Chinese Journal of Tissue Engineering Research ›› 2025, Vol. 29 ›› Issue (32): 7015-7027.doi: 10.12307/2025.797
Previous Articles Next Articles
Antioxidants from different sources and osteoarthritis: a genome-wide association analysis in European populations
Liu Xiaowu1, Liu Jinping2, Wu Ting2, He Xian2, Cai Jianxiong3
- 1Second Clinical Medical College, Guangzhou University of Chinese Medicine, Guangzhou 510120, Guangdong Province, China; 2Panyu Hospital of Traditional Chinese Medicine, Guangzhou University of Chinese Medicine, Guangzhou 511400, Guangdong Province, China; 3Second Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou 510120, Guangdong Province, China
-
Received:2024-10-14Accepted:2024-11-20Online:2025-11-18Published:2025-04-29 -
Contact:Cai Jianxiong, Attending physician, Assistant researcher, Second Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou 510120, Guangdong Province, China Co-corresponding author: He Xian, Chief physician, Panyu Hospital of Traditional Chinese Medicine, Guangzhou University of Chinese Medicine, Guangzhou 511400, Guangdong Province, China -
About author:Liu Xiaowu, MD candidate, Second Clinical Medical College, Guangzhou University of Chinese Medicine, Guangzhou 510120, Guangdong Province, China Liu Jinping, Master candidate, Panyu Hospital of Traditional Chinese Medicine, Guangzhou University of Chinese Medicine, Guangzhou 511400, Guangdong Province, China Liu Xiaowu and Liu Jinping contributed equally to this article. -
Supported by:Guangzhou Science and Technology Planning Project, No. 2024A03J0744 (to CJX); Excellent Paper Support Program of Guangdong Hospital of Traditional Chinese Medicine (to CJX)
CLC Number:
Cite this article
Liu Xiaowu, Liu Jinping, Wu Ting, He Xian, Cai Jianxiong. Antioxidants from different sources and osteoarthritis: a genome-wide association analysis in European populations[J]. Chinese Journal of Tissue Engineering Research, 2025, 29(32): 7015-7027.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
