Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (25): 3978-3986.doi: 10.3969/j.issn.2095-4344.1776
Previous Articles Next Articles
Chrysin inhibits mouse osteoclastogenesis induced by receptor activator of nuclear factor kappa B ligand
-
Revised:2019-02-26Online:2019-09-08Published:2019-09-08 -
Contact:Tian Xiaobin, Professor, Doctoral supervisor, Guizhou Medical University, Guiyang 550025, Guizhou Province, China; Sun Li, MD, Chief physician, Doctoral supervisor, Department of Orthopedics, Guizhou Provincial People’s Hospital, Guiyang 550002, Guizhou Province, China -
About author:Li Xianghe, Master candidate, Guizhou Medical University, Guiyang 550025, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 81560356 (to TXB); Social Development Project of Guizhou Province, No. (2015) 3044 (to SL)
CLC Number:
Cite this article
Li Xianghe, Luo Wei, Hu Junxian, Yang Jing, Han Xinyun, Dong Shiwu, Yang Xianteng, Li Senlei, Yan Zhihui, Nie Yingjie, Tian Xiaobin, Sun Li.
share this article
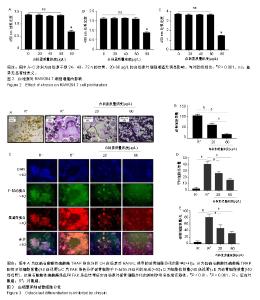
2.1 白杨素对RAW264.7细胞活性的影响 0,20,40,60,80 μg/L白杨素处理RAW264.7细胞24-72 h后,CCK8分析结果显示,白杨素质量浓度在80 μg/L时能明显抑制细胞增殖,而在质量浓度为20-60 μg/L时,细胞增殖不受影响,见图2。因此,后续研究中白杨素的质量浓度设为20和60 μg/L。 2.2 白杨素抑制RANKL诱导的破骨细胞分化和形成 用RANKL(50 μg/L)、巨噬细胞集落刺激因子(25 μg/L)和白杨素(0,20,60 μg/L)诱导RAW264.7细胞分化为破骨细胞。通过抗酒石酸酸性磷酸酶染色分析白杨素对RANKL诱导的破骨细胞分化的影响。结果显示,白杨素显著抑制RANKL诱导的破骨细胞生成,且以质量浓度依赖的方式抑制RAW264.7细胞中抗酒石酸酸性磷酸酶阳性多核细胞数量,见图3A,B。此外,通过FAK染色分析破骨细胞中F-actin环结构的形成,F-actin环结构对破骨细胞骨吸收功能是必不可少的[19]。结果显示,白杨素质量浓度为60 μg/L时,F-actin环结构的尺寸明显减小,见图3C-E。表明白杨素能有效抑制破骨细胞分化和形成。"

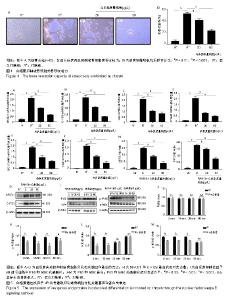
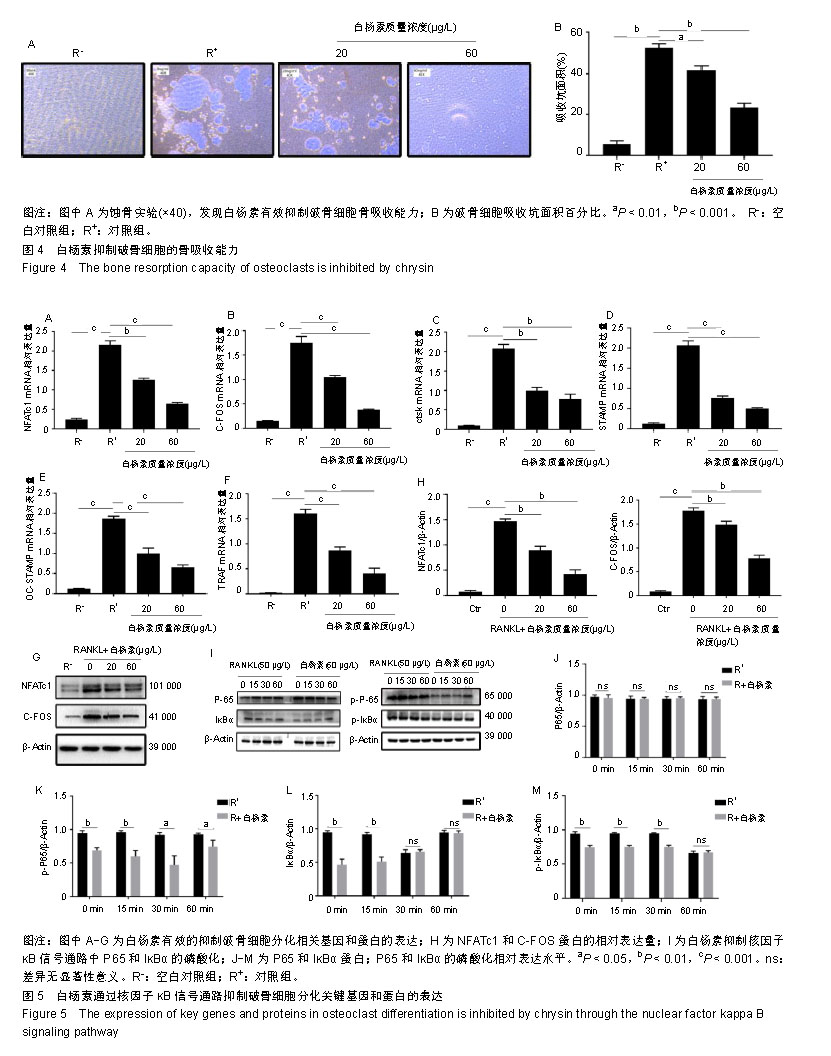
2.3 白杨素在体外降低骨吸收活性 由于白杨素抑制RANKL诱导的破骨细胞生成,实验利用骨陷窝形成分析白杨素是否能抑制破骨细胞体外骨吸收活性。结果显示,白杨素以浓度依赖的方式明显的抑制骨吸收面积,见图4。 2.4 白杨素抑制RANKL诱导的破骨细胞标志基因表达 为进一步阐明白杨素对破骨细胞分化的影响,实时定量PCR结果显示,在破骨细胞分化过程中,RANKL显著上调破骨细胞特异性基因NFATc1、C-FOS、组织蛋白酶K、抗酒石酸酸性磷酸酶、OC-STAMP、DC-STAMP的mRNA表达水平。而白杨素以浓度依赖的方式对上述mRNA表达具有抑制作用,见图5A-F。Western blot分析结果表明,RANKL明显上调了NFATc1和C-FOS的表达,而白杨素则抑制这2种蛋白的表达图5G。该结果与实时定量PCR结果一致。表明白杨素在体外可以抑制RANKL诱导的破骨细胞关键基因表达。 2.5 白杨素通过抑制核因子κB信号通路来抑制破骨细胞分化 RANKL诱导的核因子κB信号通路的激活对破骨细胞的分化和功能是必不可少的[20]。为了探讨白杨素在破骨细胞发生中的作用机制,在白杨素(60 μg/L)存在或不存在的条件下,用RANKL(50 μg/L)和巨噬细胞集落刺激因子 (25 μg/L)分别处理RAW264.7细胞0,15,30和60 min,Western blot分析结果显示,当白杨素(60 μg/L)存在时, 核因子κB p65的磷酸化表达被白杨素下调。实验进一步发现,白杨素同时还显著抑制IκBα的磷酸化和退化,见图5H-M。表明,白杨素通过减弱核因子κB信号通路的活性来抑制RANKL诱导的破骨细胞分化。"
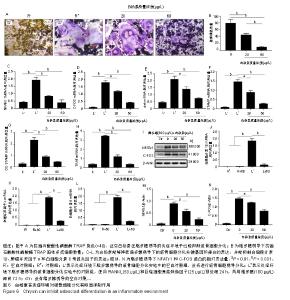
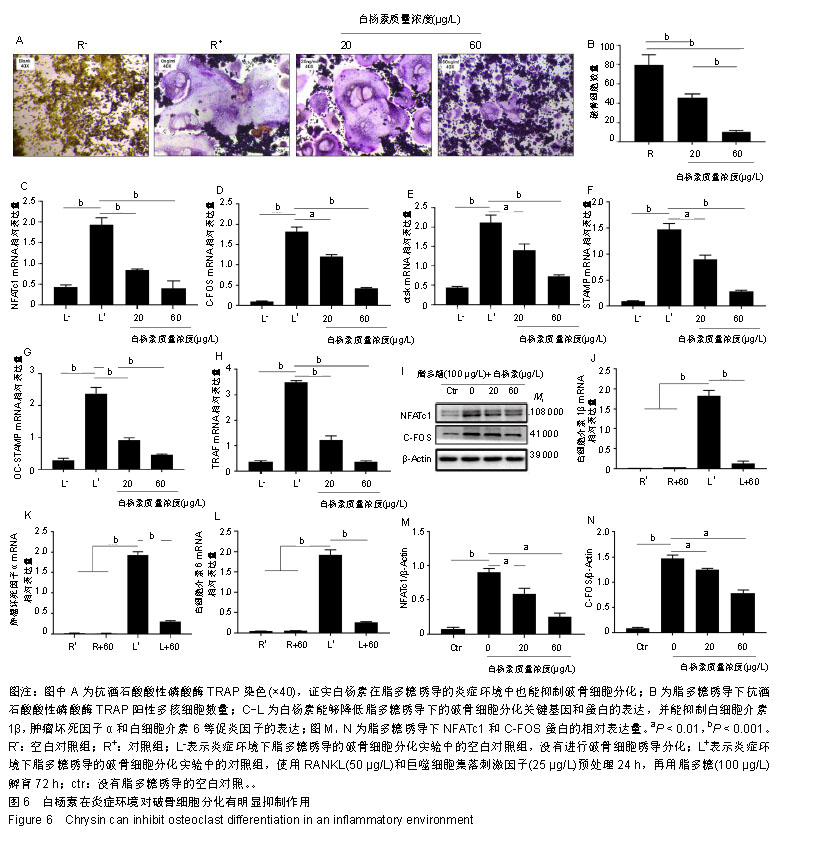
