Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (29): 4724-4729.doi: 10.3969/j.issn.2095-4344.2014.29.023
Previous Articles Next Articles
Development of articular cartilage repair technique
Ruan Zheng1, 2, Yin Qing-shui2, Zhang Yu2
- 1Graduate School of Southern Medical University, Guangzhou 510515, Guangdong Province, China; 2Department of Orthopedics, Guangzhou General Hospital of Guangzhou Military Area Command of Chinese PLA, Guangzhou 510010, Guangdong Province, China
-
Revised:2014-04-28Online:2014-07-09Published:2014-07-09 -
Contact:Yin Qing-shui, Professor, Doctoral supervisor, Department of Orthopedics, Guangzhou General Hospital of Guangzhou Military Area Command of Chinese PLA, Guangzhou 510010, Guangdong Province, China -
About author:Ruan Zheng, Studying for master’s degree, Graduate School of Southern Medical University, Guangzhou 510515, Guangdong Province, China; Department of Orthopedics, Guangzhou General Hospital of Guangzhou Military Area Command of Chinese PLA, Guangzhou 510010, Guangdong Province, China
CLC Number:
Cite this article
Ruan Zheng, Yin Qing-shui, Zhang Yu. Development of articular cartilage repair technique[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(29): 4724-4729.
share this article
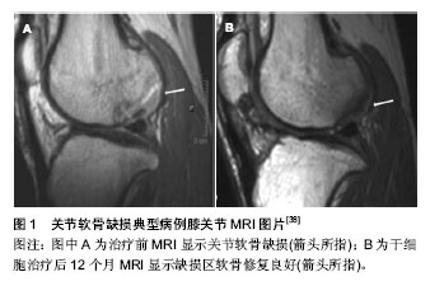
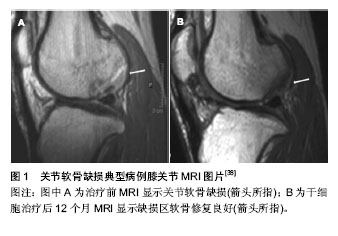
2.1 外科治疗软骨损伤修复 微骨折技术是临床上一种常见的治疗软骨损伤的方法。它是在软骨缺损区域对软骨下骨钻孔,形成局部血肿,通过骨髓间充质干细胞的增殖、分化对缺损区域进行修复[16]。这项技术最早由Steadman等[17]于1985年开始应用于临床,是对软骨下骨钻孔方法的进一步改进。微骨折典型适应证为小的(<2 cm2)单级Ⅲ、Ⅳ级软骨缺损并且不伴有软骨下骨缺损患者。Steadman在对233例患者进行了3年的临床随访后发现,有75%的患者的疼痛感减轻,随访11年后发现客观改善指数明显升高。但该方法修复形成的是纤维软骨,在机械性能和生物性能方面都较透明软骨有很大差距[18]。 