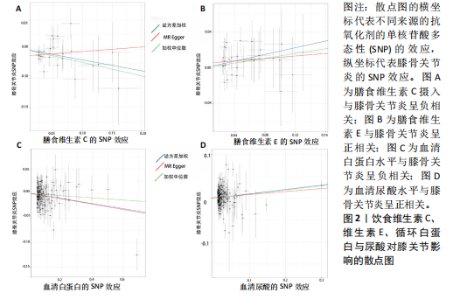
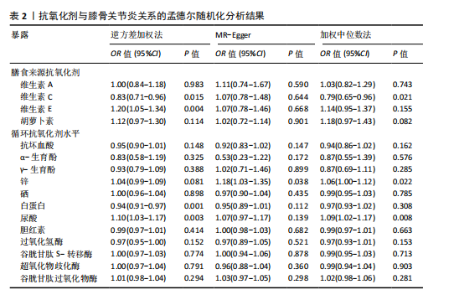
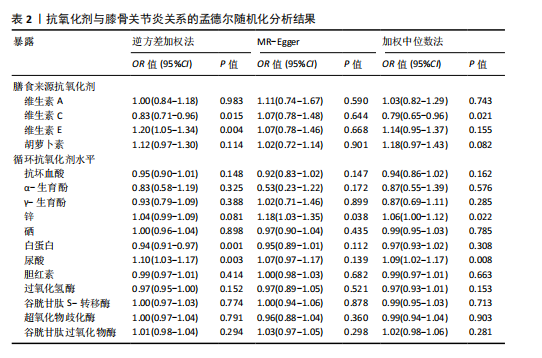
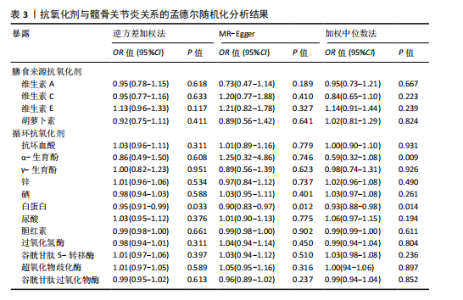
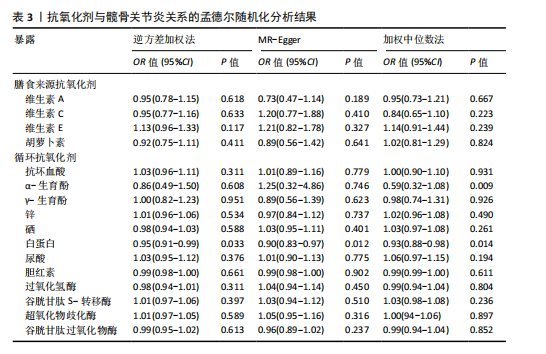
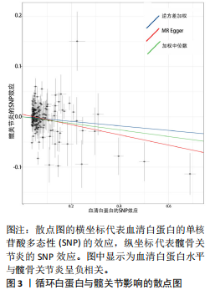
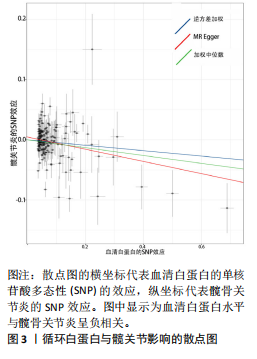
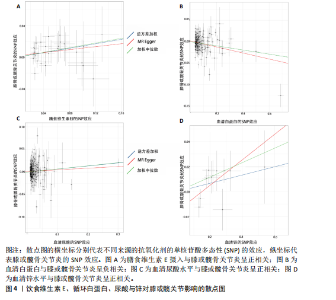
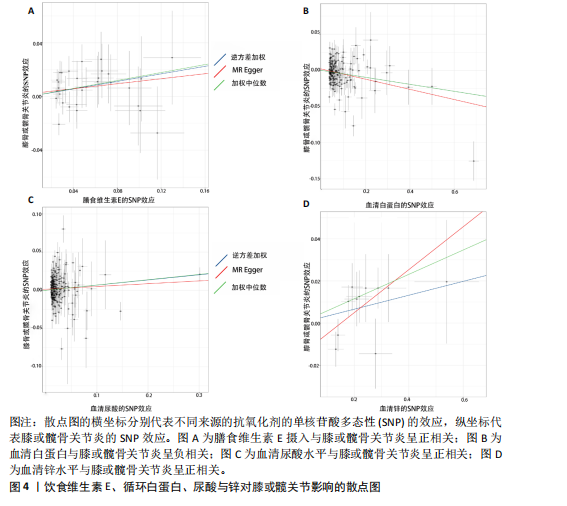
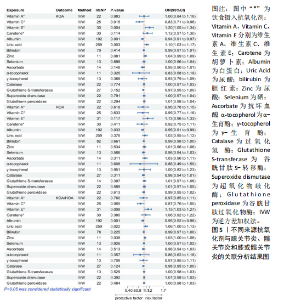
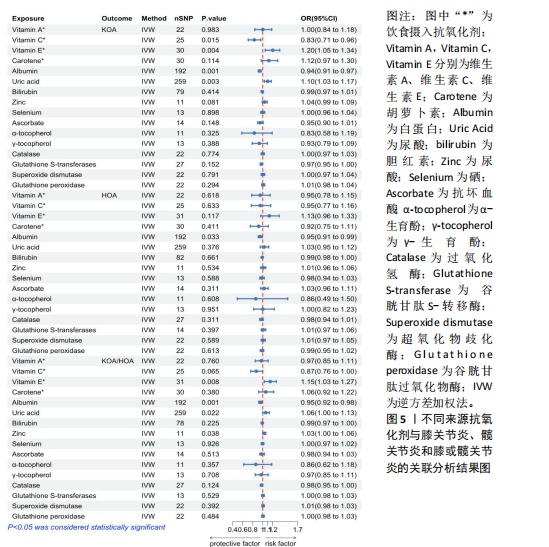
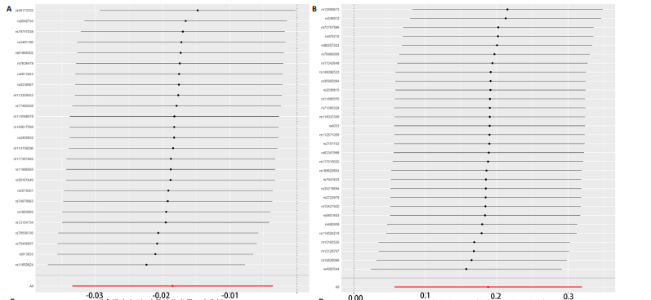
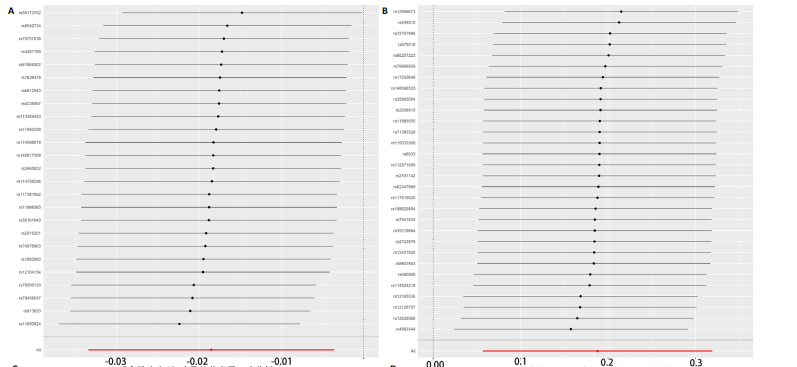
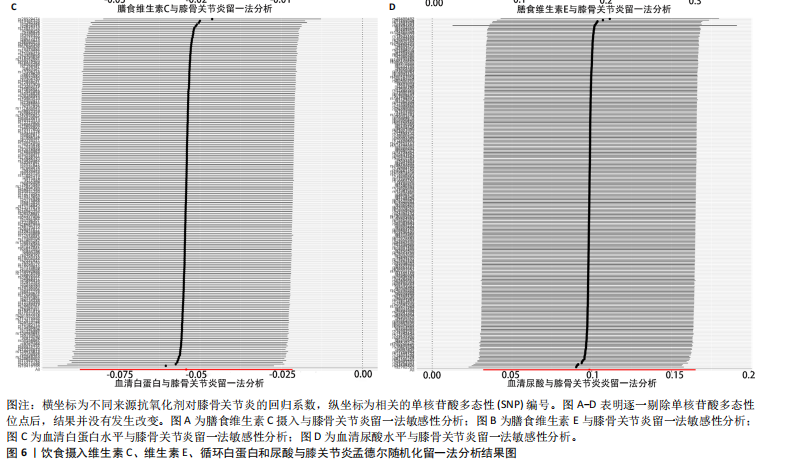
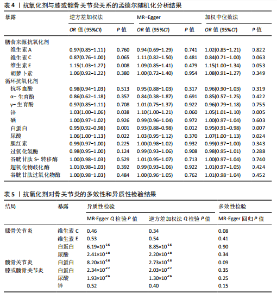
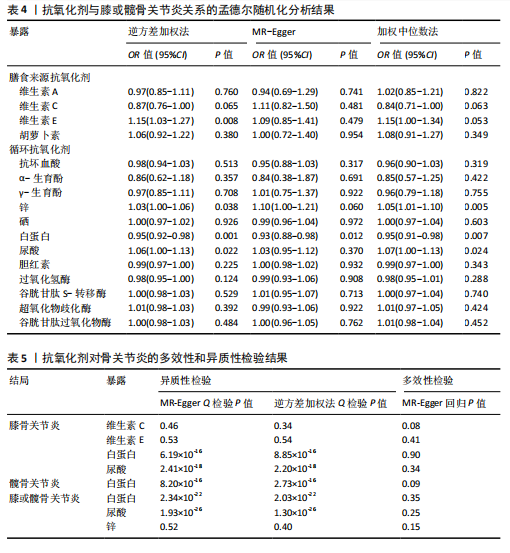
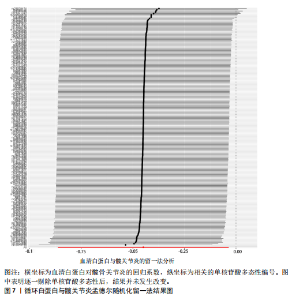
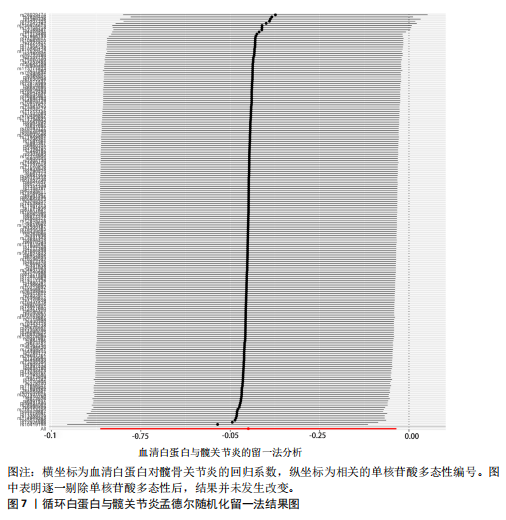
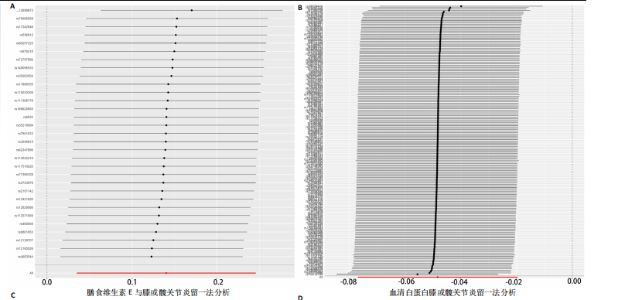
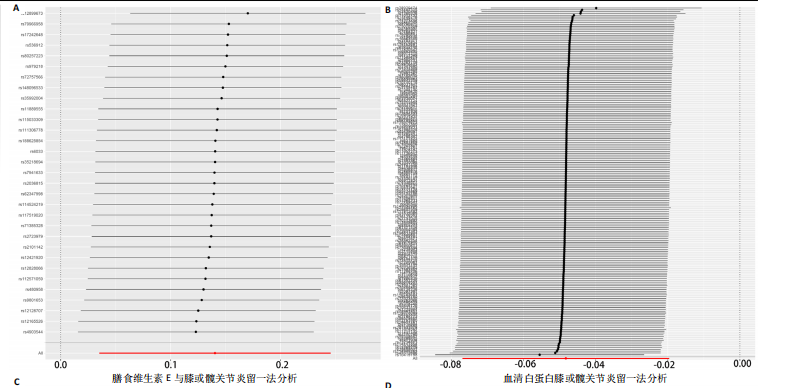
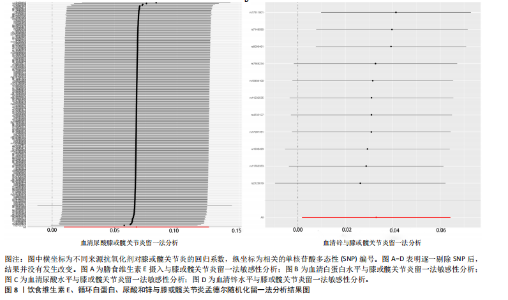
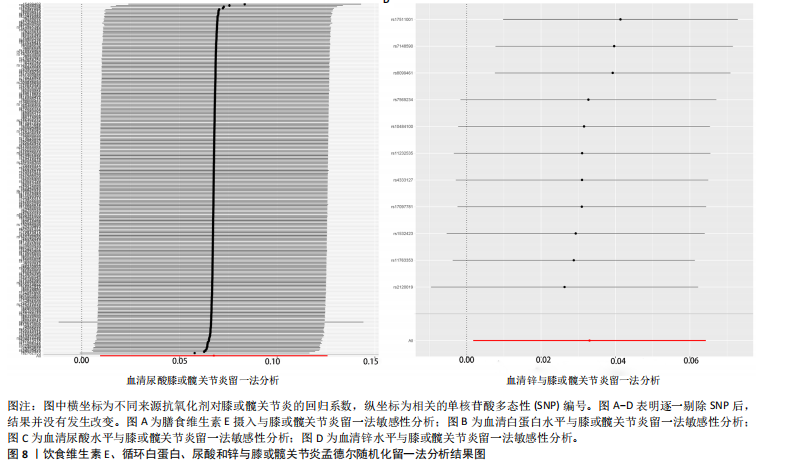
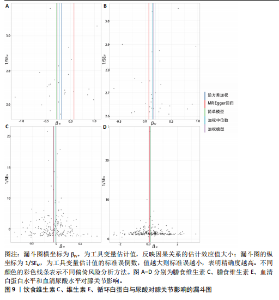
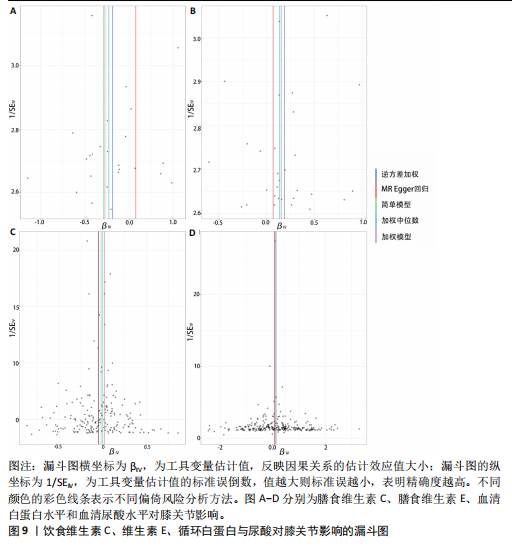
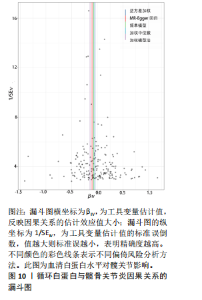
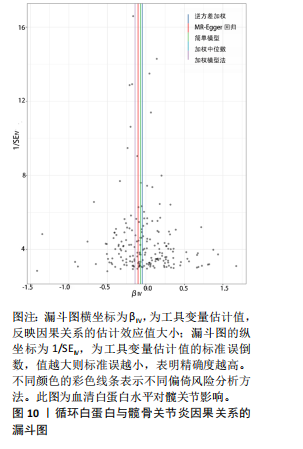
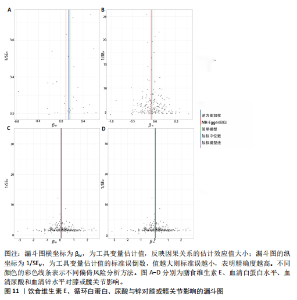
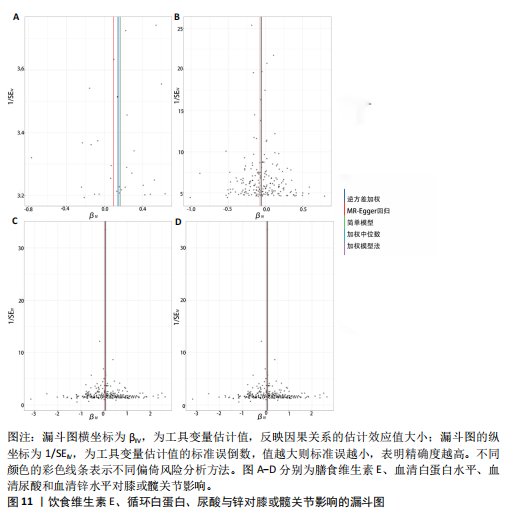
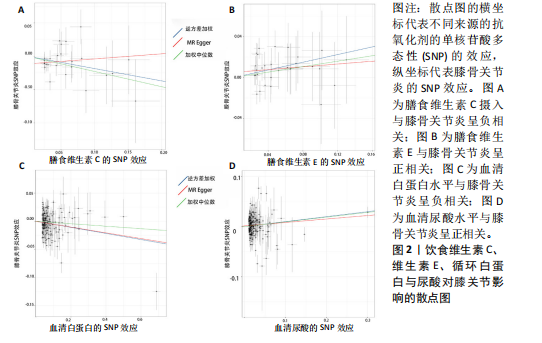
2.1 孟德尔随机化结果 2.1.1 抗氧化剂与膝骨关节炎关系 孟德尔随机化分析结果显示,膳食维生素C(OR=0.83,95%CI:0.71-0.96,P=0.015)摄入与膝骨关节炎呈负向因果关系,而维生素E摄入(OR=1.20,95%CI:1.05-1.34,P=0.004)与膝骨关节炎呈正向因果关系。循环抗氧化剂中,白蛋白(OR=0.94,95%CI:0.91-0.97,P=0.001)与膝骨关节炎呈负向因果关系,尿酸(OR=1.10,95%CI:1.03-1.17,P=0.003)与膝骨关节炎呈正向因果关系。不同抗氧化剂与膝骨关节炎的孟德尔随机化分析散点图见图2,回归线无明显偏移。未发现膳食维生素A、胡萝卜素和血清抗坏血酸、α-生育酚、γ-生育酚、谷胱甘肽S-转移酶、谷胱甘肽过氧化物酶、过氧化氢酶、超氧化物歧化酶、胆红素、锌、硒与膝骨关节炎存在因果关联(P > 0.05),见表2。 2.1.2 抗氧化剂与髋骨关节炎的关系 孟德尔随机化分析结果显示,膳食维生素A、维生素C、维生素E、胡萝卜素与髋骨关节炎无因果关系。循环抗氧化剂中白蛋白(OR=0.95,95%CI:0.91-0.99,P=0.03)与膝骨关节炎呈负向因果关系,见图3,暂未发现血清抗坏血酸、α-生育酚、γ-生育酚、谷胱甘肽S-转移酶、谷胱甘肽过氧化物酶、过氧化氢酶、超氧化物歧化酶、尿酸、胆红素、锌、硒与髋骨关节炎的存在因果关系(P > 0.05),具体见表3。 