Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (32): 8443-8453.doi: 10.12307/2025.876
Previous Articles Next Articles
Characterization and biological performance of manganese-doped hydroxyapatite/polydopamine composite materials
Yu Qiming, Di Jingyi, Zhang Hao, Chen Jilong, Xiao Hui, Hu Tuqiang
- Department of Stomatology, Shiyan Renmin Hospital (Affiliated Renmin Hospital of Hubei University of Medicine), Shiyan 442000, Hubei Province, China
-
Accepted:2026-02-04Online:2026-11-18Published:2026-04-27 -
Contact:Hu Tuqiang, MD, Chief physician, Department of Stomatology, Shiyan Renmin Hospital (Affiliated Renmin Hospital of Hubei University of Medicine), Shiyan 442000, Hubei Province, China -
About author:Yu Qiming, MS candidate, Physician, Department of Stomatology, Shiyan Renmin Hospital (Affiliated Renmin Hospital of Hubei University of Medicine), Shiyan 442000, Hubei Province, China -
Supported by:Hubei Provincial Health Research Fund, No. WJ2021M054 (to YQM); Graduate Science and Technology Innovation Project of Hubei University of Medicine, No. YC2024061 (to YQM)
CLC Number:
Cite this article
Yu Qiming, Di Jingyi, Zhang Hao, Chen Jilong, Xiao Hui, Hu Tuqiang. Characterization and biological performance of manganese-doped hydroxyapatite/polydopamine composite materials[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8443-8453.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
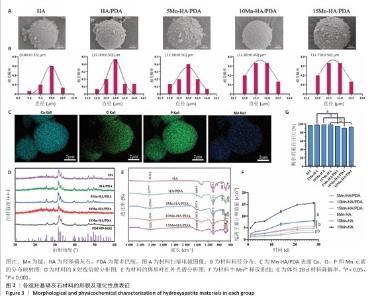
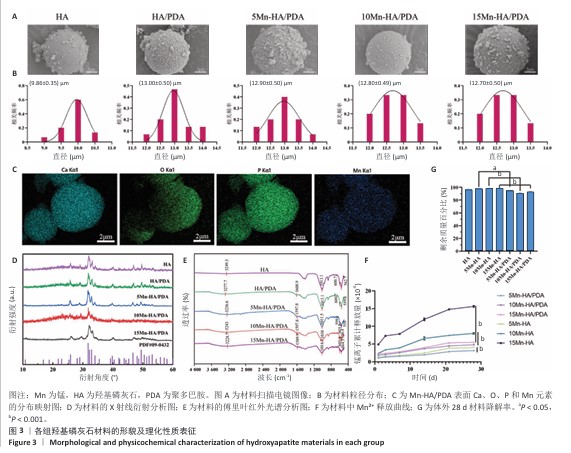
2.1.2 扫描电镜观察材料具体结构 扫描电镜下HA/PDA、5Mn-HA/PDA、10Mn-HA/PDA及15Mn-HA/PDA表面形态未见明显差异,均呈球状,表面为粗糙多孔结构;相较于纯羟基磷灰石,聚多巴胺修饰后的羟基磷灰石微球表面粗糙且见明显的颗粒状纹理(图3A),提示聚多巴胺成功黏附在羟基磷灰石微球表面。各组微球的粒径检测结果,如图3B所示。各组微球的粒径分布均较为集中,分布在9.86-13.00 μm范围内,呈正态分布趋势,说明材料尺寸具有良好的均一性;其中,纯羟基磷灰石的平均粒径最小,为(9.86±0.35) μm,经过聚多巴胺表面修饰的羟基磷灰石微球粒径均有不同程度增加。为对材料的元素组成进行分析,利用能量色散光谱对5Mn-HA/PDA进行检测,结果显示微球中可检测到Mn、Ca、P、O等元素,其中Ca、P主要来自于羟基磷灰石,O主要来自于聚多巴胺和羟基磷灰石,锰掺杂后可见锰元素均匀分布在微球中(图3C)。 2.1.3 X射线衍射对材料成分进行分析 纯羟基磷灰石样品的衍射峰与羟基磷灰石标准卡片高度一致,主要特征峰出现在2θ= 25.9°,31.8°,32.9°,34.1°,39.8°,46.7°,49.5°和53.2°处,体现出结晶性良好,并且具备较高的相纯度(图3D)。在HA/PDA组样品的图谱中各衍射峰位置均未出现变动,但峰强度略有下降,说明聚多巴胺的包覆并未改变羟基磷灰石的晶体结构,但由于聚多巴胺的无定形特性,能够对X射线信号产生一定吸收,导致衍射强度减弱。在锰掺杂样品中,随着锰掺杂量从5%逐步增加至15%,各组样品的衍射峰强度呈下降趋势且峰形逐渐增宽,尤其是在10Mn-HA/PDA组与15Mn-HA/PDA组中表现更为明显。这可能与Mn2+进入羟基磷灰石晶格后引起的晶格畸变或晶粒尺寸减小有关,但是所有锰掺杂样品的主要特征衍射峰并未发生明显位移。 