Chinese Journal of Tissue Engineering Research ›› 2016, Vol. 20 ›› Issue (31): 4693-4699.doi: 10.3969/j.issn.2095-4344.2016.31.019
Previous Articles Next Articles
Application of finite element analysis in orthopedics: new theory and new progress
Ni Peng-hui1, Zhang Ying1, Yang Jing1, Xu Zi-ang1, NuErBoLi1, Cheng Kui2, Liu Da-peng1
- 1the Fifth Affiliated Hospital of Xinjiang Medical University, Urumqi 830000, Xinjiang Uygur Autonomous Region, China; 2Xinjiang Institute of Engineering, Urumqi 830000, Xinjiang Uygur Autonomous Region, China
-
Revised:2016-05-27Online:2016-07-22Published:2016-07-22 -
Contact:Liu Da-peng, M.D., Chief physician, the Fifth Affiliated Hospital of Xinjiang Medical University, Urumqi 830000, Xinjiang Uygur Autonomous Region, China -
About author:Ni Peng-hui, Studying for master’s degree, the Fifth Affiliated Hospital of Xinjiang Medical University, Urumqi 830000, Xinjiang Uygur Autonomous Region, China -
Supported by:the Natural Science Foundation of Xinjiang Uygur Autonomous Region, No. 2014211C121
CLC Number:
Cite this article
Ni Peng-hui, Zhang Ying, Yang Jing, Xu Zi-ang, NuErBoLi, Cheng Kui, Liu Da-peng. Application of finite element analysis in orthopedics: new theory and new progress[J]. Chinese Journal of Tissue Engineering Research, 2016, 20(31): 4693-4699.
share this article
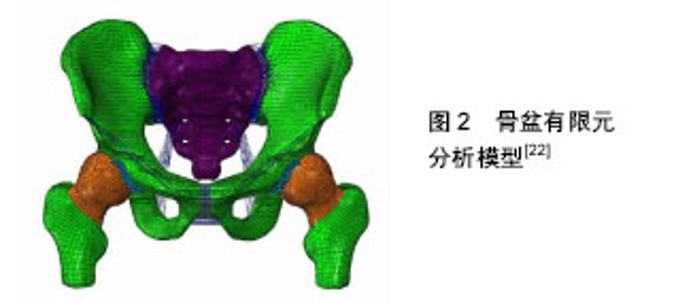
2.1 有限元分析模型的建立 2.1.1 几何模型的建立 由于人体骨骼形状和结构都非常复杂,所以建立准确的三维有限元分析模型就尤为重要,现在广泛被应用的是医学影像重建建模技术。通过CT扫描、核磁共振成像等医学影像技术对研究的对象进行逐层扫描,从而得到二维的断层影像数据,将获取的数据导入Mimics 17.0中,通过阈值分割、区域增长、空腔填充和三维建模等步骤获取人体几何模型[6-9]。 目前,Mimics是运用比较广泛的医学三维重构软件,但直接通过Mimics等软件建立骨骼三维模型往往不能直接用于有限元分析,还需要通过geomagic等软件对模型进行修改,使分析模型尽可能的是由光滑面或体构成。因为有限元分析的模型必须是连续的弹性体,单个模型中不能有分离或断开的情况出现,同时应尽可能消除模型中的破孔、尖角、毛刺等几何特征,否则在这些部位需要用大量单元进行模拟,这将消耗大量的计算时间,甚至导致模型重构失败。 这种方法的优点是,能够得到形状复杂的模型,而且能描述出模型的内部结构,精度也较高,已经成为现在骨科有限元分析模型建立的主要方法之一。 2.1.2 划分有限元网格 有限元分析法是力学分析方法中的数值法。有限元网格划分就是将整体划分为个体进行分析,最后将个体集合为整体进行研究。其中有限元网格划分的质量直接影响分析结果的精度,主要表现在以下3方面:①几何模型结构细节处理。②网格划分密度。③网格过渡。人体骨骼的形状和结构特点比较复杂,有限元分析中描述骨骼外形(包括内表面),一般会把重建三维分析模型划分成有限个四面体或六面体,这样就会造成分离出来的正面体(正四面体或正六面体)变形,其后果即是越发加强了对有限元网格划分这一操作的质量要求。 2.1.3 骨骼材料属性定义 通常对材料属性赋值有均匀赋值法、查表赋值法、蒙板赋值3种方法,而人体骨骼是由羟基磷灰石和胶原纤维组成的复合材料,骨的力学性质(弹性模量、剪切模量、黏弹性、破坏时的极限应力和应变等)不仅与复合材料本身性质有关,还与骨的结构组成、形态和胶原纤维蛋白如何组合等有关。而且即使是同一块人体骨骼上,不同位置和部位的密度也有很大的差异。针对这一特点,现在对骨组织的属性赋值主要是依据其CT值计算出骨密度值,再对骨组织进行划分有限元网格单元,根据每个网格单位CT值来对骨组织的属性赋值,从而描述骨的生物力学特性。 2.1.4 确定边界条件 人体骨骼的承载和运动是和其周围的肌肉等相关组织分不开的,由于这些结构之间的配合比较复杂,要建立一个完整的有限元分析模型是比较困难的。所以,为了便于分析求解往往会简化分析模型。待简化之后明确正确的边界条件也是有限元分析的重要环节之一。