Chinese Journal of Tissue Engineering Research ›› 2016, Vol. 20 ›› Issue (40): 5966-5972.doi: 10.3969/j.issn.2095-4344.2016.40.006
Previous Articles Next Articles
Neuroprotective effects of sufentanil preconditioning on spinal cord injury in mouse models
Yu Yu-qin1, Hu Nian-chun1, Duan Ji-an1, Li Da-peng1, Liu Chang2
- 1Department of Anesthesiology, Wuhan Central Hospital, Wuhan 430000, Hubei Province, China; 2Department of Anesthesiology, WISCO General Hospital, Wuhan 430000, Hubei Province, China
-
Revised:2016-08-23Online:2016-09-30Published:2016-09-30 -
Contact:Liu Chang, Attending physician, Department of Anesthesiology, WISCO General Hospital, Wuhan 430000, Hubei Province, China -
About author:Yu Yu-qin, Attending physician, Department of Anesthesiology, Wuhan Central Hospital, Wuhan 430000, Hubei Province, China
CLC Number:
Cite this article
Yu Yu-qin, Hu Nian-chun, Duan Ji-an, Li Da-peng, Liu Chang. Neuroprotective effects of sufentanil preconditioning on spinal cord injury in mouse models[J]. Chinese Journal of Tissue Engineering Research, 2016, 20(40): 5966-5972.
share this article
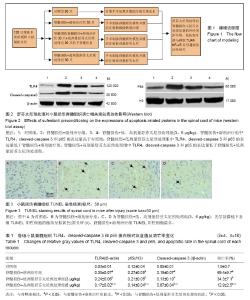
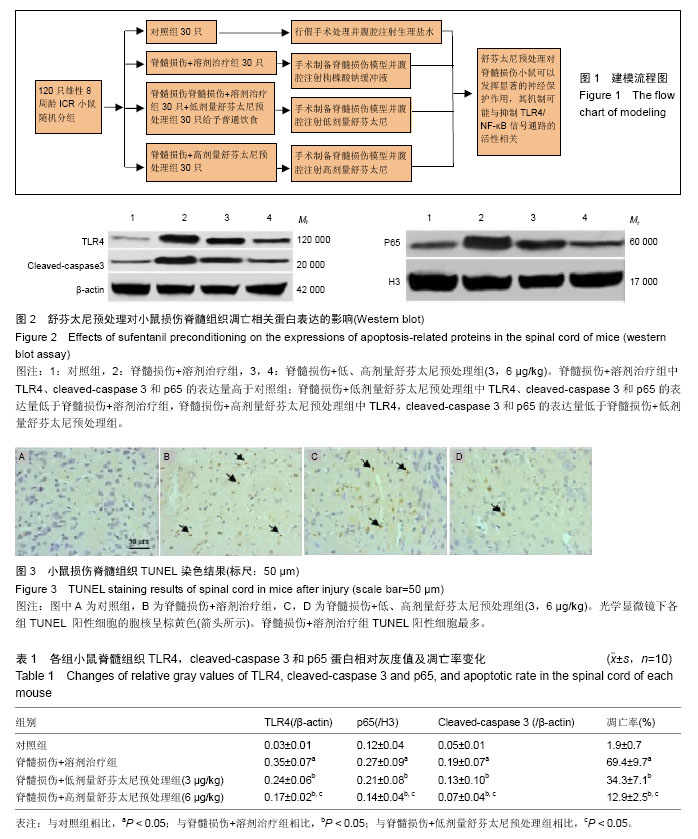
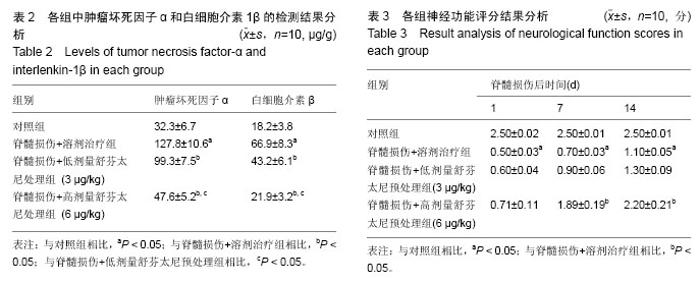
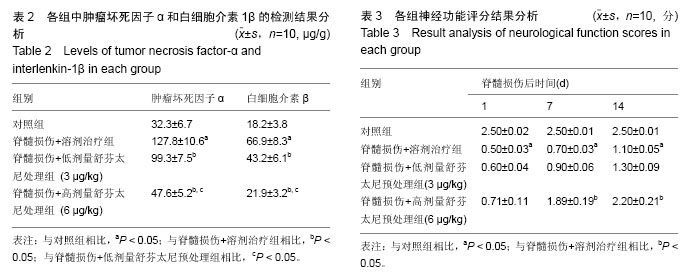
2.1 实验动物数量分析 纳入大鼠120只,进入结果分析120只,死亡和感染4只,并及时补充,无脱失值。建模流程图见图1。 2.2 舒芬太尼预处理对小鼠损伤脊髓组织细胞凋亡的影响 如图2、表1所示,与对照组相比,脊髓损伤组中cleaved-caspase 3蛋白的表达水平显著增加(P < 0.05),舒芬太尼预处理可以显著抑制cleaved-caspase 3蛋白的表达(P < 0.05);TUNEL染色结果表明(图3,表1),在对照组中,TUNEL阳性细胞个数很少;与对照组相比,溶剂治疗组中神经细胞的凋亡率显著增加(图3,P < 0.05);而腹腔注射舒芬太尼则显著减少了神经细胞的凋亡率(P < 0.05),且呈剂量依赖性。 2.3 舒芬太尼预处理对小鼠损伤脊髓组织TLR4/NF- κBp65蛋白表达的影响 免疫印迹研究结果表明(图2,表1),与对照组相比,溶剂治疗组中TLR4和p65的表达显著增加(P < 0.05);与溶剂处理组相比,舒芬太尼预处理可以显著抑制TLR4和p65的表达(P < 0.05),且呈剂量依赖性。"
2.4 舒芬太尼预处理对对小鼠损伤脊髓组织肿瘤坏死因子α和白细胞介素1β蛋白表达的影响 酶联免疫吸附法检测结果显示,与对照组相比,溶剂治疗组中肿瘤坏死因子α和白细胞介素1β的表达水平显著增加;与溶剂处理组相比,舒芬太尼预处理可以显著抑制肿瘤坏死因子α和白细胞介素1β的表达(P < 0.05),且呈剂量依赖性(表2)。 2.5 舒芬太尼预处理可以显改善脊髓损伤的神经功能评分且呈剂量依赖性 对照组的神经功能评分在14 d内稳定在2.5分,与对照组相比,脊髓损伤+溶剂治疗组的运动功能评分显著降低(表3,P < 0.05);在脊髓损伤后7和14 d,高剂量舒芬太尼预处理可以显著改善小鼠在脊髓损伤后的运动功能评分(P < 0.05)。"
| [1] Singh A, Tetreault L, Kalsi-Ryan S, et al. Global prevalence andincidence of traumaticspinal cord injury. Clin Epidemiol. 2014;6:309-331. [2] Huang XL, Yan TB. Rehabilitation Medicine. Beijing: People’s Medical Publishing House, 2013:166. [3] Chen YH, Liu M, He JH. Epidemiological survey ofpatients with spinal cord injury. J Pract Med. 2011; 27(6):1032-1034. [4] 孙兆忠,李瑞.脐血干细胞移植及电针治疗脊髓损伤大鼠神经生长因子及神经营养因子3的表达[J].