Chinese Journal of Tissue Engineering Research
Construction of tissue-engineered heart valves: Status and Prospects
Sun Yan-li1, Han Hong-guang2, Huang Dai-fa1, He Shun-chuan1, Li Yu-jie3
- 1Second Department of Cadre Medicine, 2Department of Cardiovascular Surgery, General Hospital of Shenyang Military Region, Shenyang 110016, Liaoning Province, China; 3Beijing Second Retired Cadres, Military Training Department of PLA General Staff Department, Beijing 100093, China
-
Revised:2013-10-31Online:2013-12-10Published:2013-12-10 -
Contact:Han Hong-guang, M.D., Associate chief physician Master’s supervisor, Department of Cardiovascular Surgery, General Hospital of Shenyang Military Region, Shenyang 110016, Liaoning Province, China hanxiyao@163.com -
About author:Sun Yan-li, Attending physician, Second Department of Cadre Medicine, General Hospital of Shenyang Military Region, Shenyang 110016, Liaoning Province, China 18909881676@163.com6
CLC Number:
Cite this article
Sun Yan-li, Han Hong-guang, Huang Dai-fa, He Shun-chuan, Li Yu-jie. Construction of tissue-engineered heart valves: Status and Prospects[J]. Chinese Journal of Tissue Engineering Research, doi: 10.3969/j.issn.2095-4344.2013.50.024.
share this article
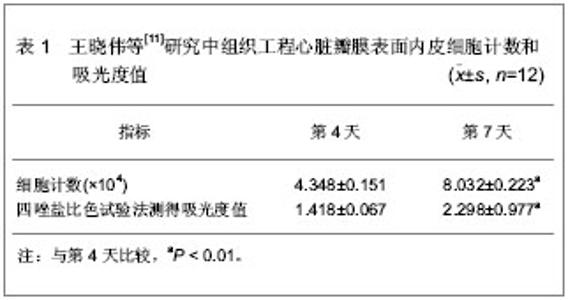
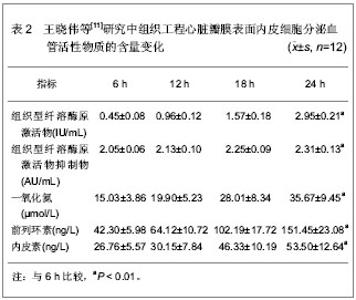
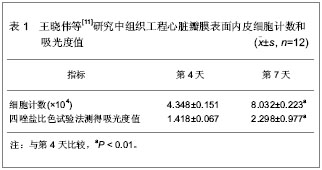
2.1 构建组织工程心脏瓣膜的种子细胞 人体正常心脏瓣膜是由特异的瓣膜上皮细胞、间质细胞和多种间质成分按照特定的排列和结构所组成的一个复合 体[9]。目前,用于构建组织工程心脏瓣膜的种子细胞有几种,从细胞的来源上看,自体来源细胞的临床应该是最佳选择。理想的种子细胞应该满足以下几点要求:①获取方便,数量足够大或者能够通过体外培养扩增。②与正常心脏瓣膜细胞有着相似的生理功能,抗炎、抗凝、无免疫原性。③黏附力强,能够牢固的黏附在瓣膜支架上生长。④生长及修复能力强,能产生细胞外基质并最终取代瓣膜支架。 2.1.1 血管内皮细胞 血管内皮细胞在心血管系统起到抗血栓形成的作用,同时抑制血小板聚集和分泌血管活性因子,是构建组织工程心脏瓣膜的种子细胞来源之一。血管内皮细胞包括静脉内皮细胞和动脉内皮细胞两种。周虹等[10]应用脂肪间充质干细胞在体外诱导分化为内皮细胞作为种子细胞来构建组织工程心脏瓣膜,并种植于支架上,研究发现支架上排列连续的单细胞层,脂肪间充质干细胞在体外向内皮细胞诱导分化后初步具有内皮细胞的功能,且生长良好,可以在体外构建组织工程心脏瓣膜。 王晓伟等[11]以脱细胞猪主动脉瓣作为支架,扩增人脐静脉血管内皮细胞并种植在瓣膜上,体外静态构建组织工程心脏瓣膜。选脱细胞瓣叶裁成直径 1.5 cm圆片置入12孔板中,胎牛血清中浸泡12 h后,种植人脐静脉血管内皮细胞3次,间隔24 h,每次数目为1×105个,再继续培养4 d,共培养7 d。第7天取瓣膜固定,行苏木精-伊红染色(×200)和扫描电镜观察(×350),血管内皮细胞Ⅷ因子免疫组织化学染色(×400)鉴定。从种植细胞的第1天算起,在开始种植细胞的第4,7天进行单个瓣叶光镜细胞计数和四唑盐比色试验。组织工程心脏瓣膜内皮细胞生理功能检测是将瓣膜移入另一个12孔板中。加入培养液 3 mL,分别于6,12,18和24 h 吸取培养液1.5 mL,3 000 r/min离心10 min,取上清-80 ℃冰箱保存。组织型纤溶酶原激活物和组织型纤溶酶原激活物抑制物活性用发色底物法,一氧化氮采用硝酸还原酶法,前列环素和内皮素采用放射免疫法进行检测。瓣叶上内皮细胞的生长状态,见表1。构建组织工程心脏瓣膜表面内皮细胞分泌的血管活性物质含量,见表2。"
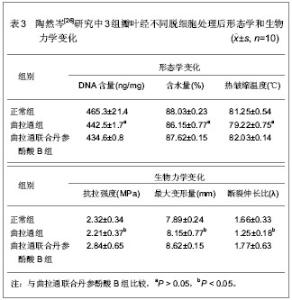
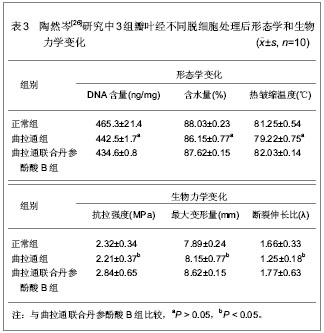
