Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (16): 2532-2537.doi: 10.3969/j.issn.2095-4344.2014.16.012
Previous Articles Next Articles
Preparation and cytocompatibility of cuttlebone-transformed hydroxyapatite
Huang Xie-shan1, Liu Ming2, Wang Jiang2
- 1Oral Center, Affiliated Haikou Hospital, Xiangya School of Medicine, Central South University, Haikou 570228, Hainan Province, China; 2Materials and Chemical Engineering Department of Hainan University, Ministry of Education Key Laboratory of Application Technology of Hainan Superior Resources Chemical Materials, Haikou 570228, Hainan Province, China
-
Online:2014-04-16Published:2014-04-16 -
Contact:Wang Jiang, M.D., Associate professor, Materials and Chemical Engineering Department of Hainan University, Ministry of Education Key Laboratory of Application Technology of Hainan Superior Resources Chemical Materials, Haikou 570228, Hainan Province, China -
About author:Huang Xie-shan, Master, Associate chief physician, Oral Center, Affiliated Haikou Hospital, Xiangya School of Medicine, Central South University, Haikou 570228, Hainan Province, China -
Supported by:the Major Science and Technology Plan of Hainan Province, No. ZDXM20120066; the Natural Science Foundation of Hainan Province, No. 511106
CLC Number:
Cite this article
Huang Xie-shan, Liu Ming, Wang Jiang. Preparation and cytocompatibility of cuttlebone-transformed hydroxyapatite[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(16): 2532-2537.
share this article
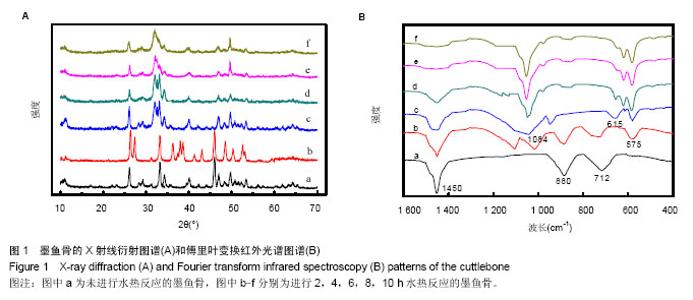
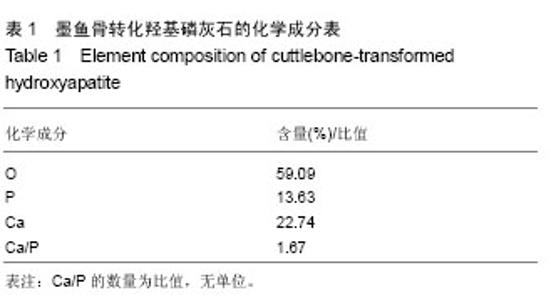
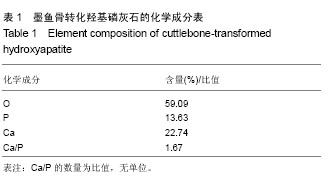
2.1 墨鱼骨转化羟基磷灰石材料的制备结果 图1A是水热反应前(原墨鱼骨)及不同水热反应时间样品粉末的X射线衍射图谱,图1A中图谱a经X射线衍射图谱处理软件JADE进行分析,得到其各衍射峰的d值与JCPDSNo.05-0453完全一致,表明构成墨鱼骨的矿物是文石相碳酸钙结构。图1中图谱b,c,d,e,f对应样品的水热反应时间分别为2,4,6,8,10 h。用MDI Jade 7.0软件分别对各图谱进行分析,得出在水热反应2 h后,出现了羟基磷灰石物相,其晶型结构也发生了变化。随着反应的进行,墨鱼骨文石矿物相逐步向羟基磷灰石物相转化,图中b,c,d是文石和羟基磷灰石的衍射峰共存。在反应时间4 h时,羟基磷灰石衍射峰强度远高于文石,说明水热反应4 h后,大部分文石相碳酸钙已转化为羟基磷灰石。图中e其各衍射峰与JCPDSNo.74-0566相符,说明在反应时间达到8 h时,文石相碳酸钙已完全转化成羟基磷灰石,图中f图谱与e图谱完全一样,说明水热反应8 h后样品的晶型结构稳定无变化。 图1B是水热反应前(原墨鱼骨)及不同水热反应时间样品粉末的傅里叶变换红外光谱图谱。图1B中图谱a与文石相碳酸钙的傅里叶变换红外光谱图相符[34],表明构成墨鱼骨的矿物是文石相碳酸钙。其CO32-不对称伸缩振动峰(1 450 cm-1强峰)、面外变形振动峰(880 cm-1中强峰)和面内变形振动峰(712 cm-1弱峰)的吸收强度随着水热反应的进行逐渐变弱(图谱b-d),在反应达到8 h(图谱e)时消失。在反应达到2 h(图谱b)时,出现了P-O振动峰(1 084 cm-1和575 cm-1)和OH-伸缩振动峰(615 cm-1),其吸收强度随着水热反应时间的进行石的傅里叶红外光谱图谱相符。