2.1.4 傅里叶红外光谱检测材料官能团 在4 000-2 000 cm-1范围内,所有样品的光谱透射率变化较小,曲线较为平坦(图3E)。在3 236.6 cm-1处,各样品均显示一个较宽的吸收峰,归结于O-H键的伸缩振动,该峰提示材料中羟基基团的存在;在1 600 cm-1以下区域,HA/PDA组光谱在1 596.9 cm-1处观察到显著的吸收峰,对应芳香环的C=C骨架振动,而在羟基磷灰石组曲线对应位置未见明显特征峰,验证了聚多巴胺涂层的成功黏附。锰掺杂后样品的光谱趋势与羟基磷灰石、HA/PDA一致。另外,所有样品在1 009.7 cm-1处观察到显著吸收峰,对应P-O键的伸缩振动,而604.3 cm-1和563.8 cm-1处呈现的2个较强的吸收峰,归属于PO?3-的弯曲振动,进一步确认了羟基磷灰石成分在复合材料中的存在,并且锰掺杂未改变羟基磷灰石的基本结构。 2.1.5 材料锰离子释放量 从图3F可以看出,第1天观测时各组均有一定程度的Mn2+释放,随着时间的推移,Mn2+累计释放量逐渐增加,并且随着锰掺杂量的增加,材料Mn2+的累计释放量也逐渐升高,其中15Mn-HA组释放水平最高。与相应未修饰组相比,聚多巴胺修饰后的各组曲线整体更为平缓,并且在整个释放周期内Mn2+的累计释放量均低于相对应的未修饰组(P < 0.001),这一差异随着锰投料比例的增加而更为明显。该结果证明聚多巴胺在一定程度上抑制了Mn2+的突释,起到控释作用。 2.1.6 材料降解速率 如图3G所示,经过28 d的体外降解,6组材料均表现出较好的结构稳定性,剩余质量百分比均超过90%,但是10Mn-HA/PDA材料剩余质量百分比小于10Mn-HA(P < 0.001),15Mn-HA/PDA剩余质量百分比小于10Mn-HA(P < 0.001),5Mn-HA/PDA材料剩余质量百分比小于5Mn-HA(P < 0.05),说明与未修饰材料相比,经过聚多巴胺表面修饰的材料表现出更显著的质量损失。 "
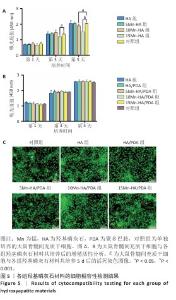
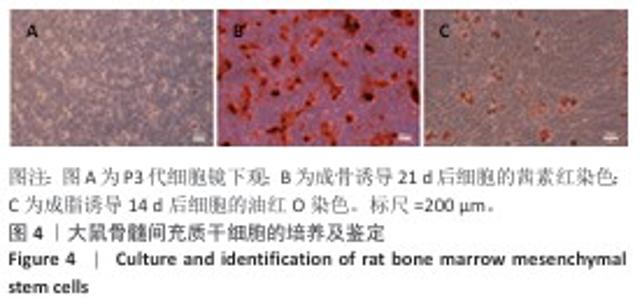
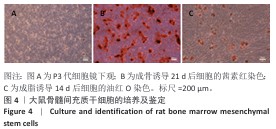
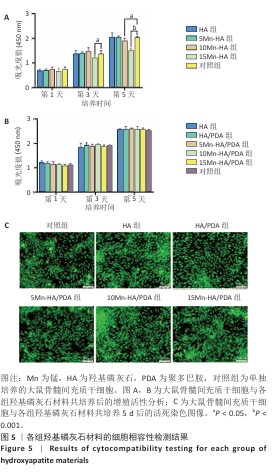
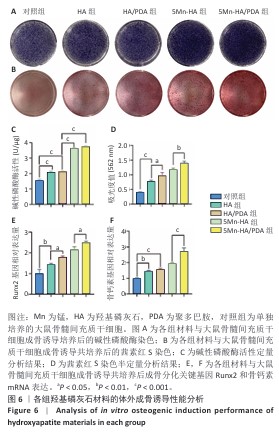
2.3 材料的细胞相容性评价结果 2.3.1 细胞增殖实验 如图5A所示,将未经聚多巴胺表面修饰的各组材料与大鼠骨髓间充质干细胞共培养,培养第1天,各组细胞增殖活性比较差异无显著性意义(P > 0.05);随培养时间延长,各组细胞呈现持续增殖趋势,至第3天,15Mn-HA组细胞增殖活性显著低于对照组(P < 0.05);培养第5天,10Mn-HA组、15Mn-HA组细胞增殖活性低于对照组(P < 0.05,P < 0.001),提示10%及15%锰掺杂羟基磷灰石可能对大鼠骨髓间充质干细胞的增殖产生一定抑制作用。如图5B所示,培养第1,3,5天,经聚多巴胺修饰后各材料组的细胞增殖活性与对照组相比差异无显著性意义(P > 0.05),提示聚多巴胺涂层能够有效调控Mn2+的释放速率,从而防止前期Mn2+突释,降低高浓度Mn2+对细胞活性的潜在负面影响。 2.3.2 细胞活死染色 为进一步验证聚多巴胺修饰后各组材料对大鼠骨髓间充质干细胞活性的影响,将各组材料与大鼠骨髓间充质干细胞共培养5 d后进行活死细胞染色,结果图5C所示。各组均可见到大量形态完整的梭形细胞,绿色荧光分布均匀,显示细胞健康的生长状态及较高的汇合度,代表死细胞的红色荧光信号极少,各组间无显著差异,进一步证明经聚多巴胺表面修饰材料具有较好的细胞相容性。这一结论与CCK-8实验的定量分析结果一致。 综合细胞增殖检测与活死染色结果,5%锰掺杂羟基磷灰石在聚多巴胺修饰前后的细胞活性与对照组相比无显著差别,因骨修复周期较长,为防止Mn2+浓度堆积所带来的潜在毒性影响,选择细胞相容性良好的5Mn-HA与5Mn-HA/PDA进行后续成骨相关实验。 "
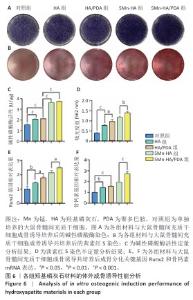
2.4 材料的体外骨诱导能力检测结果 为评价各组材料对大鼠骨髓间充质干细胞成骨分化的影响,分别通过碱性磷酸酶活性检测和茜素红S染色对成骨分化早、晚期标志物进行了定性与定量分析,见图6A-D。 2.4.1 碱性磷酸酶活性检测 碱性磷酸酶染色结果显示,与对照组相比,羟基磷灰石组与HA/PDA组染色程度略有加深,5Mn-HA 组和5Mn-HA/PDA组染色程度进一步加深,表现出更加显著的碱性磷酸酶活性。碱性磷酸酶活性定量检测结果进一步印证了染色实验的趋势:5Mn-HA组及5Mn-HA/PDA组碱性磷酸酶活性最高,均显著高于对照组、羟基磷灰石组、HA/PDA组(P < 0.001);而羟基磷灰石组及HA/PDA组碱性磷酸酶活性亦显著高于对照组(P < 0.001)。 2.4.2 茜素红S染色及定量分析 茜素红S染色结果显示,与对照组相比,羟基磷灰石组和HA/PDA组可见明显的钙盐沉积,并且HA/PDA组钙盐沉积更为显著;另外,相较于羟基磷灰石组和HA/PDA组,5Mn-HA组、5Mn-HA/PDA组钙盐沉积明显增加,其中以5Mn-HA/PDA组增加最为显著。