2.1.2 内皮祖细胞 有研究表明,内皮祖细胞可以用于构建组织工程心脏瓣膜[12]。张怀勤等[13]利用外周血内皮祖细胞制备组织工程瓣膜的可行性,分离人外周血内皮祖细胞,用酶去垢剂法去除新鲜猪主动脉瓣细胞制备去细胞瓣膜支架,将培养的人外周血内皮祖细胞接种到去细胞瓣膜上。人外周血内皮祖细胞与去细胞瓣膜共孵育2周后,细胞紧贴瓣膜表面生长形成一层连续的单细胞层,因此,外周血分离培养扩增得到的内皮祖细胞能够再内皮化去细胞猪主动脉瓣构建组织工程瓣膜,外周血内皮祖细胞是组织工程瓣膜内皮种子细胞的新的来源。顾春虎等[14]采用密度梯度离心法从羊骨髓中分离出内皮祖细胞,并诱导分化后,免疫组织化学染色鉴定。将诱导分化得到内皮祖细胞种植到去细胞猪主动脉瓣膜上。内皮祖细胞经定向诱导后,具有表达CD31、CD34和von Willcbrand因子等内皮细胞特征,诱导分化后的内皮祖细胞可以在去细胞猪主动脉瓣膜表面黏附生长,并且表达CD-31和von Willcbrand因子。 2.1.3 干细胞 骨髓基质干细胞作为组织工程心脏瓣膜的种子细胞具有以下特点:①获得的低侵入性。人骨髓基质干细胞可以通过骨髓穿刺简单获得并有在体外快速扩增的能力,这对临床实验应用是非常重要的。②独特的免疫学特征。可通过免疫诱导耐受或分子修饰调控等措施使之与异种瓣膜支架间获得更好的组织相容性[15],同时也为同种异体间基质干细胞的移植提供一种可能[16]。③多向分化能力,从理论上来讲,在适宜的条件下,基质干细胞可以分化为各种自体瓣膜细胞成分,既无需分层种植,也可以避免其它来源的种子细胞与瓣膜细胞间的差异,而这种差异对组织工程心脏瓣膜的结构和功能维系可以是至关重要的[17]。傅杰[18]通过全髓贴壁法获得数量足够的、均质的骨髓间充质干细胞;骨髓间充质干细胞在L-DMEM胎牛血清不同浓度以及不同因子的诱导条件下可以定向分化,通过此法可在体外获得大量高纯度的内皮细胞、平滑肌细胞和成纤维细胞,用于构建组织工程心脏瓣膜。此外,国外有学者研究用羊水干细胞等构建组织工程心脏瓣膜[19]。 2.2 构建组织工程心脏瓣膜的支架材料 在组织工程心脏瓣膜的研究中,生物支架材料的研究尤其关键。理想的组织工程心脏瓣膜支架材料应该具有以下特点:①有良好的可塑性和机械强度,能够支持细胞生长和细胞间的相互作用,为新生组织提供支撑,保持一定时间的生物力学特征,从而引导组织形成一个具有器官细胞外基质的功能器官。②良好的生物相容性,并允许有足够的细胞生长并形成抗血栓细胞内膜,并有利于种子细胞的黏附和生长。③生物降解率可控,降解率要与新生组织细胞生长率相适应,可以根据组织生长时间进行调控和调整,而且能在新器官生成之前提供足够的机械强度,承受快速血流所产生的张力和剪切力。④能够提供良好的材料-细胞界面,具有生物表面活性,有利于细胞黏附、生长,并能激活细胞特异基因表达,维持正常细胞表型表达。⑤具有三维立体多孔性结构特点,至少含有90%微孔,还要具有较高的面积/体积比和较大的内表面积,并组成一个相互关联的孔隙网络,有利于细胞的生长和营养代谢。⑥易于保存和消毒。组织工程心脏瓣膜支架材料有两种,天然支架材料和人工合成材料。天然支架材料包括脱细胞支架、胶原支架等,人工合成材料如聚乳酸、聚乳酸羟基乙酸、聚羟基烷酸酯等。 脱细胞方法是生物瓣支架材料的常用技术,脱细胞组织纤维支架保留了完整的细胞生存三维微环境,是细胞黏附的良好平台[20-24]。脱细胞方法在心血管领域应用包括去垢剂法、酶消化法。去垢剂方法首先用Triton X-100对组织处理,再用SDS、Triton X-100或胆脂酸钠对组织进行脱细胞。去垢剂法和酶消化法脱细胞有3个基本步骤,首先将组织浸入含蛋白酶抑制剂的低渗液内,其次,含Triton X-100高渗盐溶液处理,最后,酶溶液消化处理。单纯去垢剂法处理后的组织主要成分为胶原、弹性蛋白、纤维黏连素及层黏连蛋白,与正常组织非常相似。去垢剂法和酶消化法处理后的组织主要成分为弹性蛋白、非可溶性胶原和紧密连接的糖性蛋白。对于脱细胞方法的使用上,目前还存在着争议。国内关于组织工程心脏瓣膜支架材料的研究还不是很多,举列如下。 2.2.1 Triton X-100-SD法脱猪主动脉瓣膜细胞 李文通[25]比较不同脱细胞方法去除组织细胞,选取健康成年猪心脏,将主动脉瓣膜连同动脉管壁一并取出,生理盐水反复冲洗后无菌条件下剪取主动脉瓣叶,分为对照组、Triton X-100-SD法脱细胞处理组和去垢剂-酶消化法脱细胞处理组,每组16个新鲜猪主动脉瓣叶,处理后进行组织学观察以及DNA含量分析。发现去垢剂-酶消化法脱细胞处理组瓣叶处理后可以完全去除细胞,但瓣膜原有纤维结构发生改变。Triton X-100-SD法脱细胞处理组瓣叶处理后脱细胞完全,并能够保持瓣膜原有纤维结构,是一种较好的组织工程瓣膜生物支架制备方法。 2.2.2 丹参酚酸B处理脱细胞猪心脏瓣膜 陶然岑[26]观察曲拉通法联合丹参酚酸B处理对猪心脏瓣膜形态学以及生物力学的影响。选取六七个月龄猪,取主动脉瓣膜,将120片瓣叶随机分为曲拉通组、曲拉通联合丹参酚酸B组和正常组,每组40片,观察各种在形态学和生物力学方面的变化,见表3。"
| [1] Zilla P, Brink J, Human P, et al. Prosthetic heart valves: catering for the few.Biomaterials. 2008;29(4):385-406.[2] MURRAY G. Homologous aortic-valve-segment transplants as surgical treatment for aortic and mitral insufficiency. Angiology. 1956;7(5):466-471.[3] Krahn AD, Healey JS, Simpson CS, et al. Anticoagulation of patients on chronic warfarin undergoing arrhythmia device surgery: wide variability of perioperative bridging in Canada. Heart Rhythm. 