骨科有限元分析中,研究的常常是人体局部的骨骼,在这种状态下,分析模型中简化掉的部分结构的力学作用就需要通过添加边界条件来实现,在有限元分析中尽可能真实的反应出骨骼之间、关节周围肌肉的支撑和力传递作以及外力作用等就显得尤为重要。 2.1.5 分析与结果验证 分析作为有限元分析仿真的执行环节,主要工作是确定一种适合分析模型本身的有限元算法,这样不仅能在一定程度上提高结果的准确性,也能提高分析计算的速度。由于某些分析模型和分析类型非常复杂,同时受个人计算机硬件水平的限制往往计算的时间很长,特别是在非线性分析时。所以,选择恰当的算法对于分析过程也有很大的帮助。 在得到分析结果之后,就是判断输出结果的正确性。为了证实分析所得到的结果是否正确,需要利用所掌握的相关知识结合临床综合分析,或者进行可行性实验进行反复验证并分析,得到相对客观数据,从而归纳并分析得出其一般规律,提高所得数据的可信度。 2.2 有限元分析在临床骨科中的实际应用与发展 2.2.1 在脊柱骨折研究领域的应用 利用有限元分析法可以对脊柱骨折的生物力学功能和致病机制等进行研究及评价。Silve等[10]通过有限元法证实进行椎弓根螺钉置入时,皮质骨提供的强度占椎体强度的10%,在相对较高的压力载荷下,终板中下方的松质骨发生骨折时,椎体的强度明显下降,而骨内压则随之升高。Cao等[11]建立了三椎体腰椎有限元分析模型,分析结果标明34%的椎体压缩骨折发生在上终板或下终板附近,而63%的椎体压缩骨折发生在上下终板之间。结果认为骨折的发生与负荷类型没有关系,而与皮质骨的弹性模量成正比例关系,与松质骨的弹性模量成反比例关系。Crawford等[12]研究认为椎体屈曲强度对椎体骨折有之间影响,而椎体的前后径有对椎体的屈曲强度有影响。Dall Ara等[13]通过非线性解剖特异的定量CT数据位依据建立的有限元分析模型,预测了体内椎体的力量。随后应用离体实验验证了该模型的正确性。Matsuura等[14]利用标本研究得出有限元分析可能是预测椎体成形术后带来的原位椎体骨折应力负荷的有用工具。 2.2.2 在关节置换中的应用 有限元分析技术为了解关节置换患者术前关节功能状态、术中假体的选择、术后预测提供了一种全新的方法。Chang等[15]通过对正常股骨头外形进行有限元分析后,建议将假体股骨柄的股骨颈设计成相对较细,距股骨头顶端相对较短的形态,其理论依据是这样可以适当减少对周围骨质的破坏,从而减少骨结构重塑,同时假体稳定性可以保障。Patil等[16]通过有限元法分析全髋关节置换后髋关节的应力分布表明,当髋臼杯外展角由35°增加到55°时,髋关节内应力会逐步上升,其磨损程度也会增大;而随着髋臼杯的前倾角的增大,髋关节内应力会逐渐减小。从而论证得出髋臼杯置入位置和其磨损存在必然的联系。Sakagoshi等[17]利用有限元分析法技术来研究股骨头坏死面积、术中假体安放位置与髋关节置换后生物力学的变化三者之间的关系,其实验出现随着股骨头坏死面积的增大,骨-骨水泥界面的应力会逐渐增大;当假体内倾角处于内翻位时,骨水泥的最大应力增加。内倾角110°逐渐增大至140°时,假体的峰值应力逐步减小,从而论证出股骨头坏死面积大节假体的三维有限元模型,在股骨头中心加大关节面负荷量,采用非线性接触弹塑性分析聚乙烯内衬关节面的接触应力。从而证实表明,选择股骨头直径相对较大的髋臼假体,并严格控制假体置入在安全范围内,术后减少上小与假体位置不当均是影响髋关节置换后生物力学环境的重要因素。严世贵等[18]通过建立髋关楼等负重性活动均有利于减少全髋关节置换后对内衬的破坏。 2.2.3 在四肢与骨盆骨折中的应用 有限元分析法可以在四肢骨折固定方式选择以及术后对固定效果的评价提供相应依据。Bresina等[19]在1995年用有限元分析评估点状内固定(PC-Fix)系统的内固定效果;Rogge等[20]通过对桡骨远端骨折的三维有限元模型采用不同内固定方式所产生的不同生物力学特征进行分析,从而以寻找出最优固定方式。Qian等[21]在尸体实验中研究发现厚度、极限载荷和极限应变在更大的近端部分伸肌,机械性能的变化可能会影响预测力的传输,术前建模时应考虑伸肌装置的作用予以建模。既往骨盆骨折的治疗是基于广泛的临床经验。Fan等[22]在有限元模型的基础上,对3种固定系统的生物力学通过有效强度水平、压力分布、力转移,沿骨折线位移进行评估,发现所有3个固定系统可以用来获得有效的功能结果(图2)。前柱钢板结合四边形螺钉是最优方法丁字形的髋臼的骨折。这个固定系统可以减少很多与其他固定系统有关风险和限制。"

2.2.4 在病理机制方面的研究应用 Chen等[23]在2003年,通过有限元分析对于糖尿病患者足底应力分布改变进行了研究,这对于矫形支具的设计和材料选择都有很大的帮助[24]。Goto等[25]运用压缩屈曲理论,通过有限元仿真分析模拟出脊柱侧凸形成的过程,研究其病理机制。Campoli等[26]研究股骨的形状和密度分布及其振动固有频率之间的关系,得出形状和密度可能导致大型计算固有频率的变化,不同的有限元模型需要准确估计骨的自然频率。 2.2.5 在骨应力分析中的应用 踝关节损伤是临床上最常见的骨科疾病,以前对于踝关节闭合软组织损伤综合诊断和治疗以及踝关节骨折术后导致创伤性关节炎、退行性病变等并发症多数采用传统的生物力学进行研究。在有限元技术比较成熟的今天,对于这方面的研究多采用建立有限元分析模型来进行力学分析。采用有限元分析技术对正常步态下足踝部的解剖结构和力学接触特点进行研究,从而获取足踝部在各种受力情况下各关节面之间接触应力情况、应力分布部位以及应力值[27-32]。黄诸侯等[33]对跟骨的应力分析表明,在踝关节中立位,足背伸20°时,跟骨应力最大点为跟距外侧的关节面与由内后斜向前外方向的相交处,除此之外后距关节面和跟腱之间的部位承受应力也相对较大。"

