Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (32): 5221-5227.doi: 10.3969/j.issn.2095-4344.2809
Previous Articles Next Articles
Recent advances and applications of Kartogenin in cartilage regeneration and repair
Lu Junxu, Wang Yuanbo, Hu Yanuan, Yang Biaobing
- Institute of Plastic Surgery, Weifang Medical University, Weifang 261000, Shandong Province, China
-
Received:2020-02-10Revised:2020-02-15Accepted:2020-03-04Online:2020-11-18Published:2020-09-26 -
Contact:Yang Biaobing, Master, Professor, Institute of Plastic Surgery, Weifang Medical University, Weifang 261000, Shandong Province, China -
About author:Lu Junxu, Master candidate, Institute of Plastic Surgery, Weifang Medical University, Weifang 261000, Shandong Province, China
CLC Number:
Cite this article
Lu Junxu, Wang Yuanbo, Hu Yanuan, Yang Biaobing. Recent advances and applications of Kartogenin in cartilage regeneration and repair[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(32): 5221-5227.
share this article
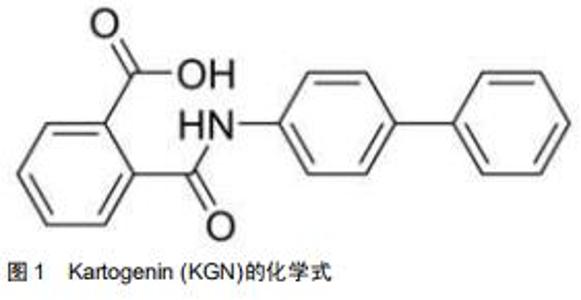
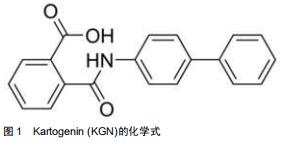
近年来,使用组织工程间充质干细胞定向分化的方法再生关节软骨已经成为了研究的热点。Kartogenin (KGN)是2012年由JOHNSON等[4]从22 000多种结构不同的杂环类药性分子中筛选出的一种能够促进人间充质干细胞成软骨分化的小分子见,图1。KGN在软骨的再生与保护、肌腱骨的愈合、创面的愈合、肢体的发育等再生领域有着广泛的应用。KGN可促进软骨修复,促进腱骨连接处软骨样过渡区形成,促进创面愈合所需的胶原合成,协调调节肢体发育。此外,依赖于这种小分子物质的比较稳定的特性,KGN可以在常温下存储和运输。自发现以来,KGN作为一种用于关节内的新型软骨生成的药物引起了广泛关注。尽管最初开发它的重点是作用与骨关节炎的治疗,但目前已被用于治疗其他疾病,包括促进椎间盘和骨腱连接的再生。文章总结了2010至2020年KGN应用于在软骨再生方面已取得的最新进展,综述了KGN分子作用的临床模型,并对其未来的前景进行了探讨和展望。 "
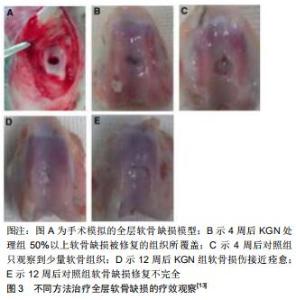
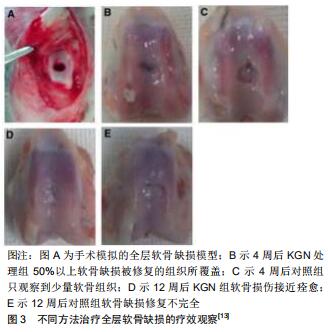
2.3.1 具有软骨再生与保护的效果 JOHNSON等[4]建立了慢性关节损伤及急性外科手术两种啮齿动物模型,并使用关节内注射KGN的方式对这两种模型进行治疗。骨关节炎患者血浆中的寡聚基质蛋白水平通常会升高[32],JOHNSON等[4]发现注射KGN后的慢性关节损伤模型中发现外周血内的寡聚基质蛋白的水平有所下降。在接受KGN治疗的急性外科手术模型组的关节组织学评分降低了50%(基于骨关节炎研究协会国际评分系统);另外,与对照组相比,接受KGN治疗显著缓解了骨关节炎引起的疼痛,循环中的Ⅱ型胶原片段(软骨损伤后释放的肽类物质)的水平也降低了1.8倍。这些实验均证明了KGN有促进软骨再生的作用。骨关节炎通过微骨折手术治疗可以缓解症状,但大多数新生的软骨组织会转变为纤维软骨,这种纤维软骨与透明软骨相比有所退化,且机械力学特性有明显的减弱[33],所以微骨折手术治疗的疗效仍不能令人满意。到目前为止,尚未有治疗方法来恢复退行性软骨的细胞表型。KGN除了增强软骨生成外,还可以保持软骨细胞表型。当KGN与微骨折手术疗法相结合时,可实现透明软骨的再生。XU等[13]诱发兔髌骨沟全层软骨缺损,并将其分为2组,分别采用不同的治疗方法:第1组行微骨折手术治疗加每周关节内注射KGN处理;第2组行微骨折治疗加二甲基亚砜注射液处理。4周后,第1组中50%以上的软骨缺损被修复的组织所覆盖,而第2组只观察到少量的纤维组织;12周后,第1组中的软骨损伤处几乎痊愈,并且有接近完整的软骨表面,见图3,第1组的国际软骨修复学会和O’Driscoll得分也比第2组更高,相比之下,第2组中的软骨损伤约有80%被裂隙状表面的纤维状组织覆盖;免疫组织化学染色显示,第1组产生了大量的Ⅱ型胶原和较少的Ⅰ型胶原,而第2组仅有少量的Ⅱ型胶原和较多的Ⅰ型胶原产生。该研究证明了微骨折手术治疗加关节内注射KGN治疗软骨损伤具有软骨再生和保护的作用。 "
