206. |
| [13] | Tang Hui, Yao Zhihao, Luo Daowen, Peng Shuanglin, Yang Shuanglin, Wang Lang, Xiao Jingang. High fat and high sugar diet combined with streptozotocin to establish a rat model of type 2 diabetic osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1207-1211. |
| [14] | Li Jiacheng, Liang Xuezhen, Liu Jinbao, Xu Bo, Li Gang. Differential mRNA expression profile and competitive endogenous RNA regulatory network in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1212-1217. |
| [15] | Chai Le, Lü Jianlan, Hu Jintao, Hu Huahui, Xu Qingjun, Yu Jinwei, Quan Renfu. Signal pathway variation after induction of inflammatory response in rats with acute spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1218-1223. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||