定量检测结果显示,5Mn-HA/PDA组吸光度值高于其余4组,HA/PDA组吸光度值高于对照组、羟基磷灰石组,并且羟基磷灰石组吸光度值高于对照组,差异均有显著性意义。说明引入锰掺杂羟基磷灰石的促成骨分化能力进一步增强,以5Mn-HA/PDA组最为显著。 2.4.3 q-PCR检测成骨相关基因表达 Runx2为成骨分化早期的关键转录因子,骨钙素为晚期成骨标志物,为从分子水平验证材料的促成骨性能,通过q-PCR检测了成骨分化的关键基因Runx2和骨钙素mRNA表达,结果如图6E,F所示。5Mn-HA/PDA组Runx2及骨钙素mRNA表达量最高,显著高于其余4组。羟基磷灰石组、HA/PDA组、5Mn-HA组Runx2及骨钙素mRNA表达表达量亦均显著高于对照组;其中,HA/PDA组Runx2表达量亦显著高于羟基磷灰石组,差异均有显著性意义,该结果进一步证实了5Mn-HA/PDA具有较好的成骨诱导活性。 "
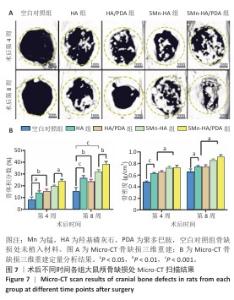
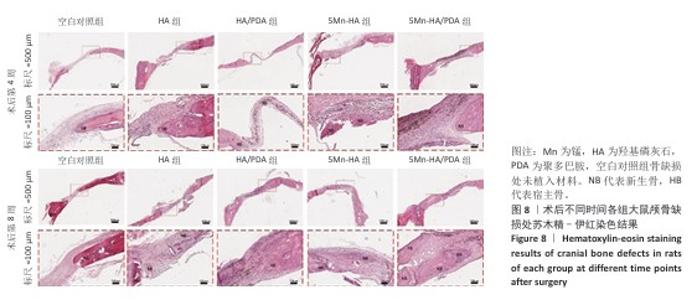
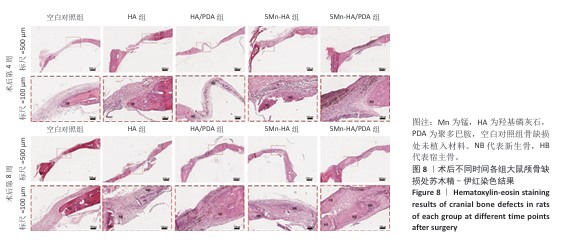
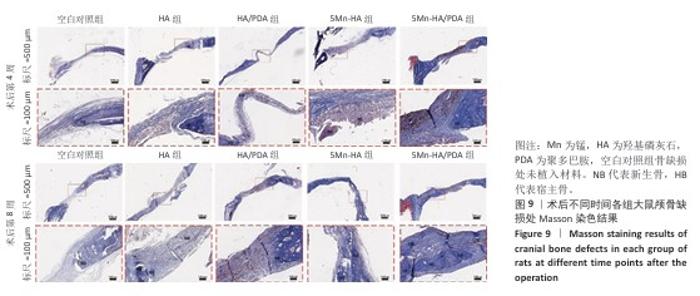
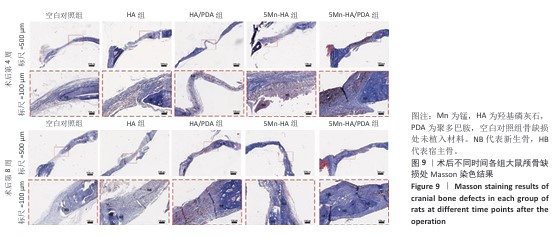
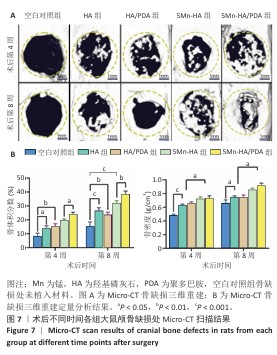
2.5 材料的动物体内骨诱导能力检测结果 2.5.1 实验动物数量分析 24只SD大鼠全部进入结果分析。 2.5.2 大鼠颅骨Micro-CT检查 术后第4,8周,各组大鼠颅骨缺损部位Micro-CT成像结果,见图7A。术后第4周,空白对照组骨缺损区内几乎无新骨形成,呈现大面积空洞;羟基磷灰石组和HA/PDA组骨缺损区内可见少量新生骨组织,5Mn-HA组、5Mn-HA/PDA组骨缺损区内可见较多新生骨组织,以5Mn-HA/PDA组新生骨组织更多。术后第8周,空白对照组骨缺损区内仅见少量新生骨组织,羟基磷灰石组和HA/PDA组骨缺损区内新生骨组织较术后4周增加,5Mn-HA组、5Mn-HA/PDA组骨缺损区内可见大量的新生骨组织,并且5Mn-HA/PDA组缺损区域基本被新生骨组织完全覆盖,与周围宿主骨形成了良好的结合。 进一步分析了新生骨的骨体积分数和骨密度,结果如图7B所示。术后第4周,5Mn-HA/PDA组骨体积分数最高,并且显著高于其余4组;5Mn-HA组骨体积分数亦明显高于空白对照组、羟基磷灰石组和HA/PDA组。同时,与空白对照组相比,羟基磷灰石组和HA/PDA组骨体积分数均显著升高。术后第8周,各组骨体积分数较第4周均呈上升趋势,其中5Mn-HA/PDA组仍为最高,显著优于其他4组;此外,羟基磷灰石组、HA/PDA组及5Mn-HA组骨体积分数均显著高于空白对照组。在骨密度方面,术后第4周和第8周,5Mn-HA组及5Mn-HA/PDA组均显著高于羟基磷灰石组与HA/PDA组,羟基磷灰石组和HA/PDA组显著高于空白对照组,差异均具有显著性意义。 2.5.3 组织学分析 苏木精-伊红染色结果直观地反映了缺损区域的组织构成和细胞状态,各组骨缺损处苏木精-伊红染色结果,见图8。术后第4周,空白对照组骨缺损区域主要为疏松的纤维结缔组织,无明显新生骨形成;羟基磷灰石组与HA/PDA组骨缺损边缘可见少量新生骨组织,但新骨形成量有限,中心区域仍为纤维组织占据,HA/PDA组材料被纤维组织包裹;相比之下,5Mn-HA组骨缺损处新生骨小梁数量显著增多,5Mn-HA/PDA组新生骨小梁数量最多,但中央仍以纤维组织为主。术后第8周,空白对照组骨修复基本停滞,羟基磷灰石组与HA/PDA组新生骨量虽有增加,但仍局限于缺损边缘,未能形成有效骨桥接;5Mn-HA组骨缺损中央观察到明显的骨岛形成,伴有成骨细胞在周围聚集,5Mn-HA/PDA组相比于5Mn-HA组修复效果更好,骨缺损中央形成了大面积骨岛,并能明显观察到有从边缘向中心生长的新骨形成。 Masson染色结果:Masson染色结果与苏木精-伊红染色相符,如图9所示。术后第4周,对照组蓝染面积稀少,纤维束松散;羟基磷灰石组与HA/PDA组在缺损边缘出现薄层蓝染向内延伸,但中央仍以浅蓝、疏松的纤维性蓝染为主,并且连续性较弱;而5Mn-HA组可见蓝染的骨基质沿边缘向内侧延伸;5Mn-HA/PDA组边缘蓝染最为明显、范围更大,小梁周围蓝染加深,但中央仍以浅蓝纤维为主,连续性尚不足,骨桥接尚未形成。术后第8周,羟基磷灰石组与HA/PDA组的蓝染范围和强度有所增加,但仍未实现骨性桥接;5Mn-HA组中央出现散在的骨岛样蓝染;5Mn-HA/PDA组则在缺损中央形成了大面积致密的蓝色骨岛,显示出向缺损中心长入的趋势,边缘与中心的骨连接性显著增强。"
