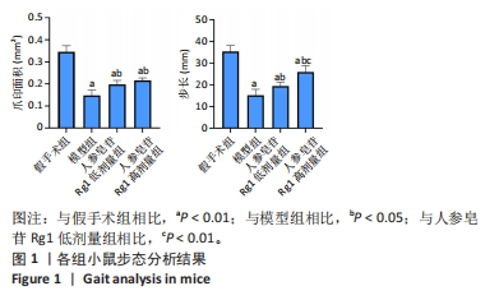
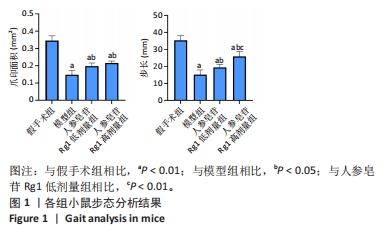
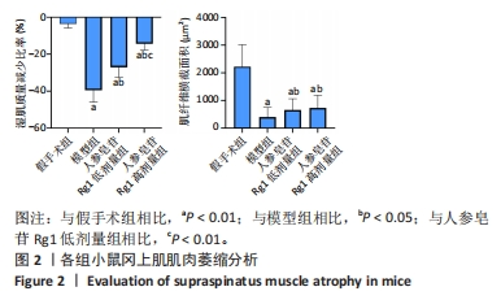
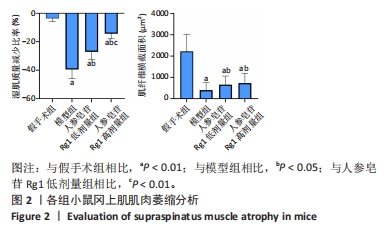
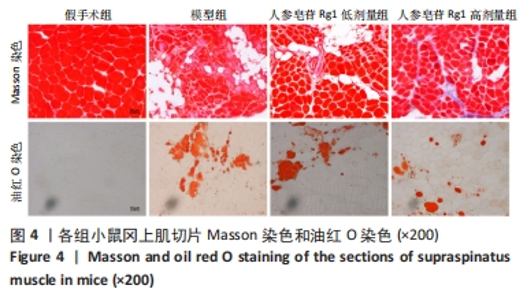
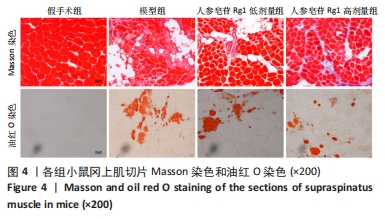
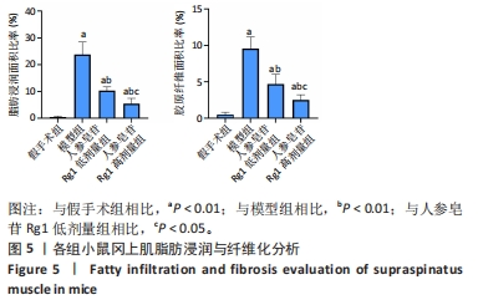
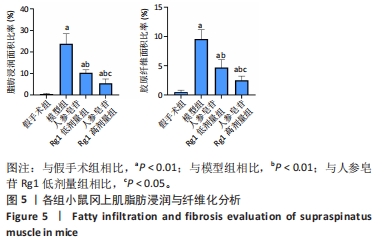
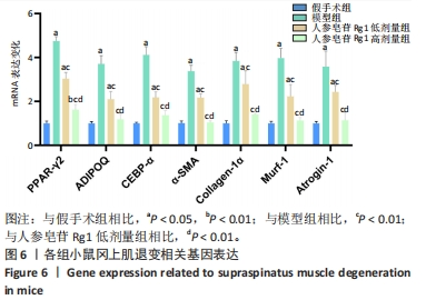
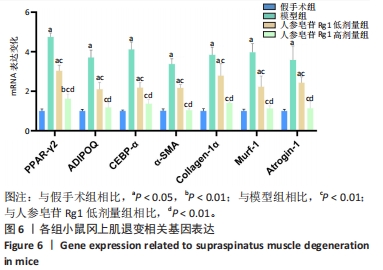
Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (32): 5136-5140.doi: 10.12307/2024.504
Previous Articles Next Articles
Effect of ginsenoside Rg1 on muscle degeneration after massive rotator cuff injury in mice
He Rongzhen1, 2, Ying Lyufang2, He Xingwen2, Chen Chuanshun1, Yin Yuesong1, Zhang Kexiang1, Wang Zili1
- 1Department of Orthopaedics, The Third Xiangya Hospital of Central South University, Changsha 410006, Hunan Province, China; 2Department of Orthopaedics, Ningbo Hangzhou Bay Hospital, Ningbo 315336, Zhejiang Province, China
-
Received:2023-08-10Accepted:2023-10-12Online:2024-11-18Published:2023-12-28 -
Contact:Wang Zili, MD, Assistant researcher, Department of Orthopaedics, The Third Xiangya Hospital of Central South University, Changsha 410006, Hunan Province, China -
About author:He Rongzhen, Master candidate, Associate chief physician, Department of Orthopaedics, Department of Orthopaedics, The Third Xiangya Hospital of Central South University, Changsha 410006, Hunan Province, China; Department of Orthopaedics, Ningbo Hangzhou Bay Hospital, Ningbo 315336, Zhejiang Province, China -
Supported by:Science and Technology Program of Traditional Chinese Medicine of Zhejiang Province, No. 2020ZB231 (to HRZ); General Guidance Subject of Hunan Provincial Health Commission, No. 202204073833 (to ZKX)
CLC Number:
Cite this article
He Rongzhen, Ying Lyufang, He Xingwen, Chen Chuanshun, Yin Yuesong, Zhang Kexiang, Wang Zili. Effect of ginsenoside Rg1 on muscle degeneration after massive rotator cuff injury in mice[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(32): 5136-5140.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
| [1] PIPER CC, HUGHES AJ, MA Y, et al. Operative versus nonoperative treatment for the management of full-thickness rotator cuff tears: a systematic review and meta-analysis. J Shoulder Elbow Surg. 2018;27(3):572-576. [2] CHEUNG S, DILLON E, THAM SC, et al. The presence of fatty infiltration in the infraspinatus: its relation with the condition of the supraspinatus tendon. Arthroscopy. 2011;27(4):463-470. [3] JEONG HY, KIM HJ, JEON YS, et al. Factors Predictive of Healing in Large Rotator Cuff Tears: Is It Possible to Predict Retear Preoperatively? Am J Sports Med. 2018;46(7):1693-1700. [4] COLLIN P, KEMPF JF, MOLÉ D, et al. Ten-Year Multicenter Clinical and MRI Evaluation of Isolated Supraspinatus Repairs. J Bone Joint Surg Am. 2017;99(16): 1355-1364. [5] GLADSTONE JN, BISHOP JY, LO IK, et al. Fatty infiltration and atrophy of the rotator cuff do not improve after rotator cuff repair and correlate with poor functional outcome. Am J Sports Med. 2007;35(5):719-728. [6] ELIASBERG