Chinese Journal of Tissue Engineering Research ›› 2015, Vol. 19 ›› Issue (14): 2278-2282.doi: 10.3969/j.issn.2095-4344.2015.14.025
Previous Articles Next Articles
Hair follicle stem cells promote the healing of skin wound
Du Wei-bin1, Quan Ren-fu2, Zheng Xuan2, Wang Tuo1
- 1Zhejiang University of TCM, Hangzhou 310053, Zhejiang Province, China; 2Xiaoshan District TCM Hospital of Hangzhou City, Hangzhou 311201, Zhejiang Province, China
-
Revised:2015-03-07Online:2015-04-02Published:2015-04-02 -
Contact:Quan Ren-fu, Professor, Doctoral supervisor, Xiaoshan District TCM Hospital of Hangzhou City, Hangzhou 311201, Zhejiang Province, China -
About author:Du Wei-bin, Studying for master’s degree, Zhejiang University of TCM, Hangzhou 310053, Zhejiang Province, China -
Supported by:the Public Benefit Technology Application Project for Social Development of Science and Technology Department of Zhejiang Province, No. 2010C330133; the Social Developmental Project of Xiaoshan District of Hangzhou, No. 2014205
CLC Number:
Cite this article
Du Wei-bin, Quan Ren-fu, Zheng Xuan, Wang Tuo. Hair follicle stem cells promote the healing of skin wound
share this article
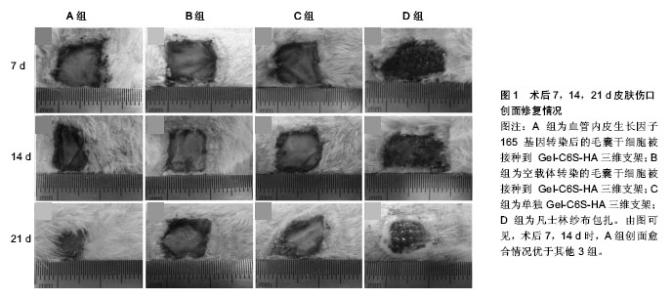
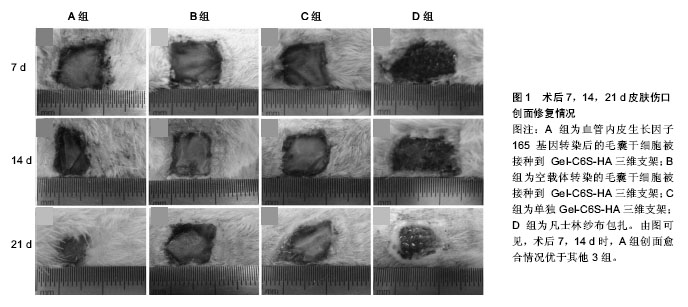
2.1 调控细胞分化功能 2.1.1 促早期血管化 血管新生参与了一系列生理病理过程,如创伤愈合、再生性循环、视觉成熟以及肿瘤形成等。目前所研究的组织工程皮肤,都不具备自身血管网结构,在植入体内后,需通过诱导周围组织血管长入,与周围组织建立血液循环,从而获得营养供给。因此,如何实现快速血管化是目前组织工程皮肤领域面临的重大难题之一[9-11]。 基因修饰毛囊干细胞:血管内皮生长因子在创伤愈合及组织工程皮肤早期血管化等方面均发挥重要作用。近年来,围绕血管内皮生长因子和毛囊干细胞开展的研究逐渐增多,其为组织工程皮肤的研究和治疗提供了新思路。血管内皮生长因子165是血管内皮细胞生长因子5种亚型之一,其活性最强,分布范围最广,是体内发挥作用的主要形式[12]。但血管内皮生长因子165的生物半衰期短,有学者利用慢病毒介导的血管内皮生长因子165基因修饰方法,对鼠毛囊干细胞进行转染,在体外得到高表达血管内皮生长因子165 mRNA和蛋白的鼠毛囊干细胞,可为组织工程构建人工毛囊、血管及皮肤等提供良好的种子细胞[13]。Quan等[14]经过改良进行体内研究,成功制备出适合毛囊干细胞培养的具有良好组织相容性和生物降解性的三维支架[15-16],再将血管内皮生长因子165基因修饰鼠毛囊干细胞接种入该支架,结果显示该方法能加速早期血管化并能促进皮肤伤口修复(图1),也提示着毛囊干细胞具有向内皮细胞分化的倾向和新生血管再生的潜能。 诱导分化成内皮细胞:内皮细胞在血管的发生和形成的过程中演绎着非常重要的角色。然而,内皮细胞自体取材来源相对有限,取材后极易引起血管的狭窄和闭塞,而使患者损伤加重,且经过原代分离培养的血管内皮细胞极易老化,增殖能力相当局限,细胞周期也较短[17-20]。利用合适的种子干细胞进行诱导,逐渐被科研人员所推崇。研究表明,绵羊毛囊干细胞中含有平滑肌前体细胞,纯化培养后可形成有收缩功能的平滑肌细胞,提示毛囊干细胞具有形成血管壁细胞的潜力[21-22]。许志成等[23]采用中性蛋白酶分离人毛囊干细胞,用含10 μg/L血管内皮生长因子、2 μg/L碱性成纤维细胞生长因子及体积分数为10%血清的EGM-2诱导液对其诱导。结果显示细胞形态逐步向内皮细胞的铺路石样形态转变,阳性表达率近80%,提示该体外诱导法可诱导人毛囊干细胞分化成血管内皮细胞,从而促进早期血管化,利于皮肤伤口修复。 2.1.2 促表皮及附属器官再生 正常状态下,毛囊干细胞不参与皮肤表皮层的自我更新。但当皮肤伤口出现时,毛囊干细胞可早于皮肤基底层表皮干细胞,率先被激活而迁移伤口边缘,来帮助伤口的修复和皮肤的再生。那么,如何利用毛囊干细胞的特性,加快伤口修复,构建良好的组织工程全层皮肤,也已成为研究领域的热点。 "