表明墨鱼骨文石相碳酸钙在磷酸盐溶液水热反应作用下,逐步转化为羟基磷灰石,并且转化程度随着反应时间的延长而增加。 文石矿物在磷酸盐溶液中水热转变为羟基磷灰石,通常被认为是发生了局部规整离子交换反应,因为羟基磷灰石(六方晶系,a=0.943 nm,b=0.688 nm,g=1 200)和文石相碳酸钙(a=0.939 nm,b=0.574 nm,g=1 170,可以描述为假六方晶系)的晶格参数相近[34],离子所处的位置大致相同[35],当文石相碳酸钙经局部规整离子交换反应转化为羟基磷灰石时,由于减少了成核作用所需结构重排的能量,从而使反应易进行,表部文石相碳酸钙直接与磷酸盐发生水热反应,内部文石相碳酸钙不与溶液直接接触,易与扩散进去的PO43-,OH-离子发生固态局部规整离子交换反应转化成羟基磷灰石,反应从外到内,所以是一个随时间逐步转化的过程。 2.2 墨鱼骨转化羟基磷灰石材料的成分和形貌结构 图2A是利用X射线光电子能谱法分析的预处理过的墨鱼骨的X射线能谱法谱图,图2B是利用X射线能谱法分析水热反应 8 h的产物的X射线能谱法谱图,结果显示墨鱼骨的主要化学成分文石相碳酸钙在水热反应8 h的产物中的C元素已经检测不到,检测到元素主要为Ca,O,P元素[36]。X射线能谱法微区定量分析结果如表1所示,通过计算得到水热反应产物的Ca/P为1.67,与羟基磷灰石Ca10(PO4)6(OH)2的化学计量比相符合。说明在水热反应过程中,经磷酸盐溶液作用,反应产物中已不存在文石相碳酸钙,完全转化为羟基磷灰石。"
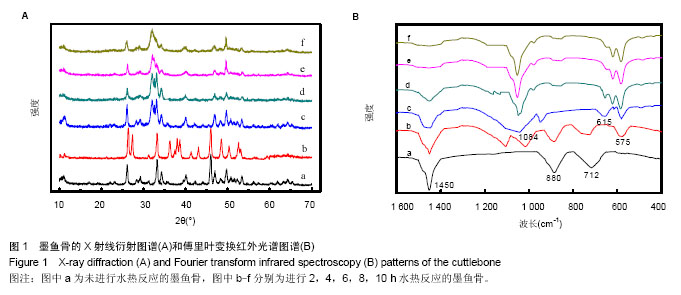
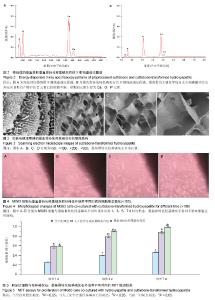
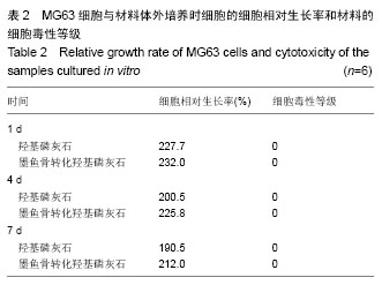
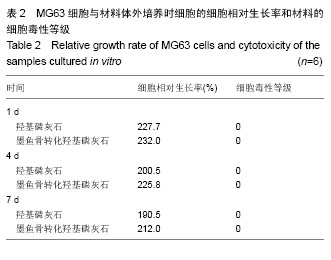
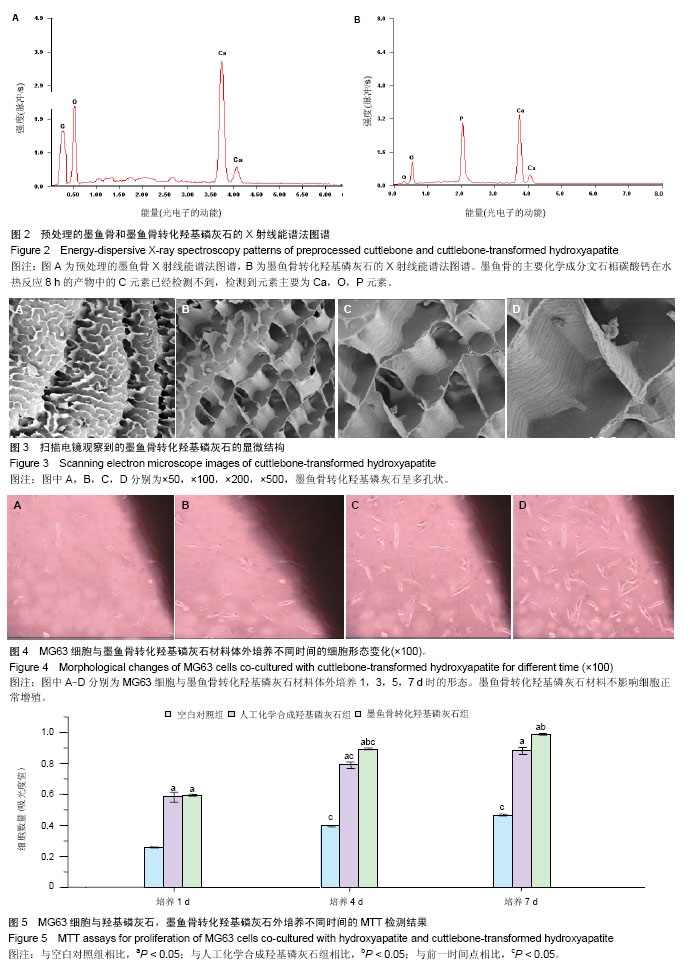
图3是墨鱼骨转化羟基磷灰石的扫描电镜照片,大体观察为瓷白色,背部呈肉眼可见的多孔状外观,纹理清晰(图3A)。可观察到有较粗大的支柱样结构,支柱之间相互平行, 即为肉眼可见之清晰纹理,相邻支柱间是与之相交排列的小梁结构,壁较薄,支柱与小梁相间排列。 表面光滑,并呈现出板层样(图3B),气孔率高达80%以上。构成规则的壁龛样网格(图3C),由“横墙”和“竖壁”规则搭建而成多孔通道结构,而且这些孔道是相通的,孔道开口大体呈长方形,大小较均匀,尺寸为200-300 μm(图3D)。 2.3 墨鱼骨转化羟基磷灰石的细胞相容性 材料与细胞复合培养1 d,光镜观察到材料周边有少量细胞,细胞形态不够清晰,细胞延展的不够充分(图4A)。材料与细胞复合培养3 d,材料周边的细胞数量有所增多,细胞形态趋于清晰,细胞贴材料周边生长,生长状态良好,但生长活力不够旺盛(图4B)。材料与细胞复合培养5 d,细胞数量明显增加,细胞贴附材料生长良好,细胞形态可分辨为梭形、三角形和多边形(图4C)。材料与细胞复合培养7 d,材料周边的细胞数量明显增多,布满材料周边胞浆透明,生长活力旺盛,细胞形态更加清晰,多为梭形、三角形和多边形,无细胞衰老及异常分裂现象(图4D),可见墨鱼骨转化羟基磷灰石材料不影响细胞正常的增殖。 图5是试验组和对照组MG63细胞的MTT检测结果,根据吸光度值随着细胞数的增加而增加,两者呈现明显的正相关关系[37],各组材料的细胞随着培养时间的延长,数量都有所增加,在各个时间点各组之间细胞的数量差异有显著性意义(P < 0.05)。 表2为MG63细胞与材料体外培养1,4和7 d时细胞的相对生长率(细胞相对生长率按下式计算:细胞相对生长率= 其中AE和AC分别是实验组和对照组的吸光度值)。按照GB/T16886.5-2003标准[38],可以看出材料的细胞毒性为0级,属于无细胞毒性的材料,从细胞相容性方面的研究结果,可知材料对MG63细胞的生长无明显影响且无明显毒性,属于无细胞毒性材料,具有较好的细胞相容性。 "
| [1] Ozin GA, Varaksa N, Coombs N, et al. Bone mimetics: a composite of hydroxyapatite and calciumdodecylphosphate lamellar phase. J Mater Chem. 1997;7:1601-1607. [2] Doremus RH. Bioceramics. J Mater Sci. 1992;27(2):285-297. [3] Suchanek W, Yoshimura M. Processing and properties of hydroxyapatite-based biomaterials for use as hard tissue replacement implants. J Mater Res. 1998;13(1):94-117. [4] Burstein FD, Cohen SR, Hudgins R, et al. The use of porous granular hydroxyapatite in secondary orbitocranial reconstruction. Plast Reconstr Surg. 1997;100(4):869-874. [5] Choi SH, Levy ML, McComb JG. A method of cranioplasty using coralline hydroxyapatite. Pediatr Neurosurg. 