在微骨折技术上改进的马赛克技术,也就是自体骨软骨移植技术,是在软骨缺损区域钻取圆形孔隙成马赛克样图案后,从自身非负重区域部位获取透明软骨后填塞入孔内进行修复[16]。骨膜和软骨膜均来自未分化的中胚层细胞,两者均具有形成软骨的能力。许多学者根据骨膜再生软骨这一原理,采用易获得的自体骨膜修复关节软骨损伤,结果显示骨膜能够再生类透明软骨,形成软骨陷窝,进而充填关节软骨缺损。自体骨软骨移植的优势在于通过移植可以使透明软骨而不是纤维软骨修复骨软骨缺损。因为非负重面软骨来源有限,自体骨软骨移植主要应用于小到中等的全厚骨软骨缺损病例(2.5 cm直径内)中并且软骨下骨深度最好不超过6 mm。 Hangody等[19-20]在19世纪90年代首先介绍了这种技术并且成功移植了自体透明软骨。Ollat等[21]对142例接受马赛克技术治疗软骨缺损的患者进行了平均时间为8年的随访,他们的平均缺损面积为2.29 cm2,有81.8%的患者在主观意识上较术前有明显的好转。自体骨软骨移植不存在移植物成活及排斥反应的问题,但其缺点是受到材料来源的限制,仍不能广泛应用。 同种异体骨软骨移植材料获得相对较易且可按需要预制成任意形状和大小,因此比自体骨软骨移植更为常用。近20年以来,关于同种异体骨软骨移植取得成功的报道相继出现。同种异体软骨移植可以修复直径大于 >3 cm,深度>1 cm的软骨损伤修复[22]。 Emans等[23]在兔的胫骨上段切除一块骨膜,以诱导骨膜来源的骨软骨形成,3 周后用诱导的骨软骨移植修复自体股骨髁的关节软骨缺损,结果证明,用这种方法诱导的骨膜来源的骨软骨能很好的修复关节软骨缺损。有人采用多个软骨片移植技术修复大面积软骨缺损,称为“马赛克镶嵌成型术”,软骨镶嵌式移植是治疗软骨缺损的新方 法[24]。 Muller等[25]对14例膝关节软骨损伤的患者采用Lysholm评分标准进行治疗前后的功能评分,显示治疗前4.7分,治疗后为7.2分,有明显的统计学意义。Gross等[26]报道胫骨平台及股骨髁移植物7.5年的存活率为85%,单纯股骨髁移植物10年随访存活率为85%,并且17年随访能观察到活的透明软骨,股骨侧10年生存率为85%,胫骨侧为80%。新鲜异体骨软骨移植取得了一定的临床疗效,但新鲜移植物存在免疫排斥、关节不稳定、移植物被吸收等并发症,导致远期有不同程度的软骨退变。 2.2 组织工程技术 自体软骨细胞移植是从人体的非负重区域的部分分离出一小块软骨后,在体外培养两三周,再移植到自身的缺损区,并覆盖上骨膜。实验证明此法可以生成以Ⅱ型胶原为主的透明软骨[27]。在此基础上衍生出了基质诱导的自体软骨细胞移植,即将含有自体软骨细胞的可降解的胶原基质移植到软骨缺损区[28]。研究发现,接受基质诱导的自体软骨细胞移植手术的患者,5年后的磁共振片显示修复的缺损区已与周围的软骨完全融合,并且疼痛较术前明显好转,但在关节活动评分上没有明显差 别[29]。目前在临床上,这两种技术都取得了比较可观的中期结果,但同时也存在着一些不足,比如:较大的花费及繁琐的细胞体外培养[18]。 2.2.1 细胞来源 软骨损伤修复组织工程研究中理想的种子细胞条件是易于分离和获取、体外增殖能力强、可以合成软骨特异性的细胞外基质成分,比如聚蛋白多糖和Ⅱ型胶原。目前研究最多的种子细胞就是软骨细胞和干细 胞[30]。 软骨细胞:软骨细胞是最显而易见的细胞来源,它可以在体外产生、维持并改造软骨的细胞外基质。NeoCart自体软骨细胞移植技术是目前兴起的第3代组织工程软骨修复技术,指在非负重区获取软骨细胞并扩增后,接种于Ⅰ型胶原构建的蜂巢样基质中,在带有生物力学刺激的生物反应器中培养。然后经小关节切口植入缺损区,采用生物黏合剂固定。在一项与微骨折技术相比较的Ⅱ期临床实验中发现,随访26个月后,其临床结果有显著性的提 高[31],但这种方法在治疗较大面积的软骨缺损时效果仍不理想。 Vijayan等[32]报道采用双层胶原膜技术治疗14例较大面积软骨缺损的患者,平均缺损面积为7.2 cm2,随访5年后,有12例患者取得了较满意的效果。