Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (47): 8255-8262.doi: 10.3969/j.issn.2095-4344.2013.47.018
Previous Articles Next Articles
Research progress and clinical practice of TiO2 nanotubes
Zhang Hang-zhou1, Wang Lin1, Tian Ang2, Sun Yu1, Bai Xi-zhuang1, Xue Xiang-xin2
- 1Department of Sport Medicine and Joint Surgery, the First Hospital of China Medical University, Shenyang 110001, Liaoning Province, China;2 School of Material and Metallurgy, Northeastern University, Shenyang 110001, Liaoning Province, China
-
Revised:2013-08-19Online:2013-11-19Published:2013-11-19 -
Contact:Bai Xi-zhuang, Ph.D., Doctoral supervisor, Chief physician, Department of Sport Medicine and Joint Surgery, the First Hospital of China Medical University, Shenyang 110001, Liaoning Province, China zpmhh@sina.com -
About author:Zhang Hang-zhou☆, Studying for doctorate, Department of Sport Medicine and Joint Surgery, the First Hospital of China Medical University, Shenyang 110001, Liaoning Province, China zhanghz1000@sina.com -
Supported by:the National Natural Science Foundation of China (General Program), No. 81071449*, L2010645*
CLC Number:
Cite this article
Zhang Hang-zhou, Wang Lin, Tian Ang, Sun Yu, Bai Xi-zhuang, Xue Xiang-xin. Research progress and clinical practice of TiO2 nanotubes[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(47): 8255-8262.
share this article

General conditions of included data There were 24 literaturesconcerning implant-related infection[1-24], 23 literaturesaddressing mechanisms, biocompatibility and clinical application of TiO2 nanotube[25-47]. There were 6 literatures about effect of TiO2 nanotube on diminishing inflammation[26-28, 37, 45-46], 14 literatures on promoting effect of TiO2 nanotube on biocompatibility and osseointegration[30-33, 36-44, 47]. Characterization of research results of included data Implant-related infection and strategy Two thirds of implant-related infection was staphylococcus aureus infection[1-5, 9]. Bacterial biofilm is a property of most bacterial infection, especially staphylococci. Gilbert et al [18] confirmed that to kill bacteria in the bacterial biofilm needed a large dose of antibiotics, about > 1 000 times of killing the same kind of bacteria in cell suspension. Another property of bacterial biofilm is to induce drug resistance of bacteria in bacterial biofilm. When drug concentration was low, and did not reach the minimal inhibitory concentration, pathogen easily had drug resistance, and increased the difficulty of treatment[19-21]. Poelstra and colleagues[22] found that the first 6 hours after the implant was implanted in the body was a key period to prevent infection. Six hours later, bacterial biofilm on the surface of the implant formed. Although aseptic processing was strictly performed and laminar flow operating room was used, the incidence of postoperative implant infection in patients undergoing joint replacement was 2%-4%[3-7]. What is more serious is that the incidence of implant infection after open fracture was 50%[20-21]. Differing from clinical common infection, the implant-related infection cannot be cured by common systemic application of antibiotics. The steel plate or artificial joint should be taken out of and surrounding tissues should be debrided and the operation should be operated again, which always brought huge pains on the soul and body and heavy economic burden. The patents have to take many risks such as long length of stay, complicated rebuilding, prosthesis failure, even death. High-concentration systemic application of antibiotics would cause adverse reactions all over the