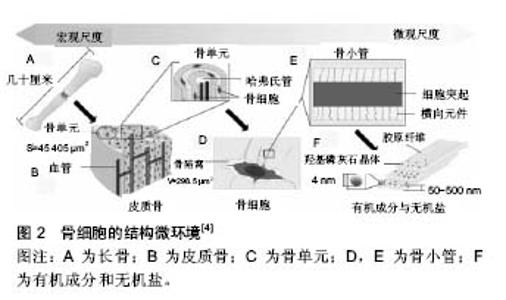
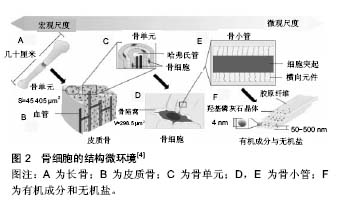
Chinese Journal of Tissue Engineering Research ›› 2017, Vol. 21 ›› Issue (7): 1137-1142.doi: 10.3969/j.issn.2095-4344.2017.07.027
Previous Articles Next Articles
Research progress of the influence of alcohol on the local microenvironment of femoral head
Xi Li-cheng1, Li Hong-yu2
- 1Guangxi University of Chinese Medicine, Nanning 530021, Guangxi Zhuang Autonomous Region, China; 2Department of Orthopedics, the People’s Hospital of Guangxi Zhuang Autonomous Region, Nanning 530021, Guangxi Zhuang Autonomous Region, China
-
Revised:2016-12-10Online:2017-03-08Published:2017-04-11 -
Contact:Li Hong-yu, M.D., Chief physician, Master’s supervisor, Department of Orthopedics, the People’s Hospital of Guangxi Zhuang Autonomous Region, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
About author:Xi Li-cheng, Studying for master’s degree, Guangxi University of Chinese Medicine, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
Supported by:the Graduate Research Innovation Program, No. YJS201651; the Natural Science Foundation of Guangxi Zhuang Autonomous Region in 2014, No. 2014jjAA40654
CLC Number:
Cite this article
Xi Li-cheng, Li Hong-yu. Research progress of the influence of alcohol on the local microenvironment of femoral head[J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1137-1142.
share this article

松质骨由交织排列的骨小梁构成,骨小梁由于应力分布疏密的不同而具有对抗来自不同方向的压应力作用,骨小梁力学强度变化与矿物质含量及其三维空间构型构成了股骨头内部骨组织承重的主要生物力学结构[7]。 2.1.2 酒精对股骨头力学和骨微结构的影响 生物力学因素在股骨头坏死的发生、发展和转归过程中具有重要作用。研究表明,长时间饮酒或者局部注射酒精能显著降低股骨头内骨组织的矿物质密度[8],改变骨小梁的三维空间结构,在局部注射酒精3周后,股骨头的骨矿密度、骨小梁数目、骨小梁间隙和厚度均显著下降。酒精致股骨头坏死后,股骨头内的松质骨弹性模量、松质骨平均应变量和压缩强度均显著下降。坏死骨质的吸收将使骨小梁的力学强度下降,而持续的应力作用将阻碍坏死后骨组织的修复,使坏死骨小梁的力学强度进一步降低,生物力学结构的失衡是导致股骨头坏死后塌陷的重要原因之一[7,9]。 此外,当酒精致股骨头局部发生坏死后,股骨头坏死区毗邻的正常骨组织结构也存在吸收、修复、结构重塑等生物学反应,骨的生物力学性能也随之降低。 酒精致股骨头发生坏死后,骨单位结构发生改变,骨陷窝数量增加,在坏死的修复期,活骨与坏死骨组织之间产生应力集中,这种应力集中效应导致骨小梁呈现微骨折,削弱了股骨头的力学强度[8]。 研究表明,大量的骨小梁组织呈拱结构形状[10],股骨头之所以能承受很高的载荷与骨小梁构成的拱形结构有密切关系,股骨头骨小粱的拱结构及其胶原纤维和矿质结晶的排列特征都为股骨头承受高载荷提供了有利条件。长期饮酒的患者,成骨细胞和骨细胞发生坏死,这种拱形结构发生改变,原产生的拱体所受的弯矩增加,承受更多的非轴向压力,而使得股骨头坏死区承载能力下降,进一步扩大坏死的范围,髋关节接触应力也随之明显增加[11]。股骨头发生坏死后,日常髋部的运动功能也将导致病理状态下的股骨头持续承受分布不均的应力作用,更容易发生应力骨折,最终导致股骨头塌陷和骨性关节炎的发生。 2.2 成骨微环境 2.2.1 骨髓间充质干细胞的成骨分化作用 骨髓间充质干细胞是一种具有多向分化潜能和稳定的自我更新能力的干细胞,可分化为脂肪细胞、成骨细胞和血管内皮细胞等。适宜的刺激能激活体内成骨微环境中的细胞信号传导系统,促进骨髓间充质干细胞的成骨分化,加速新骨的形成和骨坏死的修复。 2.2.2 酒精对骨髓间充质干细胞成骨分化的影响 研究表明,酒精性股骨头坏死的发生与发展与骨髓间充质干细胞的成骨分化功能密切相关[12-13]。