诱导分化成角质形成细胞:表皮角质形成细胞的发生在皮肤伤口修复中起到关键性的作用。在皮肤伤口的自我修复中,毛囊干细胞可一定程度分化为角质形成细胞参与其中。王瑶等用特定的培养基将毛囊干细胞定向分化为角质形成细胞,结果显示,毛囊干细胞特征性标记物CD200,CD29阴性表达,表皮分化细胞特征性角蛋白CK10阳性表达,表明其干细胞特性消失,成功诱导实现了向表皮角质形成细胞分化的特征。这些都从侧面阐明毛囊干细胞可以参与和加速表皮再生,从而促进了皮肤伤口的进一步修复[24-26]。 诱导分化成汗腺等细胞:毛发、汗腺、皮脂腺等作为皮肤的附属器官,在构建全层皮肤中有着重要的地位。毛囊干细胞通过一定手段的作用,调控毛发的增殖已无可厚非。近年来已有研究表明,通过对毛囊干细胞的定向诱导,可以将其成功分化为汗腺及皮脂腺细胞[26],这意味着以毛囊干细胞为种子细胞,通过有效的技术可以促使汗腺及皮脂腺的再生,不仅参与了皮肤伤口的修复,更促进了修复后皮肤的功能重建,为构建良好的组织工程全层皮肤提供了实验依据和种子细胞。 2.2 调控细胞内作用机制 2.2.1 调节信号通路 毛囊干细胞在细胞内信号通路的作用下,能够得到有效的增殖及分化,从而确保毛发增生与再上皮化过程的顺利进行,又能维持毛发等皮肤附属器的增殖与分化过程相对平衡,从而使皮肤组织的更新与自我修复达到生理水平。 Wnt信号通路:Wnt信号通路包括经典Wnt信号通路(Wnt/beta-catenine 信号通路)和10个非经典Wnt信号通路(Wnt /PCP、Wnt-PKA、Wnt-RYK信号通路等)。调节毛囊干细胞增殖和命运的Wnt信号通路,在毛囊循环的过程中呈一种动态变化,在生长期活性最高[27],因此Wnt信号通路在毛囊发生、干细胞聚集、迁移以及分化过程中发挥重要作用[28]。Wnt信号通路的主要信号分子β-catenin和Tcf3都能调节毛囊干细胞的分化状态,使其加快增殖,是组织再生的关键[29-32]。当β-catenin 信号通路被阻断时,可负性调节Wnt信号通路,从而促进毛囊干细胞向皮脂腺和毛囊滤泡间上皮分化。Tcf3能激活毛囊干细胞的状态,使其在毛囊基底层为替补细胞存在,最终分化参与毛囊和表皮的增殖再生。综合上述,在毛发生长早期,以Wnt信号通路为主,通过调节多种因素,能使毛囊干细胞发生加速增殖和分化,从而引起表皮相关成分的形成,促进创面的自我修复。 BMP信号通路:BMP在毛囊重建和通过调节毛发基质前体细胞的分化和增殖来控制毛囊循环中起到重要作用[33]。经典的BMP信号通过SMAD转录因子介导,也可以通过PTEN /AKT通路发生,从而调节正在进行增殖分化的干细胞,维持干细胞巢中的干细胞数量。而BMP-6、BMP抗体及滤泡抑素在BMP/TGF-β信号通路中有着重要的作用,在毛囊生成过程中,抑制毛囊循环的代谢来激活角质细胞的分化,也可以间接抑制其分泌信号的传递来促进毛发生成[34],而BMP和Wnt/β-catinen两条信号通路又相互制衡调节毛囊干细胞的毛囊周期和表皮再生[35]。 Notch信号通路:研究表明,Notch信号通路在毛囊和毛囊滤泡间的表达对毛囊干细胞的分化潜能具有一定的调控作用。毛囊干细胞内的RAM结构域可以介导Notch的胞内段与RBP-Jκ的结合,从而调节细胞分化基因,抑制毛囊干细胞向表皮细胞的分化,但这个过程却能促进其向毛囊滤泡间上皮、皮脂腺及毛囊的分化,还可通过 RBP-Jκ与其他信号通路的相互作用调节其增殖和分化的过程[36-37]。Delta-like 1(Dll1)、Jagged1和Jagged2三种配体在Notch信号通路的信号转导过程中发挥着重要作用,分别在不同的基层表达,调节毛囊干细胞的扩增和分化,从而促进创面的自我更新修复能力[38-39]。 2.2.2 调节转录因子及其他因素 LHX2转录因子:LHX2转录因子是皮肤损伤反应的重要调节器,在皮肤伤口修复中起着至关重要的作用。Mardaryev等[40]研究发现,LHX2在隆起的毛囊细胞中正调节Sox9和TCF4,并促进伤口再上皮化,并能同时负调节次级毛芽中的LGR5和抑制毛囊循环。此外,LHX2在毛囊干细胞的增殖分化中也起着关键作用。Folgueras等[41]利用全基因组染色质和转录谱等手段,显示LHX2可以反作用于那些可以协调细胞骨架动力学和附着力的基因。而LHX2缺失将导致毛囊干细胞无法维持静息状态,使得其逐步转变成皮脂腺,这将有利于皮肤的自我修复。当然有其他足够的启动信号累加时,毛囊干细胞可以被调动,从而推动组织再生和头发的生长。 Kruppel-like factor KLF4转录因子:Kruppel-like factor KLF4作为一种转录因子,在建立皮肤屏障功能中起着关键作用。Li等[42]使用KLF4/CreERTM和Rosa26RLacZ双转基因小鼠,在皮肤伤口愈合的情况下进行世系追踪,研究KLF4在参与伤口愈合中作用。结果表明,毛囊干细胞富集的小鼠皮肤角质形成细胞高表达KLF4,有利于皮肤伤口愈合。 BRG1酶类:BRG1是染色质重塑复合物的关键酶,在毛囊干细胞中动态表达,以控制组织的再生与修复。Xiong等[43]发现一个分子电路,集成了染色质重塑(BRG1),转录调控(NF-κB,Gli)和细胞间信号传导(SHH),从而在组织再生过程中控制隆起干细胞,以促进头发再生和创伤后早期表皮修复。 感觉神经元:感觉神经支配的调节减少与延迟伤口愈合有着密切的有关。Martínez-Martínez 等[44]通过使用辣椒素处理的方法来减少大鼠皮肤末梢感知,并运用体视学方法进行形态分析,推测感觉神经支配会影响毛囊干细胞的迁移和功能的发挥,而影响着皮肤伤口的愈合效率。 ILK整合素连接激酶:ILK是正常的表皮形态发生的关键所在,但其在毛囊干细胞和表皮再生中的作用有待探究。Nakrieko等[45]在毛囊干细胞的研究中,将角蛋白15中的ILK基因灭活,ILK的缺失妨碍了皮肤受损伤口愈合,同时极大地降低伤口闭合率。因此,ILK在皮肤损伤后的修复过程中扮演着重要的角色。"