2.6 白杨素下调脂多糖诱导的破骨细胞形成 为了研究白杨素在脂多糖介导的炎症条件下对破骨细胞分化的影响,RAW264.7细胞使用RANKL(50 μg/L)和巨噬细胞集落刺激因子(25 μg/L)预处理24 h。然后,这些细胞再用脂多糖(100 μg/L)和白杨素(0,20,60 μg/L)孵育72 h,诱导细胞分化为破骨细胞,抗酒石酸酸性磷酸酶染色用来分析脂多糖诱导的破骨细胞分化。作者观察到抗酒石酸酸性磷酸酶阳性多核破骨细胞呈浓度依赖性的减少,见图6A,B。 为了进一步研究白杨素对脂多糖诱导破骨细胞分化的影响,采用实时定量PCR方法对多种破骨细胞标志基因进行分析。如图6所示,白杨素明显下调这些标记基因的表达,与抗酒石酸酸性磷酸酶染色结果一致,见图6C-H。 同样,利用Western blot分析脂多糖诱导的NFATc1和C-FOS蛋白表达,也获得了相同的结果,NFATc1和C-FOS的蛋白表达均明显降低,见图6I,M,N。表明白杨素在脂多糖诱导的炎症条件下对破骨细胞的形成有负性调节作用。 因为白杨素抑制脂多糖诱导的破骨细胞形成,因此分析了在脂多糖刺激条件下,能够促进破骨细胞形成并导致骨丢失的促炎细胞因子(肿瘤坏死因子α、白细胞介素1β、白细胞介素6)的表达情况。实时定量PCR在mRNA水平上分析这些基因的表达。结果显示白杨素能显著抑制这些促炎细胞因子基因的表达,见图6J-L。"
| [1]Song D, Meng T, Xu W, et al. 5-Fluoruracil blocked giant cell tumor progression by suppressing osteoclastogenesis through NF-kappaB signals and blocking angiogenesis. Bone. 2015;78:46-54. [2]Wei CM, Liu Q, Song FM, et al. Artesunate inhibits RANKL-induced osteoclastogenesis and bone resorption in vitro and prevents LPS-induced bone loss in vivo. J Cell Physiol. 2018;233(1):476-485. [3]Zhang T, Zhao K, Han W, et al. Deguelin inhibits RANKL-induced osteoclastogenesis in vitro and prevents inflammation-mediated bone loss in vivo. J Cell Physiol. 2019; 234(3):2719-2729. [4]Chen Y, Wang X, Yang M, et al. miR-145-5p Increases Osteoclast Numbers In Vitro and Aggravates Bone Erosion in Collagen-Induced Arthritis by Targeting Osteoprotegerin. Med Sci Monit. 2018;24:5292-5300.[5]Thummuri D, Naidu VGM, Chaudhari P. Carnosic acid attenuates RANKL-induced oxidative stress and osteoclastogenesis via induction of Nrf2 and suppression of NF-κB and MAPK signalling. J Mol Med (Berl). 2017;95(10): 1065-1076. [6]Wang W, Yang L, Zhang D, et al. MicroRNA-218 Negatively Regulates Osteoclastogenic Differentiation by Repressing the Nuclear Factor-κB Signaling Pathway and Targeting Tumor Necrosis Factor Receptor 1. Cell Physiol Biochem. 2018; 48(1):339-347.[7]Song F, Wei C, Zhou L, et al. Luteoloside prevents lipopolysaccharide-induced osteolysis and suppresses RANKL-induced osteoclastogenesis through attenuating RANKL signaling cascades. J Cell Physiol. 2018;233(2): 1723-1735. [8]Kim HJ, Park C, Kim GY, et al. Sargassum serratifolium attenuates RANKL-induced osteoclast differentiation and oxidative stress through inhibition of NF-κB and activation of the Nrf2/HO-1 signaling pathway. Biosci Trends. 2018;12(3):257-265.[9]Sapkota M, Li L, Kim SW, et al. Thymol inhibits RANKL-induced osteoclastogenesis in RAW264.7 and BMM cells and LPS-induced bone loss in mice. Food Chem Toxicol. 