CD, DAR A, JENSEN AR, et al. Perivascular Stem Cells Diminish Muscle Atrophy Following Massive Rotator Cuff Tears in a Small Animal Model. J Bone Joint Surg Am. 2017;99(4):331-341. [7] DENIZ G, KOSE O, TUGAY A, et al. Fatty degeneration and atrophy of the rotator cuff muscles after arthroscopic repair: does it improve, halt or deteriorate? Arch Orthop Trauma Surg. 2014;134(7):985-990. [8] LIEM D, LICHTENBERG S, MAGOSCH P, et al. Magnetic resonance imaging of arthroscopic supraspinatus tendon repair. J Bone Joint Surg Am. 2007;89(8):1770-1776. [9] WANG Z, LIU X, DAVIES MR, et al. A Mouse Model of Delayed Rotator Cuff Repair Results in Persistent Muscle Atrophy and Fatty Infiltration. Am J Sports Med. 2018;46(12):2981-2989. [10] HAMANO N, YAMAMOTO A, SHITARA H, et al. Does successful rotator cuff repair improve muscle atrophy and fatty infiltration of the rotator cuff? A retrospective magnetic resonance imaging study performed shortly after surgery as a reference. J Shoulder Elbow Surg. 2017;26(6):967-974. [11] LAPNER PL, JIANG L, ZHANG T, et al. Rotator cuff fatty infiltration and atrophy are associated with functional outcomes in anatomic shoulder arthroplasty. Clin Orthop Relat Res. 2015;473(2):674-682. [12] ZHOU H, CHEN C, HU H, et al. High-intensity interval training improves fatty infiltration in the rotator cuff through the β3 adrenergic receptor in mice. Bone Joint Res. 2023;12(8):455-466. [13] WANG Z, LIU X, JIANG K, et al. Intramuscular Brown Fat Activation Decreases Muscle Atrophy and Fatty Infiltration and Improves Gait After Delayed Rotator Cuff Repair in Mice. Am J Sports Med. 2020;48(7):1590-1600. [14] 李赫健,李虹,金玉,等.人参皂苷Rg1、Rb1的药效及作用机制研究进展[J].武汉大学学报(理学版),2019,65(4):323-332. [15] 郝菲,韩俊婷,李晓辉,等.人参皂苷Rg1对ApoE-/-小鼠血脂、VWF及MMP-9蛋白水平的影响[J].武警后勤学院学报(医学版),2019,28(6):21-23. [16] WANG Z, FEELEY BT, KIM HT, et al. Reversal of Fatty Infiltration After Suprascapular Nerve Compression Release Is Dependent on UCP1 Expression in Mice. Clin Orthop Relat Res. 2018;476(8):1665-1679. [17] BRYNIARSKI AR, MEYER GA. Brown Fat Promotes Muscle Growth During Regeneration. J Orthop Res. 2019;37(8):1817-1826. [18] RYÖSÄ A, LAIMI K, ÄÄRIMAA V, et al. Surgery or conservative treatment for rotator cuff tear: a meta-analysis. Disabil Rehabil. 2017;39(14):1357-1363. [19] LONGO UG, CARNEVALE A, PIERGENTILI I, et al. Retear rates after rotator cuff surgery: a systematic review and meta-analysis. BMC Musculoskelet Disord. 2021;22(1):749. [20] UEZUMI A, FUKADA S, YAMAMOTO N, et al. Mesenchymal progenitors distinct from satellite cells contribute to ectopic fat cell formation in skeletal muscle. Nat Cell Biol. 2010;12(2):143-152. [21] LIU X, NING AY, CHANG NC, et al. Investigating the cellular origin of rotator cuff muscle fatty infiltration and fibrosis after injury. Muscles Ligaments Tendons J. 2016;6(1):6-15. [22] KUZEL BR, GRINDEL S, PAPANDREA R, et al. Fatty infiltration and rotator cuff atrophy. J Am Acad Orthop Surg. 2013;21(10):613-623. [23] DANG A, DAVIES M. Rotator Cuff Disease: Treatment Options and Considerations. Sports Med Arthrosc Rev. 2018;26(3):129-133. [24] WILSON A, MACLEAN SB. Fatty infiltration in the intact supraspinatus tendon; a normal physiological response with increasing age and female gender. Shoulder Elbow. 