|
[1] 邵加华,陈松,谢金硕,等. Kartogenin与软骨修复的研究进展[J]. 第二军医大学学报, 2016,37(4):471-476.
[2] TRICHE R, MANDELBAUM BR. Overview of cartilage biology and new trends in cartilage stimulation. Foot Ankle Clin. 2013; 18(1): 1-12.
[3] CHOI E, LEE J, LEE S, et al. Potential therapeutic application of small molecule with sulfonamide for chondrogenic differentiation and articular cartilage repair. Bioorg Med Chem Lett.2016;26(20):5098-5102.
[4] JOHNSON K, ZHU S, TREMBLAY MS, et al. A stem cell-based approach to cartilage repair. Science. 2012; 336(6082):717-721.
[5] KOU L, XIAO S, SUN R, et al. Biomaterial-engineered intra-articular drug delivery systems for osteoarthritis therapy.Drug Deliv. 2019; 26(1):870-885.
[6] SHERWOOD JC, BERTRAND J, ELDRIDGE SE, et al. Cellular and molecular mechanisms of cartilage damage and repair. Drug Discov Today. 2014;19(8):1172-1177.
[7] LIU C, MA X, LI T, et al. Kartogenin, transforming growth factor-β1 and bone morphogenetic protein-7 coordinately enhance lubricin accumulation in bone-derived mesenchymal stem cells. Cell BiolInt. 2015; 39(9):1026-1035.
[8] VALIANI A,IZADI MA,BAHRAMIAN H,et al.Comparison between the effect of kartogenin and TGFβ3 on chondrogenesis of human adipose- derived stem cells in fibrin scaffold.Bratisl Lek Listy. 2017;118:591-597.
[9] ONO Y, ISHIZUKA S, KNUDSON CB, et al. Chondroprotective effect of kartogenin on cd44-mediatedfunctions in articular cartilage and chondrocytes. Cartilage. 2014; 5(3):172-180.
[10] DECKER RS, KOYAMA E, ENOMOTO-IWAMOTO M, et al. Mouse limb skeletal growth and synovial joint development are coordinately enhanced by Kartogenin. Dev Biol. 2014; 395(2): 255-267.
[11] WANG Y, CHEN G, YAN J, et al. Upregulation of SIRT1 by kartogenin enhances antioxidant functions and promotes osteogenesis in human mesenchymal stem cells. Oxid Med Cell Longev. 2018;2018:1368142.
[12] WANG D, TAN H, LEBASCHI AH, et al. Kartogenin enhances collagen organization and mechanical strength of the repaired enthesis in a murine model of rotator cuff repair. Arthroscopy. 2018;34:2579-2587.