| [1] Wam B, Rybicki EF, Burdeaux BD. A new method to analyse the mechanical behaviour of skeletal parts. Acta Ortho Scand. 1972;43: 301. [2] 陈国栋,罗羽婕,王锐英.有限元分析在股骨生物力学研究中的应用[J].实用医学杂志,2011,27(2):334-336. [3] Zienkiewicz OC, Taylor RL. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials. 2005; 26(27): 5474-5491. [4] 黄若昆,谢鸣,勘武生,等.数字骨科学研究进展[J].中国矫形外科杂志,2010,18(12):1003-1005. [5] 尹庆水,万磊.数字骨科——信息化世纪的新骨科数字骨科入门(一)[J].中国骨科临床与基础研究杂志,2009,1(1): 77-78. [6] Qin JS,Wang Y,Peng XQ,et al.Three-dimensional finite element modeling of whole lumbar spine and its biomechanical analysis. J Med Biomech. 2013;28(3): 321-325. [7] Zhou J,Yang HL,Zhang W,et al.A simple intraoperative position determination method during balloon kyphoplasty in patients with osteoporotic vertebal compression fractures and MIMICS 3-dimensional reconstruction evaluation to transpedicular trocars channel and cement leakage.Bone. 2010;47(Suppl 3): 454. [8] 牛文鑫,杨云峰,俞光荣,等.人体足部三维有限元模型的有效构建方法及其合理性的实验分析研究[J],生物医学工程学杂志,2009,26(1):80-84. [9] Shi D, Wang F, Wang D, et al. 3-D finite element analysis of the influence of synovial condition in sacroiliac joint on the load transmission in human pelvic system. Med Eng Phys.2014;36(6):745-753. [10] Silve MJ, Kearveny TM, Hayes WC. Loading sharing between the shell and centrum in the lumber vertebral body. Spine. 1997;22(2): 140-150. [11] Cao KD, Grimm MJ, Yang KH. Load sharing within a human lumbar vertebral body using the finite element method. Spine. 2001;26(12): E253-E260. [12] Crawford RP, Keaveny TM. Relationship between axial and bending behaviors of the human thoracolumbar vertebra. Spine. 2004;29(20): 2248-2255. [13] Dall Ara E, Schmidt R, Pahr D, et al. A nonlinear finite element model validation study based on a novel experimental technique for inducing anterior wedge shape fractures in human vertebral bodies in vitro . J Biomech. 2010;43(12): 2374-2380. [14] Matsuura Y, Giambini H, Ogawa Y, et al. Specimen-specific nonlinear finite element modeling to predict vertebrae fracture loads after vertebroplasty. An KN.Spine (Phila Pa 1976). 2014;39(22):E1291- 1296. [15] Chang PB, Williams BJ, Bhalla KS, et al. Design and analysis of robust total joint replacements finite element model experiments with environmental variables. J Biomech Eng. 2001;123(3): 239-246. [16] Patil S, Bergula A, Chen PC, et al. Polyethylene wear and acetabular component orientation. J Bone Joint Sung Am. 2003; 85: S56-63. [17] Sakagoshi D, Kabata T, Umemoto Y, et al. A mechanical analysis of femoral resurfacing implantation for osteonecrosis of the femoral head . J Arthroplasty. 