Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (32): 8555-8562.doi: 10.12307/2026.452
Previous Articles Next Articles
4D bioprinting for regenerative medicine: a new strategy for intelligent material regulation and tissue regeneration
Liao Meixi1, Wang Zhenxing2, Lu Lili1
- 1Department of Medicine, Wuhan University of Science and Technology, Wuhan 430065, Hubei Province, China; 2Department of Plastic Surgery, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430022, Hubei Province, China
-
Accepted:2026-01-29Online:2026-11-18Published:2026-04-29 -
Contact:Lu Lili, PhD, Professor, Department of Medicine, Wuhan University of Science and Technology, Wuhan 430065, Hubei Province, China -
About author:Liao Meixi, Department of Medicine, Wuhan University of Science and Technology, Wuhan 430065, Hubei Province, China
CLC Number:
Cite this article
Liao Meixi, Wang Zhenxing, Lu Lili. 4D bioprinting for regenerative medicine: a new strategy for intelligent material regulation and tissue regeneration[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8555-8562.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

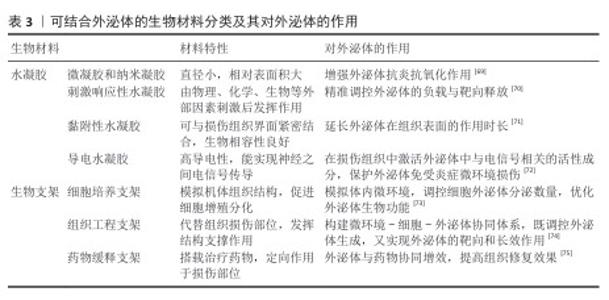
2.3 4D生物打印智能材料 用于4D生物打印智能材料需要具备2个基本品质:它们能够使用3D打印技术进行打印,需要表现出对刺激的敏感性[56-57]。该部分通过以下4个方面简单介绍4D生物打印材料。 2.3.1 形状记忆聚合物 应用于4D生物打印的刺激响应性材料具有形状记忆性,形状记忆聚合物在受到特定刺激时能够从临时形状转变为预定模式的能力[14,58]。 BASAK[59]提出聚合物中的分子链可以提高形状固定性和恢复性能,使智能材料在受到热或光等外部刺激时能够恢复其原始形态。DELAEY等[60]提出手术中应用形状记忆聚合物缝合线可在特定时间段内产生紧绷效果,有利于伤口的恢复。PFAU等[61]利用线性-ε-己内酯-二丙烯酸酯和线性-聚乳酸制作出具有温敏性的骨移植物,可以完美贴合缺损区域。此外,一些形状记忆聚合物(例如苯胺四聚体、聚-ε-己内酯和聚乙烯吡咯烷酮等)可用于药物的释放[62]。形状记忆聚合物具有较多的种类和刺激条件,具有良好的生物相容性,在生物医学领域具有巨大的应用前景。 2.3.2 形状记忆合金 形状记忆合金也是一类智能材料,当受到载荷和温度变化影响时,可以单独或组合改变形状,或被刺激后能从显著变形恢复到原始形状[63]。GHOSH 等[5]利用形状记忆合金的类弹性特性合成了正畸牵引器,用于治疗下颌牙齿畸形。近年来,基于形状记忆合金的装置,如支架、导丝和植入物,在神经学、骨科和心脏病学等生物医学领域得到了广泛应用[64]。 2.3.3 多材料4D生物打印系统 使用单一材料时,其物理、机械性能以及功能存在局限性。多材料和复合4D生物打印材料的开发开辟了新的可能性[65],提供了多功能结构的制造。IMRIE等[66]在4D打印过程中使用具有不同温度敏性的形状记忆聚合物,得到了可在不同部分能够按所需顺序响应不同温度刺激的支架。LIU等[20]利用聚己内酯/甲基丙烯酰化明胶复合材料制作关节软骨修复材料,与单独使用的水凝胶或微纤维支架相比,该修复材料的刚度增加高达54倍。KIM等[9]利用聚(D,L-丙交酯-共-碳酸三亚甲酯)、聚碳酸三亚甲酯和Fe3O4纳米粒子组成复合材料进行4D打印,该复合材料在磁场和热联合刺激下表现出多种形状变化,可用于开发机器人和生物医学领域的多功能设备。 智能材料兼具可打印性与刺激响应性,在4D生物打印中发挥基石作用。虽然单一材料已经实现了众多场景的应用,但是复合材料具备更多的功能,具有广阔的应用前景,其中多材料4D打印结构中各种材料的精确分布和相互作用仍需探索[67]。 "


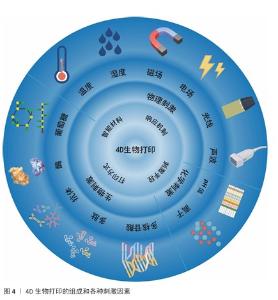
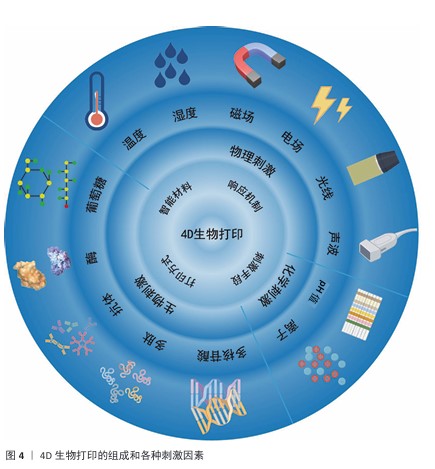
2.4.1 物理刺激 物理刺激在4D打印中具有重要应用价值。光、电场、磁场、湿度及温度等物理刺激能够调控聚合物链的内部结构与动力学,从而引发打印结构的可控形变或功能变化[5]。物理刺激不仅可增强构建体的可操作性和精确性,还为智能支架和动态植入物的开发提供了技术基础。表4为不同物理刺激响应的原理和生物医学应用总结。 2.4.2 化学刺激 pH值和离子浓度是重要的生理参数,它们的失衡可能导致多种疾病,例如心血管疾病、感染、神经退行性疾病和不同类型的癌症。因此,可以通过调控pH值、离子浓度等变化改变聚合物链的结构和动力学[5],实现材料的结构和功能与环境匹配。 pH值:pH值敏感聚合物通常含有弱酸或弱碱基团(如羧基、磷酸、吡啶等),当环境pH值改变时会使这些基团发生电离释放或质子化,引发聚合物在溶解度、降解性、构象及自组装上的变化[5,19],这一特性已被用于药物控释、葡萄糖传感器及基因递送[80]。例如,在肿瘤酸性微环境中,pH值响应系统可实现靶向药物释放;在胃肠道pH值梯度中,可实现口服药物的控释[5,81-82]。但是,天然pH值响应聚合物(壳聚糖、透明质酸、明胶等)的力学性能不足,常需与合成聚合物(如聚组氨酸、聚丙烯酸)复合以提升稳定性和可控性[83]。 离子浓度:离子浓度是人体最重要的生理参数之一。目前研究主要集中于4D打印结构在盐浓度变化时的形状转变[5,84-85]。利用多价离子如 Ca2+和 Zn2+进行交联已被证明可用于4D打印,从而获得具有合适强度和可调机械性能的支架[16]。在具有形态转变记忆的海藻酸盐和透明质酸复合水凝胶体系中,Ca2+浓度可以影响流变特性[16]。总之,该策略强调了离子刺激与生物相容性结合的优势[16]。未来,通过结合疾病导致的体内离子变化,为疾病治疗提供了替代方案。 2.4.3 生物刺激 生物刺激响材料应能够感知体内生物参数变化,通过调控水凝胶的溶胀、收缩或降解实现结构和功能的动态变化[87]。主要的生物刺激包括多肽或多核苷酸、酶以及多种生化因素[19]。例如,KIRILLOVA等[86]利用DNA作为可编程黏合剂,通过调整交联长度实现水凝胶的精确变形。此外,酶在各种生物过程(如蛋白质表达和降解、细胞黏附等)以及疾病中的特异性十分重要,研究人员通过将药物负载于基质金属蛋白酶响应水凝胶中,用于肿瘤治疗[19]。此外,研究人员已开发出含有β细胞的聚乳酸结构糖响应性水凝胶,当葡萄糖分子存在时,该结构会释放由β细胞分泌的胰岛素[43]。综上所述,生物刺激响应材料凭借对生理信号的精准感知与调控,为4D打印提供动态适应能力,在组织工程与再生医学中展现出重要应用潜力。 2.4.4 多刺激因素 近年来,多刺激响应材料的研究不断推进,旨在构建可同时感知并调控多种信号的智能体系,常见组合包括pH值-温度、pH值-磁场及温度-磁场等,主要用于药物递送与控释[88-89]。例如,聚(N-异丙基丙烯酰胺)-co-丙烯酸水凝胶在磁场与电场作用下可实现药物的按需释放[90],此类材料在4D打印中展现出模拟细胞外基质复杂环境的潜力,但应用仍需优化刺激参数,确保处于生理范围并避免细胞损伤[91]。 4D打印依托物理、化学、生物及多刺激响应材料的发展,突破了传统3D打印结构静态和功能单一的局限,实现最终结构的形态可控变化与动态功能。物理刺激材料利用光、温度、磁场等外部信号实现快速可逆形变;化学刺激材料通过pH值或离子浓度变化实现环境适应性调控;生物刺激材料精准感知对酶、葡萄糖等生理因子;多刺激响应材料进一步整合多重信号,实现复杂环境下的协同调控。这些进展显著提升了4D打印结构在动态适应性、功能集成、智能化及个性化方面的潜力,为组织工程、智能药物递送、器官模拟等提供了新思路。未来,随着刺激材料可编程性与工艺精度的提升,4D打印有望实现更高水平的生物功能模拟和临床转化。 "