PPARγ2是一种在骨髓间充质干细胞分化中起重要调控作用的成脂分化因子,它可抑制成骨过程中的核心结合因子cbfαl的表达,从而抑制成骨细胞特有基因的表达[14]。酒精通过上调过氧化酶基因的表达激活PPARγ2,使得骨髓间充质干细胞成脂分化能力显著增强,而向成骨细胞转化的能力受到明显抑制,其摄取营养物质的能力也下降[13]。 长期饮酒致股骨头坏死的患者中,股骨头内骨髓间充质干细胞数量减少,细胞活性下降,成骨分化明显减弱,而破骨细胞的活性却增强,骨形成与骨吸收的平衡遭到破坏[13]。同时,当股骨头骨髓内骨髓间充质干细胞成脂能力明显增强时,造成骨髓内脂肪组织堆积,引起骨内压增高,血流减少。骨髓间充质干细胞成骨成脂分化异常使得骨再生障碍,骨内微环境发生病理性改变,最终出现股骨头的坏死与塌陷[15]。此外,酒精除了影响骨髓间充质干细胞的成骨成脂分化作用外,对骨髓间充质干细胞向血管内皮细胞分化也具有一定的调节作用。 2.3 股骨头的微循环 2.3.1 股骨头的血运结构及特点 股骨头的血液循环不同于骨干部位的血供,其头下区域多无双重血液供应。在股骨头的骨髓松质骨内,上升至关节面软骨下的动脉已成为功能性终末动脉,因此,位于终末动脉区及其远端的骨组织容易发生血液循环不足。综上所述,由于股骨头区血液循环解剖结构的特殊性,其血供代偿能力远不如骨干部,因而容易发生缺血性坏死。此外,股骨头内为固定容积室,股骨头髓内的病变容易阻塞回流或导致血流阻断从而加重股骨头的缺血状态[10-11,16]。 2.3.2 酒精对股骨头微循环的影响 酒精性股骨头坏死的发病机制目前有多种学说,但其本质均为股骨头骨组织的血液循环障碍,骨的营养物质和氧供减少,骨组织、细胞长期缺氧而发生变性、坏死。研究表明,酒精的作用能使脂肪细胞增殖、肥大[10-11,16],而股骨头内为密闭的固定容积室,无法扩张,脂肪细胞的增殖和肥大使得股骨头内骨内压增高,骨内压持续增加后股骨头内微血管受压,微循环受到严重阻碍。 在酒精及代谢产物的长期毒性刺激作用下,骨髓内脂肪细胞发生代谢紊乱和形态学改变,终末动脉被栓塞,血管内皮细胞受到损害,前列腺素E2(PGE2)和血栓素A2(TXA2)升高引起局部血小板聚集,并导致早期动脉血管内凝血[17]。 同时,大剂量酒精摄入可引起氧化-抗氧化系统失衡,缺血再灌注性损伤产生多量氧化作用,使血管壁溶解,导致潜在微灶性出血。研究表明,长期大量饮酒后,酒精在体内代谢过程中产生的乙醛可使氧自由基增 多[18],并使氧自由基主要的清除物超氧化物歧化酶活性下降。氧自由基具有强烈的引发脂质过氧化作用,使乳过氧化物酶的反应增强,膜受体、蛋白酶和离子通道等功能发生障碍。而细胞膜和亚细胞器是过氧化脂质的重要损伤部位,过氧化脂质通过影响细胞膜的通透性最终损伤血管内皮和毛细血管壁,增加毛细血管的通透性,严重影响股骨头局部血运环境[19-20]。 研究表明,酒精的刺激使得微血管内脂质沉积[21-22],脂肪栓子水解产生的游离脂肪酸、血管内毒素及高凝血状态等因素都可以触发血管内凝血,由于血管内凝血消耗了大量凝血因子,而引起反复性出血并继发纤维蛋白溶解,导致血管内皮细胞坏死及内皮下胶原暴露,促进小动脉痉挛,血液粘滞度增加,微血栓形成,微血栓使得股骨头内微小血管堵塞,股骨头血供减少。此外,缺血再灌注损伤引发过量氧化反应,引起内皮细胞膜的损伤,进而导致微小血管破裂,缺血进一步加重,最终出现股骨头缺血、坏死及塌陷[23]。也有人认为,在慢性酒精中毒时,个体敏感者会发生局部血管炎,进而导致血管壁硬化,血管壁硬化可能是股骨头缺血坏死的重要原因之一,在血液流变学异常的情况下也会形成血栓[23-24]。 2.4 酒精对股骨头免疫与炎性微环境的影响 炎症因子广泛存在于人体各种组织中,它们可从分子水平对多种疾病的发生发展和修复过程起着重要的生物调节作用。Okazak等[25]在研究酒精性股骨头坏死时,发现酒精通过LPS相关通路能诱导白细胞介素1、白细胞介素2、白细胞介素4、白细胞介素6、粒细胞集落刺激生物因子、干扰素-γ和肿瘤坏死因子α等炎性因子的分泌,从而导致股骨头内骨细胞的破坏。另有研究表明,白细胞介素4、白细胞介素5、白细胞介素12、白细胞介素13及白细胞介素18等炎性细胞因子能影响RANKL的表达[26-27],RANKL/RANK系统是联系免疫系统与骨代谢的桥梁,在骨重建与骨塑形中通过影响成骨细胞和破骨细胞的增殖直接调控骨结构,该调节系统也与免疫细胞和免疫细胞分泌的细胞因子联系密切,RANKL/RANK系统还可直接或间接作用于骨分化,而该作用的最终结果与免疫细胞亚群、细胞因子和局部微环境等因素有关。除了成骨相关作用以外,RANKL/RANK系统还可促进破骨细胞分化及成熟,抑制破骨细胞的凋亡,从而加快骨质的破坏,加速股骨头坏死的进展和骨塌陷的发生。 同样有研究证明,酒精可通过Toll样受体4(TLR4)通路引起系统性的炎症和血管炎症反应[28-29],产生骨细胞和血管内皮细胞的细胞毒性和细胞损伤反应,从而引发血管内凝血、血管壁的增厚与毛细血管数量的减少,最终导致微循环的障碍。在血管内凝血过程中,纤维蛋白和纤维蛋白原的降解产物可诱导单核细胞分泌炎性细胞因子,如肿瘤坏死因子α、白细胞介素1,这些因子都具有骨细胞毒性作用,能直接诱导骨细胞的坏死和凋亡[30]。因此,股骨头内炎性微环境的紊乱也是导致股骨头坏死进展的重要因素之一。 此外,在股骨头松质骨坏死修复区中可见浆细胞、淋巴细胞的浸润,以往认为,T淋巴细胞在非创伤性股骨头坏死与修复过程中起重要作用;近来发现,B淋巴细胞和中性粒细胞在股骨头坏死的免疫炎性反应进程中也具有重要调控作用[31-32]。骨髓中的滤泡辅助性T细胞(Tfh)的表面表达趋化因子受体5,趋化因子受体5可诱导程序性死亡因子和辅助刺激因子的表达,并以分泌大量白细胞介素21为特征[33],白细胞介素21具有诱导活化B细胞的作用,Tfh-B细胞相互作用最终导致患者股骨头内局部免疫失衡[32]。