2010;25(8): 1282-1289. [18] 严世贵,吴浩波,陈维善,等. 全髋关节置换后聚乙烯内衬应力的弹塑性有限元分析[J]. 中华骨科杂志,2004,24(4): 211-213. [19] Bresina S,Tepic S.Finite element analysis(FEA) for the Point contact fixator: screw drive,plate design overcuts.Injury. 1995; 26(S2): s20-23. [20] Rogge RD,Adams BD,Goel VK.An analysis of bone stresses and bone stability using finite element model of simulated distal radial fractures.Hand Surg. 2002; 27(1):86. [21] Qian K, Traylor K, Lee SW, et al. Mechanical properties vary for different regions of the finger extensor apparatus.J Biomech. 2014;47(12): 3094-3099. [22] Fan Y, Lei J, Zhu F,et al. Biomechanical Analysis of the Fixation System for T-Shaped Acetabular Fracture. Comput Math Methods Med. 2015;2015:370631. [23] Chen WP, Ju CW, Tang FT. Effects of total contact insoles on the plantar stress redistribution: a finite element analysis. Clin Biomech. 2003;6: 17-24. [24] Vanderoost J, Jacecques SV, Vander Perre G, et al. Fast and accurate specimen-speciffic simulation of trabecular bone elastic modulus using novel beam-shell finite element model. J Biomech. 2011;44: 1566-1572. [25] Goto M,Kawakami N, Azegami H, et al. Buckling and bone modeling as factors in the development of idiopathic scolisis. J Biomech Eng. 2002;124: 784-790. [26] Campoli G, Baka N, Kaptein BL, et al. Relationship between the shape and density distribution of the femur and its natural frequencies of vibration. J Biomech. 2014;47(13):3334-3343. [27] Anderson DD, Gold JK, Shivanna K, et al. Intr-articular contact stress distribution at the ankle throughout stance phase-patient-specific finite element analysis as a metric of degeneration propensity. Biomech Model Mechan. 2006;3: 882-891. [28] Suckel A, Muller O, Wachter N, et al. In vitro measurement of intr-articular pressure in the ankle joint. Knee Surg Sports Tra. 2010;5: 664-668. [29] Wan L, De Asla RJ, Rubash HE, et al. Determination of in-vivo articular cartilage contact areas of human talocrural joint underweight-bearing conditions. Osteoarthritis Cartilage. 2006;37: 1294-1301. [30] Song Y, Debski RE, Musahl V, et al. a three-dimensional finite element model of the human anterior cruciate ligament: a computational analysis with experimental validation. Biomech. 2004;37: 383-390. [31] 汪强,孙俊英,赖震. 膝关节三维有限元模型的建立[J].山西医药杂志,2007,36(3):210-212. [32] Camacho DL, Ledoux WR, Rohr ES, et al. A three-dimensional, anatomically detailed foot model: a foundation for a finite element simulation and means of quantifying foot-bone position. Rehabil Res Dev. 2002; 39: 401-411. [33] 黄诸侯,李俊,陈日齐,等.