Chinese Journal of Tissue Engineering Research ›› 2015, Vol. 19 ›› Issue (11): 1782-1787.doi: 10.3969/j.issn.2095-4344.2015.11.026
Previous Articles Next Articles
Regulatory role of acupuncture and moxibustion in immunosenescence and epigenetic mechanisms
Shen Wen-bin1, Zhou Ci-li1, 2, Wu Huan-gan1, 2, Huang Yan1, 2, Zhao Chen1, Cui Yun-hua1, 2
- 1Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China; 2Shanghai Research Institute of Acupuncture and Meridian, Shanghai 200030, China
-
Revised:2015-02-12Online:2015-03-12Published:2015-03-12 -
Contact:Cui Yun-hua, M.D., Associate researcher, Master’s supervisor, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China; Shanghai Research Institute of Acupuncture and Meridian, Shanghai 200030, China Zhao Chen, M.D., Chief physician, Master’s supervisor, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China -
About author:Shen Wen-bin, Studying for master’s degree, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China -
Supported by:the National Natural Science Foundation of China, No. 81303029; National Basic Research Program of China (973 Program), No. 2009CB522900; Shanghai Science and Technology Project, No. 12ZR1429700; Shanghai Municipal Health Bureau Projects, No. 20114Y008
CLC Number:
Cite this article
Shen Wen-bin, Zhou Ci-li, Wu Huan-gan, Huang Yan, Zhao Chen, Cui Yun-hua. Regulatory role of acupuncture and moxibustion in immunosenescence and epigenetic mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(11): 1782-1787.
share this article
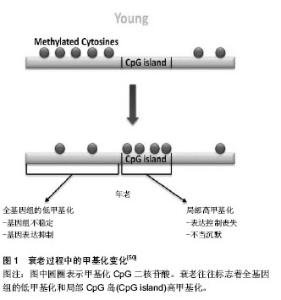
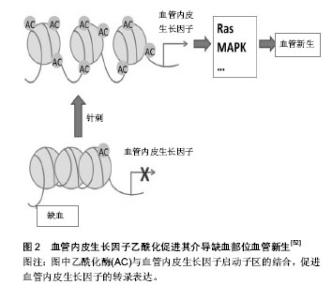
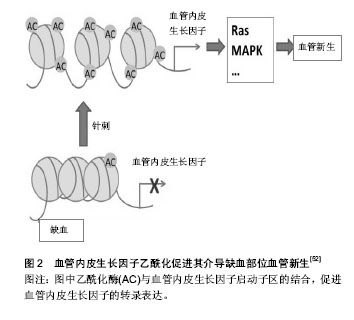
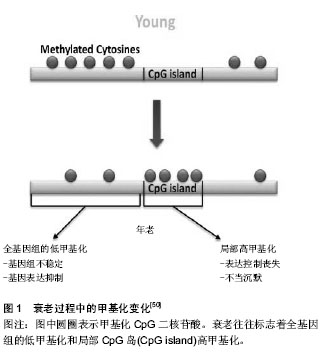
2.1 针灸对免疫衰老的调节作用 免疫衰老以固有免疫系统增龄性改变和适应性免疫系统增龄性改变并存为显著特征。大量研究已证实,针灸能提高免疫系统功能,对免疫器官、免疫细胞及细胞因子等均具有调节作用[6],从整体水平上改善免疫衰老。 2.1.1 免疫衰老的特征性表现 免疫衰老在固有免疫系统中的特征性表现:固有免疫反应是机体免疫的第一步,其功能会随着年龄增长而逐渐降低,并且可能会进一步影响到由细胞或体液介导的适应性免疫[7]。肿瘤坏死因子α主要由巨噬细胞分泌,是一种重要的炎性因子,研究发现老年大鼠的肿瘤坏死因子α水平较成年大鼠明显降低[8]。自然杀伤细胞是Ⅰ型固有淋巴细胞,无需提前致敏即具有杀死肿瘤细胞和病毒感染细胞的能力,对不同的病原体均能做出快速回应。有研究表明在衰老过程中自然杀伤细胞的频率、表型和分布会出现大量的改变[2,9-10],尽管在单细胞水平上衰老对其结合目的细胞和分泌穿孔素的能力没有产生影响,但其细胞毒性作用却严重受损[11]。另外CD16的表达及其诱发抗体依赖性自然杀伤细胞毒性作用也受到衰老的影响[12]。有研究证明自然杀伤细胞的数量和细胞毒性作用的改变与老年人发生感染和死亡的风险较高有直接关系[9,13-14]。机体内关键的早期固有免疫反应不仅依靠着中性粒细胞的活性和吞噬能力,还依靠着其向感染位点的趋化和定向迁移能力,体外实验表明老年小鼠的中性粒细胞定向迁移能力低于青年小鼠[15]。