body, including low efficiency, prolonged duration, repeated hospitalization, drug overdose, lacks of selectivity and toxicity[16]. Moreover, the drug is hard to be transferred to the site of the infection[16]. Under an ideal condition, pharmacodynamic action should be added and adverse reactions all over the body should be reduced. At present, the curative effects of local application of antibiotics were better than that of systemic application of antibiotics[8, 22-23]. Local application of antibiotics could precisely control the dose of antibiotics in the implant, high effectively release antibiotics, avoid the adverse reactions of systemic application of antibiotics, and reduce the possibility of drug resistance of the bacteria. Administration of antibiotics all over the body had poor penetration ability for necrotic tissues, but elevation of drug dose would lead to toxic complications all over the body such as liver-kidney related toxicity[8, 22-23]. Local administration of antibiotics could increase local drug concentration and avoid adverse reactions of systemic medication, instead of systemic application of antibiotics. Local application of antibiotics in the prevention of infection has been selected in the clinic in the Department of Orthopedics, such as gentamicin bone cement. To overcome the defect of traditional strategy, local drug delivery has been a promising method. Local drug delivery system has been first used in antibiotics bone cement (PMMA) in 1980s by Buchholz et al [9]. The advantages of local drug delivery system comprised prevention of adverse reactions of systemic medication, and local high performance of antibiotics[24]. Nevertheless, the main shortcoming of antibiotic bone cement is that the implants have to been taken out twice. Antibiotic bone cement is not fit for cementless prosthesis (hip and knee joint prosthesis and implant steel plate). Another shortcoming of antibiotic bone cement is that when drug release concentration is low (lower than minimal inhibitory concentration), low-dose antibiotic release can lead to the production of drug-resistant bacteria. Kendall et al [19] found that bacteria could survive on the surface of antibiotic bone cement in vitro models. Anagnostakos et al [21] confirmed that pathogen was staphylococcus aureus and methicillin-resistant staphylococcus aureus in 18 patients suffering from antibiotic bone cement infection. Therefore, it should be careful whether antibiotic bone cement was extensively used in the prevention of prosthesis infection. Development background and formation mechanism of TiO2 The development of nanotechnology provides a new drug delivery platform for local drug delivery in implant. The aperture and length of nanomaterial could be controlled. TiO2 nanotubes could be made by anodic oxidation[25] (Figure 1)."

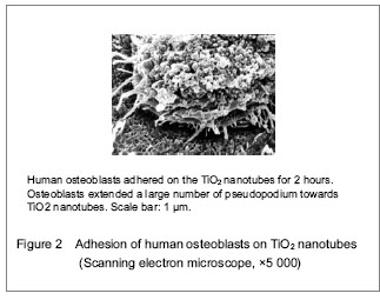
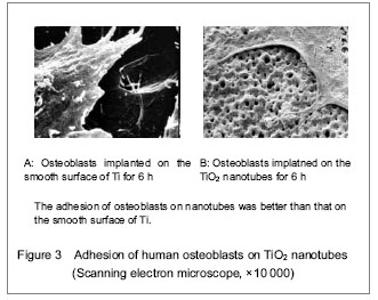
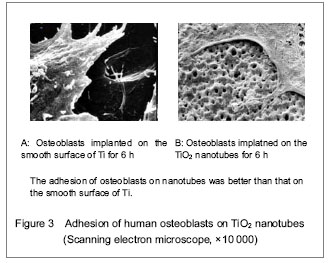
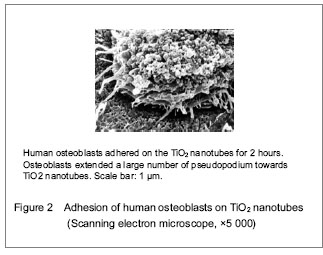
TiO2 nanotubes are promising research subjects in the field of material at present. Drug can be delivered to the therapeutic region via nanomaterials effectively, which can overcome adverse distribution in the body, and avoid the defect of systemic medication (such as low efficiency, drug overdose, lack of choice and toxicity). TiO2 nanotubes not only elevated osseointegration of the metal, cell proliferation and differentiation, but also can be utilized as a drug carrier. TiO2 nanotubes provided many cell attachment points to contribute to cell adhesion and proliferation by deposition of numerous proteins (Figures 2, 3)."