2.1.3 抗氧化剂与膝或髋骨关节炎的关系 根据孟德尔随机化分析结果,膳食维生素E(OR=1.15,95%CI:1.03-1.27,P=0.008)摄入与膝或髋骨关节炎呈正向因果关系,循环抗氧化剂中白蛋白(OR=0.95,95%CI:0.92-0.98,P=0.01)与膝或髋骨关节炎呈负向因果关系,而尿酸(OR=1.06,95%CI:1.00-1.13,P=0.022)和锌(OR=1.03,95%CI:1.00-1.06,P=0.038)与膝或髋骨关节炎呈正向因果关系。孟德尔随机化分析结果散点图,见图4,显示回归线无明显偏移。未发现膳食维生素A、维生素C、胡萝卜素和血清抗坏血酸、α-生育酚、γ-生育酚、谷胱甘肽S-转移酶、谷胱甘肽过氧化物酶、过氧化氢酶、超氧化物歧化酶、胆红素、硒与膝或髋骨关节炎有因果关联(P > 0.05),见表4。不同来源抗氧化剂与膝关节炎、髋关节炎和膝或髋关节炎关联见图5。 2.2 敏感性分析结果 文章 MR-Egger 回归截距结果均接近于0,暂未检测到潜在的水平多效性(P > 0.05),说明所选工具变量并不通过暴露以外的途径影响结局,符合排他性假设。根据Cochrane Q检验结果,在膝骨关节炎中,白蛋白、胆红素、尿酸和硒存在异质性。此外,在髋骨关节炎中,白蛋白、胆红素和尿酸存在异质性。在膝或髋骨关节炎中,胡萝卜素、白蛋白、胆红素和尿酸存在异质性,因此上述暴露采用随机效应逆方差加权模型进行因果推断,具体见表 5。依据MR多效残差和离群值分析(MR-PRESSO)的评估结果,未检测到异常的单核苷酸多态性存在。所选工具变量的特性确保了其不受潜在的水平多效性干扰,增强了研究结果的可信度与稳定性。在敏感性分析中,留一法结果显示,移除任一单核苷酸多态性均未导"
| [1] YAO Q, WU X, TAO C, et al. Osteoarthritis: pathogenic signaling pathways and therapeutic targets. Signal Transduct Target Ther. 2023;8(1):56. [2] SU S, HE J, WANG C, et al. A new dressing system reduces the number of dressing changes in the primary total knee arthroplasty: a randomized controlled trial. Front Surg. 2022;9:800850. [3] COLLABORATORS GO. Global, regional, and national burden of osteoarthritis, 1990-2020 and projections to 2050: a systematic analysis for the Global Burden of Disease Study 2021. Lancet Rheumatol. 2023;5(9):e508-e522. [4] PAESA M, ALEJO T, GARCIA-ALVAREZ F, et al.New insights in osteoarthritis diagnosis and treatment: nano-strategies for an improved disease management. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2023;15(2):e1844. [5] ZHANG Y, LIU T, YANG H, et al. Melatonin: a novel candidate for the treatment of osteoarthritis. Ageing Res Rev. 2022;78: 101635. [6] WANG J, ZHANG Y, CAO J, et al. The role of autophagy in bone metabolism and clinical significance. Autophagy. 2023;19(9):2409-2427. [7] DADSENA S, ZOLLO C, GARCÍA-SÁEZ AJ. Mechanisms of mitochondrial cell death. Biochem Soc Trans. 