2022;14(5):510-514. [25] LEE SJ, LEE EJ, KIM SH, et al. IL-17A promotes transdifferentiation of mouse myoblast cells (C2C12) into adipocytes by increasing the expression of peroxisome proliferator-activated receptor γ through CAAT/enhancer binding protein β signaling. Biotechnol Lett. 2011;33(2):229-235. [26] LUO H, ZHOU Y, HU X, et al. Activation of PPARγ2 by PPARγ1 through a functional PPRE in transdifferentiation of myoblasts to adipocytes induced by EPA. Cell Cycle. 2015;14(12):1830-1841. [27] AGHA O, DIAZ A, DAVIES M, et al. Rotator cuff tear degeneration and the role of fibro-adipogenic progenitors. Ann N Y Acad Sci. 2021;1490(1):13-28. [28] DAVIES MR, GARCIA S, LIU M, et al. Muscle-Derived Beige Adipose Precursors Secrete Promyogenic Exosomes That Treat Rotator Cuff Muscle Degeneration in Mice and Are Identified in Humans by Single-Cell RNA Sequencing. Am J Sports Med. 2022;50(8):2247-2257. [29] TIAN W, CHEN L, ZHANG L, et al. Effects of ginsenoside Rg1 on glucose metabolism and liver injury in streptozotocin-induced type 2 diabetic rats. Genet Mol Res. 2017;16(1). [30] LIU H, WANG J, LIU M, et al. Antiobesity Effects of Ginsenoside Rg1 on 3T3-L1 Preadipocytes and High Fat Diet-Induced Obese Mice Mediated by AMPK. Nutrients. 2018;10(7):830. [31] SHIN SS, YOON M. Korean red ginseng (Panax ginseng) inhibits obesity and improves lipid metabolism in high fat diet-fed castrated mice. J Ethnopharmacol. 2018;210:80-87. [32] NAIMARK M, TRINH T, ROBBINS C, et al. Effect of Muscle Quality on Operative and Nonoperative Treatment of Rotator Cuff Tears. Orthop J Sports Med. 2019; 7(8):2325967119863010. [33] GO GY, LEE SJ, JO A, et al. Ginsenoside Rg1 from Panax ginseng enhances myoblast differentiation and myotube growth. J Ginseng Res. 2017;41(4):608-614. [34] JEONG HJ, SO HK, JO A, et al. Ginsenoside Rg1 augments oxidative metabolism and anabolic response of skeletal muscle in mice. J Ginseng Res. 2019;43(3):475-481. [35] WU J, SAOVIENG S, CHENG IS, et al. Ginsenoside Rg1 supplementation clears senescence-associated β-galactosidase in exercising human skeletal muscle. J Ginseng Res. 2019;43(4):580-588. [36] LEE TXY, WU J, JEAN WH, et al. Reduced stem cell aging in exercised human skeletal muscle is enhanced by ginsenoside Rg1. Aging (Albany NY). 2021;13(12): 16567-16576. |
| [1] | Zhu Chenchen, Yin Yuan, Zhou Yingji, Ma Tingting, Su Liyao, Zhang Ming. Effects of body mass index on plantar pressure [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(18): 2808-2813. |
| [2] | Yang Wanrong, Chu Yi, Xu Yao, Li Sihui, Guo Ling. Ginsenoside Rg1 inhibits H2O2-induced apoptosis and autophagy in human periodontal ligament cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(13): 2061-2067. |