| [1] SHAH KN, KAMAL RN. Bone Graft Substitutes-What Are My Options? Hand Clin. 2024;40(1):13-23. [2] BUSER D, URBAN I, MONJE A, et al. Guided bone regeneration in implant dentistry: Basic principle, progress over 35 years, and recent research activities. Periodontology 2000. 2023;93(1):29-25. [3] GOLROKHIAN M, FAKHIMI REZAEI H, REZAEIANJAM M, et al. Recent advances and clinical potential of hyaluronic acid methacrylate (HAMA)/ceramic composites in oral and dental regenerative therapies: A comprehensive review. Biomed Pharmacother. 2025; 192:118590. [4] MCKENNA GJ, GJENGEDAL H, HARKIN J, et al. Effect of autogenous bone graft site ondental implant survival and donor site complications: a systematic review and meta-analysis. J Evid Based Dent Pract. 2022; 22(3):101731. [5] PERCIVAL MK, PAUL V, HUSSEINI AG. Recent Advancements in Bone Tissue Engineering: Integrating Smart Scaffold Technologies and Bio-Responsive Systems for Enhanced Regeneration. Int J Mol Sci. 2024;25(11):6012. [6] NICOLAE CL, PÎRVULESCU DC, NICULESCU AG, et al. An Up-to-Date Review of Materials Science Advances in Bone Grafting for Oral and Maxillofacial Pathology. Materials (Basel). 2024;17(19):4782. [7] ZHANG J, ZHANG W, YUE W, et al. Research Progress of Bone Grafting: A Comprehensive Review. Int J Nanomedicine. 2025;20:4729-4757. [8] FERRAZ MP. Bone Grafts in Dental Medicine: An Overview of Autografts, Allografts and Synthetic Materials. Materials (Basel). 2023;16(11):4117. [9] WEI H, CUI J, LIN K, et al. Recent advances in smart stimuli-responsive biomaterials for bone therapeutics and regeneration. Bone Res. 2022;10(1):17. [10] LIU W, CHEONG N, HE Z, et al. Application of Hydroxyapatite Composites in Bone Tissue Engineering: A Review. J Funct Biomater. 2025;16(4):127. [11] ZASTULKA A, CLICHICI S, TOMOAIA-COTISEL M, et al. Recent Trends in Hydroxyapatite Supplementation for Osteoregenerative Purposes. Materials (Basel). 2023;16(3):1303. [12] SHANG J, WANG Z, ZHANG Z. Composite or Modified Hydroxyapatite Microspheres as Drug Delivery Carrier for Bone and Tooth Tissue Engineering. Curr Med Chem. 2025;32(5):974-981. [13] ZENG J, XIONG S, ZHOU J, et al. Hollow Hydroxyapatite Microspheres Loaded with rhCXCL13 to Recruit BMSC for Osteogenesis and Synergetic Angiogenesis to Promote Bone Regeneration in Bone Defects. Int J Nanomedicine. 2023;18:3509-3534. [14] TANG Z, LI X, TAN Y, et al. The material and biological characteristics of osteoinductive calcium phosphate ceramics. Regen Biomater. 