2021;49(2):663-674. [8] RIEGGER J, SCHOPPA A, RUTHS L, et al. Oxidative stress as a key modulator of cell fate decision in osteoarthritis and osteoporosis: a narrative review. Cell Mol Biol Lett. 2023;28(1):76. [9] LARSSON K, HARRYSSON H, HAVENAAR R, et al. Formation of malondialdehyde (MDA), 4-hydroxy-2-hexenal (HHE) and 4-hydroxy-2-nonenal (HNE) in fish and fish oil during dynamic gastrointestinal in vitro digestion. Food Funct. 2016;7(2):1176-1187. [10] TUDORACHI NB, TOTU EE, FIFERE A, et al. The implication of reactive oxygen species and antioxidants in knee osteoarthritis. Antioxidants (Basel). 2021;10(6):985. [11] ZENG L, YANG T, YANG K, et al. Efficacy and safety of curcumin and curcuma longa extract in the treatment of arthritis: a systematic review and meta-analysis of randomized controlled trial. Front Immunol. 2022;13:891822. [12] KNAPIK JJ, TRONE DW, STEELMAN RA, et al. Associations between clinically diagnosed medical conditions and dietary supplement use: the US military dietary supplement use study. Public Health Nutr. 2023;26(6):1238-1253. [13] XU C, WANG S, TI W, et al. Role of dietary patterns and factors in determining the risk of knee osteoarthritis: a meta-analysis. Mod Rheumatol. 2022;32(4):815-821. [14] VALSAMIDOU E, AMERIKANOU C, TZAVARA C, et al. A standardized nutraceutical supplement contributes to pain relief, improves quality of life and regulates inflammation in knee osteoarthritis patients; A randomized clinical trial. Heliyon. 2023; 9(9):e20143. [15] ZHAO Q, TANG Y, ZHANG L, et al. Biological functions of selenoprotein glutathione peroxidases (GPXs) and their expression in osteoarthritis. J Inflamm Res. 2023;16:183-196. [16] JOKIĆ A, SREMCEVIĆ N, KARAGÜLLE Z, et al. Oxidative stress, hemoglobin content, superoxide dismutase and catalase activity influenced by sulphur baths and mud packs in patients with osteoarthritis. Vojnosanit Pregl. 