但免疫与炎性环境对股骨坏死的具体作用机制及作用程度仍有待于进一步研究。 2.5 氧微环境的改变 2.5.1 酒精对股骨头氧环境的影响 长期饮酒的患者血液处于一种高凝状态,再加之酒精对血管的细胞毒性反应,局部毛细血管壁增厚、微血管数量减少,使得局部代谢障碍,运输氧的血红蛋白等物质减少,氧含量降低[34-35]。细胞内及细胞外环境氧分压降低使细胞有氧呼吸作用减弱,无氧呼吸增强,胞内酸性代谢产物堆积,局部组织细胞氧供障碍进一步加重,形成恶性循环。 2.5.2 氧环境改变加速股骨头坏死 研究表明,局部氧环境对骨再生与成骨分化具有重要意义[36-37],短期且适当的缺氧可促进成骨细胞的功能,而长期缺氧则抑制成骨细胞的增殖、分化和矿化,并促进其凋亡,同时抑制骨髓间充质干细胞的成骨功能,并且随着缺氧时间的延长,成骨细胞增殖能力逐渐下降,同时成骨细胞表型标志物骨源性碱性磷酸酶(NBAP)活性及矿化结节形成数量明显降低,从而导致骨形成受阻。有研究表明,骨髓间充质干细胞对低氧应急表现出“冬眠现象”[38],1%低氧张力下骨髓间充质干细胞的增殖迅速下降并停滞于G1期,1%氧环境还可严重抑制骨髓间充质干细胞的成脂和成骨分化,而当氧张力升至3%时,细胞可重获成骨分化能力[39]。Salim等[40-41]观察到低于0.02%的氧张力在体外可抑制细胞外钙沉积和成骨前体细胞骨结节的形成,并且认为低氧能下调成骨转录因子Runx2的表达,这种调控作用是低氧抑制成骨分化的基本机制。骨髓间充质干细胞成骨分化能力减弱,骨吸收与骨形成的平衡遭到破坏,最终加剧股骨头的坏死与骨结构塌陷。 2.6 骨内压的改变 2.6.1 酒精对股骨头骨内压的影响 骨内压(IOP)是指骨的血流动力在骨质间隙内或者骨腔内所产生的压力。过量饮酒后,体内脂质代谢紊乱,总胆固醇、磷脂、甘油三脂、游离脂肪酸等均明显升高,形成高脂血症。随着饮酒时间延长,骨髓内出现脂肪细胞肥大、增生,并融合成脂肪滴,逐渐压迫和取代红骨髓,并使髓内有限空间缩小,引起骨细胞的“占位性病变”,压迫小静脉,使受压处静脉管腔骤然变窄[10,16]。管腔形态忽窄忽宽,血流压力势必忽高忽低,血流通过管腔狭窄部时阻力增加,在管腔宽处很容易形成涡流,造成血小板沉积,血浆纤维蛋白原析出,容易形成血栓。一旦小静脉回流受阻,静脉压升高,必然导致动脉灌注受阻,而明显地降低毛细血管的血流速度,在毛细血管处同样易形成“湍流”,增大回流阻力。而股骨头是一个坚硬且不能膨胀的有限骨腔室,同骨骼肌相比,股骨头内缺少促进血液回流的动力,根据Starling原理,静脉回流一旦受阻,则静脉压升高并同时引起骨内压的明显升高,而骨内压的升高又进一步加重静脉回流受阻,形成一种恶性循环。 也有实验表明,在静脉回流障碍基本清除后,骨内压力仍明显高于正常[42],若仅依靠静脉回流障碍和血黏度增高学说理论似乎难以解释骨内持续高压的原因。因此,在骨内高压状态下,骨内血管对血管活性物质的敏感性下降,骨内血管的主动调节机制受损,可能是造成骨内持续高压的主要原因。 2.6.2 骨内压升高易致股骨头缺血性坏死 酒精性股骨头坏死目前发病机制不完全统一,陈跃平等[43]研究认为,长期饮酒导致骨内压增加、血流灌注减少是发生股骨头坏死的主要原因。正常的髋关节内压约为 5.37 kPa,如压力超过26.75 kPa极易发生股骨头缺血性坏死。骨内压力增加时,进一步压闭骨内薄壁小静脉、窦状隙及毛细血管,血管外周阻力增加,骨内静脉淤滞进一步加重,股骨头内血流量显著减少[10-11]。当压力超过平均动脉压时,将引起股骨头进行性缺血。股骨头局部营养物质和氧的供给量减少,有毒代谢产物不能及时运走,造成组织细胞缺氧和酸中毒,骨细胞及成骨细胞坏死,最终加速股骨头坏死的发生。"

| [1] Wang Y,Cao Y,Li Y,et al.Genetic association of the ApoB and ApoA1 gene polymorphisms with the risk for alcohol-induced osteonecrosis of femoral head. Int J Clin Exp Pathol. 2015;8(9):11332-11339.[2] Benalla M, Palacio-Mancheno PE, Fritton SP, et al. Dynamic permeability of the lacunar-canalicular system in human cortical bone.Biomech Model Mechanobiol. 2014;13(4):801-812.[3] D'Emic MD, Benson RB. Measurement, variation, and scaling of osteocyte lacunae: a case study in birds. Bone.2013;57(1): 300-310.[4] 任丽,续惠云,骞爱荣,等.骨细胞微环境仿生模拟技术[J].生物化学与生物物理进展, 2014, 41(11): 1126-1134.[5] Dallas SL, Prideaux M, Bonewald LF. The osteocyte: an endocrine cell ... and more. Endocr Rev.2013;34(5):658-690. [6] Bonewald LF. The amazing osteocyte. J Bone Miner Res. 2011; 26(2): 229-238.