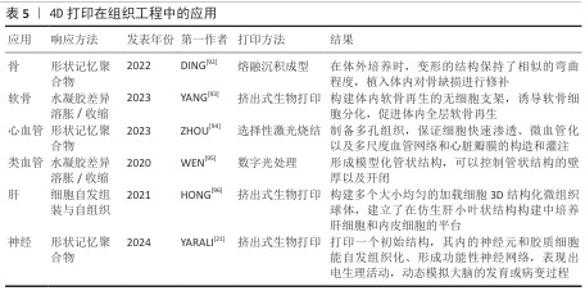
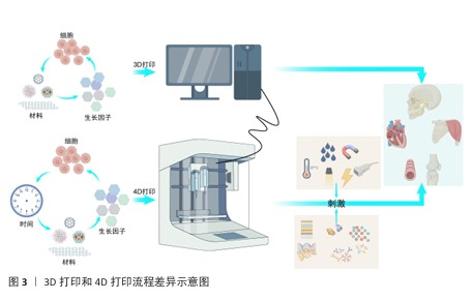
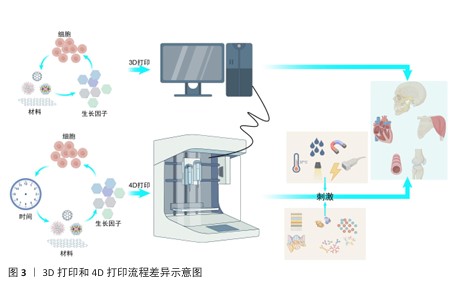
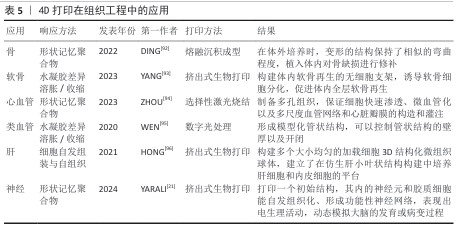
.5 4D打印在再生医学中的应用 生物制作技术的进步是为了更好地推动生物学和医学的进步。4D打印依托可响应外部刺激的智能材料,实现结构与功能的动态调控,更加契合复杂的生理环境,在再生医学中展现出独特优势。利用4D打印制品的可逆形状记忆行为、动态变化,快速成型可以实现在组织工程[22]、药物递送、生物电子及医疗等多方面的应用。 2.5.1 组织工程应用 4D打印技术通过整合智能材料与可编程构造,为组织工程提供了前所未有的动态调控能力,可应用于皮肤、骨骼、大脑及神经系统、心脏与心血管组织以及管状结构的构建(表5)。 总体而言,通过智能材料的响应特性,可实现皮肤、骨骼、神经、心血管及管状结构的动态形态调控与功能模拟,从而更精准地匹配天然组织的力学和生理特性。未来,随着材料多样性、打印精度和生物相容性不断提升,4D打印有望推动复杂组织构建向临床转化发展,实现高仿生性植入物和动态组织修复的新范式,为再生医学提供可持续技术支撑。 2.5.2 药物递送应用 4D打印利用智能响应材料实现药物释放的时空可控性,提高治疗的精确性与有效性,在药物递送中展现出显著潜力[97]。研究表明,将药物直接掺入4D打印材料中可借助外部刺激实现按需释放,例如,基于温度和pH值响应的多体系统可在水环境中精准释放药物。HSU等[98]合成的耐热水凝胶通过热刺激实现可逆收缩,调控流体通道的开闭,用于药物负载与递送。光敏与磁驱动策略进一步丰富了递送手段,CEYLAN等[99]提出了由磁力驱动的微型游泳器,可在施加光刺激后条件性释放阿霉素。4D打印的重要优势在于药物时间、剂量和地点的最佳递送,从而促进组织持续修复或再生。为此,WANG等[100]将近红外光敏感材料与石墨烯复合制备了4D近红外光敏心脏结构,可依据心脏表面曲率变化调控药物释放,为复杂器官表面递药提供了新思路。在药物筛选与个性化治疗方面,4D打印也显示出独特价值,热响应形状记忆聚合物构建的可编程细胞培养阵列,为肿瘤模型药物测试和组织学评估提供高通量平台,有效突破传统类器官筛选的局限[101-102]。总体来看,4D打印在药物递送领域正从单一刺激响应向多模态、个性化与精准治疗发展,这一技术不仅为药物释放带来了时间与空间维度上的动态调控,还能够兼顾药物的稳定性、生物利用度和靶向性,有望推动再生医学从“被动治疗”走向“主动调控”,实现治疗与再生的耦合。 2.5.3 生物电子应用 4D打印与电活性材料的结合正在推动生物电子领域的革新,为柔性器件、智能执行器及人机交互提供了全新发展契机[103]。电活性材料具有形状记忆功能,可在电或磁场刺激下产生显著形变。FANG等[104]合成了包裹大量生长因子的电磁化碳多孔纳米曲奇电子元件,可以在高频磁场下释放并促进细胞修复,用于治疗神经疾病。磁性弹性体复合材料驱动器能够实现远程控制、快速移动、无缝适应粗糙表面以及安全的人体交互。LI等[105]利用磁性弹性体复合材料驱动器制作的软体机器人,能够在人体操控下在最大变形情况下精确完成夹持。此外,将磁性弹性体复合材料驱动器与记忆导电材料相结合,在4D打印的辅助下制作电活性复合驱动器,可用作气管支架和碎骨固定,展现了在生物医学领域的巨大潜力。目前,4D打印与电活性材料的结合不仅优化了器件性能,还推动智能执行器、柔性生物电子及人机交互的发展,未来随着材料可编程性和打印精度的提升,它们可构建自适应、生物融合型电子系统。 2.5.4 医疗应用 4D打印通过智能材料的引入实现了随时间或外部刺激的形态调控,从而为医疗器械和植入物提供了新的可能[106]。目前,4D打印已在自弯曲支架、自收缩夹具等方面展现潜力,并有望改变骨科诊断与治疗规划模式。4D打印的基本流程包括:利用计算机体层成像或磁共振成像获取患者损伤处信息[107];通过软件转化为三维数字模型[108-110];并结合机械模拟优化设计[111];利用智能材料通过4D打印个性化定制具有灵活调控的植入物[112-113];植入体内。临床应用中,这类植入物因具备形状记忆和结构柔性,能更好地贴合人体解剖结构,从而缩短手术时间、减少并发症并、最小化整体手术风险,最终提升临床治疗效果[114-115]。总体而言,4D打印不仅弥补了3D打印的局限,也为再生医学、个性化医疗及未来临床转化提供了强有力的技术支撑。 2.6 挑战 4D打印的出现显著拓展了智能材料在再生医学中的应用,然而,其临床转化仍受到多重限制,需要创新思路与先进技术的推动。 2.6.1 基于材料的挑战 材料的可用性和生物兼容性是核心障碍,现有材料种类有限,并且多数难以在复杂体内环境下响应多重刺激。未来研究应集中于开发兼具打印适应性、可响应多刺激且生物相容性优良的新材料,以支持临床应用。 2.6.2 基于设计的挑战 尽管3D打印在结构构建上取得了进展,但4D打印在形态可控性方面仍有不足,主要问题在于学科交叉不充分及设计软件功能有限,难以精准调控在体内的响应时间与速率。未来需加强跨学科合作,引入高性能计算与先进建模工具,从而提升设计效率和可控性。 2.6.3 基于技术的挑战 4D打印需实现多材料集成、高精度制造及可扩展性,但现有技术各有局限:挤出式打印材料适用广但精度不足;光固化技术分辨率高却受光敏材料限制;选择性激光烧结打印难以处理水凝胶等生物材料;双光子聚合虽可达纳米精度,但效率低且难规模化。多材料打印还面临相容性差、界面结合不稳及工艺参数难以协调等问题。未来需开发适用更宽材料范围的高精度工艺,并优化打印与后处理策略。 2.6.4 成本与可扩展性问题 智能响应材料价格高、制备复杂,对设备和操作精度要求严格,使生产成本远超传统3D打印;实验室精细操作难以大规模应用,产物在稳定性和一致性上仍存在不足。临床和产业化转化需降低材料和制造成本、优化工艺流程,并建立标准化生产体系。 2.6.5 监管与伦理问题 4D打印引入智能材料和个性化设计,给监管体系带来挑战:动态可变性不能使用传统的静态质量评价;个性化制品在安全性、长期稳定性和可追溯性方面存在不确定性;涉及细胞、基因或活性分子载体时可能引发伦理争议。因此,完善法规和伦理审查体系是临床转化的关键前提。 "