中国组织工程研究,2015,19(7):61-66. [5] Assuncao-Silva RC, Gomes ED, Sousa N, et al. Hydrogels and cell based therapies in spinal cord injury regeneration. Stem Cells Int. 2015;2015:948040. [6] Polakiewicz RD, Schieferl SM, Gingras AC, et al. mu-Opioid receptor activates signaling pathways implicated in cell survivaland translational control. J Biol Chem. 1998,273(36):23534-23541. [7] Lemoine S, Zhu L, Massetti M, et al. Continuous administration of remifentanil and sufentanil induces cardioprotection in human myocardium, in vitro. Acta Anaesthesiol Scand. 2011;55(6):758-764. [8] 刘祥,景桂霞,白娟,等.舒芬太尼预处理对大鼠心肌缺血再灌注时PI3K/Akt的影响[J].南方医科大学学报,2014,34(3):335-340. [9] 陈朝板,屠伟峰,郄文斌.舒芬太尼预处理对大鼠肠道缺血再灌注后氧自由基介导性损伤的影响[J].实用医学杂志,2012,28(13):2172-2175. [10] 王美芳,孟尽海,魏华.舒芬太尼预处理对脑缺血再灌注损伤大鼠脑保护功能的影响[J].宁夏医科大学学报,2011, 33(11):1023-1025, 1032. [11] 谢丽萍,王胜,刘扬,等.c-Jun氨基末端激酶信号通路在不同剂量舒芬太尼预处理大鼠肝脏缺血-再灌注损伤中的作用[J].临床麻醉学杂志,2016,32(2):175-179. [12] 贺萱,田首元,王建刚,等.舒芬太尼后处理对缺血再灌注损伤的大鼠心肌自噬的影响及PI3K-Akt通路在其中的作用[J].中国药物与临床,2016,16(4):470-472. [13] Paterniti I, Esposito E, Mazzon E, et al. Evidence for the role of PI(3)-kinase-AKT-eNOS signalling pathway in secondary inflammatory process after spinal cord compression injury in mice. Eur J Neurosci. 2011; 33(8):1411-1420. [14] 吴晓明,高文山,王静,等.盐酸川芎嗪联合骨髓间充质干细胞移植对脊髓损伤模型大鼠的神经保护[J].中国组织工程研究,2016,20(1):95-101. [15] 李维朝.脂多糖预处理对脊髓损伤大鼠神经细胞抗凋亡的作用及相关机制研究[D].重庆:重庆医科大学,2014. [16] Basso DM, Fisher LC, Anderson AJ, et al. Basso Mouse Scale for locomotion detects differences in recovery after spinal cord injury in five common mouse strains. J Neurotrauma. 2006;23(5):635-659. [17] Minkowitz HS. A review of sufentanil and the sufentanil sublingual tablet system for acute moderate to severe pain. Pain Manag.2015;5(4):237-250. [18] Maciejewski D. Sufentanil in anaesthesiology and intensive therapy. Anaesthesiol Intensive Ther. 2012; 44(1):35-41. [19] Zhao J, Chen C, Xiao JR, et al. An Up-regulation of IRF-1 After a Spinal Cord Injury: Implications for Neuronal Apoptosis. J Mol Neurosci, 2015;57(4):595-604. [20] 王继权,赵兴长,孙平,等.依达拉奉抗大鼠脊髓损伤后脊髓细胞凋亡的机制初探[J].天津医药,2015,43(9):988-991. [21] 孙平,范仲凯,李昊天,等.硫化氢对大鼠急性脊髓损伤的保护作用[J].天津医药,2015,43(11):1271-1274. [22] Siebert JR, Eade AM, Osterhout DJ. Biomaterial approaches to enhancing neurorestoration after spinal cord injury: strategies for overcoming inherent biological obstacles. Biomed Res Int. 2015;2015:752572. [23] Houle JD, Côté MP. Axon regeneration and exercise dependent plas-ticity after spinal cord injury. Ann N Y Acad Sci. 2013;1279:154-163. [24] Tsintou M, Dalamagkas K, Seifalian AM. Advances in regenerative