[7] Mao Q, Wang W, Xu T, et al. Combination treatment of biomechanical support and targeted intra-arterial infusion of peripheral blood stem cells mobilized by granulocyte-colony stimulating factor for the osteonecrosis of the femoral head: a randomized controlled clinical trial. J Bone Miner Res.2015;30(4):647-656. [8] Zhu ZH, Gao YS, Luo SH, et al. An animal model of femoral head osteonecrosis induced by a single injection of absolute alcohol: an experimental study. Med Sci Monit.2011;17(4):97-102.[9] Sakagoshi D, Kabata T, Umemoto Y, et al. A mechanical analysis of femoral resurfacing implantation for osteonecrosis of the femoral head. J Arthroplasty. 2010;25(8):1282-1289.[10] Stronach BM, Duke JN, Rozensweig SD, et al. Subtrochanteric femur fracture after core decompression and placement of a tantalum strut for osteonecrosis of the femoral head. J Arthroplasty.2010;25(7):1168.[11] 莫坚,卢培根,贾永龙,等.兔酒精性股骨头缺血性坏死模型构建及超微结构观察[J],广西医学,2014, 36(12):1865-1867.[12] Gangji V, Hauzeur JP. Treatment of osteonecrosis of the femoral head with implantation of autologous bone-marrow cells.Surgical technique. J Bone Joint Surg Am.2005;87 Suppl 1(Pt 1): 106-112. [13] Li J, Wang Y, Li Y, et al. The effect of combined regulation of the expression of peroxisome proliferator-activated receptor-γ and calcitonin gene-related peptide on alcohol-induced adipogenic differentiation of bone marrow mesenchymal stem cells. Mol Cell Biochem.2014;392(1-2):39-48.[14] Zhang Y, Ma C, Liu X, et al. Epigenetic landscape in PPARγ2 in the enhancement of adipogenesis of mouse osteoporotic bone marrow stromal cell. Biochim Biophys Acta.2015; 1852(11):2504-2516. [15] Feng Y,Yang SH,Xiao BJ,et al.Decreased in the number and function of circulation endothelial progenitor cells in patients with avascular necrosis of the femoral head. Bone.2010;46(1): 32-40. [16] Gupta AK, Frank RM, Harris JD, et al. Arthroscopic-assisted core decompression for osteonecrosis of the femoral head. Arthrosc Tech.2013;3(1):e7-e11.[17] 王洁.前列腺素E1对炎症性血管内皮细胞损伤的保护作用及其机制[J].中国老年学杂志,2012, 32(19):4179-4180.[18] Shi X, Li Y, Hu J, et al. Tert-butylhydroquinone attenuates the ethanol-induced apoptosis of and activates the Nrf2 antioxidant defense pathway in H9c2 cardiomyocytes. Int J Mol Med.2016;38(1):123-130.[19] Wang J, Zhang Y, Liu R, et al. Geniposide protects against acute alcohol-induced liver injury in mice via up-regulating the expression of the main antioxidant enzymes. Can J Physiol Pharmacol.2015;93(4):261-267. [20] 高蒙蒙,孙桂波,斯建勇,等.