Totally new tissue culture scaffold can be obtained by changing materials’ appearance. Biologically, interface modification can simplify the complicated biological problems, and can be used as an essential manner for controlling cell behavior. Ti and its alloy surface receive interface modification using anodic oxidation, and TiO2 nanotubes coating can be prepared. Studies demonstrated that this kind of nano-structured coating can induce a series of biological effects at cellular levels. During the research, that material would be first used in the study of cell adhesion, and gradually expanded on cell proliferation, migration and differentiation[26-35]. TiO2 nanotube coating can be employed for precise control in the adhered structure of interaction between cells and materials at nanometer scale, and provide a valuable theoretic guidance for designing and developing intelligent cell culture scaffold[26-35]. Gong et al [25] first successfully prepared TiO2 nanotube array in fluorhydric acid electrolyte system in 2001. Mean diameter, length and thickness of TiO2 nanotube were controlled by parameters of anodic oxidation (oxidation time, voltage, and F-concentration)[25]. In the same electrolyte system, inner diameter of nanotubes increased with increased pressure of oxidation, and thickness of nanotube wall reduced. Nanotube length increased with prolonged oxidation time and elevated oxidation pressure. In different electrolyte systems, F-concentration and its corrosion rate on TiO2 are important factors for nanotube length[25]. In the postprocessing of TiO2 nanotube array, annealing temperature could affect the appearance and microstructure of TiO2 nanotube coating. During annealing in the air, the coating has a thermal stability in a certain range. When annealing temperature was higher than 650 °C, the structure of nanotubes collapsed. Subsequently, numerous studies have focused on clinical application of TiO2 nanotube[36-47]. TiO2 nanotube contributed to biocompatibility[36-40, 42, 45-47]. Animal studies verified that TiO2 nanotube promoted osseointegration of the implant in swine models[41, 43-44]. Biocompatibility of TiO2 nanotube TiO2 nanotubes promoted biocompatibility[36-40, 42, 45-47]. Biocompatibility of biomaterials is a core problem in the field of regenerative medicine and tissue engineering. The surface of the material is a ligament of implant inserted in the host. A totally new tissue culture scaffold was obtained by effectively changing materials’ appearance and chemical component. The surface appearance of the biomaterial directly affected the physical and chemical behaviors of the biomaterials in vivo and in vitro. Surface appearance of the material is a hot problem in the field of regenerative medicine and tissue engineering. Nanomedicine has been considered as a new field with giant potential. A whole new function of biomaterials can be found by changing the surface appearance of present materials. Ti metal and its alloy have been extensively applied in clinical medicine for over 50 years due to its excellent biocompatibility. Recent studies suggested that the alteration of the surface appearance of Ti metal (such as TiO2 nanotube) elevated its biocompatibility[36-40, 42, 45-47]. TiO2 nanotube biomaterials not only elevated biocompatibility of the materials, but also increased its therapeutic function, such as anti-infection ability. Some studies demonstrated that TiO2 nanotube surface is an effective platform of osteoblast growth and differentiation, and osteoblast viability was elevated by controlling nanotube appearance[40-45]. Cooper[30] pointed out that surface appearance of the implant influenced the osseointegration of the implant in vivo. The increase in surface appearance of the implant contributed to osteoblast viability and elevated osseointegration of the material[30]. The diameter of TiO2 nanotube varied from several nanometers to hundreds of nanometers. Different diameters of nanotubes had different effects on the functions of osteocytes and bone marrow stem cells. The main dispute is the effect of big nanotube diameter (50 nm–100 nm) on cells. Some scholarsdiscovered that the size of TiO2 nanotube diameter affected osteoblastic adhesion, proliferation and differentiation[36-39]. Park et al [39] verified that 15 nm diameter facilitated integrin aggregation, and promoted cell adhesion and reduced apoptotic rate. When nanotube diameter was longer than 50 nm, osteoblastic adhesion and diffusion were suppressed, and cell viability was noticeably reduced, resulting in cell apoptosis[36-39]. However, numerous studies had an opposite conclusion and indicated that large-diameter (80 nm-100 nm) could not cause cell apoptosis, but promote the proliferation and differentiation of bone marrow stem cells. Khang et al [32] investigated the effects of surface appearance (subnanometer, nanometer and submicron) of the implant on cells and confirmed that surface characterization of subnanometer, nanometer and submicron could selectively activate integrin receptor and induce the differentiation of bone marrow mesenchymal stem cells into osteoblasts, and surface roughness of nano-Ti contributed to osteoblast differentiation. At subnanometer and submicron scales, they studied stem cell reactions such as integrin activation, cyclin, key open gene of osteoblast differentiation and osteoblast phenotype gene-produced effects. Compared with subnanometer surface, pure nano-Ti surface has perfect activated integrin. Jayaraman et al [33] found that surface appearance of the