| [3] | Chen Modi, Sun Qibo, Xu Tianyu, Tai Guoliang, Zhao Yuxiang, Pan Zhaohui. Biomechanics of reconstruction of total calcaneus defect using fibular flap based on finite element method [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(12): 1805-1809. |
| [4] | Gu Xu, Zheng Xin, Shi Sifeng, Lu Renxiang, Cao Jie, Li Hongwei. Stability of early gait after unicompartmental knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(12): 1875-1879. |
| [5] | Huang Yongbin, Wang Tao, Lou Yuanyi, Pang Jingqun, Chen Guanghua. Application prospect of mesenchymal stem cells in promoting muscle tissue repair [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 107-112. |
| [6] | Yu Wenqiang, Ren Fuchao, Shi Guohong, Xu Yuanjing, Liu Tongyou, Xie Youzhuan, Wang Jinwu, . Methods and application of gait analysis of lower limbs after stroke [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1257-1263. |
| [7] | Liu Hongwen, Li Jiao, Xu Wenhao, Nie Hua, Liu Shaojiang, Xu Jie, Yin Li. Differential expression profiles of microRNAs in muscle tissue of denervated skeletal muscle atrophy rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 732-737. |
| [8] | Li Shihao, Li Qi, Li Zhen, Zhang Yuanyuan, Liu Miaomiao, Ouyang Yi, Xu Weiguo. Plantar pressure and gait analysis in patients with anterior cruciate ligament injury and reconstruction [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 626-631. |
| [9] | Yang Qiang, Huang Jian. Measurement research method and measurement equipment for knee-spine syndrome [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(29): 4751-4756. |
| [10] | Yang Yazhu, Du Juan, Qu Haifeng, Li Jianmin, Zhang Yuxin, Liu Junjie. Effect of ginsenoside Rg1 on learning and memory ability of brain aging mice induced by D-galactose [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(28): 4487-4493. |
| [11] | Liu Xinhao, Ma Xinran, Yang Shengyong, Wang Yanyu, Ma Shujie. Influence of Tuina manipulation on walking dysfunction in rats with sciatic nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(26): 4101-4106. |
| [12] | Cao Congcong, Ling Gengfei, Yang Chunhua. Local injection of ginsenoside Rg1 nanoparticles in the treatment of myocardial infarction in rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(25): 3977-3983. |
| [13] | Jin Naying, Wu Jiabao, Li Yuyue, Li Yingjie, Zhou Li, Li Zongheng, Zhang Yong. Gait analysis for hemiplegic patients with cerebral infarction based on RSSCAN gait system [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(23): 3609-3615. |
| [14] | Liu Gang, Ma Chao, Wang Le, Zeng Jie, Jiao Yong, Zhao Yi, Ren Jingpei, Hu Chuanyu, Xu Lin, Mu Xiaohong. Ankle-foot orthoses improve motor function of children with cerebral palsy: a Meta-analysis based on 12 randomized controlled trials [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1299-1304. |
| [15] | Shan Sharui, Huang Xuming, Zhang Mingxing, Wang Xiukun, Zheng Xiang, Bao Sairong, Hong Feng. Three-dimensional gait analysis of low-frequency repetitive transcranial magnetic stimulation for post-stroke hemiplegia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 762-767. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||