| [1] AGARWAL T, KAZEMI S, COSTANTINI M, et al. Oxygen releasing materials: Towards addressing the hypoxia-related issues in tissue engineering. Mater Sci Eng C Mater Biol Appl. 2021;122:111896. [2] MOROUÇO P, AZIMI B, MILAZZO M, et al. Four-Dimensional (Bio-)printing: A Review on Stimuli-Responsive Mechanisms and Their Biomedical Suitability. Appl Sci. 2020; 10(24):9143. [3] ALSHEBLY YS, NAFEA M, MOHAMED ALI MS, et al. Review on recent advances in 4D printing of shape memory polymers. Eur Polym J. 2021; 159:110708. [4] DEMIR KG, ZHANG Z, YANG J, et al. Computational and Experimental Design Exploration of 3D-Printed Soft Pneumatic Actuators. Adv Intell Syst. 2020;2(7):2000013. [5] GHOSH S, CHAUDHURI S, ROY P, et al. 4D Printing in Biomedical Engineering: a State-of-the-Art Review of Technologies, Biomaterials, and Application. Regen Eng Transl Med. 2023; 9(3):339-365. [6] TAMAY DG, DURSUN USAL T, ALAGOZ AS, et al. 3D and 4D Printing of Polymers for Tissue Engineering Applications. Front Bioeng Biotechnol. 2019;7:164. [7] LI Y, ZHANG F, LIU Y, et al. 4D printed shape memory polymers and their structures for biomedical applications. Sci China Technol Sci. 2020;63(4):545-560. [8] ZHAO W, YUE C, LIU L, et al. Research Progress of Shape Memory Polymer and 4D Printing in Biomedical Application. Adv Healthcare Mater. 2023;12(16):2201975. [9] KIM D, KIM M, LEE J, et al. Review on Multicomponent Hydrogel Bioinks Based on Natural Biomaterials for Bioprinting 3D Liver Tissues. Front Bioeng Biotechnol. 2022;10:764682. [10] PARK GY, TARAFDER S, EYEN SL, et al. Oxo-M and 4-PPBP Delivery via Multi-Domain Peptide Hydrogel Toward Tendon Regeneration. Front Bioeng Biotechnol. 2022;10:773004. [11] GREENE C, BEAMAN HT, STINFORT D, et al. Antimicrobial PVA Hydrogels with Tunable Mechanical Properties and Antimicrobial Release Profiles. J Funct Biomater. 2023; 14(4):234. [12] NANIZ MA, ASKARI M, ZOLFAGHARIAN A, et al. 4D printing: a cutting-edge platform for biomedical applications. Biomed Mater. 2022;17(6).doi: 10.1088/1748-605X/ac8e42. [13] LEBERFINGER AN, DINDA S, WU Y, et al. Bioprinting functional tissues. Acta Biomater. 2019;95:32-49. [14] COSTA PDC, COSTA DCS, CORREIA TR, et al. Natural Origin Biomaterials for 4D Bioprinting Tissue-Like Constructs. Adv Mater Technol. 2021;6(10):2100168. [15] MOMENI A, LIU X, JANUSZYK M, et al. Microsurgical ear replantation-is venous repair necessary?-A systematic review. Microsurgery. 2016;36(4):345-350. [16] WAN Z, ZHANG P, LIU Y, et al. Four-dimensional bioprinting: Current developments and applications in bone tissue engineering. Acta Biomater. 2020;101:26-42. [17] ZHANG Q, KUANG X, WENG S, et al. Rapid Volatilization Induced Mechanically Robust Shape-Morphing Structures toward 4D Printing. ACS Appl Mater Interfaces. 2020;12(15): 17979-17987. [18] HART LR, HE Y, RUIZ-CANTU L, et al. Chapter 15-3D and 4D printing of biomaterials and biocomposites, bioinspired composites, and related transformers. 3D and 4D Printing of Polymer Nanocomposite Materials: Elsevier, 2020:467-504. [19] MALEKMOHAMMADI S, AMINABAD NS, SABZI A, et al. Smart and Biomimetic 3D and 4D Printed Composite Hydrogels: Opportunities for Different Biomedical Applications. Biomedicines. 2021;9(11):1537. [20] LIU S, CHEN X, ZHANG Y. Chapter 14 -Hydrogels and hydrogel composites for 3D and 4D printing applications. 3D and 4D Printing of Polymer Nanocomposite Materials: Elsevier, 2020:427-465. [21] YARALI E, MIRZAALI MJ, GHALAYANIESFAHANI A, et al. 4D Printing for Biomedical Applications. Adv Mater. 