2018;5(1):43-59. [15] 魏莉,马保金,邵金龙,等.羟基磷灰石复合材料在骨组织工程中应用的研究进展[J].四川大学学报(医学版),2021,52(3):357-363. [16] MUTHUSAMY S, MAHENDIRAN B, SAMPATH S, et al. Hydroxyapatite nanophases augmented with selenium and manganese ions for bone regeneration: Physiochemical, microstructural and biological characterization. Mater Sci Eng C Mater Biol Appl. 2021;126:112149. [17] HUANG H, QIANG L, FAN M, et al. 3D-printed tri-element-doped hydroxyapatite/ polycaprolactone composite scaffolds with antibacterial potential for osteosarcoma therapy and bone regeneration. Bioact Mater. 2023;31:18-37. [18] SUN Z, SHAO Y, YAN K, et al. The Link between Trace Metal Elements and Glucose Metabolism: Evidence from Zinc, Copper, Iron, and Manganese-Mediated Metabolic Regulation. Metabolites. 2023; 13(10):1048. [19] TASKOZHINA G, BATYROVA G, UMAROVA G, et al. The Manganese-Bone Connection: Investigating the Role of Manganese in Bone Health.J Clin Med. 2024;13(16):4679. [20] RAU JV, FADEEVA IV, FOMIN AS, et al. Sic Parvis Magna: Manganese-Substituted Tricalcium Phosphate and Its Biophysical Properties. ACS Biomater Sci Eng. 2019;5(12):6632-6644. [21] BARRIONI BR, NORRIS E, LI S, et al. Osteogenic potential of sol-gel bioactive glasses containing manganese. J Mater Sci Mater Med. 2019;30(7):86. [22] 高雪钰,张文涛,孙天泽,等.金属离子在骨组织工程中的应用[J].中国组织工程研究,2024,28(3):439-444. [23] 张畅,喻正文.锰元素在生物材料中的应用:现状和问题[J].中国组织工程研究,2022,26(34):5504-5511. [24] WESTHAUSER F, WILKESMANN S, NAWAZ Q, et al. Osteogenic properties of manganese-doped mesoporous bioactive glass nanoparticles. J Biomed Mater Res A. 2020;108(9):1806-1815. [25] BAJ J, FLIEGER W, BARBACHOWSKA A, et al. Consequences of Disturbing Manganese Homeostasis. Int J Mol Sci. 2023;24(19):14959. [26] LÜTHEN F, BULNHEIM U, MÜLLER PD, et al. Influence of manganese ions on cellular behavior of human osteoblasts in vitro. Biomol Eng. 2007;24(5):531-536. [27] MAURELLI AM, DE LEO V, CATUCCI L. Polydopamine-Modified Liposomes: Preparation and Recent Applications in the Biomedical Field. ACS Omega. 2024;9(23):24105-24120. [28] DENG Z, SHANG B, PENG B. Polydopamine Based Colloidal Materials: Synthesis and Applications. Chem Rec. 2018;18(4):410-432. [29] OMIDIAN H, WILSON RL. Polydopamine Applications in Biomedicine and Environmental Science. Materials (Basel). 