但不足之处在于可获得的软骨细胞的数量非常少,从关节炎患者或者年老患者体内取出的自体软骨细胞的活力又非常的低下。最新的研究发现软骨细胞可以限制宿主的免疫反应[33],因此有研究者提出利用异体软骨进行修复,一项利用成年人的异体软骨,经过干燥和微粒化处理,移植到兔子和狒狒的动物模型中,结果证明有透明软骨的生成。而利用微骨折技术将未成年人的软骨移植到缺损区域后,也证明了有良好的疗效[34]。Tompkins[35]研究发现,利用青少年的软骨制成的颗粒可以对局限性的4级软骨损伤进行修复,修复效果较使用成年人的软骨效果明显。 干细胞:另一大热点领域就是利用间充质干细胞对软骨进行修复。间充质干细胞具有自我更新,自我维持及向多方向分化的功能,其中就包括了软骨细胞[36]。Nejadnik等[37]对72例患者进行了骨髓间充质干细胞与自体软骨细胞移植的疗效对比,在随访了24个月后发现两种方法的效果相当。骨髓间充质干细胞移植的优势在于只需要进行一次手术,并且有较低的供体并发率及较少的费用。 Haleem等[38]将间充质干细胞负载在血小板纤维蛋白胶支架上治疗5例患者的关节软骨缺损,12个月后,有3例患者的MRI检查提示缺损区软骨修复良好,与周围自身软骨平面一致(图1)。 Kasemkijwattana等[39]报道了利用骨髓间充质干细胞负载在胶原支架上后治疗两例骨性关节炎的患者,取得了满意的效果。Emadedin等[40]对6例骨性关节炎的患者单独注射骨髓间充质干细胞,6个月后在疼痛及关节活动上均取得了较好的进步。其中有3例患者的MRI显示有软骨的生成,缺损区域的软骨下骨的水肿也明显减少。最近 Lim等[41]将间充质干细胞与水凝胶支架相结合,移植入有软骨缺损的动物模型中,实验结果表明,99%的动物模型软骨缺损得到修复,其中84%生成的是透明样软骨。 浓缩骨髓含有高密度的干细胞,因此也可以用于治疗软骨损伤。Fortier等[42]利用马制造出关节损伤的动物模型,对比利用浓缩骨髓和单独使用微骨折技术的治疗效果。结果证明,利用浓缩骨髓治疗组在生成软骨的厚度及组织学表现上均优于单独微骨折治疗组。Giannini等[43]将浓缩骨髓负载于透明质酸薄膜上,用于治疗胫距关节面处的软骨损伤,随访1年后运用关节镜技术取出小块修复软骨进行组织切片分析,结果证明疗效与自体软骨细胞移植大致相仿。Gigante等[44]也证实了将浓缩骨髓与胶原蛋白支架相结合治疗软骨损伤,可以有透明软骨的生成,组织学及关节功能评分也可以得到明显的提高。 脂肪干细胞也是目前的一个研究方向,将脂肪干细胞与支架材料相结合,在治疗软骨缺损方面取得了满意的疗效[45-46]。Koh和Choi[47]研究从髌下脂肪垫分离出浓缩脂肪干细胞后用于治疗骨关节炎。将25例患者随机分成两组,实验组在清创处理后注射浓缩脂肪干细胞及富含血小板血浆,对照组在清创处理后只注射富含血小板血浆。随访16个月后,实验组患者恢复的情况更好,但和对照组相比没有差异。 滑膜干细胞是另一种可以用于治疗软骨损伤的干细胞来源,因为他和骨髓间充质干细胞相比,具有更强的向软骨分化的潜能[48],但是这方面的研究现在还处在前期临床实验阶段。有研究将骨膜瓣表面镶嵌胶原蛋白及滑膜干细胞,移植到膝关节全层软骨损伤的动物模型中,结果证明可以提高组织学评分,并与周围自身软骨结合良好。Shimomura等[49]研究报道将滑膜干细胞经过体外培养后嫁接在一种新颖的3D结构支架上,移植到动物模型的软骨缺损区域,结果证实有显著性的恢复效果,生成的软骨也接近于正常软骨。Hori等[50]在2011年提出了一项新颖的技术,即将具有磁性的滑膜干细胞注射入膝关节内,细胞可以自己聚集在软骨缺损区,并且可以完全修复关节软骨。 2.2.2 支架材料 作为细胞载体的支架材料也是研究的热点。