[13] XU X, SHI D, SHEN Y, et al. Full-thickness cartilage defects are repaired via a microfracture technique and intraarticular injection of the small-molecule compound kartogenin. Arthritis Res Ther. 2015;17(1) : 20.
[14] RAI V, DILISIO MF, DIETZ NE, et al. Recent strategies in cartilage repair: a systemic review of the scaffold development and tissue engineering. J Biomed Mater Res A. 2017;105(8): 2343-2354.
[15] LI X, DING J, ZHANG Z, et al. Kartogenin-incorporated thermogel supports stem cells for significant cartilage regeneration. ACS Appl Mater Interfaces. 2016;8(8):5148-5159.
[16] YIN H, WANG J, GU Z, et al. Evaluation of the potential of kartogenin encapsulated poly(L-lactic acid-co-caprolactone)/collagen nanofibers for tracheal cartilage regeneration. J Biomater Appl. 2017;32(3): 331-341.
[17] SHI D, XU X, YE Y, et al. Photo-cross-linked scaffold with kartogenin-encapsulated nanoparticles for cartilage regeneration. Acs Nano. 2016;10(1):1292-1299.
[18] KANG ML, JEONG SY, IM GI. Hyaluronic acid hydrogel functionalized with self-assembled micelles of amphiphilic pegylated kartogenin for the treatment of osteoarthritis. Tissue Eng Part A. 2017;23(13-14): 630-639.
[19] HU Q, DING B, YAN X, et al. Polyethylene glycol modified PAMAM dendrimer delivery of kartogenin to induce chondrogenic differentiation of mesenchymal stem cells. Nanomedicine. 2017;13(7):2189-2198.
[20] HUANG H. Editorial commentary: kartogenin promotes wounded enthesis regeneration. Arthroscopy. 2018;34(9):2588-2589.
[21] JING H, ZHANG X, GAO M, et al. Kartogenin preconditioning commits mesenchymal stem cells to a precartilaginous stage with enhanced chondrogenic potential by modulating JNK and β-catenin-related pathways. FASEB J. 2019;33:5641-5653.
[22] ZHOU Y, ZHANG J, YANG J, et al. Kartogenin with PRP promotes the formation of fibrocartilage zone in the tendon-bone interface. J Tissue Eng Regen Med. 2017;11(12):3445-3456.
[23] GRANADOS-MONTIEL J, CRUZ-LEMINI M, RANGEL-ESCAREÑO C, et al. SERPINA9 and SERPINB2: Novel Cartilage Lineage Differentiation Markers of Human Mesenchymal Stem Cells with Kartogenin.Cartilage. 2018:1947603518809403.
[24] KWON JY, LEE SH, NA HS, et al. Kartogenin inhibits pain behavior, chondrocyte inflammation, and attenuates osteoarthritis progression in mice through induction of IL-10.Sci Rep. 2018;8:13832.
[25] DORMAN LJ, TUCCI M, BENGHUZZI H. In vitro effects of bmp-2, bmp-7, and bmp-13 on proliferation and differentation of mouse mesenchymal stem cells. Biomed Sci Instrum. 2012;48(9): 81-87.
[26] PANDOLFI L, MINARDI S, TARABALLI F, et al. Composite microsphere-functionalized scaffold for the controlled release of small molecules in tissue engineering. J Tissue Eng. 2016;7: 2041731415624668.
[27] KANG ML, KO JY, KIM JE, et al. Intra-articular delivery of kartogenin-conjugated chitosan nano/microparticles for cartilage regeneration. Biomaterials. 2014;35(37):9984-9994.
[28] MASSARO M, BUSCEMI G, ARISTA L, et al. Multifunctional carrier based on halloysite/laponite hybrid hydrogel for kartogenin delivery. ACS Med Chem Lett. 2019;10:419-424.
[29] CAI JY, ZHANG L, CHEN J. Kartogenin and its application in regenerative medicine. Curr Med Sci. 2019;39:16-20.
[30] CHEN L, DONG SW, TAO X, et al. Autologous platelet-rich clot releasate stimulates proliferation and inhibits differentiation of adult rat tendon stem cells towards nontenocyte lineages. J Int Med Res. 2012; 40(4):1399-1409.
