Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (26): 6729-6735.doi: 10.12307/2026.837
Previous Articles Next Articles
Preparation and characterization of beta-tricalcium phosphate/polyvinyl alcohol and hydroxyapatite/polyvinyl alcohol bone substitute materials
Hu Zhizhao1, 2, 3, Liu Zhaoyong1, Guo Jing1, Jin Shengnan1, Zhou Qiqi2, 3, Hu Yang2, 3
- 1Department of Stomatology, Zhuhai People's Hospital (Affiliated Hospital of Beijing Institute of Technology, Zhuhai Clinical Medical College of Jinan University), Zhuhai 519000, Guangdong Province, China; 2Department of Oral Restoration and Implants, First Affiliated Hospital of Xinjiang Medical University (Affiliated Stomatological Hospital), Urumqi 830054, Xinjiang Uygur Autonomous Region, China; 3Xinjiang Uygur Autonomous Region Institute of Stomatology, Urumqi 830054, Xinjiang Uygur Autonomous Region, China
-
Accepted:2026-01-06Online:2026-09-18Published:2026-03-10 -
Contact:Hu Yang, MS, Associate professor, Associate chief physician, Department of Oral Restoration and Implants, First Affiliated Hospital of Xinjiang Medical University (Affiliated Stomatological Hospital), Urumqi 830054, Xinjiang Uygur Autonomous Region, China; Xinjiang Uygur Autonomous Region Institute of Stomatology, Urumqi 830054, Xinjiang Uygur Autonomous Region, China -
About author:Hu Zhizhao, MS, Attending physician, Department of Stomatology, Zhuhai People's Hospital (Affiliated Hospital of Beijing Institute of Technology, Zhuhai Clinical Medical College of Jinan University), Zhuhai 519000, Guangdong Province, China; Department of Oral Restoration and Implants, First Affiliated Hospital of Xinjiang Medical University (Affiliated Stomatological Hospital), Urumqi 830054, Xinjiang Uygur Autonomous Region, China; Xinjiang Uygur Autonomous Region Institute of Stomatology, Urumqi 830054, Xinjiang Uygur Autonomous Region, China -
Supported by:Key Research and Development Task Special Project of Xinjiang Uygur Autonomous Region, No. 2016B03049-2 (to HY); Natural Science Foundation Project of Xinjiang Uygur Autonomous Region, No. 2022D01C752 (to ZQQ)
CLC Number:
Cite this article
Hu Zhizhao, Liu Zhaoyong, Guo Jing, Jin Shengnan, Zhou Qiqi, Hu Yang. Preparation and characterization of beta-tricalcium phosphate/polyvinyl alcohol and hydroxyapatite/polyvinyl alcohol bone substitute materials[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6729-6735.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

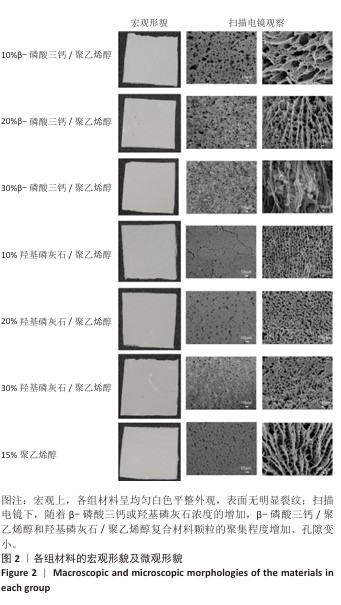
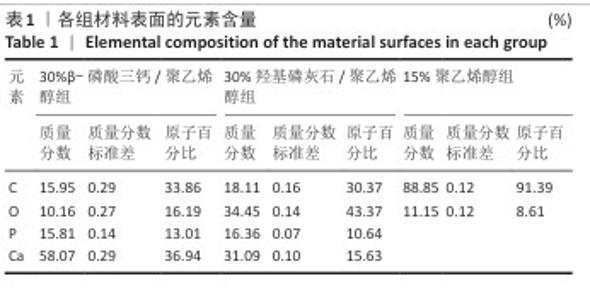
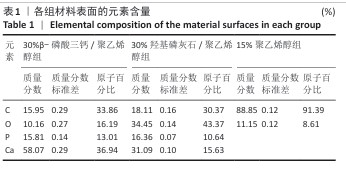
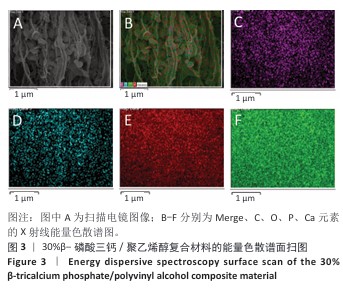