2024;36(31):2402301. [22] LIN C, ZHANG L, LIU Y, et al. 4D printing of personalized shape memory polymer vascular stents with negative Poisson’s ratio structure: A preliminary study. Sci China Technol Sci. 2020;63(4):578-588. [23] LENDLEIN A, LANGER R. Biodegradable, elastic shape-memory polymers for potential biomedical applications. Science. 2002;296(5573):1673-1676. [24] LENDLEIN A, SCHMIDT AM, LANGER R. AB-polymer networks based on oligo(epsilon-caprolactone) segments showing shape-memory properties. Proc Natl Acad Sci U S A. 2001;98(3):842-847. [25] MOHR R, KRATZ K, WEIGEL T, et al. Initiation of shape-memory effect by inductive heating of magnetic nanoparticles in thermoplastic polymers. Proc Natl Acad Sci U S A. 2006;103(10):3540-3545. [26] RAVIV D, ZHAO W, MCKNELLY C, et al. Active printed materials for complex self-evolving deformations. Sci Rep. 2014;4:7422. [27] NAVARRO G, GÓMEZ-AUTET M, MORALES P, et al. Homodimerization of CB(2) cannabinoid receptor triggered by a bivalent ligand enhances cellular signaling. Pharmacol Res. 2024;208:107363. [28] BAKARICH SE, GORKIN R, 3RD, IN HET PANHUIS M, et al. 4D Printing with Mechanically Robust, Thermally Actuating Hydrogels. Macromol Rapid Commun. 2015; 36(12):1211-1217. [29] LUO F, SUN TL, NAKAJIMA T, et al. Free Reprocessability of Tough and Self-Healing Hydrogels Based on Polyion Complex. ACS Macro Lett. 2015;4(9):961-964. [30] LUO F, SUN TL, NAKAJIMA T, et al. Oppositely charged polyelectrolytes form tough, self-healing, and rebuildable hydrogels. Adv Mater. 2015;27(17):2722-2727. [31] GE Q, SAKHAEI AH, LEE H, et al. Multimaterial 4D Printing with Tailorable Shape Memory Polymers. Sci Rep. 2016;6:31110. [32] MIAO S, CASTRO N, NOWICKI M, et al. 4D printing of polymeric materials for tissue and organ regeneration. Mater Today (Kidlington). 2017;20(10):577-591. [33] DE MARCO C, PANÉ S, NELSON BJ. 4D printing and robotics. Sci Robot. 2018;3(18):eaau0449. [34] GUO J, ZHANG R, ZHANG L, et al. 4D Printing of Robust Hydrogels Consisted of Agarose Nanofibers and Polyacrylamide. ACS Macro Lett. 2018;7(4):442-446. [35] LUI YS, SOW WT, TAN LP, et al. 4D printing and stimuli-responsive materials in biomedical aspects. Acta Biomater. 2019;92:19-36. [36] KIM SH, SEO YB, YEON YK, et al. 4D-bioprinted silk hydrogels for tissue engineering. Biomaterials. 2020;260:120281. [37] WAN X, LUO L, LIU Y, et al. Direct Ink Writing Based 4D Printing of Materials and Their Applications. Adv Sci. 2020;7(16):2001000. [38] MA LY, SOIN N. Recent Progress in Printed Physical Sensing Electronics for Wearable Health-Monitoring Devices: A Review. IEEE Sens J. 2022;22(5):3844-3859. [39] HE J, ZHANG Y, SUN X, et al. Cerebrospinal Fluid-Derived extracellular Vesicle-Inspired Multifunctional bone regeneration scaffold for cranial defect repair. Che Eng J. 2025;504: 158908. [40] AZIBERE S, BOROVIK M, HALL AF, et al. Evaluation of fused deposition modeling (FDM)-printed devices for microfluidic-based cell culture studies. Anal Bioanal Chem. 2025; 417(19):4435-4447. [41] CHEN Y, WEI J. Application of 3D Printing Technology in Dentistry: A Review. Polymers (Basel). 2025;17(7):886. [42] DELANNOY E, BURETTE A, JANEL S, et al. Gut-on-chip methodology based on 3D-printed molds: a cost-effective and accessible approach. Lab Chip. 2025; 25(17):4396-4409. [43] AGARWAL K, SRINIVASAN V, LATHER V, et al. Insights of 3D bioprinting and focusing the paradigm shift towards 4D printing for biomedical applications. J Mate Res. 2023; 38(1):112-141. [44] DHAND AP, DAVIDSON MD, BURDICK JA. Lithography-based 3D printing of hydrogels. Nat Rev Bioeng. 