therapies for spinal cord injury: a biomaterials approach. Neural Regen Res. 2015;10(5): 726-742. [25] Wei HY, Ma X. Tamoxifen reduces infiltration of inflammatory cells, apoptosis and inhibits IKK/NF-κB pathway after spinal cord injury in rats. Neurol Sci. 2014;35(11):1763-1768. [26] Esposito E, Impellizzeri D, Mazzon E, et al. The NAMPT inhibitor FK866 reverts the damage in spinal cord injury. J Neuroinflammation. 2012;9:66. [27] Bachmeier BE, Nerlich AG, Weiler C, et al. Analysis of tissue distribution of TNF-alpha, TNF-alpha-receptors, and the activating TNF- alpha-converting enzyne suggests activation of the TNF-alpha system in the ageing intervertebral disc.Ann NY Acad Sci. 2007;1096:442-541. [28] Xu R, Xu G, Shi J, et al. A correlative study of NF-kappa B activity and cytokines expression in human chronic nasal sinusitis. Laryngol Otol. 2007; 121:644-649. [29] 吴俊.促红细胞生成素对大鼠急性脊髓损伤后核因子-κB及炎症因子表达的影响[D].南京:南京医科大学,2011. [30] 白国良,周建斌,刘保江.舒芬太尼对大鼠心肌缺血再灌注中Toll样受体4以及肿瘤坏死因子α的影响[J].当代医学, 2013,19(17):18-20. [31] Sindhu S, Al-Roub A, Koshy M, et al. Palmitate-Induced MMP-9 Expression in the Human Monocytic Cells is Mediated through the TLR4-MyD88 Dependent Mechanism. Cell Physiol Biochem. 2016; 39(3):889-900. [32] Müller HD, Cvikl BB, Lussi AA, et al. Salivary pellets induce a pro-inflammatory response involving the TLR4-NF-kB pathway in gingival fibroblasts. BMC Oral Health. 2016;17(1):15. [33] Feng H, Su R, Song Y, et al. Positive Correlation between Enhanced Expression of TLR4/MyD88/NF-κB with Insulin Resistance in Placentae of Gestational Diabetes Mellitus. PLoS One. 2016;11(6):e0157185. [34] Wang Y, Chen G, Yu X, et al. Salvianolic Acid B Ameliorates Cerebral Ischemia/Reperfusion Injury Through Inhibiting TLR4/MyD88 Signaling Pathway. Inflammation. 2016;39(4):1503-1513. [35] Hajivalili M, Pourgholi F, Majidi J, et al. G2013 modulates TLR4 signaling pathway in IRAK-1 and TARF-6 dependent and miR-146a independent manner. Cell Mol Biol (Noisy-le-grand). 2016;62(4):1-5. [36] Karuppagounder V, Giridharan VV, Arumugam S, et al. Modulation of Macrophage Polarization and HMGB1-TLR2/TLR4 Cascade Plays a Crucial Role for Cardiac Remodeling in Senescence-Accelerated Prone Mice. PLoS One. 2016;11(4):e0152922. [37] Belinga VF, Wu GJ, Yan FL, et al. Splenectomy following MCAO inhibits the TLR4-NF-κB signaling pathway and protects the brain from neurodegeneration in rats. J Neuroimmunol. 2016;293:105-113. [38] Rashidian A, Muhammadnejad A, Dehpour AR, et al. Atorvastatin attenuates TNBS-induced rat colitis: the involvement of the TLR4/NF-kB signaling pathway. Inflammopharmacology. 