同样的,在人体试验中也证实了这一点,老年人的中性粒细胞表现出了定向迁移能力受损的特点[16]。在免疫衰老过程中,固有免疫系统的中性粒细胞、巨噬细胞和自然杀伤细胞等多种细胞的数量及功能会出现一些特征性改变。 免疫衰老在适应性免疫系统中的特征性表现:适应性免疫系统是机体防御系统的第三道防线,在第一、二道防线被病原体突破之后被激活,起着识别和杀灭病原体的作用。胸腺是人体重要的免疫器官,但随着年龄的增长,胸腺的大小和质量会逐步减少,中年以前每年减少3%,以后则每年减少1%[17]。胸腺中的淋巴细胞伴随年龄增长逐渐减少,脂肪组织逐渐增多,这主要是因为能够分泌胸腺激素的胸腺上皮细胞减少造成的[18]。胸腺作为T细胞发育成熟的重要场所,胸腺间质细胞为造血母细胞发育成T淋巴细胞提供了一个特殊的微环境[19-20],但在免疫衰老的过程中胸腺间质细胞的结构和功能会受到严重损伤[21-22],例如会出现胸腺囊肿,胸腺上皮细胞器丢失等。适应性免疫系统在免疫衰老过程中最常见的变化包括T细胞和B细胞增殖能力减弱,种类减少,记忆型细胞增多,幼稚细胞减少,1型辅助性T细胞(Helper T cell 1,Th1)和2型辅助性T细胞(Helper T cell 2,Th2)比例反转[23]。T淋巴细胞的减少会明显减弱机体的免疫监视功能,从而导致老年人更容易患感染性疾病、癌症及疫苗失效等[22,24-26]。调节性T细胞具有维持外周免疫耐受和调节自身免疫细胞的作用,当调节性T细胞的数量过多时,机体对感染性疾病、神经性病变和癌症的易感性也会随之增加[27-29]。Garg等[30]运用流式细胞术证实了老龄小鼠比幼年小鼠拥有更高水平的调节性T细胞,而在人体中,Gregg等[31]同样也证实了调节性T细胞在T细胞总量中所占的比例随着年龄逐渐升高,这在一定程度上解释了老年人容易患以上这些疾病的原因。有研究认为初始B细胞数目下降可能是发生免疫衰老的重要标志[32]。随着年龄增长,B细胞分泌抗体的能力下降[33],并且在一些研究中发现老年小鼠产生的抗体对目标的亲和力较低,不能有效的控制感染[34]。而一些和自身免疫性疾病相关的抗体却会随着年龄逐渐增加,例如,健康年轻人的类风湿因子水平只有5%,老年人则增加到5倍的水平,70岁以上健康老年人的抗核抗体水平要比健康成年人高很多[35]。 2.1.2 针灸对免疫衰老的调节 在免疫衰老的进程中,机体的多种免疫器官、组织、细胞和分子的功能及结构受其影响,且这种影响是多样化的,或升高或降低,很难用一种治疗方法解决。而针灸具有整体调节、双向良性调节和多靶点调节的作用特点,因此可以运用针灸的方法改善免疫衰老对机体免疫系统的影响。 固有免疫系统中针灸对免疫衰老的调节:针灸可增加固有免疫系统中相关细胞的数量,增强细胞的功能,促进细胞分泌。朱梅等[36-37]发现针刺足三里和关元穴对老年大鼠肝内巨噬细胞的影响是多方面的,首先是使巨噬细胞的数量明显增加,其次是增大了细胞体积,再次是增强了细胞的吞噬功能,并且使细胞处于激活状态。另有研究发现艾灸也具有相似作用,艾灸可以显著促进巨噬细胞对已被吞噬的细菌的杀灭和清除作用,并且能够明显下调多种炎症细胞因子的表达,如白细胞介素1、白细胞介素10等。 适应性免疫系统中针灸对免疫衰老的调节:伴随年龄的增长,免疫器官逐渐萎缩退化,组织结构逐渐发生改变,功能逐渐减退。最具有代表性的为胸腺,老年时期的胸腺已基本被脂肪组织所代替,功能也基本丧失。研究证实艾灸能明显延缓D-半乳糖造成的衰老小鼠免疫器官的萎缩退化,提高衰老小鼠的胸腺指数[38]。T淋巴细胞亚群的分布异常与多种疾病有直接关系,临床实验研究表明温和灸能明显提高老年人CD3+和CD4+T细胞的含量,恢复CD4+/CD8+的比值[39],改善老年人的免疫功能。淋巴细胞的增殖能力会出现增龄性降低,Pavão等[40]选用12个健康年轻人(平均年龄27.6岁)和12个健康老年人(平均年龄65.6岁)研究针刺对心理压力和淋巴细胞增殖能力的影响,实验开始前测得老年人淋巴细胞增殖能力明显低于年轻人(P=0.01),经过针刺干预后年轻人组的淋巴细胞增殖能力无任何改变(P=0.31),而老年人组明显提高(P=0.004),更值得注意的是,针刺干预后的老年人组淋巴细胞增殖能力水平和年轻人组基本相同,除此以外还证实了针刺对于健康老年人能够有效的缓解压力和紧张情绪。艾灸可以提高老年人T淋巴细胞总数,使由免疫衰老改变的CD4+/CD8+细胞比值恢复正常,同时能够促进白细胞介素2的分泌[41]。淋巴B细胞在受到抗原刺激后能产生多种免疫球蛋白,参与机体的免疫反应,目前所使用的大多数疫苗就是通过刺激B淋巴细胞产生抗体的,老年人对疫苗的反应性降低或许与此有关。肖凌等[42]采用百白破疫苗免疫衰老模型大鼠,“双固一通”针法进行干预,结果发现针灸能明显降低衰老大鼠的血脂水平,提高衰老大鼠对疫苗的反应性,起到改善免疫衰老的作用。 2.2 针灸调节免疫衰老的表观遗传机制探讨 表观遗传存在于人类整个生命过程中,表观遗传修饰主要包括DNA甲基化,组蛋白修饰,染色质结构变异和RNA干扰等[43]。已有研究表明表观遗传调控和免疫衰老具有很大的相关性,伴随着年龄增长出现的表观遗传修饰改变很可能是免疫衰老发生的基因机制。这也为研究针灸调节免疫衰老的机制提供了一个新的方向。 