跟骨三维有限元模型的建立及其骨折发生机制[J]. 中国骨伤,2012,25(2):97-101. [34] Pang QJ, Yu X, Guo ZH.The sustentaculum tali screw fixation for the treatment of Sanders type II calcaneal fracture: A finite element analysis. Pak J Med Sci. 2014; 30(5):1099-1103. [35] Rotariu M,Filep R,Turnea M,et al. Analyse of socket-prosthesis-blunt complex for lower limb amputee using objective measure of patient's gait cycle. Rev Med Chir Soc Med Nat Iasi.2015;119(1): 281-286. [36] Choi AH, Conway RC, Ben-Nissan B.Finite-element modeling and analysis in nanomedicine and dentistry.Nanomedicine (Lond). 2014;9(11):1681-1695. [37] Iselin LD, Wahl P, Studer P, et al. Associated lesions in posterior wall acetabular fractures: not a valid predictor of failure. J Orthop Traumatol. 2013;14(3):179-184. [38] 毕胜,李义凯,赵卫东,等.腰部推拿手法生物力学和有限元比较研究[J].中华物理医学与康复杂志,2002,24(9): 524-526. [39] 陈浩,徐海涛,张美超,等.坐位旋转手法对腰椎内在应力的实时监测[J] .中国临床解剖杂志,2005,23(4):420-422. [40] Wu YD, Cai XH, Liu XM, et al. Biomechanical analysis of the acetabular buttressplate: are complex acetabular fractures in the quadrilateral area stable after treatment with anterior construct plate-1/3 tube buttress plate fixation? Clinics. 2013;68(7):1028-1033. |
| [1] | Shi Bin, An Jing, Chen Long-gang, Zhang Nan, Tian Ye . Influencing factors for pain after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 993-997. |
| [2] | Wang Xian-xun. Impact of local compression cryotherapy combined with continuous passive motion on the early functional recovery after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 998-1003. |
| [3] | Yuan Wei, Zhao Hui, Ding Zhe-ru, Wu Yu-li, Wu Hai-shan, Qian Qi-rong. Association between psychological resilience and acute mental disorders after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1015-1019. |
| [4] | Chen Qun-qun, Qiao Rong-qin, Duan Rui-qi, Hu Nian-hong, Li Zhao, Shao Min. Acu-Loc®2 volar distal radius bone plate system for repairing type C fracture of distal radius [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1025-1030. |
| [5] | Huang Xiang-wang, Liu Hong-zhe. A new low elastic modulus of beta titanium alloy Ti2448 spinal pedicle screw fixation affects thoracic stability: biomechanical analysis [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1031-1035. |
| [6] | Xie Qiang. Three-dimensional finite element model for biomechanical analysis of stress in knee inversion and external rotation after posterior cruciate ligament rupture [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1036-1040. |