2.2 各组材料X射线能量色散谱分析结果 各组材料表面的Mapping元素面扫分布,见图3-5。各组材料表面的元素含量半定量分析结果,见表1。 结果显示,30%β-磷酸三钙/聚乙烯醇组材料中含有C、Ca、P、O元素,各元素在材料表面广泛分布且较为均匀,并未检测出其他元素,说明该复合材料在制备过程中并未被杂质污染,符合此次表征实验要求。30%羟基磷灰石/聚乙烯醇组材料表面所含元素与30%β-磷酸三钙/聚乙烯醇组相比未发生改变,仍是Ca、P、C、O四种元素,未检测出其他杂质元素污染,符合实验目的。15%聚乙烯醇组材料表面所含元素种类相比30%β-磷酸三钙/聚乙烯醇组、30%羟基磷灰石/聚乙烯醇组减少,仅含有C、O两种元素,C、O两种元素在材料各区域分布均匀。 "
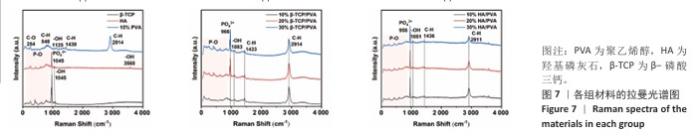
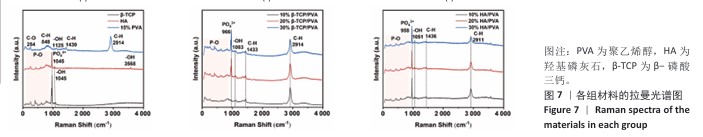
2.4 各组材料的拉曼光谱分析结果 各组材料的拉曼光谱图,见图7。在200-900 cm-1处出现P-O的弯曲振动峰,960 cm-1和924 cm-1处为P-O的伸缩振动峰,这些峰表示了磷酸根离子与氧原子之间的化学键的振动;3 568 cm-1和1 045 cm-1处出现-OH的伸缩振动峰,254 cm-1处的峰是聚乙烯醇中C-O的伸缩振动,848 cm-1处的峰对应于聚乙烯醇中C-H的伸缩振动,1 125 cm-1处的峰是-OH的弯曲振动,1 430 cm-1处的峰代表聚乙烯醇中CH2基团C-H的变形振动,2 914 cm-1处的峰对应于聚乙烯醇中CH2基团C-H的伸缩振动。对比单纯的羟基磷灰石、β-磷酸三钙、聚乙烯醇,各组复合材料的弯曲振动峰、伸缩振动峰均无改变,可以再次推断复合材料成功制备。"
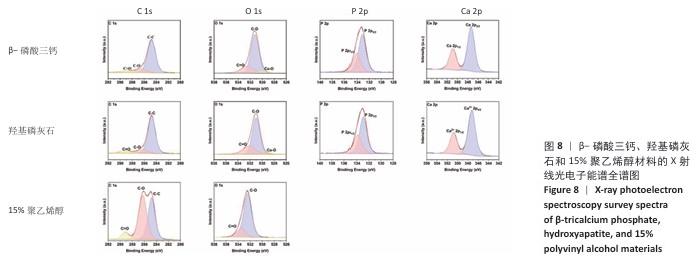
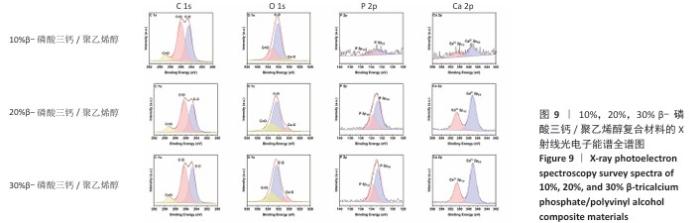
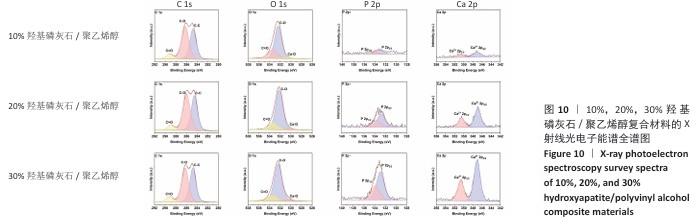
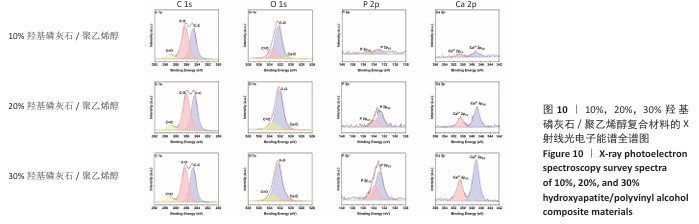
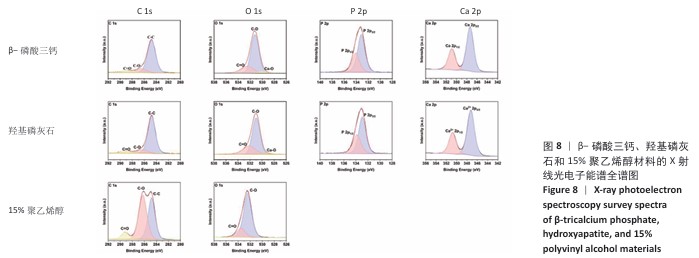
2.5 各组材料的X射线光电子能谱分析结果 各组材料的X射线光电子能谱全谱图,见图8-10。在β-磷酸三钙/聚乙烯醇复合材料的X射线光电子能谱C1s、Ca 2p、O1s和P 2p谱图中,可见明显的β-磷酸三钙和聚乙烯醇特征峰,并且随着β-磷酸三钙颗粒浓度的增加,Ca和P元素的强度明显增加,说明10%,20%,30%β-磷酸三钙/聚乙烯醇复合材料制备成功。在羟基磷灰石/聚乙烯醇复合材料的X射线光电子能谱C1s、Ca 2p、O1s和P 2p谱图中,可见明显的羟基磷灰石和聚乙烯醇特征峰,随着羟基磷灰石掺杂量的增加,Ca和P元素的强度明显增加,说明10%,20%,30%羟基磷灰石/聚乙烯醇复合材料制备成功。在X射线光电子能谱谱图中并未出现新的化学键,表明β-磷酸三钙、羟基磷灰石及聚乙烯醇物质相互之间化学状态相对稳定。"
| [1] MARIAN D, TORO G, D’AMICO G, et al. Challenges and Innovations in Alveolar Bone Regeneration: A Narrative Review on Materials, Techniques, Clinical Outcomes, and Future Directions. Medicina (Kaunas). 2024;61(1):20. [2] XU Z, WANG J, GAO L, et al. Hydrogels in Alveolar Bone Regeneration. ACS Biomater Sci Eng. 2024;10(12):7337-7351. [3] SHEKARIAN M, AGHAJANI F, HEJAZI ZSM, et al. Tooth Graft and Platelet-Rich Fibrin Mixture for Oral Bone Reconstruction and Preservation: A Scoping Review. Clin Exp Dent Res. 2025;11(4):e70160. [4] LI L, XU J, YE C, et al. Biomaterials-based strategy for dental-oral tissue regeneration: current clinical application, laboratory development, and future direction. Biomaterials. 2025;326:123714. [5] CHEN Y, ZHANG X, TANG Y, et al. The effects of FOXC2-gene-manipulated human periodontal ligament stem cells on bone regeneration of craniofacial bone defect. Dent Mater. 2025;41(6):708-720. [6] HUANG K, LI Q, LIU Y, et al. 3D-printed functionalized strontium-silk fibroin-hydroxyapatite scaffolds facilitate bone regeneration via immunomodulatory and sequential angiogenic-osteogenic coupling. Bioact Mater. 2026;55:271-289. [7] WANG FZ, LIU S, GAO M, et al. 3D-Printed Polycaprolactone/Hydroxyapatite Bionic Scaffold for Bone Regeneration. Polymers(Basel). 