2010;67(7):573-578. [17] KRACHLER M, DOMEJ W. Clinical laboratory parameters in osteoarthritic knee-joint effusions correlated to trace element concentrations. Biol Trace Elem Res. 2001; 79(2):139-148. [18] COSTA-CAVALCANTI RG, DE SÁ-CAPUTO DDC, MOREIRA-MARCONI E, et al. Effect of auriculotherapy on the plasma concentration of biomarkers in individuals with knee osteoarthritis. J Acupunct Meridian Stud. 2018;11(4):145-152. [19] VEEN L, HANTIKAINEN E, BELLOCCO R, et al. Dietary antioxidants, non-enzymatic antioxidant capacity and the risk of osteoarthritis in the Swedish National March Cohort. Eur J Nutr. 2021;60(1): 169-178. [20] CHAGANTI RK, TOLSTYKH I, JAVAID MK, et al. High plasma levels of vitamin C and E are associated with incident radiographic knee osteoarthritis. Osteoarthritis Cartilage. 2014;22(2):190-196. [21] NOOH MH, ALSHEHRI MS, ALZAHRANI ZS, et al. The efficacy and safety of intra-articular low molecular weight fraction of human serum albumin for the management of moderate to moderately severe knee osteoarthritis: a systematic review and meta-analysis. Cureus.2023;15(6):e41240. [22] CHEN D, XU H, SUN L, et al. Assessing causality between osteoarthritis with urate levels and gout: a bidirectional Mendelian randomization study. Osteoarthritis Cartilage. 2022;30(4):551-558. [23] LI H, ZENG C, WEI J, et al. Associations between dietary antioxidants intake and radiographic knee osteoarthritis. Clin Rheumatol. 2016;35(6):1585-1592. [24] DAVIES NM, HOLMES MV, DAVEY SMITH G. Reading Mendelian randomisation studies: a guide, glossary, and checklist for clinicians. Bmj. 2018;362:k601. [25] HUANG L, XIE Y, JIN T, et al. Diet-derived circulating antioxidants and risk of knee osteoarthritis, hip osteoarthritis and rheumatoid arthritis: a two-sample Mendelian randomization study. Front Med (Lausanne). 2023;10:1147365. [26] XU J, ZHANG S, TIAN Y, et al. Genetic causal association between iron status and osteoarthritis: a two-sample mendelian randomization. Nutrients. 