| [1] 鲁彦玮,王志军.组织工程皮肤种子细胞和支架材料的研究现状[J].中国美容整形外科杂志,2011,22(6):373-376. [2] Cotsarelis G. Epithelial stem cells: a folliculocentric view. J Invest Dermatol. 2006;126(7):1459-1468. [3] 吴家昌,杨彤涛,许啸,等.人骨髓间充质干细胞体外向软骨细胞分化的实验研究[J].医学研究生学报,2010, 23(2):128-132. [4] Taylor G, Lehrer MS, Jensen PJ, et al. Involvement of follicular stem cells in forming not only the follicle but also the epidermis. Cell. 2000;102(4):451-461. [5] Yu H, Fang D, Kumar SM, et al. Isolation of a novel population of multipotent adult stem cells from human hair follicles. Am J Pathol. 2006;168(6):1879-1888. [6] Liu JY, Peng HF, Andreadis ST. Contractile smooth muscle cells derived from hair-follicle stem cells. Cardiovasc Res. 2008;79(1):24-33. [7] Xu ZC, Zhang Q, Li H. Human hair follicle stem cell differentiation into contractile smooth muscle cells is induced by transforming growth factor-β1 and platelet-derived growth factor BB. Mol Med Rep. 2013;8(6):1715-1721. [8] Xu ZC, Zhang Q, Li H. Differentiation of human hair follicle stem cells into endothelial cells induced by vascular endothelial and basic fibroblast growth factors. Mol Med Rep. 2014;9(1):204-210. [9] Wang X, Li Q, Hu X, et al. Fabrication and characterization of poly(L-lactide-co-glycolide) knitted mesh-reinforced collagen-chitosan hybrid scaffolds for dermal tissue engineering. J Mech Behav Biomed Mater. 2012;8:204-215. [10] Novosel EC, Kleinhans C, Kluger PJ. Vascularization is the key challenge in tissue engineering. Adv Drug Deliv Rev. 2011; 63(4-5):300-311. [11] Cassell OC, Hofer SO, Morrison WA, et al. Vascularisation of tissue-engineered grafts: the regulation of angiogenesis in reconstructive surgery and in disease states. Br J Plast Surg. 2002;55(8):603-610. [12] Robinson CJ, Stringer SE.The splice variants of vascular endothelial growth factor (VEGF) and their receptors. J Cell Sci. 2001;114(Pt 5):853-865. [13] 郑宣,许世超,全仁夫.慢病毒载体介导VEGF165基因修饰大鼠毛囊干细胞的实验研究[J].中国修复重建外科杂志,2014,28(2): 155-161. [14] Quan R, Zheng X, Xu S, et al. Gelatin-chondroitin-6- sulfate-hyaluronic acid scaffold seeded with vascular endothelial growth factor 165 modified hair follicle stem cells as a three-dimensional skin substitute. Stem Cell Res Ther. 2014;5(5):118. [15] Macneil S. Biomaterials for tissue engineering of skin. Mater Today. 