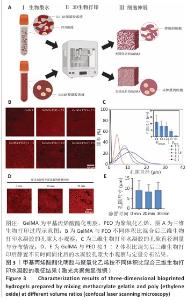
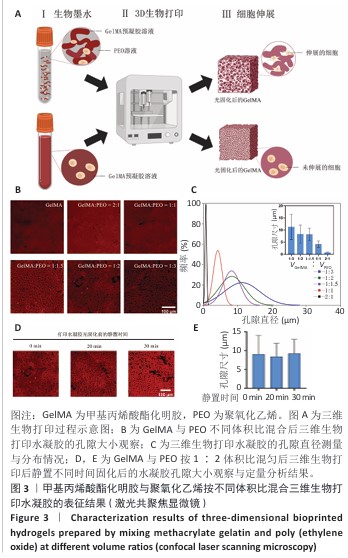
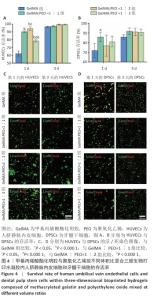
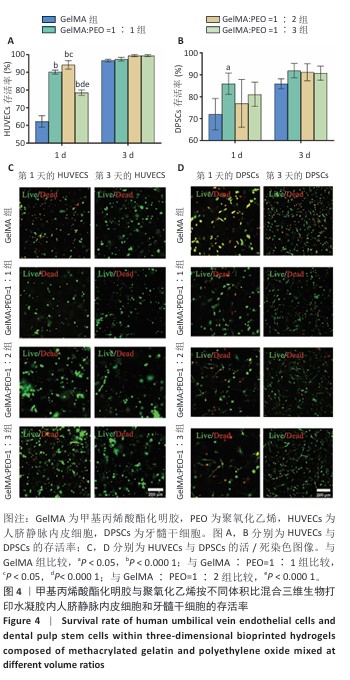
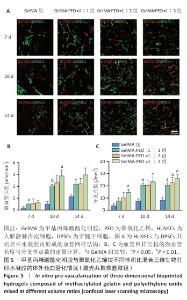
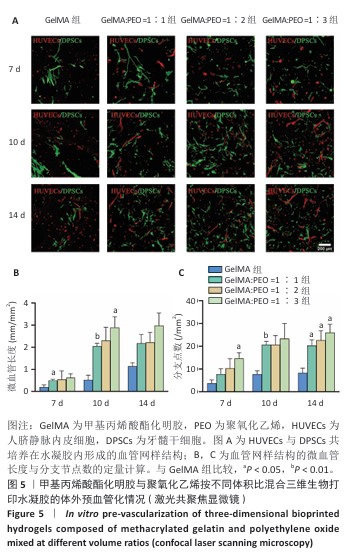
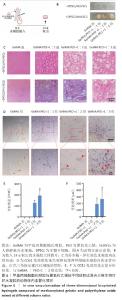
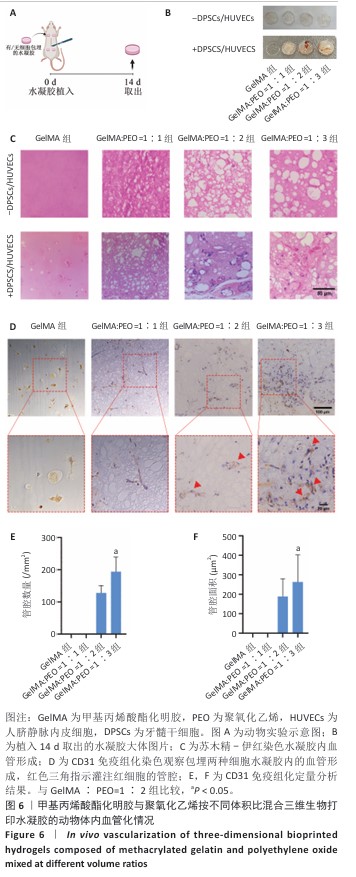
[1] KHADEMHOSSEINI A, LANGER R. A decade of progress in tissue engineering. Nat Protoc. 2016;11(10):1775-1781.
[2] HENDRICKX B, VRANCKX JJ, LUTTUN A. Cell-Based Vascularization Strategies for Skin Tissue Engineering. Tissue Eng Part B Rev. 2011;17(1):13-24.
[3] SUN F, LU Y, WANG Z, et al. Vascularization Strategies for Tissue Engineering for Tracheal Reconstruction. Regen Med. 2021;16(6):549-566.