2018;120:418-429.[10]Choi JK, Jang YH, Lee S, et al. Chrysin attenuates atopic dermatitis by suppressing inflammation of keratinocytes. Food Chem Toxicol. 2017;110:142-150.[11]Zeinali M, Rezaee SA, Hosseinzadeh H. An overview on immunoregulatory and anti-inflammatory properties of chrysin and flavonoids substances. Biomed Pharmacother. 2017; 92:998-1009.[12]Ramírez-Espinosa JJ, Saldaña-Ríos J, García-Jiménez S, et al. Chrysin Induces Antidiabetic, Antidyslipidemic and Anti-Inflammatory Effects in Athymic Nude Diabetic Mice. Molecules. 2017;23(1):E67. [13]George MY, Esmat A, Tadros MG, et al. In vivo cellular and molecular gastroprotective mechanisms of chrysin; Emphasis on oxidative stress, inflammation and angiogenesis. Eur J Pharmacol. 2018;818:486-498. [14]Mani R, Natesan V, Arumugam R. Neuroprotective effect of chrysin on hyperammonemia mediated neuroinflammatory responses and altered expression of astrocytic protein in the hippocampus. Biomed Pharmacother. 2017;88:762-769.[15]Zheng W, Tao Z, Cai L, et al. Chrysin Attenuates IL-1β-Induced Expression of Inflammatory Mediators by Suppressing NF-κB in Human Osteoarthritis Chondrocytes. Inflammation. 2017;40(4):1143-1154.[16]Jiang Y, Gong FL, Zhao GB, et al. Chrysin suppressed inflammatory responses and the inducible nitric oxide synthase pathway after spinal cord injury in rats. Int J Mol Sci. 2014;15(7):12270-12279. [17]Eldutar E, Kandemir FM, Kucukler S, et al. Restorative effects of Chrysin pretreatment on oxidant-antioxidant status, inflammatory cytokine production, and apoptotic and autophagic markers in acute paracetamol-induced hepatotoxicity in rats: An experimental and biochemical study. J Biochem Mol Toxicol. 2017;31(11):e21960.[18]Chen Y, Dou C, Yi J, et al. Inhibitory effect of vanillin on RANKL-induced osteoclast formation and function through activating mitochondrial-dependent apoptosis signaling pathway. Life Sci. 2018;208:305-314. [19]Tang R, Yi J, Yang J, et al. Interleukin-37 inhibits osteoclastogenesis and alleviates inflammatory bone destruction. J Cell Physiol. 2019;234(5):7645-7658. [20]Dharmapatni AASSK, Algate K, Coleman R, et al. Osteoclast-Associated Receptor (OSCAR) Distribution in the Synovial Tissues of Patients with Active RA and TNF-α and RANKL Regulation of Expression by Osteoclasts In Vitro. Inflammation. 2017;40(5):1566-1575. [21]Crockett JC, Rogers MJ, Coxon FP, et al. Bone remodelling at a glance. J Cell Sci. 2011;124(Pt 7):991-998.