[31] LI J, LEE WY, WU T, et al. Near-infrared light-triggered release of small molecules for controlled differentiation and long-term tracking of stem cells in vivo using upconversion nanoparticles. Biomaterials. 2016; 110: 1-10.
[32] MARX RE. Platelet-rich plasma: evidence to support its use. J Oral Maxillofac Surg. 2004;62(4):489-496.
[33] ZHANG J, WANG JH. Kartogenin induces cartilage-like tissue formation in tendon-bone junction. Bone Res. 2014; 2(1):61-70.
[34] HIBINO N, HAMADA Y, SAIRYO K, et al. Callus formation during healing of the repaired tendon-bone junction. A rat experimental model. J Bone Joint Surg Br. 2007;89(11):1539-1544. [35] FAN W, LI J, YUAN L, et al. Intra-articular injection of kartogenin-conjugated polyurethane nanoparticles attenuates the progression of osteoarthritis. Drug Deliv 2018;25:1004-1012.
[36] CANAL F, SANCHIS J, VICENT MJ, et al. Polymer–drug conjugates as nano-sized medicines. Curr Opin Biotechnol. 2011;22(6): 894-900.
[37] WANG J, WANG Y, SUN X, et al. Biomimetic cartilage scaffold with orientated porous structure of two factors for cartilage repair of knee osteoarthritis. Artif Cells Nanomed Biotechnol. 2019;47:1710-1721.
[38] KANG ML, KIM JE, IM GI, et al. Thermo-responsive nanospheres with independent dual drug release profiles for the treatment of osteoarthritis. Acta Biomater. 2016;39:65-78.
[39] ZHANG J, YUAN T, ZHENG N, et al. The combined use of kartogenin and platelet-rich plasma promotes fibrocartilage formation in the wounded rat achilles tendon enthuses. Bone Joint Res. 2017;6(4): 231-244.
[40] 郑权,徐宏光,汪景,等.小分子药物kartogenin保护终板软骨退变的实验研究[J].中国临床药理学与治疗学,2014, 19(8): 846-850.
[41] HUANG Y, JIANG T, CHEN J, et al. Effects of kartogenin on the attenuated nucleus pulposus cell degeneration of intervertebral discs induced by interleukin-1β and tumor necrosis factor-α. Int J Mol Med. 2018;41:749-756.
[42] ZHU Y, TAN J, ZHU H, et al. Development of kartogenin-conjugated chitosan-hyaluronic acid hydrogel for nucleus pulposus regeneration. Biomater Sci. 2017;5:784-791.
[43] ZHANG J, XIA W, LIU P, et al. Chitosan modification and pharmaceutical/biomedical applications. Mar Drugs. 2010;8(7) : 1962-1987.
[44] KOU L, XIAO S, SUN R, et al. Biomaterial-engineered intra-articular drug delivery systems for osteoarthritis therapy. Drug Deliv. 2019; 26:870-885.
[45] SPAKOVA T, PLSIKOVA J, HARVANOVA D, et al. Influence of Kartogenin on Chondrogenic Differentiation of Human Bone Marrow-Derived MSCs in 2D Culture and in Co-Cultivation with OA Osteochondral Explant. Molecules. 2018; 23(1). pii: E181.
[46] MAUDENS P, SEEMAYER CA, THAUVIN C, et al. Nanocrystal-polymer particles: extended delivery carriers for osteoarthritis treatment. Small. 2018;14. doi: 10.1002/smll.201703108.
[47] LI H, JIANG J, WU Y, et al. Potential mechanisms of a periosteum patch as an effective and favourable approach to enhance tendon-bone healing in the human body. Int Orthop. 2012;3(3): 665-669.
[48] WONG MW, QIN L, TAI JK, et al. Engineered allogeneic chondrocyte pellet for reconstruction of fibrocartilage zone at bone-tendon junction-a preliminary histological observation. J Biomed Mater Res B Appl Biomater. 2004;70(2):362-367.
[49] ZHOU Q, ZHANG JH, YUAN S, et al. A new insight of kartogenin induced the mesenchymal stem cells (mscs) selectively differentiate into chondrocytes by activating the bone morphogenetic protein 7 (BMP-7)/Smad5 Pathway. Med Sci Monit. 2019;25:4960-4967.