2008; 11:26-35. [16] Wang TW, Sun JS, Wu HC,et al. Evaluation and biological characterization of bilayer gelatin/chondroitin-6-sulphate/hyaluronic acid membrane. J Biomed Mater Res B Appl Biomater. 2007;82(2):390-399. [17] Ge S, Pachter JS. Isolation and culture of microvascular endothelial cells from murine spinal cord. J Neuroimmunol. 2006;177(1-2):209-214. [18] Wu Z, Hofman FM, Zlokovic BV. A simple method for isolation and characterization of mouse brain microvascular endothelial cells. J Neurosci Methods. 2003;130(1):53-63. [19] Parkinson FE, Hacking C. Pericyte abundance affects sucrose permeability in cultures of rat brain microvascular endothelial cells. Brain Res. 2005;1049(1):8-14. [20] Cha ST, Talavera D, Demir E, et al. A method of isolation and culture of microvascular endothelial cells from mouse skin. Microvasc Res. 2005;70(3):198-204. [21] Amoh Y, Li L, Yang M, et al. Nascent blood vessels in the skin arise from nestin-expressing hair-follicle cells. Proc Natl Acad Sci U S A. 2004;101(36):13291-13295. [22] Liu JY, Peng HF, Andreadis ST. Contractile smooth muscle cells derived from hair-follicle stem cells. Cardiovasc Res. 2008;79(1):24-33. [23] 许志成,李宏,张群.毛囊干细胞体外诱导分化为血管内皮细胞的实验研究[J].组织工程与重建外科杂志,2012,8(5):241-244. [24] Abbas O, Mahalingam M. Epidermal stem cells: practical perspectives and potential uses. Br J Dermatol. 2009; 161(2):228-236. [25] Adameyko I, Lallemend F, Aquino JB, et al. Schwann cell precursors from nerve innervation are a cellular origin of melanocytes in skin. Cell. 2009;139(2):366-379. [26] 王瑶.毛囊干细胞的多分化潜能及其向汗腺细胞分化的实验研究[D].天津:南开大学,2013:52-95. [27] Merrill BJ, Gat U, DasGupta R, et al. Tcf3 and Lef1 regulate lineage differentiation of multipotent stem cells in skin. Genes Dev. 2001;15(13):1688-1705. [28] Watt FM, Estrach S, Ambler CA. Epidermal Notch signalling: differentiation, cancer and adhesion. Curr Opin Cell Biol. 2008; 20(2):171-179. [29] Demehri S, Kopan R. Notch signaling in bulge stem cells is not required for selection of hair follicle fate. Development. 2009; 136(6):891-896. [30] Cai J, Lee J, Kopan R, et al. Genetic interplays between Msx2 and Foxn1 are required for Notch1 expression and hair shaft differentiation. Dev Biol. 2009;326(2):420-430. [31] Choi YS, Zhang Y, Xu M, et al. Distinct functions for Wnt/β-catenin in hair follicle stem cell proliferation and survival and interfollicular epidermal homeostasis. Cell Stem Cell. 2013;13(6):720-733. [32] Deschene ER, Myung P, Rompolas P, et al. β-Catenin activation regulates tissue growth non-cell autonomously in the hair stem cell niche. Science. 