1998; 29(6): 324-327. [6] Boden SD, Martin GJ Jr, Morone M, et al. The use of coralline hydroxyapatite with bone marrow, autogenous bone graft, or osteoinductive bone protein extract for posterolateral lumbar spine fusion. Spine (Phila Pa 1976). 1999;24(4):320-327. [7] Wasielewski RC, Sheridan KC, Lubbers MA. Coralline hydroxyapatite in complex acetabular reconstruction. Orthopedics. 2008;31(4):367. [8] Thalgott JS, Giuffre JM, Fritts K, et al. Instrumented posterolateral lumbar fusion using coralline hydroxyapatite with or without demineralized bone matrix, as an adjunct to autologous bone. Spine J. 2001;1(2):131-137. [9] Ono I, Gunji H, Kaneko F, et al. Treatment of extensive cranial bone defects using computer-designed hydroxyapatite ceramics and periosteal flaps. Plast Reconstr Surg. 1993; 92(5): 819-830. [10] Fu K, Meng ZB, Li J, et al. Repairing the defect of benign bone tumor with the coralline hydroxyapatite. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2008;33(5):421-424. [11] Wolfe SW, Pike L, Slade JF 3rd, et al. Augmentation of distal radius fracture fixation with coralline hydroxyapatite bone graft substitute. J Hand Surg Am. 1999;24(4):816-827. [12] Thalgott JS, Klezl Z, Timlin M, et al. Anterior lumbar interbody fusion with processed sea coral (coralline hydroxyapatite) as part of a circumferential fusion. Spine (Phila Pa 1976). 2002; 27(24):E518-527. [13] Gosain AK, Riordan PA, Song L, et al. A 1-year study of osteoinduction in hydroxyapatite-derived biomaterials in an adult sheep model: part II. Bioengineering implants to optimize bone replacement in reconstruction of cranial defects. Plast Reconstr Surg. 2004;114(5):1155-1165. [14] Ning Y, Wei T, Defu C, et al. The research of degradability of a novel biodegradable coralline hydroxyapatite after implanted into rabbit. J Biomed Mater Res A. 2009;88(3):741-746. [15] Cohen AJ, Dickerman RD, Schneider SJ. New method of pediatric cranioplasty for skull defect utilizing polylactic acid absorbable plates and carbonated apatite bone cement. J Craniofac Surg. 2004;15(3):469-472. [16] Roy DM, Linnehan SK. Hydroxyapatite formed from coral skeletal carbonate by hydrothermal exchange. Nature. 