生物支架的作用就是为种子细胞提供一个舒适的环境,通过促进细胞合成软骨基质去替换自体软骨损伤处的不利于软骨形成的细胞外基质,达到促进软骨修复的目的。 良好的生物支架需要符合以下几点要求:①在体内可以被生物降解,并且没有毒性和副作用。②材料为多孔性的,利于营养物质的进入和废弃物的排除。③可以促进细胞的生存、增殖、分化和细胞外基质的产生。④经固定后可以与软骨缺损处的组织一体化。⑤可以提供有效的机械支持。多种天然或合成的高分子材料已经用于软骨组织工程的研究当中[51]。 目前在软骨修复组织工程中应用最广泛的支架材料为聚-α-羟基脂类,尤其是聚乳酸和聚乙醇酸。其生物降解能力强,无毒副作用,并且已经被美国食品药品监督局(FDA)批准用于临床使用[52]。但是它的缺点在于不利于维持细胞表现为软骨细胞的形态,产生的细胞外基质的性能不高[53]。 最近,负载有鞘氨基醇及基质细胞衍生因子的支架被应用于动物试验中,结果证明有透明软骨的生成。单独负载鞘氨基醇及基质细胞衍生因子的支架也被证明可以诱导形成Ⅱ型胶原纤维,但没有联合负载的支架的效果良好。目前有许多方法可以等比例的放大支架结构,以便更好的修复较小面积的局部软骨损伤。脂剂微泡壳或者微泡管用做水凝胶支架的致孔剂被发现可以减小对溶质扩散的移植,并可以提高机械性能[54]。采用变形加载的方法可以使得修复的软骨在纤维平行度上与自身软骨更加的相似[55]。最近研究出了一种具有最有利结构的多聚甲醛胶体,他可以被用于靶向传递药物及释放组织抑制剂[56]。 近年来,水凝胶材料因其和软骨相似的双极性,及可以提供细胞生长并分化的环境而成为研究的热点。同时体外实验研究还发现将脂肪干细胞包裹在水凝胶体内可以促进干细胞向软骨方向的分化[57]。最近一种可生物降解的低聚糖复合水凝胶材料已被用于动物实验中,进行对软骨修复的研究[58]。这种水凝胶以含有胰岛素样生长因子1,转化生长因子β3的明胶颗粒的形式移植入体内,明胶颗粒可以起到缓释的作用。12周后,使用水凝胶及诱导因子组修复的软骨在形态及组织学得分上明显优于对照组。Ahearne等[59]也研究发现,将用纤维蛋白,琼脂糖或者吉兰糖胶制成的水凝胶缓释微球与脂肪干细胞相结合,在软骨缺损的动物模型体内也取得了良好的效果。 Sharma等[60]研究出了一种具有光反应性的水凝胶材料,与微骨折术相结合,证明有透明软骨的修复。该技术尤其在不规则软骨缺损时具有明显的优势。最近有研究发现一种新颖的微量3D编织技术用以制造编织物支架结构,并结合软骨细胞与水凝胶的混合物。这种结构表现出和自体软骨有相同的抗压强度,拉伸力和接触性能[61]。尽管这种支架结构还处于体外实验阶段,但其潜在的意义是深远的。其可以使得每个人根据自己的解剖特点而制定个体化的修复支架,并且支持立即负重及软骨的修复再生。"
| [1] Temenoff JS, Mikos AG.Review: tissue engineering for regeneration of articular cartilage. Biomaterials. 2000;21: 431-440
[2] Engel A.Osteoarthritis and body measurements. Vital Health Stat.1968;11:1-37
[3] Mano JF, Reis RL.Osteochondral defects: present situation and tissue engineering approaches.J Tissue Eng Regen Med. 2007;1(4):261-273.
[4] Swieszkowski W, Tuan BH, Kurzydlowski KJ, et al.Repair and regeneration of osteochondral defects in the articular joints. Biomol Eng.2007;24(5):489-495.