implant influenced osteoblast proliferation, differentiation and extracellular matrix protein expression. Nanometer structure promoted fibronectin expression, and contributed to osseointegration of implant and bone tissue. Some scholars pointed out that TiO2 nanotubes accelerated osteoblastic adhesion and osseointegration[40-44]. The ability of accelerating osseointegration depends on the size of the diameter of TiO2 nanotubes. Nanotubes of big diameter showed better ability on bone formation compared with nanotubes of small diameter. The nanotubes of small diameter (about 30 nm) contributed to osteoblastic adhesion, but nanotubes of big diameter (70-100 nm) induced cell differentiation and resulted in an elevated activation of alkaline phosphatase. Popat et al [45] performed histological analysis at 4 weeks after implantation in Lewis rats and found that TiO2 nanotubes promoted cell adhesion, cell viability, including alkaline phosphatase and calcium contents. Biocompatibility results demonstrated that TiO2 nanotubes could not induce chronic inflammation or fibrosis. von Wilmowsky et al [43] implanted TiO2 nanotubes into pig models for 3, 7, 14, 30 and 90 days, conducted histological examination, and found a significant high expression of type I collagen after implantation of TiO2 nanotubes at 7 and 14 days. Nanotubes of 30 nm diameter enhanced osteoblast function and affected bone formation and development. The study showed that TiO2 nanotubes contributed to cell adhesion and elevated cell viability and promoted osseointegration. Moreover, TiO2 nanotubes exhibited a good and stable structure, and were not destroyed by shearing force. Bjursten et al [44] confirmed that nanotubes excessively promoted osseointegration in vivo. Study contained nanometer group (80 nm in diameter and 2 μm in length) and micrometer group (2 μm). Using rabbit models, study verified that nanotubes facilitated osseointegration. The contact rate of bone tissue and material was greater in the nanometer group than that in the non-nanometer group. The contact rates of bone tissue and material were (78.3±33.3)% and (21.7±24.7)% in the nanometer group and the micrometer group, respectively. Stretch forces were (10.8±3.1) N and (1.2±2.7) N in the nanometer group and micrometer group, respectively. Studies confirmed a large amount of new bone formation on the surface of nanotubes. Calcium and phosphorus contents were obviously increased on the surface of nanometer structure compared with the micrometer group. These characteristics benefited for the application of biotype implant in clinic, such as biotype artificial hip knee joint prosthesis and intravascular stent. Application of TiO2 nanotubes as drug carrier At present, orthopedic implant only can be used for 10–15 years due to various reasons including infection and poor osseointegration. Recent study results demonstrated that TiO2 nanotube smear layer not only promoted osseointegration in implants, but also can be utilized as drug carriers such as antibiotics and bone morphogenetic protein. TiO2 nanotubes not only have good biocompatibility and contributed to ossification, but also can be used as a carrier to load other drugs such as growth factor and antibiotics. Peng et al [37] employed TiO2 nanotubes to carry drugs (protein, rapamycin and paclitaxel). In this study, the size of TiO2 nanotubes varied to elute albumin and common micromolecule drugs. Drugs on nanotubes eluted bioactivity and reduced cell proliferation in vitro. Nanotube height and diameter deeply affected dynamics of eluting. Small nanotube diameter and long nanotube height prolonged the time of drug release. Molecular weight of the drugs also affected the time of drug release. The release time of drugs with big molecules was longer than that of micromolecules. This study suggested that TiO2 nanotubes are promising smear layer of implants. A previous study demonstrated that TiO2 nanotubes are promising drug carrier of implants in the Department of Orthopedics[37]. Popat et al [45] loaded TiO2 nanotubes with various doses of gentamicin. TiO2 nanotubes were 80 nm in diameter and 400 nm in length, loaded with 200, 400 and 600 μg gentamicin. They studied release dynamics of these nanotubes and staphylococcus epidermidis adhering gentamicin. In addition, the study evaluated MC3T3-E1 effects on cell function by observing cytological behavior of MC3T3-E1 on these nanotubes. Study results indicated that TiO2 nanotubes carrying gentamicin not only contributed to osteocyte viability, but also significantly diminished the adhesion of staphylococcus epidermidis. Presently, TiO2 nanotubes carrying dexamethasone, penicillin and streptomycin reduced infection and inflammation. George et al [44] verified that TiO2 nanotubes carrying drugs (streptomycin, penicillin and dexamethasone) could promote osteoblast viability. A recent study confirmed that TiO2 nanotubes had apparent inhibitory effects on glial cells[34]. A study successfully prepared hydroxyapatite/TiO2 nanotube complex smear layer by simulating body fluid soaking method[35], first investigated the biological behavior of glial cells on the surface of Ti nanotube array, and explored the effects of nanotubes of different diameters on survival rate and morphology of glial cells such as U87 cells and C6 cells. With increasing diameter of nanotubes, cell number and viability showed a decreased tendency on the surface of TiO2 nanotubes smear layer. Cell apoptosis would appear if the nanotube diameter exceeded critical size (50 nm in this experiment)."