2025;3(2):108-125. [45] VIGOGNE M, AESCHBACH C, BERNHARDT R, et al. Step Test for Rapid Screening of Material and Process Parameters for Resin Development in DLP 3D Printing. Angew Chem Int Ed Engl. 2025;64(33):e202504154. [46] ZHENG Y, FU L, ZHANG Z, et al. Three-Dimensional Bioprinting of Growth Differentiation Factor 5-Preconditioned Mesenchymal Stem Cell-Derived Exosomes Facilitates Articular Cartilage Endogenous Regeneration. ACS Nano. 2025;19(16): 15281-15301. [47] DU X, LIU Y, ZHAO W, et al. 3D printing of heat-resistant thermosetting polyimide composite with high dimensional accuracy and mechanical property. Compos Part B Eng. 2025;298:112394. [48] YAN Z, DENG Y, HUANG L, et al. Biopolymer-based bone scaffold for controlled Pt (IV) prodrug release and synergistic photothermal-chemotherapy and immunotherapy in osteosarcoma. J Nanobiotechnology. 2025; 23(1):286. [49] SEOANE-VIAÑO I, PÉREZ-RAMOS T, LIU J, et al. Visualizing disintegration of 3D printed tablets in humans using MRI and comparison with in vitro data. J Control Release. 2024; 365:348-357. [50] MA X, XU M, CUI X, et al. Hybrid 3D Bioprinting of Sustainable Biomaterials for Advanced Multiscale Tissue Engineering. Small. 2025: e2408947. doi: 10.1002/smll.202408947. [51] LI K, DENG X, LIU W, et al. Surface Energy-Confined Multi-Layer Inkjet Printing for Customizable Optical Microstructures. Adv Mater. 2025:e09818. doi: 10.1002/adma. 202509818. [52] YANG J, LU J, HAN D, et al. Direct ink writing of aerogels: Fundamentals, strategies, applications, and perspectives. Prog Mater Sci. 2025;152:101462. [53] PAK K, YANG JC, SIM JY, et al. Fabrication of multifunctional wearable interconnect E-textile platform using direct ink writing (DIW) 3D printing. Npj Flex Electron. 2025;9(1):48. [54] AI A, HASANZADEH E, SAFSHEKAN F, et al. Enhanced spinal cord regeneration by gelatin/alginate hydrogel scaffolds containing human endometrial stem cells and curcumin-loaded PLGA nanoparticles in rat. Life Sci. 2023;330: 122035. [55] HASANZADEH E, SEIFALIAN A, MELLATI A, et al. Injectable hydrogels in central nervous system: Unique and novel platforms for promoting extracellular matrix remodeling and tissue engineering. Mater Today Bio. 2023; 20:100614. [56] ZHOU W, QIAO Z, NAZARZADEH ZARE E, et al. 4D-Printed Dynamic Materials in Biomedical Applications: Chemistry, Challenges, and Their Future Perspectives in the Clinical Sector. J Med Chem. 2020;63(15):8003-8024. [57] LE DUIGOU A, CORREA D, UEDA M, et al. A review of 3D and 4D printing of natural fibre biocomposites. Mater Des. 2020;194:108911. [58] KUANG X, ROACH DJ, WU J, et al. Advances in 4D Printing: Materials and Applications. Adv Func Mater. 2019;29(2):1805290. [59] BASAK S. Investigating entanglement-driven shape memory property: Insights and structure-property relationships from recent developments. Smart Mater Methods. 2024;1(1):48-84. [60] DELAEY J, DUBRUEL P, VAN VLIERBERGHE S. Shape-Memory Polymers for Biomedical Applications. Adv Funct Mater. 