[5] Oldershaw RA. Cell sources for the regeneration of articular cartilage: the past, the horizon and the future. Int J Exp Pathol. 2012;93(6):389-400.
[6] Goldring MB, Otero M, Plumb DA,et al.Roles of inflammatory and anabolic cytokines in cartilage metabolism: signals and multiple effectors converge upon MMP-13 regulation in osteoarthritis. Eur Cell Mater.2011;21:202-220.
[7] Leong DJ, Hardin JA, Cobelli NJ, et al.Mechanotransduction and cartilage integrity. Ann N Y Acad Sci.2011;1240:32-37.
[8] Rosa SC, Rufino AT, Judas FM,et al.Role of glucose as a modulator of anabolic and catabolic gene expression in normal and osteoarthritic human chondrocytes. J Cell Biochem, 2011;112(10):2813-2824.
[9] Ito S, Sato M, Yamato M,et al.Repair of articular cartilage defect with layered chondrocyte sheets and cultured synovial cells. Biomaterials.2012;33(21):5278-5286.
[10] Detterline AJ, Goldstein JL, Rue JP,et al.Evaluation and treatment of osteochondritis dissecans lesions of the knee. J Knee Surg.2008;21(2):106-115.
[11] Kreuz PC, Steinwachs MR, Erggelet C,et al. Results after microfracture of full-thickness chondral defects in different compartments in the knee. Osteoarthr Cartil. 2006;14: 1119-1125.
[12] Redman SN, Oldfield SF, Archer CW.Current strategies for articular cartilage repair. Eur Cell Mater.2005;9:23-32.
[13] Bentley G, Biant LC, Carrington RW, et al. A prospective, randomised comparison of autologous chondrocyte implantation versus mosaicplasty for osteochondral defects in the knee.J Bone Joint Surg Br.2003;85:223-230.
[14] Hunziker EB.Articular cartilage repair: basic science and clinical progress. A review of the current status and prospects. Osteoarthr Cartil.2002;10:432-463.
[15] Hunziker EB.The elusive path to cartilage regeneration. Adv Mater.2009;21:3419-3424.
[16] Panseri S, Russo A, Cunha C, et al.Osteochondral tissue engineering approaches for articular cartilage and subchondral bone regeneration. Knee Surg Sports Traumatol Arthrosc. 2012;20(6):1182-1191.
[17] Steadman JR, Rodkey WG, Rodrigo JJ. Microfracture: surgical technique and rehabilitation to treat chondral defects. Clin Orthop Relat Res. 2001;(391 Suppl):S362-369.
[18] Grande DA, Sgaglione NA.Regenerative medicine: self-directed articular resurfacing: a new paradigm? Nat Rev Rheumatol. 2010;6(12):677-678.
[19] Hangody L, Kárpáti Z. New possibilities in the management of severe circumscribed cartilage damage in the knee. Magy Traumatol Ortop Kezseb Plasztikai Seb.1994;37(3):237-243.
[20] Hangody L, Kish G, Kárpáti Z, et al.Arthroscopic autogenous osteochondral mosaicplasty for the treatment of femoral condylar articular defects. Knee Surg Sports Traumatol Arthrosc. 1997;5(4):262-267.
[21] Ollat D, Lebel B, Thaunat M,et al.Mosaic osteochondral transplantations in the knee joint, midterm results of the SFA multicenter study. Orthop Traumatol Surg Res.2011;97 (8 Suppl): S160-166.
[22] Beaver RJ, Mahomed M, Backstein D,et al.Fresh osteochondral allografts for post-traumatic defects in the knee. A survivorship analysis. J Bone Joint Surg Br.1992;74(1): 105-110.
[23] Emans PJ, Hulsbosch M, Wetzels GM, et al.Repair of osteochondral defects in rabbits with ectopically produced cartilage.Tissue Eng.2005;11(11-12):1789-1796.
[24] Bartha L, Vajda A, Duska Z, et al. Autologous osteochondral mosaicplasty grafting. J Orthop Sports Phys Ther. 2006; 36(10):739-750.