2022; 14(18):3683. [27] SHIN SY, FAUMAN EB, PETERSEN AK, et al. An atlas of genetic influences on human blood metabolites. Nat Genet. 2014;46(6): 543-550. [28] SUN BB, MARANVILLE JC, PETERS JE, et al. Genomic atlas of the human plasma proteome. Nature. 2018;558(7708):73-79. [29] EVANS DM, ZHU G, DY V, et al. Genome-wide association study identifies loci affecting blood copper, selenium and zinc. Hum Mol Genet. 2013;22(19):3998-4006. [30] SAKAUE S, KANAI M, TANIGAWA Y, et al. A cross-population atlas of genetic associations for 220 human phenotypes. Nat Genet. 2021;53(10):1415-1424. [31] TACHMAZIDOU I, HATZIKOTOULAS K, SOUTHAM L, et al. Identification of new therapeutic targets for osteoarthritis through genome-wide analyses of UK Biobank data. Nat Genet. 2019;51(2):230-236. [32] SUDLOW C, GALLACHER J, ALLEN N, et al. UK biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med. 2015;12(3):e1001779. [33] PANOUTSOPOULOU K, SOUTHAM L, ELLIOTT KS, et al. Insights into the genetic architecture of osteoarthritis from stage 1 of the arcOGEN study. Ann Rheum Dis. 2011;70(5):864-867. [34] LABRECQUE J, SWANSON SA. Understanding the assumptions underlying instrumental variable analyses: a brief review of falsification strategies and related tools. Curr Epidemiol Rep. 2018;5(3):214-220. [35] YUAN K, XIE X, HUANG W, et al. Elucidating causal relationships of diet-derived circulating antioxidants and the risk of osteoporosis: a Mendelian randomization study. Front Genet. 2024;15:1346367. [36] ZOU M, LIANG Q, ZHANG W, et al. Diet-derived circulating antioxidants and risk of inflammatory bowel disease: a Mendelian randomization study and meta-analysis. Front Immunol. 2024;15:1334395. [37] ZHANG X, ZHAO H, MAN J, et al. Investigating causal associations of diet-derived circulating antioxidants with the risk of digestive system cancers: a mendelian randomization study. Nutrients. 2022;14(15):3237.