1974; 247(5438):220-222. [17] Hollinger JO, Brekke J, Gruskin E, et al. Role of bone substitutes. Clin Orthop Relat Res. 1996;(324):55-65. [18] Lane JM, Tomin E, Bostrom MP. Biosynthetic bone grafting. Clin Orthop Relat Res. 1999;(367 Suppl):S107-117. [19] Voor MJ, Arts JJ, Klein SA, et al. Is hydroxyapatite cement an alternative for allograft bone chips in bone grafting procedures? A mechanical and histological study in a rabbit cancellous bone defect model. J Biomed Mater Res B Appl Biomater. 2004; 71(2):398-407. [20] Chen F, Mao T, Tao K, et al. Bone graft in the shape of human mandibular condyle reconstruction via seeding marrow-derived osteoblasts into porous coral in a nude mice model. J Oral Maxillofac Surg. 2002;60(10):1155-1159. [21] Sivakumar M, Kumar TS, Shantha KL, et al. Development of hydroxyapatite derived from Indian coral. Biomaterials. 1996; 17(17):1709-1714. [22] Birchall JD, Thomas NL. On the architecture and function of cuttlefish bone. J Mater Sci. 1983;18(7):2081-2086. [23] Kumar GS, Girija EK, Thamizhavel A, et al. Synthesis and characterization of bioactive hydroxyapatite-calcite nanocomposite for biomedical applications. J Colloid Interface Sci. 2010;349(1):56-62. [24] Battistella E, Mele S, Foltran I, et al. Cuttlefish bone scaffold for tissue engineering: a novel hydrothermal transformation, chemical-physical, and biological characterization. J Appl Biomater Funct Mater. 2012;10(2):99-106. [25] Kannan S, Rocha JH, Agathopoulos S, et al. Fluorine-substituted hydroxyapatite scaffolds hydrothermally grown from aragonitic cuttlefish bones. Acta Biomater. 2007;3(2):243-249. [26] Jia X, Qian W, Wu D, et al. Cuttlebone-derived organic matrix as a scaffold for assembly of silver nanoparticles and application of the composite films in surface-enhanced Raman scattering. Colloids Surf B Biointerfaces. 2009;68(2): 231-237. [27] Milovac D, Gallego Ferrer G, Ivankovic M, et al. PCL-coated hydroxyapatite scaffold derived from cuttlefish bone: morphology, mechanical properties and bioactivity. Mater Sci Eng C Mater Biol Appl. 2014;34:437-445. [28] Fernandez JM, Molinuevo MS, Cortizo MS, et al. Development of an osteoconductive PCL-PDIPF-hydroxyapatite composite scaffold for bone tissue engineering. J Tissue Eng Regen Med. 2011;5(6): e126-135. [29] Arafat MT, Lam CX, Ekaputra AK, et al. Biomimetic composite coating on rapid prototyped scaffolds for bone tissue engineering. Acta Biomater. 