[22]Suda T, Nakamura I, Jimi E, et al. Regulation of osteoclast function. J Bone Miner Res. 1997;12(6):869-879.[23]Muruganandan S, Dranse HJ, Rourke JL, et al. Chemerin neutralization blocks hematopoietic stem cell osteoclastogenesis. Stem Cells. 2013;31(10):2172-2182. [24]Wu X, Chim SM, Kuek V, et al. HtrA1 is upregulated during RANKL-induced osteoclastogenesis, and negatively regulates osteoblast differentiation and BMP2-induced Smad1/5/8, ERK and p38 phosphorylation. FEBS Lett. 2014;588(1):143-150.[25]Kim JY, Min JY, Baek JM, et al. CTRP3 acts as a negative regulator of osteoclastogenesis through AMPK-c-Fos-NFATc1 signaling in vitro and RANKL-induced calvarial bone destruction in vivo. Bone. 2015;79:242-251. [26]Jeong E, Choi HK, Park JH, et al. STAC2 negatively regulates osteoclast formation by targeting the RANK signaling complex. Cell Death Differ. 2018;25(8):1364-1374. [27]Park JH, Lee NK, Lee SY. Current Understanding of RANK Signaling in Osteoclast Differentiation and Maturation. Mol Cells. 2017;40(10):706-713.[28]Obaid R, Wani SE, Azfer A, et al. Optineurin Negatively Regulates Osteoclast Differentiation by Modulating NF-κB and Interferon Signaling: Implications for Paget's Disease. Cell Rep. 2015;13(6):1096-1102. [29]Krauss JL, Zeng R, Hickman-Brecks CL,et al. NLRP12 provides a critical checkpoint for osteoclast differentiation. Proc Natl Acad Sci U S A. 2015;112(33):10455-10460.[30]Gu Z, Wang H, Xia J, et al. Decreased ferroportin promotes myeloma cell growth and osteoclast differentiation. Cancer Res. 2015;75(11):2211-2221.[31]Jie Z, Shen S, Zhao X, et al. Activating β-catenin/Pax6 axis negatively regulates osteoclastogenesis by selectively inhibiting phosphorylation of p38/MAPK. FASEB J. 2018. doi: 10.1096/fj.201801977R.[32]Zhang HQ, Wang YJ, Yang GT, et al. Taxifolin Inhibits Receptor Activator of NF-κB Ligand-Induced Osteoclastogenesis of Human Bone Marrow-Derived Macrophages in vitro and Prevents Lipopolysaccharide-Induced Bone Loss in vivo. Pharmacology. 2019;103(1-2):101-109. [33]Wang W, Huang M, Hui Y, et al. Cryptotanshinone inhibits RANKL-induced osteoclastogenesis by regulating ERK and NF-κB signaling pathways. J Cell Biochem. 