2014;343(6177):1353-1356. [33] Botchkarev VA, Kishimoto J. Molecular control of epithelial-mesenchymal interactions during hair follicle cycling. J Investig Dermatol Symp Proc. 2003;8(1):46-55. [34] 耿松海,王剑利,王万卷,等.毛囊干细胞定位和体外向表皮分化[J].中国医学科学院学报, 2006, 28(3): 360-363. [35] 邬宗周,邓辉,袁定芬.毛囊干细胞参与创伤修复及相关信号通路[J].中国组织工程研究与临床康复,2009,13(23):4581-4584. [36] Batts SA, Shoemaker CR, Raphael Y. Notch signaling and Hes labeling in the normal and drug-damaged organ of Corti. Hear Res. 2009;249(1-2):15-22. [37] Paus R, Arck P, Tiede S. (Neuro-)endocrinology of epithelial hair follicle stem cells. Mol Cell Endocrinol. 2008;288(1-2): 38-51. [38] Sugiyama-Nakagiri Y, Akiyama M, Shimizu H. Hair follicle stem cell-targeted gene transfer and reconstitution system. Gene Ther. 2006;13(8):732-737. [39] Demehri S, Kopan R. Notch signaling in bulge stem cells is not required for selection of hair follicle fate. Development. 2009;136(6):891-896. [40] Mardaryev AN, Meier N, Poterlowicz K, et al. Lhx2 differentially regulates Sox9, Tcf4 and Lgr5 in hair follicle stem cells to promote epidermal regeneration after injury. Development. 2011;138(22):4843-4852. [41] Folgueras AR, Guo X, Pasolli HA, et al. Architectural niche organization by LHX2 is linked to hair follicle stem cell function. Cell Stem Cell. 2013;13(3):314-327. [42] Li J, Zheng H, Wang J, et al. Expression of Kruppel-like factor KLF4 in mouse hair follicle stem cells contributes to cutaneous wound healing. PLoS One. 2012;7(6):e39663. [43] Xiong Y, Li W, Shang C, et al. Brg1 governs a positive feedback circuit in the hair follicle for tissue regeneration and repair. Dev Cell. 2013;25(2):169-181. [44] Martínez-Martínez E, Galván-Hernández CI, Toscano-Márquez B, et al. Modulatory role of sensory innervation on hair follicle stem cell progeny during wound healing of the rat skin. PLoS One. 2012;7(5):e36421. [45] Nakrieko KA, Rudkouskaya A, Irvine TS, et al. Targeted inactivation of integrin-linked kinase in hair follicle stem cells reveals an important modulatory role in skin repair after injury. Mol Biol Cell. 2011;22(14):2532-2540. |