2011;7(2):809-820. [30] Kim BS, Yang SS, Lee J. A polycaprolactone/cuttlefish bone-derived hydroxyapatite composite porous scaffold for bone tissue engineering. J Biomed Mater Res B Appl Biomater. 2013. in press. [31] Li X, Zhao Y, Bing Y, et al. Biotemplated syntheses of macroporous materials for bone tissue engineering scaffolds and experiments in vitro and vivo. ACS Appl Mater Interfaces. 2013;5(12):5557-5562. [32] Jia X, Qian W. Simultaneous synthesis and assembly of gold nanoparticles in cuttlebone-derived organic matrix: a "green" pathway for gold nanocomposite. J Nanosci Nanotechnol. 2008; 8(9):4370-4376. [33] Ogasawara W, Shenton W, Davis SA, et al. Template mineralization of ordered macroporous chitin−silica composites using a cuttlebone-derived organic matrix. Chem Mater. 2000;12(10):2835-2837. [34] Taves DR. Similarity of octacalcium phosphate and hydroxyapatite structures. Nature. 1963;200:1312-1313. [35] 刘金华,王大志.乌贼骨及其水热改性制备羟基磷灰石的研究[J].无机材料学报,2006,21(2):433-438. [36] 胡浩,朱胜利,崔振铎,等.TiZrCuPd金属玻璃表面沉积钙磷层的研究[J].功能材料,2010,41(10):1845-1847. [37] 杨浩,王春婷,吴玉梅,等.MTT试验中细胞特性状态及细胞数与OD值的关系探讨[J].动物医学进展,2002,23(5):49-53. [38] 国家药品监督管理局.GB/T 16886.5-2003 医疗器械生物学评价 第5部分:体外细胞毒性试验[S].北京:中国标准出版社,2003 |
| [1] | Song Hua, Ren Xiang-qian, Wei Dong-xing. Nano-hydroxyapatite effects on bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(8): 1155-1159. |
| [2] | Xue Zhen, Niu Li-yuan, An Gang, Guo Ya-shan, Lv Song-cen. Feasibility of nano-hydroxyapatite/chitosan/calcium sulphate hemihydrate as an injectable bone tissue engineering scaffold [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(8): 1160-1164. |
| [3] | Sun Qing-zhi . Nano-hydroxyapatite/silk fibroin bone substitute for repairing bone defects [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(8): 1190-1194. |
| [4] | Li Xuan, Shi Chun-lai, Wang Xue-yan, Zhang Ming. Cardiovascular stent biocompatibility: bibliometric analysis of the international developmental trend [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(8): 1267-1271. |
| [5] | He Yan-ping, Ma De-chun, Li Lei, Zhang Li, Zheng Shuang, Dong Ke-xin. Hemocompatibility and surface modification of artificial blood vessel materials [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(8): 1272-1276. |