2025;17(7):858. [8] MANDATORI D, D’AMICO E, ROMASCO T, et al. A 3D in vitro model of biphasic calcium phosphate (BCP) scaffold combined with human osteoblasts, osteoclasts, and endothelial cells as a platform to mimic the oral microenvironment for tissue regeneration. J Dent. 2024;151:105411. [9] DU Y, LIU T, DING T, et al. Adhesive lipophilic gels delivering rapamycin prevent oral leukoplakia from malignant transformation. Mater Today Bio. 2024;29: 101305. [10] HUANG Q, HAN L, WANG R, et al. Electrospun Nanofibers Loaded with Concentrated Growth Factors and Nanohydroxyapatite for the Healing of Alveolar Bone in Tooth Extraction Wounds. ACS Biomater Sci Eng. 2025;11(10):5975-5990. [11] AMER AA, KARKAR Y, BINGLE L, et al. Fast-Disintegrating Oral Films Containing Nisin-Loaded Niosomes. Molecules. 2025;30(18):3715. [12] MASLII Y, HERBINA N, DENE L, et al. Mucoadhesive polymeric film with plant-based compounds for dental applications: formulation, characterization and evaluation. Pharm Dev Technol. 2025;30(4):505-520. [13] DOŁOWACKA-JÓŹWIAK A, NAWROT-HADZIK I, MATKOWSKI A, et al. Mucoadhesive PVA Film for Sustained Resveratrol Delivery: Formulation, Characterization, and Release Profile. Molecules. 2025;30(12):2642. [14] ECKERMANN C, KLEIN CJ, SCHÄFER F, et al. Probiotics-embedded polymer films for oral health: Development, characterization, and therapeutic potential. Colloids Surf B Biointerfaces. 2025;255:114886. [15] DONGWEN L, DAPENG M, JIAZHI Y, et al. Hydrogels in Oral Disease Management: A Review of Innovations in Drug Delivery and Tissue Regeneration. Med Sci Monit. 2025;31:e946122. [16] CETIN GENC C, YILMAZ-DAGDEVIREN HD, DENIZ Y, et al. Unveiling Bone and Dental Regeneration Potential of Quince Seed Mucilage-Nanohydroxyapatite Scaffolds in Rabbit Mandibles. J Biomed Mater Res B Appl Biomater. 2025; 113(4):e35570. [17] KHOSRONEJAD A, ARABION H, IRAJI A, et al. Mandibular bone defect healing using polylactic acid-nano-hydroxyapatite-gelatin scaffold loaded with hesperidin and dental pulp stem cells in rat. Tissue Cell. 2025;93:102700. [18] FISCHER NG, DE SOUZA ARAÚJO IJ, DAGHRERY A, et al. Guidance on biomaterials for periodontal tissue regeneration: Fabrication methods, materials and biological considerations. Dent Mater. 2025;41(3):283-305. [19] ZHANG X, CONTESSI NEGRINI N, CORREIA R, et al. Generating Tooth Organoids Using Defined Bioorthogonally Cross-Linked Hydrogels. ACS Macro Lett. 2024; 13(12):1620-1626. [20] ZHANG Q, HE J, ZHU D, et al. Genetically modified organoids for tissue engineering and regenerative medicine. Adv Colloid Interfac. 