[38] PIERCE BL, BURGESS S. Efficient design for Mendelian randomization studies: subsample and 2-sample instrumental variable estimators. Am J Epidemiol. 2013; 178(7):1177-1184. [39] BOWDEN J, DAVEY SMITH G, HAYCOCK PC, et al. Consistent estimation in mendelian randomization with some invalid instruments using a weighted median estimator. Genet Epidemiol. 2016;40(4): 304-314. [40] AU YEUNG SL, GILL D. Standardizing the reporting of Mendelian randomization studies. BMC Med. 2023;21(1):187. [41] 谭文岳,韦柏安,王树声,等.甲状腺功能障碍与前列腺癌的因果关联:两样本孟德尔随机化研究[J].重庆医科大学学报,2023,48(8):935-939. [42] ANSARI MY, AHMAD N, HAQQI TM. Oxidative stress and inflammation in osteoarthritis pathogenesis: role of polyphenols. Biomed Pharmacother. 2020;129:110452. [43] CHIN KY, IMA-NIRWANA S. The role of vitamin E in preventing and treating osteoarthritis - a review of the current evidence. Front Pharmacol. 2018;9:946. [44] LIU L, ZHANG W, LIU T, et al. The physiological metabolite α-ketoglutarate ameliorates osteoarthritis by regulating mitophagy and oxidative stress. Redox Biol. 2023;62:102663. [45] COLLETTI A, CICERO AFG. Nutraceutical approach to chronic osteoarthritis: from molecular research to clinical evidence. Int J Mol Sci. 2021;22(23):12920. [46] CHIU PR, HU YC, HUANG TC, et al. Vitamin c protects chondrocytes against monosodium iodoacetate-induced osteoarthritis by multiple pathways. Int J Mol Sci. 2016; 18(1):38. [47] BARLIAN A, JUDAWISASTRA H, ALFARAFISA NM, et al. Chondrogenic differentiation of adipose-derived mesenchymal stem cells induced by L-ascorbic acid and platelet rich plasma on silk fibroin scaffold. Peer J. 2018;6:e5809. [48] CHIN KY. Updates in the skeletal and joint protective effects of tocotrienol: a mini review. Front Endocrinol (Lausanne). 2024; 15:1417191. [49] SANCHEZ-LOPEZ E, CORAS R, TORRES A, et al. Synovial inflammation in osteoarthritis progression. Nat Rev Rheumatol. 2022; 18(5):258-275. [50] CAROCHO M, FERREIRA IC. A review on antioxidants, prooxidants and related controversy: natural and synthetic compounds, screening and analysis methodologies and future perspectives. Food Chem Toxicol. 2013;51:15-25. [51] PEARSON P, LEWIS SA, BRITTON J, et al. The pro-oxidant activity of high-dose vitamin E supplements in vivo. BioDrugs. 2006;20(5):271-273. [52] SATO T, IIDA K, OHKAWA T, et al. Relationship between ankle-foot-complex mobility during static loading and frontal moment impulses of knee and hip joints during the stance phase. Gait Posture. 2024;108: 301-306. [53] HALL M, VAN DER ESCH M, HINMAN RS, et al. How does hip osteoarthritis differ from knee osteoarthritis? Osteoarthritis Cartilage. 2022;30(1):32-41. [54] LU Z, PU C, ZHANG Y, et al. Oxidative stress and psychiatric disorders: evidence from the bidirectional mendelian randomization study. Antioxidants (Basel). 2022;11(7):1386. [55] CHOE H, KOBAYASHI N, ABE K, et al. Evaluation of serum albumin and globulin in combination with c-reactive protein improves serum diagnostic accuracy for low-grade periprosthetic joint infection. J Arthroplasty. 2023;38(3):555-561. [56] LI T, ZENG J, MIAO X, et al. Association between serum albumin with geriatric nutritional risk index and osteopenia in Chinese elderly men: a nested case-control study. Asia Pac J Clin Nutr. 2024;33(4):569-580. [57] TANG Y, XU X, ZHANG S, et al. Genetic liability for diet-derived circulating antioxidants, oxidative stress, and risk of osteoarthritis: a Mendelian randomization study. Front Nutr. 2023;10:1233086. [58] XU Y, CAO X, ZHAO H, et al. Impact of camellia japonica bee pollen polyphenols on hyperuricemia and gut microbiota in potassium oxonate-induced mice. Nutrients. 2021;13(8):2665. [59] ZHOU H, SHEN X, YAN C, et al. Extracellular vesicles derived from human umbilical cord mesenchymal stem cells alleviate osteoarthritis of the knee in mice model by interacting with METTL3 to reduce m6A of NLRP3 in macrophage. Stem Cell Res Ther. 2022;13(1):322. [60] NISHIZAWA H, MAEDA N, SHIMOMURA I. Impact of hyperuricemia on chronic kidney disease and atherosclerotic cardiovascular disease. Hypertens Res. 2022;45(4):635-640. [61] CIOSEK Ż, KOT K, ROTTER I. Iron, zinc, copper, cadmium, mercury, and bone tissue. Int J Environ Res Public Health. 2023; 20(3):2197. [62] WEN X, WANG J, PEI X, et al. Zinc-based biomaterials for bone repair and regeneration: mechanism and applications. J Mater Chem B. 2023;11(48):11405-11425. [63] SCHOOFS H, SCHMIT J, RINK L. Zinc toxicity: understanding the limits. Molecules. 2024; 29(13):3130. [64] LI G, CHENG T, YU X. The impact of trace elements on osteoarthritis. Front Med (Lausanne). 2021;8:771297. [65] KITABAYASHI C, FUKADA T, KANAMOTO M, et al. Zinc suppresses Th17 development via inhibition of STAT3 activation. Int Immunol. 