| [1] Darouiche RO. Treatment of infections associated with surgical implants. New Engl J Med. 2004;350(14): 1422-1429.
[2] Gulati K, Aw MS, Findlay D, et al. Local drug delivery to the bone by drug-releasing implants: perspectives of nano-engineered titania nanotube arrays. Ther Deliv. 2012; 3(7):857-873.
[3] Dale H, Hallan G, Hallan G, et al. Increasing risk of revision due to deep infection after hip arthroplasty. Acta Orthop. 2009;80(6):639-645.
[4] Lentino JR: Prosthetic joint infections: bane of orthopedists, challenge for infectious disease specialists. Clin Infect Dis. 2003;36(9):1157-1161.
[5] Kurtz SM, Lau E, Schmier J, et al. Infection burden for hip and knee arthroplasty in the United States. J Arthroplasty. 2008;23(7):984-991.
[6] Green SA. Complications of external skeletal fixation. Clin Orthop Relat Res. 1983;(180):109-116.
[7] Green SA, Ripley MS. Chronic osteomyelitis in pin tracks. J Bone Jt Surg. 1984;66(7):1092-1098.
[8] Diefenbeck M, Mückley T, Hofmann GO. Prophylaxis and treatment of implant-related infections by local application of antibiotics. Injury. 2006;37 Suppl 2:S95-104.
[9] Buchholz HW, Elson RA, Engelbrecht E, et al. Management of deep infection of total hip-replacement. J Bone Joint Surg Br. 1981;63(3):342-353.
[10] Donlan RM, Costerton JW. Biofilms: survival mechanisms of clinically relevant microorganisms. Clin Microbiol Rev. 2002;15(2):167-193.
[11] Hall-Stoodley L, Costerton JW, Stoodley P. Bacterial biofilms: from the natural environment to infectious diseases. Nat Rev Microbiol. 2004;2(2):95-108.
[12] Hetrick EM, Schoenfisch MH. Reducing implant-related infections: active release strategies. Chem Soc Rev. 2006;35(9):780-789.
[13] Ribeiro M, Monteiro FJ, Ferraz MP. Infection of orthopedic implants with emphasis on bacterial adhesion process and techniques used in studying bacterial-material interactions. Biomatter. 2012;2(4):176-194.
[14] Costerton JW, Stewart PS, Greenberg EP. Bacterial biofilms: a common cause of persistent infections. Science. 1999; 284:1318-1322.
[15] Mah TF, O’Toole GA. Mechanisms of biofilm resistance to antimicrobial agents. Trends Microbiol. 2001;9(1):34-39.
[16] Davies D. Understanding biofilm resistance to antibacterial agents. Nat Rev Drug Discov. 2003;2(2):114-122.
[17] Smith AW. Biofilms and antibiotic therapy: is there a role for combating bacterial resistance by the use of novel drug delivery systems? Adv Drug Deliv Rev. 2005;57(10): 1539-1550.
[18] Gilbert P, Collier PJ, Brown MR. Influence of growth rate on susceptibility to antimicrobial agents: biofilms, cell cycle, dormancy, and stringent response. Antimicrob Agents Chemother. 1990;34(10):1865-1868.
[19] Kendall RW, Duncan CP, Smith JA, et al. Persistence of bacteria on antibiotic loaded acrylic depots.A reason for caution. Clin Orthop Relat Res. 1996;(329):273-280.
[20] Tunney MM, Ramage G, Patrick S, et al. Antimicrobial susceptibility of bacteria isolated from orthopaedic implants following revision hip surgery. Antimicrob Agents Chemother. 1998;42(11):3002-3005.
[21] Anagnostakos K, Hitzler P, Pape D, et al. Persistence of bacterial growth on antibiotic-loaded beads: is it actually a problem? Acta Orthop. 2008;79(2):302-307.
[22] Poelstra KA, Barekzi NA, Rediske AM, et al. Prophylactic treatment of gram-positive and gram-negative abdominal implant infections using locally delivered polyclonal antibodies. J Biomed Mater Res. 2002;60(1):206-215.