2025;335:103337. [21] ABEDI N, SADEGHIAN A, KOUHI M, et al. Immunomodulation in Bone Tissue Engineering: Recent Advancements in Scaffold Design and Biological Modifications for Enhanced Regeneration. Adv Colloid Interfac. 2025;11(3): 1269-1290. [22] ZHAO X, YAO M, WANG Y, et al. Neuroregulation during Bone Formation and Regeneration: Mechanisms and Strategies. ACS Appl Mater Interfaces. 2025; 17(5):7223-7250. [23] WANG J, CHEN X, LI R, et al. Standardization and consensus in the development and application of bone organoids. Theranostics. 2025;15(2):682-706. [24] 丁刘闯,赵小琦,韩祥祯,等. 3D打印PVA/nHA支架与SF/PVA/nHA支架的性能比较[J].口腔医学,2018,38(7):598-602. [25] 周琦琪,韩祥祯,宋艳艳,等. 3D打印羊椎骨粉/聚乙烯醇支架、纳米级羟基磷灰石/聚乙烯醇支架、羊椎骨粉/聚乙烯醇无孔骨板的性能比较[J]. 中国组织工程研究,2016,20(52):7851-7857. [26] ZHU L, DU X, FU G, et al. Efficacy of different forms of concentrated growth factors combined with deproteinized bovine bone minerals in guided bone regeneration: a randomized clinical trial. BMC Oral Health. 2025;25(1):320. [27] SHEN Z, ZHU YW, WEI YW, et al. Enhanced Osteogenic Activity of a Titanium Mesh Modified with Magnesium-Doped Nanowires for Peri-Implant Guided Bone Regeneration: In Vitro and In Vivo. ACS Biomater Sci Eng. 2025;11(5):2664-2676. [28] YIN Z, CHEN Y, YANG G, et al. 3D Printed HA/β-TCP Scaffold: A Macroscopic Microscopic Analysis and Biological Validation Study of the Effect of the Component Ratio on Performance. ACS Appl Bio Mater. 2025;8(11):9807-9823. [29] AYDIN A, ULAG S, NOURI S, et al. Production of Polyvinyl Alcohol/Amoxicillin - Chitosan/Collagen Hybrid Bilayer Membranes for Regeneration of Gingival Tissues. Macromol Biosci. 2025;25(1):e2400331. [30] BARIK B, SATAPATHY BS, ACHARYA B, et al. Therapeutic Potential of AgNP-Infused Patches in Periodontal Disease: An Observational Study in Albino Rats. Int J Nanomed. 2025;20: 5337-5352. [31] JIANG K, LUO C, LI YM, et al. An immunomodulatory and osteogenic bacterial cellulose scaffold for bone regeneration via regulating the immune microenvironment. Int J Biol Macromol. 2024;281(Pt 3):136375. [32] KHADDOUR AS, DRĂGHICI EC, IONESCU M, et al. Bone Regeneration in Defects Created on Rat Calvaria Grafted with Porcine Xenograft and Synthetic Hydroxyapatite Reinforced with Titanium Particles-A Microscopic and Histological Study. J Funct Biomater. 2025;16(4):146. [33] DE MORAES MARCONDES G, PARETSIS NF, DA SILVA DCB, et al. Bone Tissue Engineering With Chitosan, Carbon Nanotubes, and Hydroxyapatite Biomaterials Enriched With Mesenchymal Stem Cells: A Radiographic and Histological Evaluation in a Sheep Model Undergoing Ostectomy (Bone Tissue Engineering in a Sheep Model). J Appl Biomater Biom. 2025;113(1):e35523. [34] WANG Z, SHANG J, ZHANG Z. Composite or Modified Hydroxyapatite Microspheres as Drug Delivery Carrier for Bone and Tooth Tissue Engineering. Curr Med Chem. 2025;32(5):974-981. [35] YANG W, ZOU Q, WANG C, et al. Enhancing Bone Regeneration and Osteogenic Quality by n-HA Internalized Osteoblasts Synergized with ON Protein: Mechanistic Insights. ACS Appl Mater Interfaces. 