2.2.1 表观遗传调控与免疫衰老的相关性 表观遗传调控主要通过修饰基因和蛋白影响免疫衰老相关基因的功能和状态,从而调控免疫衰老。例如表观遗传调控对免疫反应相关基因和衰老差异表达相关基因起到了非常重要的调节作用,其中最具代表性的基因为白细胞介素2基因和γ干扰素基因[44]。老年小鼠中CD3+和CD4+细胞DNA甲基化水平只有年轻小鼠的20%和25%[30]。人类整个基因组的5-甲基胞嘧啶在衰老过程中会逐渐减少并且和人类潜在的最长寿命成反比[45-46]。运用基因敲除的方法敲除小鼠的DNA甲基转移酶1基因,与未敲除的小鼠对比后发现当这种基因敲除的小鼠生长到了老年阶段时,发现在其肝脏和唾液腺中自身抗体滴度较低,且衰老相关的淋巴细胞浸润较少;18个月大的DNA甲基转移酶1基因缺失小鼠不如正常对照小鼠的T细胞免疫老化明显,且更好的保持了T细胞免疫功能, T细胞发展成记忆T细胞的速度慢,分泌白细胞介素2的水平更高,T细胞增殖反应更好;另一方面,DNA甲基转移酶1基因缺失小鼠甲基胞嘧啶结合蛋白2表达水平增加,这在蛋白机制上解释了这些小鼠中的DNA甲基化水平出现差异的原因[47]。组蛋白H4乙酰化和H3磷酸乙酰化发生在促炎分子信号出现之后,组蛋白的乙酰化又进一步使DNA招募到的核转录因子κB增多[48],核转录因子κB只有在活化之后才能进入细胞核参与转录调控,在多种老年组织中可观察到活化的核转录因子κB异常增多,这个现象也被认为是免疫衰老的最初原因[49](图1)。 2.2.2 针灸调节免疫衰老与表观遗传调控机制研 究 表观遗传调控最突出的特征是所有的修饰均是在不改变基因序列的前提下进行的,通过修饰基因、改变蛋白结构、控制DNA与其它分子的结合多途径、多层次的影响和调节遗传信息的表达,最终在机体的整体水平上起到调控作用。而从以上有关针灸对免疫衰老的调节可以看出针灸能够从器官、组织、细胞和分子多种水平上全面整体的改善免疫衰老,这和表观遗传调控的特征有很大程度的契合。因此,可以将表观遗传调控引入针灸调节免疫衰老的机制中来,这有利于更好的揭示针灸调节免疫衰老的机制。 表观遗传调控就像是控制着大门的控制器,在生命的各个进程中控制着大门的开放和关闭,但这个控制器很容易被所处的环境改变[50-58],针灸对于免疫衰老的调节可能和针灸改变了这个控制器有关。有研究已证实艾灸可以增强小鼠心肌细胞中组蛋白乙酰化酶p300的表达,激活了心肌细胞中的抗细胞凋亡蛋白Bcl-2蛋白的表达,起到了保护心肌细胞的作用[59]。另一项研究则证实针刺能明显增加缺血性心肌梗死模型大鼠组蛋白H3K9乙酰化酶与血管内皮生长因子启动子区的结合,促进血管内皮生长因子的转录表达,介导缺血心肌血管新生,起到了保护缺血心肌的作用[60](图2)。这2项研究表明了针灸确实通过调控表观遗传修饰起到调节机体功能的作用,这为今后针灸调节免疫衰老的表观遗传学机制研究提供了借鉴。 多项研究证实针灸能有效改善免疫衰老,表观遗传学与免疫衰老密切相关。例如,随着年龄增加,巨噬细胞吞噬能力下降,活性降低,表观遗传学研究表明衰老小鼠巨噬细胞的组蛋白去乙酰化酶水平比年轻小鼠高[61],而针刺能增强老年大鼠巨噬细胞功能[36-37]。初始T细胞中,由于白细胞介素2基因启动子区的高甲基化状态而处于静息态。但在激活信号传递给T细胞后20 min,白细胞介素2基因甲基化水平逐渐降低[62],白细胞介素2基因开始转录,白细胞介素2分泌增加。研究证实艾灸能明显提高衰老大鼠血清白细胞介素2含量,至于艾灸是否是通过改变白细胞介素2基因甲基化水平增加白细胞介素2含量的,今后作者会进一步关注。 "
| [1] Ademokun A, Wu YC, Dunn-Walters D. The ageing B cell population: composition and function. Biogerontology. 2010; 11(2):125-137. [2] Solana R, Tarazona R, Gayoso I, et al. Innate immunosenescence: effect of aging on cells and receptors of the innate immune system in humans. Semin Immunol. 2012; 24(5):331-341. [3] Hajishengallis G. Too old to fight? Aging and its toll on innate immunity. Mol Oral Microbiol. 2010;25(1):25-37. [4] Weiskopf D, Weinberger B, Grubeck-Loebenstein B. The aging of the immune system. Transpl Int. 2009;22(11):1041-1050. [5] Weksler ME, Pawelec G, Franceschi C. Immune therapy for age-related diseases. Trends Immunol. 2009;30(7):344-350. [6] 骆永珍,张燕华,周荣兴.针灸与免疫[M].