2020;30(44):1909047. [61] PFAU MR, MCKINZEY KG, ROTH AA, et al. Shape memory polymer (SMP) scaffolds with improved self-fitting properties. J Mater Chem B. 2021;9(18):3826-3837. [62] ATOUFI Z, ZARRINTAJ P, MOTLAGH GH, et al. A novel bio electro active alginate-aniline tetramer/ agarose scaffold for tissue engineering: synthesis, characterization, drug release and cell culture study. J Biomater Sci Polym Ed. 2017;28(15):1617-1638. [63] AIZARNA-LOPETEGUI U, BITTINGER SC, ÁLVAREZ N, et al. Stimuli-responsive hybrid materials for 4D in vitro tissue models. Mater Today Bio. 2025;33:102035. [64] JOSHI S, RAWAT K, C K, et al. 4D printing of materials for the future: Opportunities and challenges. Appl Mater Today. 2020;18:100490. [65] ZHOU X, REN L, SONG Z, et al. Advances in 3D/4D printing of mechanical metamaterials: From manufacturing to applications. Compos Part B Eng. 2023;254:110585. [66] IMRIE P, JIN J. Polymer 4D printing: Advanced shape-change and beyond. J Polym Sci. 2022; 60(2):149-174. [67] WAN X, HE Y, LIU Y, et al. 4D printing of multiple shape memory polymer and nanocomposites with biocompatible, programmable and selectively actuated properties. Addit Manuf. 2022;53:102689. [68] ZAREK M, MANSOUR N, SHAPIRA S, et al. 4D Printing of Shape Memory-Based Personalized Endoluminal Medical Devices. Macromol Rapid Commun. 2017;38(2). doi: 10.1002/marc.201600628. [69] SENATOV FS, NIAZA KV, ZADOROZHNYY MY, et al. Mechanical properties and shape memory effect of 3D-printed PLA-based porous scaffolds. J Mech Behav Biomed Mater. 2016;57:139-148. [70] LAI J, XIONG T, CHEN S, et al. Facile Single-Nanocomposite 4D Bioprinting of Dynamic Hydrogel Constructs with Thickness-Controlled Gradient. Adv Sci (Weinh). 2025; 12(39):e09449. [71] GOODARZI HOSSEINABADI H, BISWAS A, BHUSAL A, et al. 4D-Printable Photocrosslinkable Polyurethane-Based Inks for Tissue Scaffold and Actuator Applications. Small. 2024;20(6):e2306387. [72] MIRASADI K, YOUSEFI MA, JIN L, et al. 4D Printing of Magnetically Responsive Shape Memory Polymers: Toward Sustainable Solutions in Soft Robotics, Wearables, and Biomedical Devices. Adv Sci. 2025:e13091. doi: 10.1002/advs.202513091. [73] YANG F, ZHAO R, JIA J, et al. Homogeneous distribution of Fe(3)O(4) nanoparticles in poly(L-lactic acid)/thermoplastic polyurethane bone scaffold for boosting magnetic-response shape memory. Int J Biol Macromol. 2025; 322(Pt 4):147009. [74] NABIPOUR M, MELLATI A, ABASI M, et al. Preparation of bilayer tissue-engineered polyurethane/poly-L-lactic acid nerve conduits and their in vitro characterization for use in peripheral nerve regeneration. J Biol Eng. 2024;18(1):16. [75] DONG SL, HAN L, DU CX, et al. 3D Printing of Aniline Tetramer-Grafted-Polyethylenimine and Pluronic F127 Composites for Electroactive Scaffolds. Macromol Rapid Commun. 2017;38(4). doi: 10.1002/marc. 201600551. [76] YANG Z, LIU H, ZHANG X, et al. Photo-Responsive Dynamic Organic Room-Temperature Phosphorescence Materials Based on a Functional Unit Combination Strategy. Adv Mater. 2024;36(3):e2306784. [77] BROWN TE, ANSETH KS. Spatiotemporal hydrogel biomaterials for regenerative medicine. Chem Soc Rev. 2017;46(21): 6532-6552. [78] LIU J, ZHAO Z, DENG C, et al. Nanoscale Donor-Acceptor Covalent Organic Frameworks for Mitochondria-Targeted Sonodynamic Therapy and Antitumor Immunity. J Am Chem Soc. 2025;147(29):25622-25634. [79] NASEER SM, MANBACHI A, SAMANDARI M, et al. Surface acoustic waves induced micropatterning of cells in gelatin methacryloyl (GelMA) hydrogels]. Biofabrication. 