2024;16(50):68967-68982. [36] XIANG M, ZHANG G, LIU Y, et al. Polydopamine-functionalized nanohydroxyapatite coated exosomes with enhanced cytocompatibility and osteogenesis for bone regeneration. Biomed Mater Eng. 2025;36(2):98-109. [37] QIU W, ZHANG K, WU M, et al. Tri-Layer Citrate-Based Hydroxyapatite Composite Scaffold Promoting Osteogenesis and Gingival Tissue Regeneration for Periodontal Bone Defect Repair. Adv Healthc Mater. 2025;14(13):e2501002. [38] PITOL-PALIN L, MOURA J, FRIGÉRIO PB, et al. A preliminary study of cell-based bone tissue engineering into 3D-printed β-tricalcium phosphate scaffolds and polydioxanone membranes. Sci Rep. 2024;14(1):31184. [39] VALDEC S, BOSSHARD FA, PATRIZI A, et al. Beta-Tricalcium Phosphate (β-TCP) for Customized Bone Regeneration (CBR) in the Aesthetic Area of the Maxillary Anterior Alveolar Ridge. Int J Oral Maxillofac Implants. 2025:1-18. doi: 10.11607/jomi.11352. [40] ARORA V, JOSHI A, ANTHWAL N, et al. Bone regeneration using PRF, PRP and β-TCP in bone defects. Bioinformation. 2025;21(4):793-796. [41] YU F, ZHAO X, ZHANG S, et al. Regulation of T Cell Glycosylation by MXene/β-TCP Nanocomposite for Enhanced Mandibular Bone Regeneration. Adv Healthc Mater. 2025;14(6):e2404015. [42] PONGSETTAKUL A, LEEPONG N, SUTTAPREYASRI S. Repairing Dehiscence Defects at Implant Sites Using β-Tricalcium Phosphate/Calcium Sulfate Versus Xenograft Combined With Membrane: A Randomized Clinical Trial. J Oral Implantol. 2025; 51(1):6-13. [43] CHAD MAB, CENCI EDS, ORLANDINI RK, et al. Socket preservation following tooth extraction: an experimental study comparing β-Tricalcium phosphate and F18 bioglass in rats. J Oral Maxil Surg. 2024;29(1):16. [44] WEI L, SUN Y, YU D, et al. The Clinical Efficacy and Safety of ErhBMP-2/BioCaP/β-TCP as a Novel Bone Substitute Using the Tooth-Extraction-Socket-Healing Model: A Proof-of-Concept Randomized Controlled Trial. J Clin Periodontol. 2025;52(2):299-309. [45] SHI A, SHI Y, LI J, et al. Advancements in 3D gel culture systems for enhanced angiogenesis in bone tissue engineering. J mater Chem B. 2025;13(11): 3516-3527. [46] WU B, LI X, WANG R, et al. Biomimetic Mineralized Collagen Scaffolds for Bone Tissue Engineering: Strategies on Elaborate Fabrication for Bioactivity Improvement. Small. 2025;21(3):e2406441. [47] VILLANI C, MURUGAN P, GEORGE A. Exosome-Laden Hydrogels as Promising Carriers for Oral and Bone Tissue Engineering: Insight into Cell-Free Drug Delivery. Int J Mol Sci. 2024;25(20):11092. [48] PEI Z, XU H, GUO M, et al. A soft-hard hybrid scaffold for osteochondral regeneration through integration of composite hydrogel and biodegradable magnesium. Biomaterials. 2026;324:123493. |