| [1] | Wu Yan, Huang Lan . Bone morphogenetic protein 9-induced osteogenic differentiation of dental follicle cells in vitro [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2255-2260. |
| [2] | Huang Zhe. Correlation between Bmi-1 and clinicopathological features of colorectal cancer [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 894-899. |
| [3] | Zhang Bao-hua, Qiu Fu-cheng, Dong Ci1, Han Rui, Zhang Yong-zhi, Liu Hui-miao,Xie Bing-chuan, Zhang Li-na, Wang Wen-ting, Wang Yan-yong, Zhang Zhen-qing, Gu Ping,Yan Bao-yong. Neural stem cell transplantation for central nervous system diseases via the cerebrospinal fluid [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 974-978. |
| [4] | Liu Yang, Li Xiao-yan, Chen Pan-pan, Li Jun . Bone marrow stem cell transplantation for improving heart function of patients with acute myocardial infarction: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(41): 6689-6695. |
| [5] | Tang Ying, Liu Li-na, Wang Qing-min, Liu Chang, Yang Yi-li. Performance evaluation of a cerebrovascular stent coated with bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(34): 5503-5508. |
| [6] | Zhang Jia, Hu Su-qin, Yan Lu, Wang Gui-ling. Clinical application of chitin medical wound dressing paste [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(34): 5555-5560. |
| [7] | Ruan Zheng, Yin Qing-shui, Zhang Yu. Development of articular cartilage repair technique [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(29): 4724-4729. |
| [8] | Chen Peng, Zhang Jie, Rong Dong-ming, Han Zhong-yu, Yuan Si-jie, Tian Jing. Effects of non-dextran coated superparamagnetic iron oxide nanoparticles on proliferation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(16): 2526-2531. |
| [9] | Bai Shu-meng, Liu Xi. A three-dimensional nanofiber scaffold provides an appropriate microenvironment for stem cell regulation [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(16): 2594-2600. |
| [10] | Qian Xiao-ling, Zhou Xue-qin, Yin Xiao-Li, Yang Xue-mei, Zhang Xuan-fen, Li Xiao-hui, Zhang Qing-xia. Effect of vacuum sealing drainage on nerve growth factor and microvessels in chronic wound [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(11): 1647-1652. |
| [11] | Li Ye, Bao Xu, Chen Xi, Jia Xin-ru, Xu Song-shan, Che Yong-zhe. Thymosin beta4 increases mouse hair regeneration [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(11): 1687-1693. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||