2017;9(1): 015020. [80] BAGHERIFAM S, SKJELDAL FM, GRIFFITHS G, et al. pH-Responsive Nano Carriers for Doxorubicin Delivery. Pharm Res. 2015;32(4): 1249-1263. [81] SENAPATI S, MAHANTA AK, KUMAR S, et al. Controlled drug delivery vehicles for cancer treatment and their performance. Signal Transduct Target Ther. 2018;3:7. [82] ASHFAQ UA, RIAZ M, YASMEEN E, et al. Recent Advances in Nanoparticle-Based Targeted Drug-Delivery Systems Against Cancer and Role of Tumor Microenvironment. Crit Rev Ther Drug Carrier Syst. 2017;34(4):317-353. [83] RHEE S, PUETZER JL, MASON BN, et al. 3D Bioprinting of Spatially Heterogeneous Collagen Constructs for Cartilage Tissue Engineering. ACS Biomater Sci Eng. 2016; 2(10):1800-1805. [84] WANG YG, WANG CS, LI Y, et al. Digitization of Endocytic pH by Hybrid Ultra-pH-Sensitive Nanoprobes at Single-Organelle Resolution. Adv Mater. 2017;29(1):10.1002/adma.201603794. doi: 10.1002/adma. 201603794. [85] THÉRIEN-AUBIN H, WU ZL, NIE ZH, et al. Multiple Shape Transformations of Composite Hydrogel Sheets. J Am Chem Soc. 2013;135(12):4834-4839. [86] KIRILLOVA A, MAXSON R, STOYCHEV G, et al. 4D Biofabrication Using Shape-Morphing Hydrogels. Adv Mater. 2017;29(46). doi:10.1002/adma.201703443. [87] FERREIRA NN, FERREIRA LMB, CARDOSO VMO, et al. Recent advances in smart hydrogels for biomedical applications: From self-assembly to functional approaches. Eur Polym J. 2018;99: 117-133. [88] SHIGEMITSU H, HAMACHI I. Design Strategies of Stimuli-Responsive Supramolecular Hydrogels Relying on Structural Analyses and Cell-Mimicking Approaches. Acc Chem Res. 2017;50(4):740-750. [89] SUGIMOTO T, YAMAZAKI N, HAYASHI T, et al. Preparation of dual-stimuli-responsive liposomes using methacrylate-based copolymers with pH and temperature sensitivities for precisely controlled release. Colloids Surf B Biomedicines. 2017;155: 449-458. [90] MIRVAKILI SM, LANGER R. Wireless on-demand drug delivery. Nat Electron, 2021;4(7):464-477. [91] LI YC, ZHANG YS, AKPEK A, et al. 4D bioprinting: the next-generation technology for biofabrication enabled by stimuli-responsive materials. Biofabrication. 2016; 9(1):012001. [92] DING A, LEE SJ, AYYAGARI S, et al. 4D biofabrication via instantly generated graded hydrogel scaffolds. Bioact Mater. 2022;7:324-332. [93] YANG Y, ZHAO X, WANG S, et al. Ultra-durable cell-free bioactive hydrogel with fast shape memory and on-demand drug release for cartilage regeneration. Nat Commun. 2023; 14(1):7771. [94] ZHOU Z, TANG W, YANG J, et al. Application of 4D printing and bioprinting in cardiovascular tissue engineering. Biomater Sci. 2023;11(19): 6403-6420. [95] WEN H, LI J, PAYNE GF, et al. Hierarchical patterning via dynamic sacrificial printing of stimuli-responsive hydrogels. Biofabrication. 2020;12(3):035007. [96] HONG G, KIM J, OH H, et al. Production of Multiple Cell-Laden Microtissue Spheroids with a Biomimetic Hepatic-Lobule-Like Structure. Adv Mater. 2021;33(36):e2102624. [97] GOYAL R, SAHU S, MITRA S, et al. Nanocellulose-Reinforced 4D Printed Hydrogels: Thermoresponsive Shape Morphing and Drug Release. ACS Appl Polym Mater. 2024;6(2):1348-1361. [98] HSU L, JIANG X. ‘Living’ Inks for 3D Bioprinting. Trends Biotechnol. 2019;37(8):795-796. [99] CEYLAN H, YASA IC, YASA O, et al. 3D-Printed Biodegradable Microswimmer for Theranostic Cargo Delivery and Release. ACS Nano. 2019; 13(3):3353-3362. [100] WANG Y, CUI H, WANG Y, et al. 4D Printed Cardiac Construct with Aligned Myofibers and Adjustable Curvature for Myocardial Regeneration. ACS Appl Mater Interfaces. 2021;13(11):12746-12758. [101] CHADWICK M, YANG C, LIU L, et al. Rapid Processing and Drug Evaluation in Glioblastoma Patient-Derived Organoid Models with 4D Bioprinted Arrays. iScience. 