[23] Wu P, Grainger DW. Drug/device combinations for local drug therapies and infection prophylaxis. Biomaterials. 2006;27(11):2450-2467.
[24] Zilberman M, Elsner JJ. Antibiotic-eluting medical devices for various applications. J Control Release. 2008;130(3): 202–215.
[25] Gong DW, Grimes CA, Varghese OK, et al. Titanium oxide nanotube arrays prepared by anodic oxidation. J Mater Res. 2001;16(12):3331-3334.
[26] Losic D, Simovic S. Self-ordered nanopore and nanotube platforms for drug delivery applications. Expert Opin Drug Deliv. 2009;6(12):1363-1381.
[27] Moseke C, Hage F, Vorndran E, et al. TiO2 nanotube arrays deposited on Ti substrate by anodic oxidation and their potential as a long-term drug delivery system for antimicrobial agents. Applied Surface Science. 2012; 258(14):5399-5404.
[28] Song YY, Schmidt-Stein F, Bauer S, et al. Amphiphilic TiO2 nanotube arrays: an actively controllable drug delivery system. J Am Chem Soc. 2009;131(12):4230-4232.
[29] Yang BC, Uchida M, Kim HM, et al. Preparation of bioactive titanium metal via anodic oxidation treatment. Biomaterials. 2004;25(6):1003-1010.
[30] Cooper LF. A role for surface topography in creating and maintaining bone at titanium endosseous implants. J Prosthet Dent. 2000;84(5):522-534.
[31] Webster TJ, Ejiofor JU. Increased osteoblast adhesion on nanophase metals: Ti, Ti6Al4V, and CoCrMo. Biomaterials. 2004;25(19):4731-4739.
[32] Khang D, Choi J, Im YM, et al. Role of subnano-, nano- and submicron-surface features on osteoblast differentiation of bone marrow mesenchymal stem cells. Biomaterials. 2012; 33(26):5997-6007.
[33] Jayaraman M, Meyer U, Bühner M, et al. Influence of titanium surfaces on attachment of osteoblast-like cells in vitro. Biomaterials. 2004;25(4):625-631.
[34] Yang H, Qin X, Tian A, et al. Nano Size Effects of TiO2 Nanotube Array on the Glioma Cells Behavior. Int J Mol Sci. 2012;14(1):244-254.
[35] Zhang D, Tian A, Xue X, et al. The effect of temozolomide/ poly(lactide-co-glycolide) (PLGA)/nano-hydroxyapatite microspheres on glioma U87 cells behavior. Int J Mol Sci. 2012;13(1):1109-1125.
[36] Park J, Bauer S, Schmuki P, et al. Narrow window in nanoscale dependent activation of endothelial cell growth and differentiation on TiO2 nanotube surfaces. Nano Lett. 2009;9(9):3157-3164.
[37] Peng L, Mendelsohn AD, Latempa TJ, et al. Long-term small molecule and protein elution from TiO(2) nanotubes. Nano Lett. 2009;9(5):1932-1936.
[38] Park J, Bauer S, Von Der Mark K, et al. Nanosize and vitality: TiO2 nanotube diameter directs cell fate. Nano Lett. 2007;6:1686-1691.
[39] Park J, Bauer S, Schlegel KA, et al. TiO(2) nanotube surfaces: 15 nm-an optimal length scale of surface topography for cell adhesion and differentiation. Small. 2009; 5(6):666-671.
[40] Oh S, Brammer KS, Li YSJ, et al. Stem cell fate dictated solely by altered nanotube dimension. Proc Natl Acad Sci USA. 2009;106(7):2130-2135.
[41] Brammer KS, Seunghan O, Cobb CJ, et al. Improved bone-forming functionality on diameter-controlled TiO2 nanotube surface. Acta Biomater. 2009;5(8):3215-3223.
[42] Popat KC, Leoni L, Grimes CA et al. Influence of engineered titaniananotubular surfaces on bone cells. Biomaterials. 2007;28(21):3188-3197.
[43] von Wilmowsky C, Bauer S, Lutz R et al. In vivo evaluation of anodic TiO2 nanotubes: an experimental study in the pig. J Biomed Mater Res B. 2009;89(1):165-171.
[44] Bjursten LM, Rasmusson L, Oh S, et al. Titanium dioxide nanotubes enhance bone bonding in vivo. J Biomed Mater Res. 2010;92(3):1218-1224..