| [1] | Shao Ziyu, Li Qian, Qumanguli·Abudukelimu, Han Youjun, Hu Yang. Preparation and characterization properties of three different ratios of biphasic calcium phosphate [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1952-1961. |
| [2] | Zhang Huifang, Li Hankai, Huang Haozhe, Liu Min, Wang Pin, Huang Haixia, Sun Libo, Lan Yuyan. Osteogenic and antibacterial properties of polyether ether ketone modified by multifunctional composite coating [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6719-6728. |
| [3] | Zheng Hao, Zhou Tianqi, Pan Jiazhaо, He Jialin, Zou Zihao, Teng Jianxiang, Xie Mengli, Yang Long, Tian Xiaobin. Sandwich-like nanofiber membrane loaded with salidroside regulates macrophage polarization and promotes angiogenesis in diabetic wounds [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5152-5166. |
| [4] | Zhan Lei, Wu Lina, Li Huan, Liu Min, Chen Tao, Pu Xiaobing, Zhou Changchun. Silk fibroin hydrogel loaded with icariin to promote tendon-bone healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5178-5787. |
| [5] | Li Congcong, Wufanbieke·Baheti, Zhao Li, Chen Xiaotao, Kong Chuifan, Yu Min. Physicochemical properties and biocompatibility of hydroxyapatite/graphene oxide/interleukin-4 composite coating materials [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(2): 404-413. |
| [6] | Yuan Qian, Zhang Hao, Pang Jie. Characterization and biological properties of naringin-loaded chitosan/beta-tricalcium phosphate scaffold [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(2): 424-432. |
| [7] | Li Qian, Qumanguli · Abudukelimu, Shao Ziyu, Hu Yang. Hard template construction of nano-beta-tricalcium phosphate and nano-hydroxyapatite root canal sealing materials [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(14): 3597-3608. |
| [8] | Liang Jiyao, Zhou Honghai, Wei Guikang, Su Shaoting, Chen Longhao, He Xinyu, Liu Liangpu. Quantification of in vivo biomechanics and analysis of influencing factors in cervical spine fixed-point rotation manipulation [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(3): 486-492. |
| [9] | Yang Xiaoxiao, Xu Yuanjing, Li Wentao, Wang Wenhao, Ma Zhenjiang, Wang Jinwu. Experimental study on treatment of Achilles tendinitis with ultrasound-guided phacoemulsification [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||