| [7] | He Ze-dong, Zhao Jing, Chen Liang-yu, Li Ke, Weng Jie. Multilevel finite element analysis on the biological tribology damage of water on bone tissue [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1041-1045. |
| [8] | Jiang Zi-wei, Huang Feng, Cheng Si-yuan, Zheng Xiao-hui, Sun Shi-dong, Zhao Jing-tao, Cong Hai-chen,Sun Han-qiao, Dong Hang. Design and finite element analysis of digital splint [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1052-1056. |
| [9] | Wang Fei, Liu Zhi-bin, Tao Hui-ren, Zhang Jian-hua, Li Chang-hong, Cao Qiang, Zheng Jun, Liu Yan-xiong, Qu Xiao-peng. Clinical efficacy of preoperative osteotomy designs using paper-cut technology versus photoshop software for ankylosing spondylitis with kyphosis [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1057-1063. |
| [10] | Li Hui, Ma Jun-yi, Ma Yuan, Zhu Xu . Establishment of a three-dimensional finite element model of ankylosing spondylitis kyphosis [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1069-1073. |
| [11] | Ling Guan-han, Ou Zhi-xue, Yao Lan, Wen Li-chun, Wang Guo-xiang, Lin Heng-feng. Establishment of simulating three-dimensional model of China-Japan Friendship Hospital Classification for L type osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1074-1079. |
| [12] | Fu Wei-min, Wang Ben-jie. Assessing the degree of necrotic femoral head, and association of blood supply with pathlogical changes: study protocol for a diagnostic animal trial [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1086-1091. |
| [13] | Zhang Wen-qiang, Ding Qian, Zhang Na. Associations between alpha angle and herniation pit on oblique axial magnetic resonance imaging in asymptomatic hip joints of adults [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1098-1103. |
| [14] | Sun Xiao-xin1, Zhou Wei2, Zuo Shu-ping3, Liu Hao1, Song Jing-feng1, Liang Chun-yu1. Morphological characteristics for the magnetic resonance imaging assessment of discoid lateral meniscal tears in children [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1104-1109. |
| [15] | Lin Han-wen, Wen Jun-mao, Huang Chao-yuan, Zhou Chi, Tang Hong-yu. Correlation between the changes in lower limb power line and pain area in the knee osteoarthritis patients: imaging evaluation [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1110-1114. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||