2018. doi: 10.1002/jcb.28008. [34]Rehman MU, Ali N, Rashid S, et al. Alleviation of hepatic injury by chrysin in cisplatin administered rats: probable role of oxidative and inflammatory markers. Pharmacol Rep. 2014;66(6):1050-1059. [35]Kats A, Gerasimcik N, Näreoja T, et al. Aminothiazoles inhibit osteoclastogenesis and PGE2 production in LPS-stimulated co-cultures of periodontal ligament and RAW 264.7 cells, and RANKL-mediated osteoclastogenesis and bone resorption in PBMCs. J Cell Mol Med. 2019;23(2):1152-1163. [36]Kim HJ, Yoon HJ, Kim SY, et al. A medium-chain fatty acid, capric acid, inhibits RANKL-induced osteoclast differentiation via the suppression of NF-κB signaling and blocks cytoskeletal organization and survival in mature osteoclasts. Mol Cells. 2014;37(8):598-604. [37]Kaneko K, Miyamoto Y, Tsukuura R, et al. 8-Nitro-cGMP is a promoter of osteoclast differentiation induced by RANKL. Nitric Oxide. 2018;72:46-51. [38]Chai L, Zhou K, Wang S, et al. Psoralen and Bakuchiol Ameliorate M-CSF Plus RANKL-Induced Osteoclast Differentiation and Bone Resorption Via Inhibition of AKT and AP-1 Pathways in Vitro. Cell Physiol Biochem. 2018;48(5): 2123-2133. |
| [1] | Chai Le, Lü Jianlan, Hu Jintao, Hu Huahui, Xu Qingjun, Yu Jinwei, Quan Renfu. Signal pathway variation after induction of inflammatory response in rats with acute spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1218-1223. |
| [2] | Ruan Guangping, Yao Xiang, Liu-Gao Miyang, Cai Xuemin, Li Zian, Pang Rongqing, Wang Jinxiang, Pan Xinghua. Umbilical cord mesenchymal stem cell transplantation for traumatic systemic inflammatory response syndrome in tree shrews [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3994-4000. |
| [3] | Li Li, Zhuo Jin, Zheng Ling, Zhou Ling, Wang Qisong, Luo Lan, Luo Kai. Effects of different induced polarization methods on the proliferation, apoptosis and phagocytosis of rat bone marrow-derived macrophages [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 4032-4037. |
| [4] | Liao Yuan, Qu Mengjian, Liu Jing, Zhong Peirui, Zeng Yahua, Wang Ting, Xiao Hao, Li Lan, Liu Danni, Yang Lu, Zhou Jun. Effects of ultrashort wave on inflammatory response in rats with acute lung injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 253-257. |
| [5] | Luo Yicai, Li Hao. Effect of enhanced aryl hydrocarbon receptor expression on inflammatory response and healing of alveolar bone defects in diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(14): 2166-2171. |
| [6] | Yuan Baohong, Zhao Weishan, Wang Ruotian, Guan Aoran, Wang Yu, Li Ruhong. Bone marrow mesenchymal stem cells from pigs inhibit inflammatory response induced by lipopolysaccharide and improve islet cell dysfunction in pigs [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 1969-1975. |