红车轴草总黄酮对H2O2诱导的血管内皮细胞损伤的保护作用[J]. 中国药理学通报,2015, 29(2): 201-207.[21] 王大伟,史宝明,张爽.构建酒精性股骨头坏死动物模型的理论依据及造模方法[J].中国组织工程研究与临床康复,2010, 14(50): 9413-9416.[22] Tanaka F, Kawai N, Sato M, et al. Effect of transcatheter arterial embolization with a mixture of n-butyl cyanoacrylate, lipiodol, and ethanol on the vascular wall: macroscopic and microscopic studies. Jpn J Radiol.2015;33(7):404-409. [23] Kang PD,Shen B,Yang J,et al.Platelet and endothelial cell-derived microparticles in steroid-induced osteonecrosis of the femoral head of rabbit model. Zhonghua Yi Xue Za Zhi. 2007;87(29):2045-2049.[24] Ding H, Gao YS, Hu C, et al. HIF-1α transgenic bone marrow cells can promote tissue repair in cases of corticosteroid- induced osteonecrosis of the femoral head in rabbits. PLoS One.2013;8(5):e63628.[25] Okazaki S,Nishitani Y,Nagoya S, et al.Femoral head osteonecro-sis can be caused by disruption of the systemic immune response via the toll-like receptor 4 signalling pathway. Rheumatology (Oxford).2009;48(3):227-232.[26] Freire MS,Cantuária AP,Lima SM,et al.NanoUPLC-MS(E) proteomic analysis of osteoclastogenesis downregulation by IL-4. J Proteomics.2016;131:8-16. [27] Horwood NJ, Elliott J, Martin TJ, et al.IL-12 alone and in synergy with IL-18 inhibits osteoclast formation in vitro. J Immunol, 2001, 166(8):4915-4921.[28] Agematsu K. Memory B-cells and CD27. Histol Histopathol. 2000;15(2): 573-576.[29] Donnadieu-Rigole H, Mura T, Portales P, et al. Effects of alcohol withdrawal on monocyte subset defects in chronic alcohol users.J Leukoc Biol.2016, pii: jlb.[30] Li J, Zhang FQ, Du ZN, et al. Protective effect of HO-1 transfection against ethanol-induced osteoblast damage. J Huazhong Univ Sci Technolog Med Sci.2015; 35(3):374-377. [31] Kaymaz B, Büyükdogan K, Kaymaz N, et al. Neutrophil to lymphocyte ratio may be a predictive marker of poor prognosis in Legg-Calvé-Perthes disease. Hip Int. 2016, 23:0.[32] 丁福鹏,戚长林,高 辉,等.外周血滤泡辅助性T细胞与B细胞在酒精性股骨头坏死患者中的作用[J].中国免疫学杂志, 2015, 31(8): 1109-1112.[33] Qi H, Chen X, Chu C, et al. Tfh cell differentiation and their function in promoting B-cell responses. Adv Exp Med Biol. 2014;841:153-180. [34] Rao PS, Kumar S. Chronic Effects of Ethanol and/or Darunavir/Ritonavir on U937 Monocytic Cells: Regulation of Cytochrome P450 and Antioxidant Enzymes, Oxidative Stress, and Cytotoxicity. Alcohol Clin Exp Res.2016; 40(1):73-82. [35] 齐振熙,喻灿明.酒精性股骨头缺血坏死模鼠的血流变及氧自由基代谢特征[J].福建中医学院学报, 2005, 15 (4):25-26.