2024;17(16):3916. [30] LI H, JIA Y, BAI S, et al. Metal-chelated polydopamine nanomaterials: Nanoarchitectonics and applications in biomedicine, catalysis, and energy storage. Adv Colloid Interface Sci. 2024;334:103316. [31] 李承瑜,丁自友,韩颖超.锰掺杂纳米羟基磷灰石的体外抗菌-促成骨性能研究[J].无机材料学报,2024,39(3):313-320. [32] SPICER PP, KRETLOW JD, YOUNG S, et al. Evaluation of bone regeneration using the rat critical size calvarial defect. Nat Protoc. 2012;7(10):1918-1929. [33] ZHANG Y, FAN M, ZHANG Y. Revolutionizing bone defect healing: the power of mesenchymal stem cells as seeds. Front Bioeng Biotechnol. 2024;12:1421674. [34] ORYAN A, KAMALI A, MOSHIRI A, et al. Role of Mesenchymal Stem Cells in Bone Regenerative Medicine:What Is the Evidence? Cells Tissues Organs. 2017;204(2):59-83. [35] SABERIAN E, JENČA A, ZAFARI Y, et al. Scaffold Application for Bone Regeneration with Stem Cells in Dentistry: Literature Review. Cells. 2024;13(12):1065. [36] LIU Z, TANG M, ZHAO J, et al. Looking into the Future: Toward Advanced 3D Biomaterials for Stem-Cell-Based Regenerative Medicine. Adv Mater. 2018;30(17):e1705388. [37] LI N, WANG J, FENG G, et al. Advances in biomaterials for oral-maxillofacial bone regeneration: spotlight on periodontal and alveolar bone strategies. Regen Biomater. 2024;11:rbae078. [38] WU H, ZHANG Q, ZHU J, et al.Biomaterials targeting senescent cells for bone regeneration: State-of-the-art and future perspectives. Bioact Mater. 2025;54:686-714. [39] ZHAO R, MENG X, PAN Z, et al. Advancements in nanohydroxyapatite: synthesis, biomedical applications and composite developments. Regen Biomater. 2024;12:rbae129. [40] BRISTY NS, KAWSAR M, SAHADAT HOSSAIN M. Effects of different types of modifiers on structural variation of nano-hydroxyapatite for efficient application. Nanoscale Adv. 2025;7(17):5133-5160. [41] DILEEPKUMAR VG, SRIDHAR MS, ARAMWIT P, et al.A Review on the Synthesis and Properties of Hydroxyapatite for Biomedical Applications. J Biomater Sci Polym Ed. 2022;33(2):229-261. [42] ALKARON W, ALMANSOORI A, BALÁZSI K, et al. Hydroxyapatite-Based Natural Biopolymer Composite for Tissue Regeneration. Materials (Basel). 