北京:人民卫生出版社, 2002. [7] Mills KH. Designer adjuvants for enhancing the efficacy of infectious disease and cancer vaccines based on suppression of regulatory T cell induction. Immunol Lett. 2009;122(2):108-111. [8] Dimitrijevi? M, Stanojevi? S, Kuštrimovi? N, et al. The influence of aging and estradiol to progesterone ratio on rat macrophage phenotypic profile and NO and TNF-α production. Exp Gerontol. 2013;48(11):1243-1254. [9] Camous X, Pera A, Solana R, et al. NK cells in healthy aging and age-associated diseases. J Biomed Biotechnol. 2012; 2012:195956. [10] Gayoso I, Sanchez-Correa B, Campos C, et al. Immunosenescence of human natural killer cells. J Innate Immun. 2011;3(4):337-343. [11] Campos C, Pera A, Sanchez-Correa B, et al. Effect of age and CMV on NK cell subpopulations. Exp Gerontol. 2014;54: 130-137. [12] Solana R, Mariani E. NK and NK/T cells in human senescence. Vaccine. 2000;18(16):1613-1620. [13] DelaRosa O, Pawelec G, Peralbo E, et al. Immunological biomarkers of ageing in man: changes in both innate and adaptive immunity are associated with health and longevity. Biogerontology. 2006;7(5-6):471-481. [14] Grubeck-Loebenstein B, Della Bella S, Iorio AM, et al. Immunosenescence and vaccine failure in the elderly. Aging Clin Exp Res. 2009;21(3):201-209. [15] Nomellini V, Brubaker AL, Mahbub S, et al. Dysregulation of neutrophil CXCR2 and pulmonary endothelial icam-1 promotes age-related pulmonary inflammation. Aging Dis. 2012;3(3):234-247. [16] Sapey E, Greenwood H, Walton G, et al. Phosphoinositide 3-kinase inhibition restores neutrophil accuracy in the elderly: toward targeted treatments for immunosenescence. Blood. 2014;123(2):239-248. [17] George AJ, Ritter MA. Thymic involution with ageing: obsolescence or good housekeeping? Immunol Today. 1996; 17(6):267-272. [18] Mocchegiani E, Giacconi R, Cipriano C, et al. Are zinc-bound metallothionein isoforms (I+II and III) involved in impaired thymulin production and thymic involution during ageing? Immun Ageing. 2004;1(1):5. [19] Griffith AV, Fallahi M, Nakase H, et al. Spatial mapping of thymic stromal microenvironments reveals unique features influencing T lymphoid differentiation. Immunity. 2009; 31(6): 999-1009. [20] Chen L, Xiao S, Manley NR. Foxn1 is required to maintain the postnatal thymic microenvironment in a dosage-sensitive manner. Blood. 