[4] AGHAZADEH Y, KHAN ST, NKENNOR B, et al. Cell-based therapies for vascular regeneration: Past, present and future. Pharmacol Ther. 2022;231:107976.
[5] GEHLEN J, QIU W, SCHADLI GN, et al. Tomographic volumetric bioprinting of heterocellular bone-like tissues in seconds. Acta Biomater. 2023;156:49-60.
[6] LIU X, CHEN W, ZHANG C, et al. Co-Seeding Human Endothelial Cells with Human-Induced Pluripotent Stem Cell-Derived Mesenchymal Stem Cells on Calcium Phosphate Scaffold Enhances Osteogenesis and Vascularization in Rats. Tissue EngPart A. 2017;23(11-12): 546-555.
[7] NGUYEN BB, MORIARTY RA, KAMALITDINOV T, et al. Collagen hydrogel scaffold promotes mesenchymal stem cell and endothelial cell coculture for bone tissue engineering. J Biomed Mater Res A. 2017;105(4):1123-1131.
[8] CHU Y, ZUO J, ZHANG Y, et al. Co‐culture with chorionic villous mesenchymal stem cells promotes endothelial cell proliferation and angiogenesis via ABCA9‐AKT pathway. The FASEB J. 2022;36(10):e22568.
[9] ZHU W, MA X, GOU M, et al. 3D printing of functional biomaterials for tissue engineering. Curr Opin Biotechnol. 2016;40:103-112.
[10] 徐溶蔚,王浩,付秋月,等.骨再生过程中炎症因素与牙髓干细胞的双向作用[J].中国组织工程研究,2023,27(33):5385-5393.
[11] BOK JS, BYUN SH, PARK BW, et al. The Role of Human Umbilical Vein Endothelial Cells in Osteogenic Differentiation of Dental Follicle-Derived Stem Cells in In Vitro Co-cultures. Int J Med Sci. 2018;15(11):1160-1170.
[12] YUAN C, WANG P, ZHU S, et al. Overexpression of ephrinB2 in stem cells from apical papilla accelerates angiogenesis. Oral Dis. 2019;25(3):848-859.
[13] DISSANAYAKA W L, ZHAN X, ZHANG C, et al. Coculture of Dental Pulp Stem Cells with Endothelial Cells Enhances Osteo-/Odontogenic and Angiogenic Potential In Vitro. J Endod. 2012;38(4):454-463.
[14] PIARD C, JEYARAM A, LIU Y, et al. 3D printed HUVECs/MSCs cocultures impact cellular interactions and angiogenesis depending on cell-cell distance. Biomaterials. 2019;222:119423.
[15] MADL CM, HEILSHORN SC, BLAU HM. Bioengineering strategies to accelerate stem cell therapeutics. Nature. 2018;557(7705):335-342.
[16] ZHANG X-Y, FANG G, ZHOU J. Additively Manufactured Scaffolds for Bone Tissue Engineering and the Prediction of their Mechanical Behavior: A Review. Materials. 2017;10(1):50.
[17] ZHU Y, YU X, LIU H, et al. Strategies of functionalized GelMA-based bioinks for bone regeneration: Recent advances and future perspectives. Bioact Mater. 2024;38:346-373.
[18] ZHU Y, WANG W, CHEN Q, et al. Bioprinted PDLSCs with high-concentration GelMA hydrogels exhibit enhanced osteogenic differentiation in vitro and promote bone regeneration in vivo. Clin Oral Investig. 2023;27(9): 5153-5170.
[19] PEPELANOVA I, KRUPPA K, SCHEPER T, et al. Gelatin-Methacryloyl (GelMA) Hydrogels with Defined Degree of Functionalization as a Versatile Toolkit for 3D Cell Culture and Extrusion Bioprinting. Bioengineering. 2018;5(3):55.
[20] XIN S, WYMAN OM, ALGE DL. Assembly of PEG Microgels into Porous Cell‐Instructive 3D Scaffolds via Thiol‐Ene Click Chemistry. Adv Healthc Mater. 2018;7(11):e1800160.
[21] CHEN YC, LIN RZ, QI H, et al. Functional Human Vascular Network Generated in Photocrosslinkable Gelatin Methacrylate Hydrogels. Adv Funct Mater. 2012;22(10):2027-2039.
[22] HUEBSCH N, LIPPENS E, LEE K, et al. Matrix elasticity of void-forming hydrogels controls transplanted-stem-cell-mediated bone formation. Nat Mater. 2015; 14(12):1269-1277.