[25] Muller S, Breederveld RS, Tuinebreijer WE. Results of Osteochondral Autologous Transplantation in the Knee. Open Orthop J. 2010;4:111-114.
[26] Gross AE, Aubin P, Cheah HK, et al. A fresh osteochondral allograft alternative. J Arthroplasty.2002;17(4 Suppl 1):50-53.
[27] Vasiliadis HS, Wasiak J. Autologous chondrocyte implantation for full thickness articular cartilage defects of the knee. Cochrane Database Syst Rev. 2010;(10):CD003323.
[28] Kon E, Filardo G, Di Matteo B,et al.Matrix assisted autologous chondrocyte transplantation for cartilage treatment: A systematic review. Bone Joint Res.2013;2(2):18-25.
[29] Ventura A, Memeo A, Borgo E,et al.Repair of osteochondral lesions in the knee by chondrocyte implantation using the MACI(R) technique. Knee Surg Sports Traumatol Arthrosc. 2012;20(1):121-126.
[30] Chung C, Burdick JA.Engineering cartilage tissue. Adv Drug Deliv Rev.2008;60:243-262.
[31] Crawford DC, DeBerardino TM, Williams RJ 3rd.Williams, NeoCart, an autologous cartilage tissue implant, compared with microfracture for treatment of distal femoral cartilage lesions: an FDA phase-II prospective, randomized clinical trial after two years. J Bone Joint Surg Am.2012;94(11):979-989.
[32] Vijayan S, Bartlett W, Bentley G, et al.Autologous chondrocyte implantation for osteochondral lesions in the knee using a bilayer collagen membrane and bone graft: a two-to eight-year follow-up study. J Bone Joint Surg Br.2012;94(4): 488-492.
[33] Adkisson HD, Milliman C, Zhang X, et al.Immune evasion by neocartilage derived chondrocytes: Implications for biologic repair of joint articular cartilage. Stem Cell Res. 2010;4(1): 57-68.
[34] Kruse DL, Ng A, Paden M, et al.Arthroscopic De Novo NT((R)) juvenile allograft cartilage implantation in the talus: a case presentation. J Foot Ankle Surg.2012;51(2):218-221.
[35] Tompkins M, Hamann JC, Diduch DR,et al.Preliminary Results of a Novel Single-Stage Cartilage Restoration Technique: Particulated Juvenile Articular Cartilage Allograft for Chondral Defects of the Patella. Arthroscopy. 2013;29(10): 1661-1670..
[36] Filardo G, Madry H, Jelic M,et al.Mesenchymal stem cells for the treatment of cartilage lesions: from preclinical findings to clinical application in orthopaedics.Knee Surg Sports Traumatol Arthrosc.2013;21(8):1717-1729.
[37] Nejadnik H, Hui JH, Feng Choong EP, et al.Autologous bone marrow-derived mesenchymal stem cells versus autologous chondrocyte implantation: an observational cohort study. Am J Sports Med.2010;38(6):1110-1116.
[38] Haleem AM, Singergy AA, Sabry D,et al.The Clinical Use of Human Culture-Expanded Autologous Bone Marrow Mesenchymal Stem Cells Transplanted on Platelet-Rich Fibrin Glue in the Treatment of Articular Cartilage Defects: A Pilot Study and Preliminary Results.Cartilage.2010;1(4): 253-261.
[39] Kasemkijwattana C, Hongeng S, Kesprayura S,et al. Autologous bone marrow mesenchymal stem cells implantation for cartilage defects: two cases report. J Med Assoc Thai.2011;94(3):395-400.
[40] Emadedin M, Aghdami N, Taghiyar L,et al.Intra-articular injection of autologous mesenchymal stem cells in six patients with knee osteoarthritis. Arch Iran Med.2012;15(7):422-428.