2009;6(9):1276-1279.[4] 邱雪峰,董念国.组织工程心脏瓣膜的种子细胞[J].中国组织工程研究与临床康复,2008,12(29):5719-5722.[5] Nemeth NL, Butler CE. Complex torso reconstruction with human acellular dermal matrix:long-term clinical follow-up. Plast Reconstr Surg.2009;123(1):192-196.[6] Freed LE, Vunjak-Novakovic G, Biron RJ, et al. Biodegradable polymer scaffolds for tissue engineering. Biotechnology(NY). 1994;12(7):689-693.[7] 万方数据库.万方医学网[DB/OL].2013-08-28. https://med.wanfangdata.com.cn[8] Medline.Pubmed[DB/OL].2013-08-28.http://www.ncbi.nlm.nih.gov/pubmed/[9] 谢甲琦,魏旭峰,易定华.心脏瓣膜的结构--构建组织工程心脏瓣膜的蓝图[J].中华胸心血管外科杂志,2009,25(5):289-291.[10] 周虹,张涛.脂肪间充质干细胞分化为内皮细胞并体外构建组织工程心脏瓣膜[J].中国组织工程研究,2012,16(27):4979-4984.[11] 王晓伟,叶福林,徐志云,等.人脐静脉血管内皮细胞构建组织工程心脏瓣膜及其生理功能[J].江苏医药,2007,33(2):156-159.[12] Cebotari S, Lichtenberg A, Tudorache I, et al. Clinical application of tissue engineered human heart valves using autologous progenitor cells. Circulation.2006;114(1 Suppl): I132-137.[13] 张怀勤,肖刚峰,孙成超,等.内皮祖细胞作为组织工程瓣膜内皮种子细胞的可行性研究[J].中国生物医学工程学报,2007,26(1): 110-115.[14] 顾春虎,王云雅,易定华,等.内皮祖细胞和去细胞猪主动脉瓣构建组织工程瓣膜的研究[J].中华实验外科杂志,2007,24(3): 263-265.[15] Prockop DJ. Marrow stromal cells as stem cells for nonhematopoietic tissues. Science.1997;276(5309):71-74.[16] Xiao YF, Min JY, Morgan JP. Immunosuppression and xenotransplantation of cells for cardiac repair. Ann Thorac Surg. 2004;77(2):737-744.[17] Roy A, Brand NJ, Yacoub MH. Molecular characterization of interstitial cells isolated from human heart valves. J Heart Valve Dis. 2000;9(3):459-465.[18] 傅杰.骨髓间充质干细胞定向分化心脏瓣膜构成细胞的实验研究[D].重庆:重庆医科大学,2010:1-15.[19] Weber B, Emmert MY, Behr L, et al. Prenatally engineered autologous amniotic fluid stem cell-based heart valves in the fetal circulation. Biomaterials. 2012;33(16):4031-4043.[20] Van lieshout M, Peters G, Rutten M, et al. A knitted, fibrin-covered polycaprolactone scaffold for tissue engineering of the aortic value. Tissue Eng. 2006;12(3): 481-487.[21] Kasimir MT, Rieder E, Seebacher G, et al. Comparison of different decelluarization procedures of porcine heart valves. Int J Artif Organs.2003;26(5):421-427.[22] Mirsadraee S, Wilcox HE, Korossis SA, et al. Development and characterization of an acellular human pericardial matrix for tissue engineering. Tissue Eng.2006;12(4):763-773.[23] 杨岷,陈长志,成少飞,等.牛细胞组织工程支架脱细胞方法的比较[J].中华胸心血管外科杂志,2005,21(6):349-351.[24] 王克学,张镜方,詹秋鹏,等.胰蛋白酶法和Triton-X法去除瓣膜细胞的效果比较[J].第一军医大学学报,2005,25(1):22-25.