| [7] | Liao Yuan, Qu Mengjian, Liu Jing, Zhong Peirui Zeng Yahua, Wang Ting, Xiao Hao, Li Lan, Liu Danni, Yang Lu, Zhou Jun. Effects of ultrashort wave on inflammatory response in rats with acute lung injury [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(在线): 2-. |
| [8] | Zhang Sheng, Bai Jijia, Xu Yanping, Wang Xiaohong. Constructing and assessing a rat model of sepsis-induced myocardial dysfunction [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(8): 1249-1253. |
| [9] | Wei Jianghong, Jia Aijun, Ma Libing, Wang Yueling, Qiu Lulu, Xiao Bing. Th-17 regulatory cytokines promote interleukins-17A and 17F production by neutrophils during asthma [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 5044-5051. |
| [10] | He Qiang, Qian Weiqing, Yao Nianwei, Zhou Mengling, Liu Yixin, Yin Hong. Protection of inflammatory osteoblasts in neonatal rats using catalpol from the root of Rehmannia glutinosa [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(29): 4626-4631. |
| [11] | Yin Lian, Zhao Jin, Lei Xuemei, Li Miaomiao, Wang Kun, Zhang Tingran, Luo Jiong. Effect of exercise-induced irisin on myocardial fibrosis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(23): 3730-3736. |
| [12] | Huang Tian, Huang Xin, Lai Peilong, Geng Suxia, Chen Xiaomei, Wang Yulian, Guo Liyan, Zeng Gaochun, Han Fengzhen, Li Xiaohong, Du Xin, Weng Jianyu. Interferon-gamma combined with lipopolysaccharide polarizes human umbilical cord-derived mesenchymal stem cells to a MSC2 phenotype [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(13): 2020-2027. |
| [13] | Liu Ying, Gao Jie, Wu Buling. Biological characteristics of dental pulp stem cells induced by lipopolysaccharide [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(13): 2028-2033. |
| [14] | Xu Huijun, Shi Dongmei, Zhang Mi, Wu Saixuan, Dong Ming, Lu Ying, Niu Weidong. Effect of sex combing protein 1 on proliferation and differentiation of osteoblasts in inflammatory microenvironment#br# [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(1): 130-135. |
| [15] | Liu Dayong, Zhou Weiwei, Xiao Rui, Wang Meirui, Zhao Mengming, Jia Zhi. Lipopolysaccharides affect osteogenic differentiation of bone marrow mesenchymal stem cells derived from ob/ob mice [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(5): 703-709. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||