2009;113(3):567-574. [21] Gray DH, Seach N, Ueno T, et al. Developmental kinetics, turnover, and stimulatory capacity of thymic epithelial cells. Blood. 2006;108(12):3777-3785. [22] Lynch HE, Goldberg GL, Chidgey A, et al. Thymic involution and immune reconstitution. Trends Immunol. 2009;30(7): 366-373. [23] Malaguarnera L, Cristaldi E, Lipari H, et al. Acquired immunity: immunosenescence and physical activity. Eur Rev Aging Phys Act. 2008;5(2):61–68. [24] Linton PJ, Dorshkind K. Age-related changes in lymphocyte development and function. Nat Immunol. 2004;5(2): 133-139. [25] Naylor K, Li G, Vallejo AN, et al. The influence of age on T cell generation and TCR diversity. J Immunol. 2005;174(11): 7446-7452. [26] Nikolich-Zugich J, Slifka MK, Messaoudi I. The many important facets of T-cell repertoire diversity. Nat Rev Immunol. 2004;4(2):123-132. [27] Richardson BC. Role of DNA methylation in the regulation of cell function: autoimmunity, aging and cancer. J Nutr. 2002; 132(8 Suppl):2401S-2405S. [28] Mendez S, Reckling SK, Piccirillo CA, et al. Role for CD4(+) CD25(+) regulatory T cells in reactivation of persistent leishmaniasis and control of concomitant immunity. J Exp Med. 2004;200(2):201-210. [29] Sharma S, Dominguez AL, Lustgarten J. High accumulation of T regulatory cells prevents the activation of immune responses in aged animals. J Immunol. 2006;177(12):8348-8355. [30] Garg SK, Delaney C, Toubai T, et al. Aging is associated with increased regulatory T-cell function. Aging Cell. 2014;13(3): 441-448. [31] Gregg R, Smith CM, Clark FJ, et al. The number of human peripheral blood CD4+ CD25high regulatory T cells increases with age. Clin Exp Immunol. 2005;140(3):540-546. [32] Colonna-Romano G, Bulati M, Aquino A, et al. B cell immunosenescence in the elderly and in centenarians. Rejuvenation Res. 2008;11(2):433-439. [33] Geiger H, Van Zant G. The aging of lympho-hematopoietic stem cells. Nat Immunol. 2002;3(4):329-333. [34] Nicoletti C. Antibody protection in aging: influence of idiotypic repertoire and antibody binding activity to a bacterial antigen. Exp Mol Pathol. 1995;62(2):99-108. [35] Hasler P, Zouali M. Immune receptor signaling, aging, and autoimmunity. Cell Immunol. 2005;233(2):102-108. [36] 朱梅,金华,孙晓冬,等.