[23] GUO Y, MOU S, SUO L, et al. Porous granular hydrogel scaffolds biofabricated from dual-crosslinked hydrogel microparticles for breast tissue engineering. Mater Today Bio. 2025;33:102006.
[24] JALILINEJAD N, BAHEIRAEI N, AZAMI M, et al. Fabrication and characterizations of 3D printed GelMA-Gel/bioactive glass scaffolds containing cerium for bone damage repair. Sci Rep. 2025;15(1):28156.
[25] SHI B, ZHU T, LUO Y, et al. Three-dimensional bioprinted cell-adaptive hydrogel with anisotropic micropores for enhancing skin wound healing. Int J Biol Macromol. 2024;280(Pt 4):136106.
[26] AYDIN D, KIZILEL S. Water-in-Water Emulsion Based Synthesis of Hydrogel Nanospheres with Tunable Release Kinetics. JOM. 2016;69(7):1185-1194.
[27] PETERS EB, CHRISTOFOROU N, LEONG KW, et al. Poly(Ethylene Glycol) Hydrogel Scaffolds Containing Cell-Adhesive and Protease-Sensitive Peptides Support Microvessel Formation by Endothelial Progenitor Cells. Cell Mol Bioeng. 2015;9(1):38-54.
[28] YING G, MANRÍQUEZ J, WU D, et al. An open-source handheld extruder loaded with pore-forming bioink for in situ wound dressing. Mater Today Bio. 2020;8:100074.
[29] JIA L, HUA Y, ZENG J, et al. Bioprinting and regeneration of auricular cartilage using a bioactive bioink based on microporous photocrosslinkable acellular cartilage matrix. Bioact Mater. 2022;16:66-81.
[30] YING GL, JIANG N, MAHARJAN S, et al. Aqueous Two‐Phase Emulsion Bioink‐Enabled 3D Bioprinting of Porous Hydrogels. Adv Mater. 2018;30(50): e1805460.
[31] PACKER E, INGRAHAM J, SCHER S. Factors affecting the rate oF killing of escherichia coli by repeated freezing and thawing. J Bacteriol. 1965; 89(3):718-724.
[32] SLEIGHT SC, WIGGINTON NS, LENSKI RE. Increased susceptibility to repeated freeze-thaw cycles in Escherichia coli following long-term evolution in a benign environment. BMC Evol Biol. 2006;6:104.
[33] WANG W, YUAN C, GENG T, et al. EphrinB2 overexpression enhances osteogenic differentiation of dental pulp stem cells partially through ephrinB2-mediated reverse signaling. Stem Cell Rese Ther. 2020;11(1):40.
[34] SCHULIK J, SALEHI S, BOCCACCINI A, et al. Comparison of the Behavior of 3D-Printed Endothelial Cells in Different Bioinks. Bioengineering. 2023; 10(7):751.
[35] LIU S, CHEN G, CHEN Z, et al. Research progress on stiffness controllable scaffolds based on gelatin methacryloyl hydrogels for tissue repair and reconstruction. Int J Biol Macromol. 2025;321(Pt 3):146485.
[36] LI N, ZHANG Z, ZHANG L, et al. Establishment of a Cochlear Organoid Platform for Remodeling the Extracellular Matrix. ACS Nano. 2025;19(29): 26542-26561.
[37] IM GB, LIN RZ. Bioengineering for vascularization: Trends and directions of photocrosslinkable gelatin methacrylate hydrogel. Front Bioeng Biotechnol. 2022;10:1053491.
[38] THAKAR H, SEBASTIAN SM, MANDAL S, et al. Biomolecule-Conjugated Macroporous Hydrogels for Biomedical Applications. ACS Biomater Sci Eng. 2019;5(12):6320-6341.
[39] CHOUDHARY H, ZHOU C, RAGHAVAN SR. A better picker-upper: Superabsorbent “gel sheets” with fabric-like flexibility. Matter. 2023;6(2): 521-536.
[40] SILVESTRO I, SERGI R, SCOTTO D’ABUSCO A, et al. Chitosan scaffolds with enhanced mechanical strength and elastic response by combination of freeze gelation, photo-crosslinking and freeze-drying. Carbohydr Polym. 2021;267: 118156.
[41] HANNINK G, ARTS JJ. Bioresorbability, porosity and mechanical strength of bone substitutes: What is optimal for bone regeneration? Injury. 2011; 42:S22-S25.
[42] LEE HP, STOWERS R, CHAUDHURI O. Volume expansion and TRPV4 activation regulate stem cell fate in three-dimensional microenvironments. Nat Commun. 2019;10(1):529.
|