2020;23(8):101365. [102] PARKER RN, CAIRNS DM, WU WA, et al. Smart Material Hydrogel Transfer Devices Fabricated with Stimuli-Responsive Silk-Elastin-Like Proteins. Adv Healthc Mater. 2020;9(11):2000266. [103] LI D, LAI WY, ZHANG YZ, et al. Printable Transparent Conductive Films for Flexible Electronics. Adv Mater. 2018;30(10):1704738. [104] FANG JH, HSU HH, HSU R, et al. 4D printing of stretchable nanocookie@conduit material hosting biocues and magnetoelectric stimulation for neurite sprouting. NPG Asia Mater. 2020;12(1):61. [105] LI M, PAL A, AGHAKHANI A, et al. Soft actuators for real-world applications. Nat Rev Mater. 2022;7(3):235-249. [106] MANDAL A, CHATTERJEE K. 4D printing for biomedical applications. J Mater Chem B. 2024;12(12):2985-3005. [107] HALEEM A, JAVAID M. 3D scanning applications in medical field: A literature-based review. Clin Epidemiol Glob Health. 2019;7(2):199-210. [108] RIPLEY B, KELIL T, CHEEZUM MK, et al. 3D printing based on cardiac CT assists anatomic visualization prior to transcatheter aortic valve replacement. J Cardiovasc Comput Tomogr. 2016;10(1):28-36. [109] HERRMANN KH, GÄRTNER C, GÜLLMAR D, et al. 3D printing of MRI compatible components: Why every MRI research group should have a low-budget 3D printer. Med Eng Phys. 2014; 36(10):1373-1380. [110] MEYER-SZARY J, LUIS MS, MIKULSKI S, et al. The Role of 3D Printing in Planning Complex Medical Procedures and Training of Medical Professionals-Cross-Sectional Multispecialty Review. Int J Environ Res Public Health. 2022;19(6):3331. [111] KHALID MY, ARIF ZU, AHMED W. 4D Printing: Technological and Manufacturing Renaissance. Macromol Mater Eng. 2022;307(8):2200003. [112] WANG Y, CUI H, ESWORTHY T, et al. Emerging 4D Printing Strategies for Next-Generation Tissue Regeneration and Medical Devices. Adv Mater. 2022;34(20):2109198. [113] BODAGHI M, DAMANPACK AR, LIAO WH. Triple shape memory polymers by 4D printing. Smart Mater Struct. 2018;27(6):065010. [114] AWASTHI S, VERMA T, AGARWAL M, et al. Developing effective health communication messages for community acquired pneumonia in children under five years of age: A rural North Indian qualitative study. Clin Epidemiol Glob Health. 2017;5(3):107-116. [115] AWASTHI S, PANDEY N. Rural background and low parental literacy associated with discharge against medical advice from a tertiary care government hospital in India. Clin Epidemiol Glob Health. 2015;3(1):24-28. |
| [1] | Zhu Jisheng, Teng Jianxiang, Zou Zihao, Pan Jiazhao, Zhou Tianqi, Shu Xiaolin, He Cheng, Yuan Daizhu, Tian Xiaobin . Application of photocrosslinkable nanofiber scaffold loaded with decellularized cartilage matrix in cartilage tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8402-8412. |
| [2] | Zhou Pengfei, Lin Jing, Chen Yuying, Lin Minkui. Canine dental pulp stem cells-polyglycolic acid scaffold complex for canine periodontal tissue defect [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5526-5531. |
| [3] | Yu Xingge, Lin Kaili. Application of nanocomposite hydrogels in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5441-5446. |
| [4] |
Ji Hangyu, Gu Jun, Xie Linghan, Wu Xiaotao.
Application of stem cells, tissue engineering scaffolds and neurotrophic factors in the treatment of spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(25): 4088-4093. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||