[25] 李文通.Triton+X-100-SD法与去垢剂-酶消化法脱猪主动脉瓣膜细胞的对比研究[D].江西:南昌大学,2012:5-15.[26] 陶然岑.丹参酚酸B后处理脱细胞猪心脏瓣膜的形态学及生物力学研究[D].山西:山西医科大学,2012:1-11.[27] 徐磊.PEG纳米微球改性去细胞瓣构建组织工程心脏瓣膜的实验研究[D].湖北:华中科技大学,2010:3-5.[28] Bader A, Schilling T, Teebken OE, et al .Tissue engineering of heart valves--human endothelial cell seeding of detergent acellularized porcine valves.Eur J Cardiothorac Surg. 1998; 14(3):279-284.[29] Steinhoff G, Stock U, Karim N, et al. Tissue engineering of pulmonary heart valves on allogenic acellular matrix conduits: in vivo restoration of valve tissue.Circulation. 2000;102 (19 Suppl 3):III50-55.[30] Kasimir MT, Rieder E, Seebacher G, et al. Comparison of different decellularization procedures of porcine heart valves.Int J Artif Organs. 2003;26(5):421-427.[31] Dohmen PM, Ozaki S, Verbeken E, et al. Tissue engineering of an auto-xenograft pulmonary heart valve.Asian Cardiovasc Thorac Ann. 2002;10(1):25-30.[32] Shinoka T, Breuer CK, Tanel RE, et al.Tissue engineering heart valves: valve leaflet replacement study in a lamb model.Ann Thorac Surg. 1995;60(6 Suppl):S513-516.[33] 史峰,邓诚,胡行健,等.生物反应器内构建组织工程心脏瓣膜[J].中华实验外科杂志,2013,30(2):315-316.[34] 李大刚,程光存,施益民,等.类金刚石涂层瓣膜与犬血管内皮细胞的相容性[J].中国组织工程研究,2012,16(21):3891-3894.[35] 康凯,曲辉,汤断权,等.利用人骨髓基质干细胞构建组织工程心脏瓣膜的体外实验[J].中国组织工程研究,2013,17(6):957-962. |
| [1] | Zhao Weibiao, He Ziwei, Li Ji, Li Yi. Application value of 3D printing guide plate in SuperPATH technology for elderly hip arthroplasty: retrospective study and evidence analysis of literature retrieval [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(9): 1324-1330. |
| [2] | Gao Yang, Bai Shengchao, Chen Shengju, Wang Ruiyuan, Li Junping. Effect of exercise-induced muscle damage on desmin aggregates [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(8): 1243-1248. |
| [3] | Chen Juan, Zhang Ting, Wu Yidan, Lu Yan, Ouyang Zhaolian. China’s strengths in basic research in the main subfields of tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(8): 1267-1271. |
| [4] | Wang Gang, Li Donghui, Bai Zhiming. Evolution and progress of replacement therapy, materials and reconstruction of long ureteral injuries [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(8): 1299-1305. |
| [5] |
Xie Fei, Li Yanle, Lin Xinxiao, Hu Haiwei, Sang Zhicheng, Sun Yongsheng, Jiang Kewei, Cheng Ying, Wen Guannan, Wen Jianmin, Sun Weidong.
Mechanism underlying mechanical stress regulating fibroblasts-derived
exosomes at the osteotomized end following hallux valgus correction |