[45] Popat KC, Eltgroth M, Latempa TJ, et al. Decreased Staphylococcus epidermis adhesion and increased osteoblast functionality on antibiotic-loaded titania nanotubes. Biomaterials. 2007;28(32):4880-4888.
[46] George EA, Chang Y, Thomas JW. Enhanced osteoblast adhesion to drug-coated anodized nanotular titanium safaces. Int J Nanomed. 2008;3(2):257-264.
[47] Park J, Bauer S, PittrofA et al. Synergistic control of mesenchymal stem cell differentiation by nanoscale surface geometry and immobilized growth factors on TiO2 nanotubes. Small. 2012;8(1):98-107. |
| [1] |
Dong Xiling, Zhang Xiaoming, Liu Tongbin.
Recombinant human parathyroid hormone (1-34): pro-osteogenic action and application in oral field [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(26): 4231-4236. |
| [2] | Li Hongcai, Ma Zhuang, Guo Youling. Biological behaviors of fibroblasts on the surface of TiO2 nanotubes with different diameters after annealing treatment [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(22): 3504-3509. |
| [3] | Fan Shifeng, Qie Hui, Zhang Wenxuan, Hou Xiaowei, Wang Lijie, Zheng Yu. Comparison of the effects of topical application of zoledronic acid and silver nanoparticles on osseointegration of immediate implant [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(10): 1547-1552. |
| [4] | Yuan Bo, Wang Zhiwei, Tang Yifan, Zhou Shengyuan, Chen Xiongsheng, Jia Lianshun. Construction of polycaprolactone-tricalcium phosphate with different mixture ratios using three-dimensional printing technology and its osteoinductivity in vitro [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(6): 821-826. |
| [5] | Wu Yeke, Gao Ranran, Zhao Lixing. Orthodontic micro-implant stability under continuous or intermittent loading: a histomorphometric and biomechanical evaluation [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(22): 3488-3494. |
| [6] | Wei Wei, Liu Yanfei, Zhang Ling, Xiong Na . Self-assembling peptide hydrogel: hemostatic effect and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(2): 310-316. |
| [7] | Li Ying, You Yapeng, Li Baoe, Song Yunjia, Ma Aobo, Chen Bo, Han Wen, Li Changyi. Type I collagen combined titanium dioxide nanotube composite coating modified titanium surface improves osteoblast adhesion and osseointegration [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(14): 2169-2176. |
| [8] | Zhang Minbo, Peng Qifeng, Ma Yaping, Kong Weijun, Liao Wenbo. Physical properties and biocompatibility of 3D printed bone microparticle/poly(lactic-co-glycolic acid) scaffold [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(14): 2215-2222. |
| [9] | Li Zhi, Tan Chunhua, Cai Xianhua, Wang Huasong, Ding Xiaoming, Zhao Yanhong. Fabrication and biocompatibility assessment of the scaffold with biomimetic interconnected macropore structure [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(14): 2223-2227. |
| [10] | Bai Yulong, Gao Yufeng, Zhong Hongbin, Zhao Yantao, Guo Ruizhou, Li Li. Allogeneic and xenogeneic tissue repair materials: how to choose a suitable virus inactivation process [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(14): 2261-2268. |
| [11] | Luo Kai, Yang Yafeng, Ma Teng, Xia Bing, Huang Liangliang, Huang Jinghui, Luo Zhuojing. Effects of perfluorotributylamine/alginate/bioglass biomaterials on viability and osteogenic differentiation of adipose-derived stem cells [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(13): 1995-2001. |
| [12] | Wang Yuanyuan, Song Wenshan, Yu Dejun, Dai Yuankun, Li Bafang. Preparation and evaluation of fish skin acellular dermal matrix for oral guided tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(10): 1526-1532. |
| [13] | Liu Shaoyang, Zou Hanlin . Biocompatibility of anodic titanium oxide: an experimental research [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(10): 1575-1580. |
| [14] | Cai Yuhui, Hu Kesu, Zhang Yi. Biosafety evaluation of chitosan lactate/hyaluronate sponge [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(10): 1558-1563. |
| [15] | Zhang Lei, Zhou Song, Cheng Bao-chang. Actuality and challenge of biomaterials in annulus fibrosus repair [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(6): 958-963. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||