[41] Lim CT, Ren X, Afizah MH, et al.Repair of osteochondral defects with rehydrated freeze-dried oligo[poly(ethylene glycol) fumarate] hydrogels seeded with bone marrow mesenchymal stem cells in a porcine model. Tissue Eng Part A.2013;19(15-16):1852-1861.
[42] Fortier LA, Potter HG, Rickey EJ,et al.Concentrated bone marrow aspirate improves full-thickness cartilage repair compared with microfracture in the equine model. J Bone Joint Surg Am.2010;92(10):1927-1937.
[43] Giannini S, Buda R, Cavallo M, et al.Cartilage repair evolution in post-traumatic osteochondral lesions of the talus:from open field autologous chondrocyte to bone-marrow-derived cells transplantation. Injury.2010;41(11):1196-1203.
[44] Gigante A, Calcagno S, Cecconi S, et al.Use of collagen scaffold and autologous bone marrow concentrate as a one-step cartilage repair in the knee: histological results of second-look biopsies at 1 year follow-up. Int J Immunopathol Pharmacol.2011;24(1 Suppl 2):69-72.
[45] Dragoo JL, Carlson G, McCormick F,et al.Healing full-thickness cartilage defects using adipose-derived stem cells. Tissue Eng.2007;13(7):1615-1621.
[46] Im GI, Lee JH. Repair of osteochondral defects with adipose stem cells and a dual growth factor-releasing scaffold in rabbits. J Biomed Mater Res B Appl Biomater. 2010;92(2): 552-560.
[47] Koh YG, Choi YJ. Infrapatellar fat pad-derived mesenchymal stem cell therapy for knee osteoarthritis. Knee.2012;19(6): 902-907.
[48] De Bari C, Dell'Accio F, Karystinou A, et al.A biomarker-based mathematical model to predict bone-forming potency of human synovial and periosteal mesenchymal stem cells. Arthritis Rheum.2008;58(1): 240-250.
[49] Shimomura K, Ando W, Tateishi K,et al.The influence of skeletal maturity on allogenic synovial mesenchymal stem cell-based repair of cartilage in a large animal model. Biomaterials.2010;31(31):8004-8011.
[50] Hori J, Deie M, Kobayashi T,et al.Articular cartilage repair using an intra-articular magnet and synovium-derived cells.J Orthop Res.2011;29(4):531-538.
[51] Ahmed TA, Hincke MT. Strategies for articular cartilage lesion repair and functional restoration. Tissue Eng Part B Rev. 2010; 16(3):305-329.
[52] Yoon DM, Fisher JP.Chondrocyte signaling and artificial matrices for articular cartilage engineering. Adv Exp Med Biol. 2006;585:67-86.
[53] Chen G, Sato T, Ushida T,et al. The use of a novel PLGA fiber/collagen composite web as a scaffold for engineering of articular cartilage tissue with adjustable thickness.J Biomed Mater Res A.2003;67:1170-1180.
[54] Wang X, Wenk E, Zhang X,et al.Growth factor gradients via microsphere delivery in biopolymer scaffolds for osteochondral tissue engineering.J Control Release.2009; 134(2):81-90.
[55] Kelly TA, Ng KW, Wang CC, et al.Spatial and temporal development of chondrocyte-seeded agarose constructs in free-swelling and dynamically loaded cultures.J Biomech. 2006;39(8):1489-1497.
[56] Wu J, Chu CC.Block copolymer of poly(ester amide) and polyesters: synthesis, characterization, and in vitro cellular response.Acta Biomater.2012;8(12):4314-4323.
[57] Popa E, Reis R, Gomes M. Chondrogenic phenotype of different cells encapsulated in kappa-carrageenan hydrogels for cartilage regeneration strategies. Biotechnol Appl Biochem.2012;59(2):132-141.
[58] Kim K, Lam J, Lu S, et al.Osteochondral tissue regeneration using a bilayered composite hydrogel with modulating dual growth factor release kinetics in a rabbit model. J Control Release.2013;168(2):166-178.
[59] Ahearne M, Kelly DJ. A comparison of fibrin, agarose and gellan gum hydrogels as carriers of stem cells and growth factor delivery microspheres for cartilage regeneration. Biomed Mater.2013;8(3):035004.