2016;24(2-3):109-118. [39] Lv W, Chen N, Lin Y, et al. Macrophage migration inhibitory factor promotes breast cancer metastasis via activation of HMGB1/TLR4/NF kappa B axis. Cancer Lett. 2016;375(2):245-255. [40] Wang D, Tao K, Xion J, et al. TAK-242 attenuates acute cigarette smoke-induced pulmonary inflammation in mouse via the TLR4/NF-κB signaling pathway. Biochem Biophys Res Commun. 2016; 472(3):508-515. [41] Gong Y, Tao L, Jing L, et al. Association of TLR4 and Treg in Helicobacter pylori Colonization and Inflammation in Mice. PLoS One. 2016;11(2):e0149629. [42] Kang L, Zhu G, Ouyang Q, et al. Expression and clinical significance of TLR4 and NF-κB in cholesterol pneumonia. Zhonghua Bing Li Xue Za Zhi. 2015;44(11): 790-791. [43] Zhang DS, Bai XH, Yao YJ, et al. Effect of Berberine on the Insulin Resistance and TLR4/IKKbeta/ NF-kappaB Signaling Pathways in Skeletal Muscle of Obese Rats with Insulin Resistance. Sichuan Da Xue Xue Bao Yi Xue Ban. 2015;46(6):827-831. [44] Hung CC, Lin CH, Chang H, et al. Astrocytic GAP43 Induced by the TLR4/NF-κB/STAT3 Axis Attenuates Astrogliosis-Mediated Microglial Activation and Neurotoxicity. J Neurosci. 2016;36(6):2027-2043. [45] Antón M, Alén F, Gómez de Heras R, et al. Oleoylethanolamide prevents neuroimmune HMGB1/TLR4/NF-kB danger signaling in rat frontal cortex and depressive-like behavior induced by ethanol binge administration. Addict Biol. 2016. [46] Wang Y, Su L, Morin MD, et al. TLR4/MD-2 activation by a synthetic agonist with no similarity to LPS. Proc Natl Acad Sci U S A. 2016;113(7):E884-E893. [47] Qin DP, Zhou YJ, Zhang SZ, et al. Study on effects of Tripterygium wilfordii polycoride in resisting macrophage inflammation and regulating inflammation via TLR4/NF-κB. Zhongguo Zhong Yao Za Zhi. 2015; 40(16):3256-3261. [48] Lee SI, Kim HS, Koo JM, et al. Lactobacillus acidophilus modulates inflammatory activity by regulating the TLR4 and NF-κB expression in porcine peripheral blood mononuclear cells after lipopolysaccharide challenge. Br J Nutr. 2016;115(4):567-575. [49] Previtera ML, Sengupta A. Substrate Stiffness Regulates Proinflammatory Mediator Production through TLR4 Activity in Macrophages. PLoS One. 2015;10(12):e0145813. [50] Zhao H, Zheng Q, Hu X, et al. Betulin attenuates kidney injury in septic rats through inhibiting TLR4/NF-κB signaling pathway. Life Sci. 2016;144:185-193. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||