| [6] | Zhang Shengmin, Liu Chao. Research progress in osteogenic differentiation of adipose-derived stem cells induced by bioscaffold materials [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(7): 1107-1116. |
| [7] | Gao Yang, Liang Xiaotian, Wang Bo, Li Junping, Wang Ruiyuan. Characterization of aggrephagy in exercise-induced muscle damage [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(5): 720-725. |
| [8] | Jiang Biantong, Zhang Zhigang, Jin Xiu, Wang Haiye, Wu Yuchen, Zhang Caiyun. Effects of different calcium supplementation pathways on calcium ion in calcium-free regional citrate anticoagulation-continuous renal replacement therapy: study protocol for a prospective randomized cross-over trial [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(5): 726-730. |
| [9] | Yan Shu, Lu Yan, Ouyang Zhaolian. Analysis of programs on tissue engineering funded by the National Natural Science Foundation of China between 2013 and 2018 [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(5): 731-735. |
| [10] | Hu Chaoran, Qiu Bing, Zhou Zhuxing, Yang Yang, Li Jia. In vitro biocompatibility of 3D printed polycaprolactone/nano-hydroxyapatite composite scaffold with bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 589-595. |
| [11] | Tang Jingfeng, Zhang Jun, You Qi, Liu Yi. The role and mechanism of graphene and its derivatives-related composites in cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 619-624. |
| [12] | Chen Jia, Yang Yiqiang, Hu Chen, Chen Qi, Zhao Tian, Yong Min, Ma Dongyang, Ren Liling. Fabrication of prevascularized osteogenic differentiated cell sheet based on human bone marrow mesenchymal stem cells and human umbilical vein endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 4934-4940. |
| [13] | Wang Yanghao, Wang Weizhou, Duan Hao, Zhong Zongyu, Li Xiaozhuang, Tang Zhihong, He Fei. Different material factors affect the proliferation and osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(28): 4429-4436. |
| [14] | Zhang Weizhong, Li Lei, He He, He Xin. Application and significance of nanofibrous macroporous scaffold preparation technology for bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(28): 4437-4444. |
| [15] | Shi Jun, Yang Lin, Guo Zhibin, Cui Yutao, Liu He. How does extracellular matrix-based growth factor delivery system promote osteogenesis? [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(28): 4445-4451. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||