针刺“足三里”、“关元”穴区对老年大鼠肝脏内巨噬细胞形态计量学影响[J].中国老年学杂志, 2004, 24(5):454-455. [37] 朱梅,高洪泉,刘瑞丰,等.针刺“足三里”、“关元”穴区对老年大鼠肝脏内巨噬细胞功能影响的实验研究[J].针灸临床杂志, 2003, 19(6):54-55. [38] 高希言,王燕.艾灸强壮要穴对衰老小鼠免疫功能的影响[J].河南中医,2005,25(11):24-26. [39] 李艳梅,宋立中,陈少宗.择时温针灸关元、足三里对老年人超氧化物歧化酶、T细胞亚群的影响[J].辽宁中医杂志, 2014,41(4): 779-781. [40] Pavão TS, Vianna P, Pillat MM, et al. Acupuncture is effective to attenuate stress and stimulate lymphocyte proliferation in the elderly. Neurosci Lett. 2010;484(1):47-50. [41] Zhang CY, Yang L, Chen HP, et al. Clinical study on anti-aging action of herbal cake-partition moxibustion. J Acupunct Tuina Sci. 2009,7(1):37-40. [42] 肖凌,明平红,宁勇,等.针灸调节衰老大鼠对疫苗反应性的实验研究[J].湖北中医药大学学报,2012,14(2):10-12. [43] Esteller M. The necessity of a human epigenome project. Carcinogenesis. 2006;27(6):1121-1125. [44] Yano S, Ghosh P, Kusaba H, et al. Effect of promoter methylation on the regulation of IFN-gamma gene during in vitro differentiation of human peripheral blood T cells into a Th2 population. J Immunol. 2003;171(5):2510-2516. [45] Richardson B. Impact of aging on DNA methylation. Ageing Res Rev. 2003;2(3):245-261. [46] Bollati V, Schwartz J, Wright R, et al. Decline in genomic DNA methylation through aging in a cohort of elderly subjects. Mech Ageing Dev. 2009;130(4):234-239. [47] Grolleau-Julius A, Ray D, Yung RL. The role of epigenetics in aging and autoimmunity. Clin Rev Allergy Immunol. 2010; 39(1): 42-50. [48] Saccani S, Natoli G. Dynamic changes in histone H3 Lys 9 methylation occurring at tightly regulated inducible inflammatory genes. Genes Dev. 2002;16(17):2219-2224. [49] Salminen A, Huuskonen J, Ojala J, et al. Activation of innate immunity system during aging: NF-kB signaling is the molecular culprit of inflamm-aging. Ageing Res Rev. 2008; 7(2):83-105. [50] Johnson AA, Akman K, Calimport SR, et al. The role of DNA methylation in aging, rejuvenation, and age-related disease. Rejuvenation Res. 2012;15(5):483-494. [51] Foulks JM, Parnell KM, Nix RN, et al. Epigenetic drug discovery: targeting DNA methyltransferases. J Biomol Screen. 2012;17(1):2-17. [52] Wang ZG, Wu JX. DNA methyltransferases: classification, functions and research progress. Yi Chuan. 