2024;17(16):4117. [43] ULLAH I, SIDDIQUI MA, LIU H, et al. Mechanical, Biological, and Antibacterial Characteristics of Plasma-Sprayed (Sr,Zn) Substituted Hydroxyapatite Coating. ACS Biomater Sci Eng. 2020;6(3):1355-1366. [44] AOKI K, IDETA H, KOMATSU Y, et al. Bone-Regeneration Therapy Using Biodegradable Scaffolds: Calcium Phosphate Bioceramics and Biodegradable Polymers. Bioengineering (Basel). 2024;11(2):180. [45] SANTORO A, VOTO A, FORTINO L, et al.Bone Defect Treatment in Regenerative Medicine: Exploring Natural and Synthetic Bone Substitutes. Int J Mol Sci. 2025;26(7):3085. [46] JIN P, LIU L, CHENG L, et al. Calcium-to-phosphorus releasing ratio affects osteoinductivity and osteoconductivity of calcium phosphate bioceramics in bone tissue engineering. Biomed Eng Online. 2023; 22(1):12. [47] 王鑫杰,王国栋,郑中忍,等.骨修复领域的含锰生物陶瓷材料[J].中国组织工程研究,2023,27(34):5570-5576. [48] LI R, ZHU Z, ZHANG B, et al. Manganese Enhances the Osteogenic Effect of Silicon-Hydroxyapatite Nanowires by Targeting T Lymphocyte Polarization. Adv Sci (Weinh). 2024;11(4):e2305890. [49] KOLMAS J, GROSZYK E, PIOTROWSKA U. Nanocrystalline hydroxyapatite enriched in selenite and manganese ions: physicochemical and antibacterial properties. Nanoscale Res Lett. 2015;10(1):989. [50] LI J, DENG C, LIANG W, et al. Mn-containing bioceramics inhibit osteoclastogenesis and promote osteoporotic bone regeneration via scavenging ROS. Bioact Mater. 2021;6(11):3839-3850. [51] VILLASEÑOR-CERÓN LS, MENDOZA-ANAYA D, LÓPEZ-ORTIZ S, et al. Biocompatibility analysis and chemical characterization of Mn-doped hydroxyapatite. J Mater Sci Mater Med. 2023;34(8):40. [52] HE J, WANG G, ZHOU Y, et al. Recent advances in polydopamine-coated metal-organic frameworks for cancer therapy. Front Bioeng Biotechnol. 2025;13:1553653. [53] DU J, ZHOU Y, BAO X, et al. Surface polydopamine modification of bone defect repair materials: Characteristics and applications. Front Bioeng Biotechnol. 2022;10:974533. [54] WANG Z, DENG T, CHANG H, et al. Translating m6A-Glycolysis Discovery into Therapy: GOQD-Based Multifunctional Bioactive Scaffolds Rejuvenates Bone Repair in Senescence. Small. 2025; 21(43):e07315. [55] WANG Z, ZOU Y, LI Y, et al. Metal-Containing Polydopamine Nanomaterials: Catalysis, Energy, and Theranostics. Small. 2020; 16(18):e1907042. [56] 谭帼馨,欧阳孔友,周蕾,等.