| [6] | Tan Wei, Lv Hai, Zhou Chu-song. Acellular matrix scaffold for tissue-engineered intervertebral disc which is closest to the normal three-dimensional structure of the nucleus pulposus [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(8): 1289-1294. |
| [7] | Xian Ai-ming, Zhang Xiao-gang, Cao Li, Xu En-jie, Peng Li-bin, Hu Yang. In vitro antibacterial activity and biological safety of nano-silver composite coating on the surface of titanium base [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(52): 8387-8392. |
| [8] | Lu Miao, Wang Xiang-ming, Wang Sen, Zhou Chuan-wei. Effects of different drug eluting stents on inflammatory factors in elderly patients with non-ST-segment elevation acute coronary syndrome [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(52): 8414-8419. |
| [9] | Fan Ke-xia, Ma Yuan2, Yu Si-xun, Kuang Yong-qin, Yang Li-bin, Xia Xun, Zhao You-guang, Yang Wei-zhong, Gu Jian-wen. Preparation and in vitro biocompatibility of hydroxyapatite/polyphenylene sulfide composites [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(52): 8443-8449. |
| [10] | Feng Chao, Li Zhe, Lv Xiang-guo, Xu Yue-min, Fu Qiang. In vitro preparation and biochemical evaluation of oxygen generative keratin/silk fibroin compound biomaterial [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(52): 8480-8486. |
| [11] | Wang Teng-bin, Zhu Hui, Li Tian-shi . Development of chitosan and its derivative scaffolds for tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(52): 8498-8503. |
| [12] | Geng Hai-xia, Guo Xiu-juan, Qian Jun-rong, Feng Wei . Imaging evaluation of hydroxyapatite/gel nano-composite in rabbit skull defect repair [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(34): 5413-5417. |
| [13] |
Zeng Xian-li, Yang Chun-lu.
Repairing critical-size segmental defects: morphology of tissue engineering bone scaffolds and its effects on cell loading
|
| [14] | Ruan Zheng, Yin Qing-shui, Zhang Yu. Development of articular cartilage repair technique [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(29): 4724-4729. |
| [15] | Li Jia-feng, Cui Qun, Sun Xiu-ying, Xu Lei, Sun Jin-hu, Han Jian-guo. Nano-hydroxyapatite/polycaprolactone electrospinning scaffolds repair bone defects around the immediate implant [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(16): 2557-2562. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||