[36] Atashi F, Modarressi A, Pepper MS.The role of reactive oxygen species in mesenchymal stem cell adipogenic and osteogenic differentiation: a review. Stem Cells Dev.2015; 24(10):1150-1163.[37] Zhang ZM, Jiang LS, Jiang SD, et al. Osteogenic potential and responsiveness to leptin of mesenchymal stem cells between postmenopausal women with osteoarthritis and osteoporosis. J Orthop Res.2009;27(8):1067-1073.[38] 李宁,吴桂英,李启明,等.不同氧浓度微环境对大鼠骨髓间充质干细胞成骨及成脂肪分化的影响[J].重庆医学,2009, 38(19): 2448-2450.[39] Holzwarth C, Vaegler M, Gieseke F, et al. Low physiologic oxygen tensions reduce proliferation and differentiation of human multipotent mesenchymal stromal cells. BMC Cell Biol.2010, 11:11. [40] Amini AR, Xu TO, Chidambaram RM, et al. Oxygen Tension-Controlled Matrices with Osteogenic and Vasculogenic Cells for Vascularized Bone Regeneration In Vivo. Tissue Eng Part A.2016;22(7-8):610-620.[41] Salim A, Nacamuli RP, Morgan EF, et al. Transient changes in oxygen tension inhibit osteogenic differentiation and Runx2 expression in osteoblasts.J Biol Chem.2004;279(38): 40007-40016. [42] 陆洪英,金成文,王益光,等.骨内高压发生机制的实验研究[J].潍坊医学院学报,2001, 23(4):301.[43] 陈跃平,高辉,陈亮,等.乙醇对股骨头髓内脂肪细胞的作用[J].中国组织工程研究,2013, 17(35):6221-6227. |
| [1] | Shi Bin, An Jing, Chen Long-gang, Zhang Nan, Tian Ye . Influencing factors for pain after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 993-997. |
| [2] | Wang Xian-xun. Impact of local compression cryotherapy combined with continuous passive motion on the early functional recovery after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 998-1003. |
| [3] | Yuan Wei, Zhao Hui, Ding Zhe-ru, Wu Yu-li, Wu Hai-shan, Qian Qi-rong. Association between psychological resilience and acute mental disorders after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1015-1019. |
| [4] | Chen Qun-qun, Qiao Rong-qin, Duan Rui-qi, Hu Nian-hong, Li Zhao, Shao Min. Acu-Loc®2 volar distal radius bone plate system for repairing type C fracture of distal radius [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1025-1030. |
| [5] | Huang Xiang-wang, Liu Hong-zhe. A new low elastic modulus of beta titanium alloy Ti2448 spinal pedicle screw fixation affects thoracic stability: biomechanical analysis [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1031-1035. |
| [6] | Xie Qiang. Three-dimensional finite element model for biomechanical analysis of stress in knee inversion and external rotation after posterior cruciate ligament rupture [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1036-1040. |