[60] Sharma B, Fermanian S, Gibson M, et al.Human cartilage repair with a photoreactive adhesive-hydrogel composite. Sci Transl Med.2013;5(167):167ra6.
[61] Moutos FT, Freed LE, Guilak F.A biomimetic three-dimensional woven composite scaffold for functional tissue engineering of cartilage.Nat Mater.2007;6(2):162-167. |
| [1] | Li Xuan, Shi Chun-lai, Wang Xue-yan, Zhang Ming. Cardiovascular stent biocompatibility: bibliometric analysis of the international developmental trend [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(8): 1267-1271. |
| [2] | Tan Wei, Lv Hai, Zhou Chu-song. Acellular matrix scaffold for tissue-engineered intervertebral disc which is closest to the normal three-dimensional structure of the nucleus pulposus [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(8): 1289-1294. |
| [3] | Wu Yan, Huang Lan . Bone morphogenetic protein 9-induced osteogenic differentiation of dental follicle cells in vitro [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2255-2260. |
| [4] |
Du Wei-bin, Quan Ren-fu, Zheng Xuan, Wang Tuo.
Hair follicle stem cells promote the healing of skin wound
|
| [5] | Zhang Shi-song, Zhang Zhi-feng, Huang Jian. Connective tissue growth factor and articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(11): 1755-1760. |
| [6] | Zhang Bao-hua, Qiu Fu-cheng, Dong Ci1, Han Rui, Zhang Yong-zhi, Liu Hui-miao,Xie Bing-chuan, Zhang Li-na, Wang Wen-ting, Wang Yan-yong, Zhang Zhen-qing, Gu Ping,Yan Bao-yong. Neural stem cell transplantation for central nervous system diseases via the cerebrospinal fluid [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 974-978. |
| [7] | Huang Zhe. Correlation between Bmi-1 and clinicopathological features of colorectal cancer [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 894-899. |
| [8] | Zhang Ping-ping, Xiang Chuan. Bone marrow mesenchymal stem cells for osteoarthritis: its possibility and future [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 968-973. |
| [9] | Wang Teng-bin, Zhu Hui, Li Tian-shi . Development of chitosan and its derivative scaffolds for tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(52): 8498-8503. |
| [10] | Li Bing-ting, Jia Ying-zhen, Liu Zhi-fang, Song Yuan, Hou Xiao-wei. Effect of freeze-dried bone xenograft and platelet-rich fibrin compound on osteogenesis and osseointegration of alveolar bone defects [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(52): 8376-8381. |
| [11] | Lu Miao, Wang Xiang-ming, Wang Sen, Zhou Chuan-wei. Effects of different drug eluting stents on inflammatory factors in elderly patients with non-ST-segment elevation acute coronary syndrome [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(52): 8414-8419. |
| [12] | Su Da-ming, Zhao Jun-hua, Huang Zhi-yuan, Li Da-lian, Liu Jian-jun, Wang Min, Li Hua. Effect of Jianxi Qianggu Pill on the pathological changes of articular cartilage in knee osteoarthritis rabbits [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(5): 657-662. |
| [13] | Liu Xue-guang, Qiu Yong, Sun Zhen-zhong, Qian Bang-ping, Wang Shou-feng. Culture and identification of chondrocytes isolated from the vertebral endplate of patients with type I neurofibromatosis associated with atrophic changes in vitro [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(46): 7396-7400. |
| [14] | Liu Yang, Li Xiao-yan, Chen Pan-pan, Li Jun . Bone marrow stem cell transplantation for improving heart function of patients with acute myocardial infarction: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(41): 6689-6695. |
| [15] | Liu Chen, Wang Sheng-hao, Wang Yi-bin, Tang Xue-bin, Yang Hui-lin, Li Bin. Biocompatibility of annulus fibrosus-derived stem cells with porcine decellularized annulus fibrosus matrix [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(41): 6655-6660. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||