2009;31(9): 903-912. [53] Li KK, Li F, Li QS, et al. DNA methylation as a target of epigenetic therapeutics in cancer. Anticancer Agents Med Chem. 2013;13(2):242-247. [54] Yang X, Lay F, Han H, et al. Targeting DNA methylation for epigenetic therapy. Trends Pharmacol Sci. 2010;31(11):536-546. [55] Leppert S, Matarazzo MR. De novo DNMTs and DNA methylation: novel insights into disease pathogenesis and therapy from epigenomics. Curr Pharm Des. 2014;20(11): 1812-1818. [56] Berletch JB, Andrews LG, Tollefsbol TO. A method to detect DNA methyltransferase I gene transcription in vitro in aging systems. Methods Mol Biol. 2007;371:73-80. [57] Jin B, Robertson KD. DNA methyltransferases, DNA damage repair, and cancer. Adv Exp Med Biol. 2013;754:3-29. [58] Luczak MW, Jagodziński PP. The role of DNA methylation in cancer development. Folia Histochem Cytobiol. 2006;44(3): 143-154. [59] 孔叶平.组蛋白乙酰化在艾灸抗大运动量后心肌细胞自由基中的作用机制研究[D].南京:南京中医药大学,2013. [60] Fu SP, He SY, Xu B, et al. Acupuncture promotes angiogenesis after myocardial ischemia through H3K9 acetylation regulation at VEGF gene. PLoS One. 2014;9(4):e94604. [61] 项洋.衰老小鼠巨噬细胞microRNA表达及其调控机制研究[D].北京:北京协和医学院,2011. [62] Bruniquel D, Schwartz RH. Selective, stable demethylation of the interleukin-2 gene enhances transcription by an active process. Nat Immunol. 2003;4(3):235-240. [63] García-Domínguez P, Dell'aversana C, Alvarez R, et al. Synthetic approaches to DNMT inhibitor SGI-1027 and effects on the U937 leukemia cell line. Bioorg Med Chem Lett. 2013;23(6):1631-1635. [64] Oike T, Komachi M, Ogiwara H, et al. C646, a selective small molecule inhibitor of histone acetyltransferase p300, radiosensitizes lung cancer cells by enhancing mitotic catastrophe. Radiother Oncol. 2014;111(2):222-227. |
| [1] | Shi Hua, Zhang Pu, Guo Xin, Jie Xiao-su. Effect of acupuncture on cerebral amino acids and neurotransmitters in the immature rat model of cerebral palsy [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(40): 5959-5965. |
| [2] | Chen Shu, Yong Chun-yan, Chen Heng, Chu Xiao, Zhang Chao, Tan Cheng, Ye Lan, Li Jiang-shan. Response of gastric-related neurons in the hypothalamic paraventricular nucleus to acupuncture at Neiguan and Zusanli in a rat model of gastric distension [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(5): 675-680. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||