2010;22(5):375-386. [66] DENOBLE AE, HUFFMAN KM, STABLER TV, et al. Uric acid is a danger signal of increasing risk for osteoarthritis through inflammasome activation. Proc Natl Acad Sci U S A. 2011;108(5):2088-2093. [67] SHI H, WANG H, YU M, et al. wai Serum trace elements and osteoarthritis: a meta-analysis and Mendelian randomization study. J Trace Elem Med Biol. 2024;86:127520. [68] CUI A, XIAO P, WANG P, et al. No genetic causal association between circulating alpha-tocopherol levels and osteoarthritis, a two-sample Mendelian randomization analysis. Sci Rep. 2024;14(1):10099. [69] PATTISON DJ, SILMAN AJ, GOODSON NJ, et al. Vitamin C and the risk of developing inflammatory polyarthritis: prospective nested case-control study. Ann Rheum Dis. 2004;63(7):843-847. [70] CANTER PH, WIDER B, ERNST E. The antioxidant vitamins A, C, E and selenium in the treatment of arthritis: a systematic review of randomized clinical trials. Rheumatology (Oxford). 2007;46(8): 1223-1233. [71] SEKI T, HASEGAWA Y, YAMAGUCHI J, et al. Association of serum carotenoids, retinol, and tocopherols with radiographic knee osteoarthritis: possible risk factors in rural Japanese inhabitants. J Orthop Sci. 2010;15(4):477-484. |
| [1] | Chen Jiayong, Tang Meiling, Lu Jianqi, Pang Yan, Yang Shangbing, Mao Meiling, Luo Wenkuan, Lu Wei, Zhou Jiatan. Based on Mendelian randomization, the causal relationship between 1400 metabolites and sarcopenia and the correlation analysis of cardiovascular disease were investigated [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(在线): 1-11. |
| [2] | Zhang Yibo, Lu Jianqi, Mao Meiling, Pang Yan, Dong Li, Yang Shangbing, Xiao Xiang. Exploring the causal relationship between rheumatoid arthritis and coronary atherosclerosis: a Mendel randomized study involving serum metabolites and inflammatory factors [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(在线): 1-9. |
| [3] | Ma Chi, Wang Ning, Chen Yong, Wei Zhihan, Liu Fengji, Piao Chengzhe. Application of 3D-printing patient-specific instruments combined with customized locking plate in opening wedge high tibial osteotomy [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(9): 1863-1869. |
| [4] | Sun Yundi, Cheng Lulu, Wan Haili, Chang Ying, Xiong Wenjuan, Xia Yuan. Effect of neuromuscular exercise for knee osteoarthritis pain and function: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(9): 1945-1952. |
| [5] | Dong Tingting, Chen Tianxin, Li Yan, Zhang Sheng, Zhang Lei. Causal relationship between modifiable factors and joint sports injuries [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(9): 1953-1962. |
| [6] | Chen Shuai, Jin Jie, Han Huawei, Tian Ningsheng, Li Zhiwei . Causal relationship between circulating inflammatory cytokines and bone mineral density based on two-sample Mendelian randomization [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(8): 1556-1564. |
| [7] | Wang Peiguang, Zhang Xiaowen, Mai Meisi, Li Luqian, Huang Hao. Generalized equation estimation of the therapeutic effect of floating needle therapy combined with acupoint embedding on different stages of human knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(8): 1565-1571. |
| [8] | He Guanghui, Yuan Jie, Ke Yanqin, Qiu Xiaoting, Zhang Xiaoling. Hemin regulates mitochondrial pathway of oxidative stress in mouse chondrocytes [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1183-1191. |
| [9] |
Zhao Wensheng, Li Xiaolin, Peng Changhua, Deng Jia, Sheng Hao, Chen Hongwei, Zhang Chaoju, He Chuan.
Gut microbiota and osteoporotic fractures #br#
#br#
[J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1296-1304.
|
| [10] | Ma Haoyu, Qiao Hongchao, Hao Qianqian, Shi Dongbo. Causal effects of different exercise intensities on the risk of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1305-1311. |
| [11] | Li Jiatong, Jin Yue, Liu Runjia, Song Bowen, Zhu Xiaoqian, Li Nianhu . Association between thyroid function levels and phenotypes associated with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1312-1320. |
| [12] | Wu Guangtao, Qin Gang, He Kaiyi, Fan Yidong, Li Weicai, Zhu Baogang, Cao Ying . Causal relationship between immune cells and knee osteoarthritis: a two-sample bi-directional Mendelian randomization analysis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(5): 1081-1090. |
| [13] | Liu Ning, Sun Yingjin, Huang Long, Feng Shuo, Chen Xiangyang. Optimal rotational alignment of the tibial component during Oxford unicompartmental knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(33): 7158-7164. |
| [14] | Wu Zhenhua, Zhang Xiwei, Wang Yipin, Li Qianqian. Relationship between seven serum lipid traits and osteoarthritis: a large sample analysis of European population in IEU OPEN GWAS database [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(32): 7004-7014. |
| [15] | Zhang Bochun, Li Wei, Li Guangzheng, Ding Haoqin, Li Gang, Liang Xuezhen, . Association between neuroimaging changes and osteonecrosis: a large sample analysis from UK Biobank and FinnGen databases [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(30): 6574-6582. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||