[50] 王一帆,何帆. Kartogenin在制备提高骨髓间充质干细胞抗氧化能力药物的应用. 申请/专利号:CN201810071064.9,公开/公告号: CN108451940A.
[51] LIU F, XU H. A novel kartogenin-platelet-rich plasma gel enhances chondrogenesis of bone marrow mesenchymal stem cells in vitro and promotes wounded meniscus healing in vivo.Stem Cell Res Ther. 2019;10:201.
[52] BLIDDAL H, HENRIKSEN M. Osteoarthritis: Time to put steroid injections behind us? Nat Rev Rheumatol. 2017;13(9): 519-520.
[53] 丁伟,谭洪波.KGN修复软骨、促进腱-骨愈合作用的研究进展[J].山东医药,2017, 57(16):102-104.
[54] BISHOP J, KLEPPS S, LO IK, et al. Cuff integrity after arthroscopic versus open rotator cuff repair:arospective study. J Shoulder Elbow Surg. 2006;3(3):290-299. [55] MARINI JC, FORLINO A. Replenishing cartilage from endogenous stem cells. New Eng J Med. 2012;26(26):2522-2524. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Xu Feng, Kang Hui, Wei Tanjun, Xi Jintao. Biomechanical analysis of different fixation methods of pedicle screws for thoracolumbar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1313-1317. |
| [3] | Jiang Yong, Luo Yi, Ding Yongli, Zhou Yong, Min Li, Tang Fan, Zhang Wenli, Duan Hong, Tu Chongqi. Von Mises stress on the influence of pelvic stability by precise sacral resection and clinical validation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1318-1323. |
| [4] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [5] | Zhang Yu, Tian Shaoqi, Zeng Guobo, Hu Chuan. Risk factors for myocardial infarction following primary total joint arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1340-1345. |
| [6] | Wei Wei, Li Jian, Huang Linhai, Lan Mindong, Lu Xianwei, Huang Shaodong. Factors affecting fall fear in the first movement of elderly patients after total knee or hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1351-1355. |
| [7] | Wang Jinjun, Deng Zengfa, Liu Kang, He Zhiyong, Yu Xinping, Liang Jianji, Li Chen, Guo Zhouyang. Hemostatic effect and safety of intravenous drip of tranexamic acid combined with topical application of cocktail containing tranexamic acid in total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1356-1361. |
| [8] | Xiao Guoqing, Liu Xuanze, Yan Yuhao, Zhong Xihong. Influencing factors of knee flexion limitation after total knee arthroplasty with posterior stabilized prostheses [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1362-1367. |
| [9] | Peng Zhihao, Feng Zongquan, Zou Yonggen, Niu Guoqing, Wu Feng. Relationship of lower limb force line and the progression of lateral compartment arthritis after unicompartmental knee arthroplasty with mobile bearing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1368-1374. |
| [10] | Huang Zexiao, Yang Mei, Lin Shiwei, He Heyu. Correlation between the level of serum n-3 polyunsaturated fatty acids and quadriceps weakness in the early stage after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1375-1380. |
| [11] | Zhang Chong, Liu Zhiang, Yao Shuaihui, Gao Junsheng, Jiang Yan, Zhang Lu. Safety and effectiveness of topical application of tranexamic acid to reduce drainage of elderly femoral neck fractures after total hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1381-1386. |
| [12] | Wang Haiying, Lü Bing, Li Hui, Wang Shunyi. Posterior lumbar interbody fusion for degenerative lumbar spondylolisthesis: prediction of functional prognosis of patients based on spinopelvic parameters [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1393-1397. |
| [13] | Lü Zhen, Bai Jinzhu. A prospective study on the application of staged lumbar motion chain rehabilitation based on McKenzie’s technique after lumbar percutaneous transforaminal endoscopic discectomy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1398-1403. |
| [14] | Chen Xinmin, Li Wenbiao, Xiong Kaikai, Xiong Xiaoyan, Zheng Liqin, Li Musheng, Zheng Yongze, Lin Ziling. Type A3.3 femoral intertrochanteric fracture with augmented proximal femoral nail anti-rotation in the elderly: finite element analysis of the optimal amount of bone cement [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1404-1409. |
| [15] | Du Xiupeng, Yang Zhaohui. Effect of degree of initial deformity of impacted femoral neck fractures under 65 years of age on femoral neck shortening [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1410-1416. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||