聚多巴胺螯合钙离子对钛表面的修饰及修饰后的细胞相容性[J].无机材料学报,2015,30(10): 1075-1080. [57] KANG J, MENG S, LIU C, et al. Polydopamine-assisted dual metal ion modification of titanium: Enhancing osseointegration and antibacterial performance. Colloids Surf B Biointerfaces. 2025;253:114717. [58] WESTHAUSER F, WILKESMANN S, NAWAZ Q, et al. Effect of manganese, zinc and copper on the biological and osteogenic properties of mesoporous bioactive glass nanoparticles. J Biomed Mater Res A. 2021;109(8):1457-1467. [59] ZHANG K, LI H, WANG T, et al. Mechanisms of bone regeneration repair and potential and efficacy of small molecule drugs. Biomed Pharmacother. 2025;187:118070. [60] ŁUCZAK JW, PALUSIŃSKA M, MATAK D, et al. The Future of Bone Repair: Emerging Technologies and Biomaterials in Bone Regeneration. Int J Mol Sci. 2024;25(23):12766. [61] KAUSHIK N, NHAT NGUYEN L, KIM JH, et al. Strategies for Using Polydopamine to Induce Biomineralization of Hydroxyapatite on Implant Materials for Bone Tissue Engineering. Int J Mol Sci. 2020;21(18):6544. |
| [1] | Yang Yanjun, Zhu Lin, Gu Yongchun, Yan Zhanjun. Mechanism by which magnesium implant-activated integrin α10β1 promotes osteogenic differentiation of periosteal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8319-8326. |
| [2] | Yang Ping, Qi Xiaoyang, Lei Zhijie, Chen Yixin, Qiu Xusheng. Novel collagen membrane in repairing skull bone defects in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8364-8371. |
| [3] | Fan Haixia, Tan Qingkun, Wang Hong, Cheng Huanzhi, Liu Xue, Ching-chang Ko, Geng Haixia. Rabbit skull defects repaired by the hydroxyapatite/geltin scaffold combined with bone marrow mesenchymal stem cells and umbilical vein endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1495-1499. |
| [4] | Shi Qianhui, Wu Chao, Zhou Qian, Cheng Yuting, Li Fang, Huo Hua, Qi Yuhan, Huang Xiaolin, Wang Yong, Liao Jian. Prevention and treatment of implant periapical lesions [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5434-5440. |
| [5] | Zhang Huxiong, Li Wei, Yang Wupeng, New Suyaratu. 3D-printed icariin/decalcified bone matrix material promotes the repair of femoral condyle defects in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5461-5466. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||