| [7] | He Ze-dong, Zhao Jing, Chen Liang-yu, Li Ke, Weng Jie. Multilevel finite element analysis on the biological tribology damage of water on bone tissue [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1041-1045. |
| [8] | Jiang Zi-wei, Huang Feng, Cheng Si-yuan, Zheng Xiao-hui, Sun Shi-dong, Zhao Jing-tao, Cong Hai-chen,Sun Han-qiao, Dong Hang. Design and finite element analysis of digital splint [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1052-1056. |
| [9] | Wang Fei, Liu Zhi-bin, Tao Hui-ren, Zhang Jian-hua, Li Chang-hong, Cao Qiang, Zheng Jun, Liu Yan-xiong, Qu Xiao-peng. Clinical efficacy of preoperative osteotomy designs using paper-cut technology versus photoshop software for ankylosing spondylitis with kyphosis [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1057-1063. |
| [10] | Li Hui, Ma Jun-yi, Ma Yuan, Zhu Xu . Establishment of a three-dimensional finite element model of ankylosing spondylitis kyphosis [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1069-1073. |
| [11] | Ling Guan-han, Ou Zhi-xue, Yao Lan, Wen Li-chun, Wang Guo-xiang, Lin Heng-feng. Establishment of simulating three-dimensional model of China-Japan Friendship Hospital Classification for L type osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1074-1079. |
| [12] | Fu Wei-min, Wang Ben-jie. Assessing the degree of necrotic femoral head, and association of blood supply with pathlogical changes: study protocol for a diagnostic animal trial [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1086-1091. |
| [13] | Zhang Wen-qiang, Ding Qian, Zhang Na. Associations between alpha angle and herniation pit on oblique axial magnetic resonance imaging in asymptomatic hip joints of adults [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1098-1103. |
| [14] | Sun Xiao-xin1, Zhou Wei2, Zuo Shu-ping3, Liu Hao1, Song Jing-feng1, Liang Chun-yu1. Morphological characteristics for the magnetic resonance imaging assessment of discoid lateral meniscal tears in children [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1104-1109. |
| [15] | Lin Han-wen, Wen Jun-mao, Huang Chao-yuan, Zhou Chi, Tang Hong-yu. Correlation between the changes in lower limb power line and pain area in the knee osteoarthritis patients: imaging evaluation [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1110-1114. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||